Abstract
Background:
Recombinant activated factor VIIa (rFVIIa) is a prohemostatic agent initially approved for use in hemophilia patients and has also been used for a diverse range of off-label indications in the context of massive uncontrolled blood loss; however, no convincing evidence exists regarding the optimal dose of rFVIIa to treat uncontrolled bleeding in surgical patients.
Aim:
To evaluate the effects and safety of a very low dose of rFⅦa in patients with uncontrolled perioperative bleeding in the surgical intensive care unit (ICU).
Methods:
55 patients from Beijing Hospital, who received rFⅦa between July 2004 and November 2018 for uncontrolled perioperative bleeding were included. The controls were matched for age, sex, severity, and operation type. The baseline demographics, survival, changes in bleeding and transfusion, coagulation parameters and complications were analyzed.
Results:
A low dose of rFⅦa (2.0∼3.6 mg, with a median dose of 39.02 μg/kg) appears to be effective in controlling massive hemorrhage (with an effective rate of 74.55%), and can reduce volume of red blood cell transfusion, improve coagulation status, while has a relatively low risk of thromboembolic complications (3.6%).
Conclusion:
In patients with uncontrolled perioperative bleeding, a low dose of rFⅦa could be used when traditional methods are ineffective.
Keywords
Introduction
Death from hemorrhage represents a substantial global problem; hemorrhage is not only the leading cause of trauma-related mortality but also is a very common perioperative complication that is very challenging to doctors. 1 -3 In many cases, the use of surgical hemostasis and blood products to correct potential coagulation dysfunction and to control critical bleeding is feasible. However, in a few cases, the bleeding remains uncontrollable, and mortality in patients with massive uncontrolled hemorrhage and coagulation dysfunction still remains very high. 4,5
As a prohemostatic agent, recombinant factor Ⅶa (rFⅦa) has been approved to treat bleeding in patients with hemophilia, those with inhibiting antibodies against factor Ⅷ or Ⅸ, those with Glanzmann thrombasthenia, etc., 6 and has achieved remarkable curative effects. rFⅦa has also been gradually applied to other congenital or acquired uncontrolled hemorrhagic diseases in patients without hemophilia and has also achieved good results. 7 In 2017, European guidelines on the management of bleeding following major trauma and surgery, rFⅦa is suggested only if major bleeding and coagulopathy persist when all conventional methods have failed. 4,8
However, the dose of rFⅦa for massive bleeding, which varies from 20 µg/kg to 200 μg/kg, remains greatly controversial. 9,10 The recommended optimum dose of 90 µg/kg came from the prescribing information to treat bleeding patients with hemophilia A or B. Although many publications reported an effective dose similar to the standard recommendation or even a much higher dose, 10 some studies indicate that lower doses might also have therapeutic effects. 9,11 -14
On the other hand, the incidence of rFⅦa side effects has been reported to range from <1% to 44%, 15,16 predominantly thromboembolic events (9.0%∼9.4%). 15,17,18 Studies show that rFⅦa treatments increased the incidence of arterial thromboembolic events, particularly among elderly patients, and those with cardiovascular comorbidities. 18 -20 Furthermore, rFⅦa dosage was significantly correlated with the rate of arterial thromboembolic events in patients with central nervous system bleeding. 18
Based on the mechanisms of rFⅦa in hemostasis 20,21 and previous pilot studies, 9 and additionally, due to its high economic burden for Chinese patients, we began using low-dose rFⅦa (usually a fixed dose of 2.0∼3.6 mg for all patients) to treat uncontrolled critical bleeding since 2004. In this study, we retrospectively reviewed 55 patients who used rFⅦa for uncontrolled perioperative massive bleeding to evaluate the effects and safety of a low dose rFⅦa. We hypothesized that a low dose of rFⅦa is efficacious in controlling massive bleeding with a low complication rate.
Materials and Methods
Study Design
This is a single-center retrospective observational study at Beijing Hospital, a tertiary care public hospital in Beijing, P.R. China. All patients who underwent surgery and had uncontrolled bleeding treated with rFⅦa from July 2004 to November 2018 were enrolled. All included patients were from the Department of Surgical Intensive Care Medicine, from other surgical departments including operation room, or from consultations with our group.
