Abstract
Objectives
Following subarachnoid hemorrhage (SAH), long-lasting inflammation triggered by activated glial cells has adverse effects on neurological recovery. As an α2 adrenoceptor agonist commonly utilized for sedative purposes, dexmedetomidine (DEX) has demonstrated the ability to confer neuroprotective effects across diverse physiological or pathological conditions. This study was designed to determine whether DEX protects against SAH by altering astrocytic reactivity.
Methods
Eight-week-old male C57BL/6 mice were subjected to experimental SAH. They were treated with DEX in the presence or absence of the α2 adrenoceptor antagonist atipamezole (ATI) via intraperitoneal injection. Neurological function was evaluated on the basis of a modified Garcia score and beam balance test. TUNEL staining was conducted to assess neuronal apoptosis. Western blotting was carried out to determine the expression of Bcl-2, Bax, and cleaved caspase-3 in the hippocampus and ZO-1 and occludin in the cortex, and ELISA was conducted to measure TNF-α, IL-6, IL-1β, and HMGB1 expression. The wet‒dry method was employed to measure the water content in the brain tissue. The permeability of the blood‒brain barrier (BBB) was assessed via Evans blue staining. Primary astrocytes were treated with S100A4 and/or DEX. The expression levels of GFAP, C3, GBP2, Serping 1, PTX3, S100A10, S100A4, and the NF-κB pathway were also determined.
Results
DEX improved early neurological deficits in SAH mice, mitigated the permeability of the BBB, and reduced the brain water content. DEX attenuated neuronal apoptosis and proinflammatory cytokine (TNF-α, IL-6, IL-1β and HMGB1) expression in the cortex. However, DEX-mediated protective effects were attenuated by ATI administration. Additionally, DEX attenuated GFAP, C3, Serping1, S100A4, and NF-κB pathway activation in the brain and in S100A4-treated primary astrocytes, whereas ATI reversed the effects of DEX.
Conclusion
DEX has neuroprotective and anti-inflammatory effects in SAH through the inhibition of S100A4-mediated astrocytic “A1” polarization via the activation of the α2A adrenoceptor.

1. Introduction
Subarachnoid hemorrhage (SAH), a severe disorder of the nervous system and the third most frequently occurring type of stroke, affects more than 8 million people globally. 1 It is caused primarily by head trauma or ruptured aneurysms and has a high mortality rate—approximately one in four patients die before hospital admission.2,3 Unlike other stroke types, SAH predominantly affects younger people, with significant disability rates imposing substantial financial burdens through rehabilitation costs. 4 Neurological deficits occurring within 72 hours of SAH are clinically termed early brain injury (EBI), which is correlated with poor prognosis. 5 This critical phase involves pathological changes such as blood‒brain barrier (BBB) leakage, cerebral edema, and neuronal damage. 6 While interventions such as oral nimodipine and surgical procedures, including hematoma evacuation or hemispherectomy, may reduce mortality, their clinical efficacy remains unclear. 7 These findings underscore the urgent need for innovative therapeutic strategies.
Astrocytes are the most abundant and complex glial cells in the central nervous system. During early-stage cerebral hemorrhage (typically within hours to days after bleeding), they play a “dual role”: while potentially exacerbating injury through multiple mechanisms, they may also initiate protective responses. 8 The BBB is a critical structure that maintains the stability of the brain microenvironment, with astrocyte foot processes tightly enveloping cerebral capillary endothelial cells, forming essential components of the BBB. 9 Growing evidence indicates that astrocytes play a pivotal role in SAH. Following SAH onset, astrocytes become activated and exhibit “A1” neurotoxicity, characterized by upregulated expression of biomarkers such as C3, Serping1, and GBP2, along with the release of proinflammatory factors (IL-1β, TNF-α, and IL-6) and chemokines (CXCL1 and CXCL2). These cellular activities recruit immune cells, including microglia and neutrophils, to injury sites, intensifying local inflammatory infiltration and mediating BBB damage.10,11 Astrocytes interact with other SAH components—including neurons, endothelial cells, pericytes, and microglia—through various mechanisms, potentially assisting in influencing early SAH-induced injury.3,12-14 Therefore, inhibiting “A1” astrocyte activation may represent a novel therapeutic strategy for SAH management.