With the Electronic Transfusion Database, we also matched massive transfusion (≥1,000ml/day) case-control patients with patients who received rFⅦa based on severity. The control group was matched according to age, sex, Acute Physiology and Chronic Health Evaluation Ⅱ (APACHE Ⅱ) score and volume of transfusion. All data were obtained retrospectively through a review of the electronic medical records and Electronic Transfusion Database and documented by the trained physician(s). A standardized data collection form was used. Patients were excluded from the analysis if they had missing baseline or outcome data. The incidence of arterial thrombosis, acute myocardial infarction (AMI), ischemic cerebral vascular events, deep venous thrombosis (DVT), pulmonary embolus (PE), and death were determined in both groups during their stay in hospital. DVT was routinely screened for by point-of-care ultrasound. The study followed the principles of the Declaration of Helsinki and was approved by Beijing Hospital Ethics Committee, written informed consent was waived by the Beijing Hospital Ethics Committee because of the retrospective nature of the study and data are anonymized (2020BJYYEC-121-01).
Protocol and Indications for rFⅦa Use
Off-label use of rFⅦa was considered for patients with intractable life-threatening bleeding that could not be stopped by surgical or radiological methods and/or when conventional comprehensive coagulation therapy failed. 4,8 For massive hemorrhage, conventional hemostatic methods, including angiography artery embolization and surgical hemostasis, were considered the priority. Other traditional hemostatic measures included transfusion of allogenic blood products (fresh frozen plasma (FFP), platelets (PLTs)), replacement of coagulation factors (fibrinogen, prothrombin complex concentrate (PCC)), desmopressin (DDAVP), vitamin K for liver insufficiency, tranexamic acid for trauma patients, proton pump inhibitors for stress ulcer bleeding, and somatostatin treatment for portal hypertension patients.
Before applying rFⅦa, the following criteria should be met to maintain stable clotting and to ensure a favorable hemostatic effect: 22 -24 level of fibrinogen ≥1 g/L, hematocrit (HCT) ≥24%, platelets ≥50 × 1012/L, correcting acidosis and hypoxia, a pH value ≥7.20, and a normal concentration of calcium ions. The exclusion criteria included situations that are unlikely to be successfully treated, such as end-stage metastatic cancer and non-compensated disseminated intravascular coagulation (DIC), and absolute contraindications, such as AMI, and acute ischemic cerebral stroke. 25,26
Dose and Evaluation of rFⅦa
Dosing: The first dose was 2 mg (100 kiu) or 2.4 mg (120 kiu) or 3.6 mg (180 kiu), 39.02 μg/body weight i.v. If bleeding persists, the same dose was repeated after 60 to 120 minutes. Although a third dose was not advised, 15 we used 5 doses in 1 case, and the patient was successfully treated.
Coagulation tests (level of fibrinogen (Fib), prothrombin time (PT), activated partial thromboplastin time (APTT), international normalized ratio (INR), thrombin time (TT) and prothrombin activity (AT)) were routinely performed before the use of rFⅦa and 1 hour after use. The clinical effects were evaluated after administering rFⅦa: change in bleeding (reduced or stopped vs unchanged) was assessed and documented as qualitative parameters; and bleeding rates (ml per hour, when available), transfusion volume, etc. were recorded as quantitative data. For the parameters that could not be evaluated, such as bleeding rates or volume, we used objective observations of the bleeding signs, such as suction speed, the number of blood-soaked gauze pads, frequency of hematemesis, hemodynamic stability and serial hemoglobin measurements.
Statistical Analysis
Clinical and laboratory data were collected into a Microsoft Excel (Redmond, USA) worksheet. The endpoints were the change in bleeding (reduced or stopped vs unchanged), volume of blood product transfusion (red blood cells (RBCs), FFP and PLT), change in coagulation parameters (PT, APTT, INR, TT, AT), thromboembolic events and in-hospital mortality.
Statistical analysis was performed with SPSS 11.0 (IBM, USA). For continuous variables, the Kolmogorov-Smirnov test was used to examine the normality of the distribution. For nonnormally distributed data, a nonparametric test was used to analyze the quantitative variables. The Mann-Whitney U test was used to compare independent samples, and the Wilcoxon’s signed rank test was used for paired samples. T-test were used for normally distributed data, while chi-square test were used for dichotomous variables. A P value < 0.05 was considered statistically significant.
This manuscript adheres to the applicable STROBE guidelines.
Results
Baseline Characteristics of Included Patients
In total, 55 patients with uncontrolled massive perioperative bleeding who received rFⅦa during the past 15 years were included in this study. Another 51 patients with massive perioperative bleeding who received standard treatments, including blood transfusion hemostatic support, fluid resuscitation, and supportive care, were matched according to age, sex, APACHE Ⅱ score, bleeding volume, and use of fibrinogen and PCC. The baseline characteristics of the included and matched patients are shown in Table 1. No significant differences were observed between the rFⅦa group and the control group in terms of sex, age, weight, APACHE Ⅱ score, medical history, or the main causes of bleeding. 3 patients in rFⅦa group and 4 patients in control group received radiologic or surgical hemostasis (χ2 = 0.011, P = 0.918).
Baseline Characteristics of Included Patients [Median (IQR), n (%)].
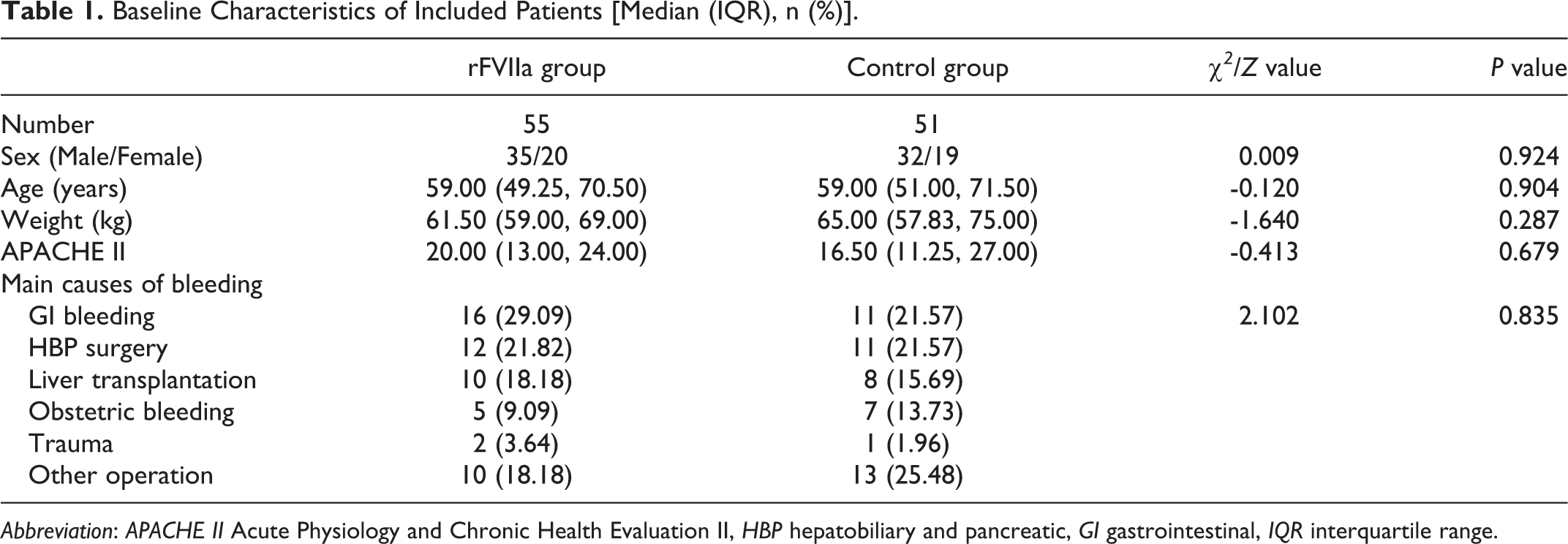
Abbreviation: APACHE Ⅱ Acute Physiology and Chronic Health Evaluation Ⅱ, HBP hepatobiliary and pancreatic, GI gastrointestinal, IQR interquartile range.