Dexmedetomidine (DEX), a highly selective α2-adrenergic receptor (α2-AR) agonist, is clinically used for general anesthesia and in specific surgical populations. 15 Recent studies have demonstrated that DEX has protective effects against traumatic brain injury (TBI), SAH, and ischemia‒reperfusion (I/R)-induced brain damage, highlighting its potential as a neuroprotective agent. 16 The mechanism of DEX-mediated neuroprotective effects is multifaceted and is primarily achieved through the activation of central α2-ARs, particularly the α2A subtype. Upon binding to α2A-ARs, DEX exerts central sympathetic inhibition by reducing norepinephrine release, thereby mitigating catecholamine-induced neurotoxicity and oxidative stress. 17 Recent studies have indicated that DEX exerts neuroprotective effects by mediating astrocytic function. The activation of astrocytic α2A-AR reduces neuroinflammation and preserves BBB integrity.18-20 For example, in a mouse model of sepsis-associated encephalopathy, DEX activated hippocampal astrocytic α2A-ARs, thereby suppressing hippocampal astrocytic reactivity, restoring the expression of the glutamate transporter GLT-1 and GLAST, and reducing glutamate-induced excitotoxicity and synaptic damage. 19 Additionally, the neuroprotective mechanisms of DEX in SAH involve reducing neuroinflammatory responses and oxidative stress, protecting the BBB, maintaining hemostatic–anticoagulant balance, and preventing vasospasm. 21 For instance, in models of TBI and postoperative cognitive impairment, DEX downregulates the activation of nuclear factor κB (NF-κB) and the NLRP3 inflammasome, reducing the release of proinflammatory cytokines such as IL-1β and TNF-α and thereby mitigating inflammation-mediated secondary injury.22,23 In a rat model of SAH, DEX inhibited microglial pyroptosis via the PI3K/AKT/GSK3β pathway. It reduces the release of proinflammatory cytokines such as IL-1β and IL-18, thereby mitigating early brain injury following SAH. 24
Traditional anti-inflammatory strategies, including nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, have been shown to reduce vasospasm following SAH. 25 However, they demonstrate limited efficacy in reducing the risk of death and are associated with significant safety concerns, which restrict their clinical application.26,27 On the basis of the potential neuroprotective effects of DEX, particularly its central sympatholytic effects, astroglial regulation, and BBB protection via α2A adrenergic receptors, we hypothesized that DEX could regulate “A1” astrocyte polarization via α2A adrenergic receptors to improve early-stage brain injury associated with SAH. By administering DEX and the α2 adrenergic receptor antagonist atipamezole (ATI) to SAH model mice, we evaluated neurological deficits, neuronal damage, astrocyte polarization, and pathway alterations. This research aims to elucidate the neuroprotective mechanisms of DEX in SAH-mediated brain injury, providing theoretical insights for clinical drug development in SAH treatment.
2. Materials and Methods
2.1. Establishment of the SAH Model
Seventy adult male C57BL/6 mice (8 weeks old, 22–24 g) were purchased from Wenzhou Medical University Experimental Animal Center (Wenzhou, China) and maintained at a controlled temperature (25 ± 1°C) and humidity (65 ± 5%) with a 12-hour light/dark cycle and free access to water and food for at least one week. All experimental procedures and animal care protocols were approved by the Ethics Committee of Lishui Hospital of Wenzhou Medical University (Approval No. LL-22-5043) in compliance with the Chinese National Guidelines for the Care and Use of Laboratory Animals. Animal studies were conducted from March 2023 to June 2024. The mice were randomly divided into four groups: the sham group (n=10), the SAH + saline group (n=20), the SAH + DEX group (n=20), and the SAH + DEX + ATI group (n=20). An SAH mouse model was established via endovascular perforation techniques. 28 The mice were anesthetized with a 5% isoflurane/oxygen mixture and maintained under 2% isoflurane/oxygen anesthesia. The mice were placed in the supine position, and a midline cervical incision was made to dissect the common carotid artery, external carotid artery, and internal carotid artery. A 4-0 silk suture was inserted from the left external carotid artery into the internal carotid artery and advanced until resistance was encountered at the target vessel. The suture was promptly removed, the external carotid artery was ligated, and the wound was closed. The sham group (n=8) included control mice that underwent the same surgical steps but without vascular perforation. The severity of SAH in the brain was evaluated 24 hours after surgery, and mice with SAH scores ≤ 7 were excluded. 29
2.2. Drug Administration
Dexmedetomidine (DEX, #HY-12719, MCE, USA) and atipamezole (ATI) (#A9611; Sigma‒Aldrich, St. Louis, MO, USA) were dissolved in dimethyl sulfoxide (DMSO). Four groups of mice were established: sham, SAH+saline, SAH+DEX, and SAH+DEX+ATI. Thirty minutes before surgery, the sham and SAH groups received 0.2 ml of physiological saline via intraperitoneal injection. The same volume of 50 μg/kg DEX was administered intraperitoneally to SAH+DEX group mice.18,24 The SAH+DEX+ATI group received an equivalent volume of DEX (100 μg/kg) via intracerebroventricular injection. Following SAH modeling, ATI was immediately administered via intracerebroventricular injection to each mouse (25 nmol per mouse). 18 The mice were anesthetized with 2% isoflurane and positioned in a prone posture within a stereotactic head frame. The location of the injection site was defined relative to the bregma as follows: 1.00 mm in the mediolateral plane, −0.3 mm in the anteroposterior plane, and −2.5 mm in the dorsoventral plane. A 26-G Hamilton syringe needle was used to administer the injection over a 1-minute period. Following the injection, the needle was kept in position for 30 seconds prior to removal.
2.3. Short-Term Neurological Performance Evaluation
Neural function assessments were conducted 24 hours after mouse model establishment via an enhanced Garcia and beam balance test, with all evaluations performed by the same observer. The Garcia test was used to evaluate cage-based spontaneous activity, limb motor symmetry, forelimb extension, climbing ability, proprioception, and whisker touch response. Scores range from 3 to 18, where higher values indicate milder neurological impairment. 30
2.4. BBB Permeability Assay
To assess BBB permeability, the mice were intravenously injected with Evans blue solution (2%, 4 ml/kg) via the tail vein. After 1 h, the mice were anesthetized, and their left ventricles were perfused with 0.1 M PBS to remove the circulating dye. The brain samples were then extracted and weighed. Brain homogenates were prepared by centrifuging the supernatant with an equal volume of 50% trichloroacetic acid overnight, followed by centrifugation at 4°C for 15 minutes at 14,000 rpm. The absorbance of the supernatant was measured via a spectrophotometer at 620 nm.
2.5. Immunofluorescence Staining
Twenty-four hours after the mouse model was established, the mice were euthanized. The brain tissue was perfused with PBS solution and processed through fixation, dehydration, embedding, and cryopreservation to obtain 15-micron-thick sections. The sections were incubated overnight with primary antibodies at 4°C. The following rabbit anti-GFAP antibody (1:1000; product number ab68428; Abcam) was used. After three washes with PBS, the tissue samples were incubated with Alexa Fluor 488-conjugated goat anti-rabbit IgG (1:1000; product number ab150073; Abcam) for 2 hours, followed by 5 minutes of DAPI staining. The sections were observed under a fluorescence microscope, and images were captured.