Hemostatic Treatment before the Application of rFⅦa
The hemostatic treatments used to control bleeding before using rFⅦa are shown in Table 2. Fibrinogen was used in 56.36% of the patients. The amount of fibrinogen ranged from 2.00 to 10.00 g, and the median dosage was 5.00 g. PCC was used in 60.00% of the patients. The amount of PCC ranged from 1,200.00 to 6,000.00 units, and the median amount was 2,000 units. As shown in Table 2, the number and dosage of hemostatic drugs was not significantly different between the 2 groups. Before administering rFⅦa, the average level of total calcium was adjusted to 2.0∼2.4 mmol/L.
Hemostatic Drugs Employed for the Included Patients [Median (IQR), n (%)].

Abbreviation: PCC prothrombin complex concentrate.
Application of rFⅦa
In total, 46 patients (83.64%, 46/55) received a single dose of rFⅦa. The first dose of rFⅦa was 2.0∼3.6 mg (100∼180 kiu, 2 or 3 ampules), ranging from 24.74 µg/kg to 72.00 µg/kg, and the median dosage was 39.02 (34.78, 43.64) μg/kg. Nine patients received a second administration of rFⅦa, and the repeated dose was 2.4∼3.6 mg (120∼180 kiu), ranging from 30.77 µg/kg to 62.07 µg/kg. The median dosage was 38.71 (35.04, 49.67) μg/kg. Only 1 patient received 5 repeated doses of rFⅦa, and each dose was 62.07 µg/kg.
Effects of the Use of rFⅦa on Major Clinical Outcomes
After treatment with rFⅦa, 41 patients had reduced bleeding or stopped bleeding, and the effective rate was 74.55% (41/55, Table 3). The overall in-hospital mortality rate was 45.45% (25/55); 10 patients died from bleeding or from direct complications caused by bleeding, and 15 patients stopped bleeding but still died from other causes (primary diseases, multiple organ dysfunction syndrome (MODS), etc.). A total of 26 patients in the treatment group were treated in the intensive care unit (ICU), with a median length of ICU stay of 8.00 (IQR: 2.25-11.00) days. The median length of hospital stay was 26.00 (IQR: 14.25-45.75) days.
Effects of Using rFⅦa on Major Clinical Outcomes [Median (IQR), n (%)].

Abbreviation: ICU intensive care unit(s).
We did not compare the hemostatic effects between the rFⅦa group and the control group because the use of rFⅦa was only indicated in patients with uncontrolled bleeding that could not be stopped by conventional hemostatic measures. Thus, rFⅦa was administered only if the bleeding could not be stopped by all the other conventional hemostatic measures (such as the measures that the control group patients responded to); otherwise, we did not use rFⅦa.
Effects of the Use of rFⅦa on Blood Transfusion
Blood transfusions were performed before and after the application of rFⅦa, as shown in Table 4 and Figure 1. The volume of the allogenic RBC transfusion was significantly lower after the use of rFⅦa. The volume of transfused FFP and PLT was not significantly different before and after rFⅦa use.
Blood Products Transfusions Before and After rFⅦa Use [Median (IQR)].

Abbreviation: RBC red blood cells, FFP fresh frozen plasma, PLTs Platelets.

Changes in blood transfusion before and after rFⅦa use. (A) The volume of red blood cell transfusion (unit(s)) received was significantly lower after the use of rFⅦa (Wilcoxon's Signed Rank Test Z = -3.247, P = 0.001). (B) The volume of fresh frozen plasma transfusion (ml) was not significantly different before and after rFⅦa use (Wilcoxon's Signed Rank Test Z = -1.161, P = 0.246). (C) The volume of platelets transfusion (unit(s)) was not significantly different before and after rFⅦa use (Wilcoxon's Signed Rank Test Z = -0.848, P = 0.396).
Effects of the Use of rFⅦa on Coagulation Parameters
With the included data, the coagulation parameters of each patient before and after rFⅦa administration were compared, and the results showed significant differences. Compare to those before the application of rFⅦa, the PT, TT, INR, and APTT were significantly decreased while the AT was significantly increased after the application of rFⅦa. The coagulation studies showed rapid normalization (Fib, TT, AT, APTT, INR) and even shortening (PT) of the coagulation parameters. Detailed data of the coagulation parameters before and after administering the first dose of rFⅦa are shown in Table 5 and Figure 2.
Effects of the Use of rFⅦa on Coagulation Parameters [Median (IQR)].
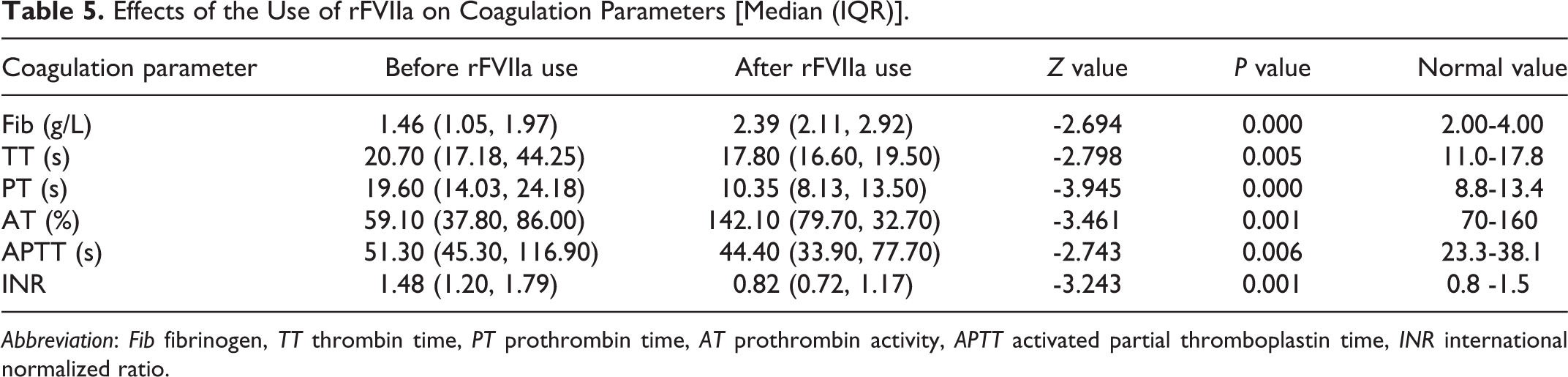
Abbreviation: Fib fibrinogen, TT thrombin time, PT prothrombin time, AT prothrombin activity, APTT activated partial thromboplastin time, INR international normalized ratio.

Effects of the use of rFⅦa on coagulation parameters. (A) Boxplot of fibrinogen level (g/L) before and after rFⅦa use. Compare to the level before the application of rFⅦa, the fibrinogen was significantly increased after the application of rFⅦa (Wilcoxon Signed Ranks Test Z = -2.694, P = 0.000). (B) Boxplot of thrombin time (TT) level (s) before and after rFⅦa use. Compare to the level before the application of rFⅦa, the TT was significantly decreased after the application of rFⅦa (Wilcoxon’s Signed Rank Test Z = -2.798, P = 0.005). (C) Boxplot of prothrombin time (PT) level (s) before and after rFⅦa use. Compare to the level before the application of rFⅦa, the PT was significantly decreased after the application of rFⅦa (Wilcoxon’s Signed Rank Test Z = -3.945, P = 0.000). (D) Boxplot of activated partial thromboplastin time (APTT) level (s) before and after rFⅦa use. Compare to the level before the application of rFⅦa, the APTT was significantly decreased after the application of rFⅦa (Wilcoxon’s Signed Rank Test Z = -2.743, P = 0.006). (E) Boxplot of prothrombin activity (AT) level (%) before and after rFⅦa use. Compare to the level before the application of rFⅦa, the AT was significantly increased after the application of rFⅦa (Wilcoxon’s Signed Rank Test Z = -3.461, P = 0.001). (F) Boxplot of international normalized ratio (INR) before and after rFⅦa use. Compare to the level before the application of rFⅦa, the INR was significantly decreased after the application of rFⅦa (Wilcoxon’s Signed Rank Test Z = -3.243, P = 0.001).
Side Effects of Using rFⅦa
Two thromboembolic complications (2/55, 3.64%) possibly related to the use of rFⅦa occurred. An 87-year-old male patient with lower GI bleeding and hemorrhagic shock was diagnosed with vascular malformation of the colon. After partial colectomy, he still presented with lower GI bleeding. The bleeding was finally stopped by a single dose of rFⅦa (2.4 mg). The patient had symptoms of paralysis 2 days later, and a Computed Tomography (CT) scan confirmed a cerebral infarction; however, we could not exclude shock-induced hypoperfusion as a potential cause. The other patient was a 26-year-old male who had right renal cell carcinoma, a 24.2 cm x 16.4 cm x 21.8 cm tumor that invaded into the liver, with retroperitoneal and peri-aortic lymph node metastasis, and a whole inferior vena cava (IVC) tumor thrombus. The patient underwent right nephrectomy, right hepatectomy (segment Ⅵ, Ⅶ, Ⅷ), cholecystectomy, and inferior vena cava tumor thrombectomy. During the operation, he had massive bleeding (over 10,000 ml) due to diffuse errhysis and severe coagulopathy. He received rFⅦa (60 µg/kg), and the bleeding decreased significantly. During the following 5 hours after the operation, the abdominal drainage level increased to 600.0 ml/h, and the hemoglobin (Hb) decreased to 57 g/L (Hb). The patient was prescribed a second dose of rFⅦa (60 µg/kg), and the drainage decreased significantly. During the following 24 hours, 3 doses of rFⅦa were given based on the changes in drainage and Hb levels, and the bleeding finally stopped on the third day. The patient was discharged from the ICU on day 7, and he developed a left femoral thrombus on day 8. The thrombus disappeared after 10 days of anticoagulation therapy.
There were no significant differences between the 2 groups regarding thrombotic complications, including DVT, AMI and cerebral infarction (Table 6). No PE was found in either group.
Side Effects in Patients Who Did and Did Not Receive rFⅦa [n (%)].

Abbreviation: DVT deep venous thrombosis, AMI acute myocardial infarction.
Discussion
In this study, we report a series of 55 patients with uncontrolled perioperative bleeding from a single center. We found that treatment with low-dose rFⅦa (fixed dose of 2.0∼3.6 mg, median 39.02 µg/kg) could significantly reduce the volume of perioperative blood loss with an effective rate of 74.55% (41/55) and the RBC transfusion volume (median 16.5 units, dropped to 7 units) (Table 4). The coagulation status improved, and the fibrinogen levels increased (Table 5), indicating the beneficial effect of using low-dose rFⅦa for uncontrolled bleeding that cannot be stopped by conventional methods. Our results showed a therapeutic effect on perioperative bleeding similar to that previously reported with a higher dosage. 18,27
Most of our patients (83.64%) received a single dose of rFⅦa, while another 9 patients received a repeated dose; only 1 patient received more than 2 doses. The overall mortality rate was 45.45% (25/55) at day 30, including 10 patients who died from bleeding or from direct complications caused by bleeding, and 15 patients who stopped bleeding but still died from other causes (primary diseases, MODS, etc.), which is slightly higher than the reported hospital mortality rate(approximately 32%). 27,28 Howes JL 15 et al. retrospectively reviewed 36 injured patients who received rFⅦa in a level 1 trauma center and found a significantly higher mortality rate in patients who received rFⅦa than in the matched control patients (52.8% vs. 22.2%, P = 0.014). However, mortality cannot be easily compared across different studies because of heterogeneous severity, different causes of hemorrhage, and varying patient characteristics. In our series, the indication for using rFⅦa as a therapeutic is refractory life-threatening hemorrhage when all conventional treatment options (surgical intervention and embolization as well as other hemostatic drugs) have failed; 4,25 thus, the administration of rFⅦa is considered relatively late, as some studies implied that the effects might be improved if rFⅦa is administered before hemostasis is severely compromised, 29 which is in accordance with our team’s experience. Additionally, due to the severity of our patients (average APACHE Ⅱ of 20), the mortality in our series is relatively high. Furthermore, there was one case of metastatic cancer, and compassionate use of rFⅦa was prescribed.
Furthermore, in our case series, only 2 out of 55 patients (3.63%) had thromboembolic complications, although these complications could not be confirmed to be directedly related to the use of rFⅦa. Schmid et al. 28 retrospectively evaluated intentional low-dose (56–71 µg/kg) rFⅦa use, but still employed a dosage higher than our median dosage, and found that patients with life-threatening hemorrhage in a single center had a surprisingly low rate of thromboembolic events: only 2 (2.7%) patients were confirmed to have thromboembolic complications. Even if both of our 2 recorded complications were subjectively considered to be directly caused by the administration of rFⅦa, the proportion is still at the lower end of the incidence range reported. 15,16,18,28 According to the meta-analysis by Levy et al., 18 even in placebo groups, the rate of arterial thromboembolic complications was still 3.2%.
Although some research showed that high-dose factor Ⅶa was hemostatically effective but did not systemically activate coagulation, after application of rFⅦa in our series, the coagulation studies showed rapid normalization (Fib, TT, AT, APTT, INR) and even shortened (PT) coagulation parameters; these results are slightly different from Schmid et al., 28 who found typical normalization of PT. rFⅦa could theoretically amplify and limit the coagulation activation cascade at the site of injury through 2 mechanisms. 30,31 The main mechanism is that rFⅦa can directly form complex compounds with the tissue factor (TF) at sites of disrupted endothelium and then serially activate the common coagulation cascade via the activation of factor Ⅹ. Alternatively, rFⅦa could directly bind to the surface of activated platelets, thus also concentrating activated factor Ⅹ to the sites of damage. Both mechanisms generate large amounts of activated factor Ⅹ, drive the thrombin burst, initiate the formation of the fibrinogen meshwork, and finally improve coagulation and clot stabilization. The underlying rationale for the application of low-dose rFⅦa is that 39.02 μg/kg could also lead to a plasmatic FⅦa concentration higher than the minimal effective level, 20 possibly reducing the theoretical side effects of excessive thrombin generation caused by high rFⅦa doses. 21
Despite the previous consensus that rFⅦa is only used when surgical and radiotherapy are ineffective 32,33 and as the last resort, there are still 2 cases where rFⅦa was used before surgical reintervention (the bleeding still continued after rFⅦa use). One patient was successfully treated with reoperation, and the other patient stopped bleeding after arterial embolization. rFⅦa was used in these rare but difficult clinical scenarios when the surgeons insisted that the bleeding was not caused by surgery or when the patients’ family refused a second operation. Ultimately, surgical intervention is considered again based on the opinions of the surgeon or family.
In summary, our present study indicates that a low dose (median 39.02 μg/kg) of rFⅦa is also reasonably effective in controlling massive hemorrhage but has a relatively low risk of thromboembolic complications. However, there were several limitations to the study. First, our report is a retrospective observational single-center study, although randomized placebo-controlled trials are extremely difficult in real clinical scenarios. Second, the number of patients included is relatively small due to the rare incidence of massive uncontrolled hemorrhage. Finally, as a retrospective analysis, the missing data might also affect the power of our findings. Although robust conclusions about efficacy or safety of rFVIIa in this setting is difficult to make by this descriptive study, it adds to the literature of off-label use of rFVIIa for uncontrolled bleeding, while provided a promising method of choice during clinical emergency with 15 years of experience.
Footnotes
Abbreviations
Authors’ Contributions
Z C, Y L, W C, X C, J H, D L and J G created the database and collected the data; Z C, Y L, Q H, Z F, and S X performed the study. Z C and D L performed the statistical analysis and interpreted the data; Z C and X C wrote the manuscript; all authors provided final approvals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the China MOHRSS: Supporting Scheme for Returned Overseas Chinese Students’ Scientific and Technological Programs. [grant numbers: BJ-2016-060].