2.6. HE Staining
The brains were dissected, fixed, and embedded in paraffin. After deparaffinization and rehydration, the slides were stained with a hematoxylin and eosin (HE) staining kit (C0105S; Beyotime, China) to evaluate brain tissue damage. Images were acquired under a microscope (Nikon, Tokyo, Japan).
2.7. Brain Water Content Assessment
The brain water content in intact brain tissue was measured using a wet-dry method. Immediately after extraction, the brains were weighed, and their wet weights were recorded. The tissues were subsequently dried at 110°C for a 48-hour period and reweighed to obtain their dry weight. The brain water content was calculated as (weightWET− weightDRY)/weightWET× 100%.
2.8. TUNEL Staining
Following the protocol of the TUNEL kit, paraffin sections of dewaxed and hydrated brain tissue were incubated in a 3% hydrogen peroxide solution at room temperature for 15 minutes. Then, 0.1% Triton X-100 citric acid buffer was added, followed by a 3-minute ice immersion reaction. Each sample received 50 μl of TUNEL reaction mixture and was incubated at 37°C in a humid chamber for 1 hour. Subsequently, 8 minutes of DAPI staining (100 μg/L) was performed for nuclear visualization. The number of apoptotic cells was analyzed via ImageJ software (version 2.9.0/1.53t) by a blinded observer.
2.9. Nissl Staining
The brain sections were dewaxed in xylene, rehydrated with alcohol, and stained for 5 minutes with Nissl staining solution (catalog #C0117; Beyotime, China). After staining, the sections were mounted and observed under a microscope for imaging. Compared with normal neurons, damaged neurons presented shrunken cell bodies and darker nuclear staining.
2.10. Primary Astrocyte Culture
Within 48 hours, newborn C57BL/6 mice were used for this experiment. The soft meninges were excised, and the cerebral cortex was extracted. After dissection, the brain tissue was placed in a solution containing 0.25% trypsin for digestion. The collected cultures were resuspended in DMEM/F12 medium supplemented with 20% fetal bovine serum (FBS; #SH30070; HyClone) and maintained at 37°C with 5% CO2. The cells were cultured under specific conditions until they reached 70% confluence. The cells were subsequently digested with trypsin and transferred to culture flasks or dishes for further cultivation. When the cell density exceeded 90%, the plates were placed in a 37°C constant temperature shaker for 12 hours at 200 r/min to purify the cells. Primary astrocytes were inoculated into 24-well or 6-well plates. Protein blot analysis was performed when the culture confluency reached 70%. To induce astrocyte activation, the cells were treated with exogenous mouse recombinant S100A4 protein (80 ng/ml) (#CSB-YP020632MO; CUSABIO, Wuhan, China) for 24 hours. 31 Six hours prior to S100A4 treatment, the cells were pretreated with DEX (1 μM) 32 or ATI (100 nM). 33 The “A1” activation status of primary astrocytes was detected via immunofluorescence with GFAP+C3 labeling.
2.11. Quantitative Real-Time PCR
Primer Sequences

2.12. ELISA
In accordance with the manufacturers’ instructions for ELISA kits containing mouse Il-1β (#CSB-E08054m), Il-6 (#CSB-E04639m), TNF-α (#CSB-E04741m), and Hmgb1 (#CSB-E08225m) (CUSABIO, Wuhan, China), the detection of brain tissue homogenates or cell supernatants was performed as follows: 1. A 96-well plate precoated with the corresponding antibodies was placed into the kit. The samples and standards were added and then incubated at 37°C in a light-shielding box for 90 minutes. 2. The plates were subsequently washed 5 times with buffer, after which the biotinylated antibody working solution was added. The mixture was incubated at 37°C under light protection for 60 minutes. 3. The plates were subsequently washed 5 times, after which the enzyme conjugate working solution was added. The mixture was incubated at 37°C under light protection for 30 minutes. 4. The plates were subsequently washed 5 more times, chromogenic substrate was added, and the samples were incubated at 37°C under light protection for 15 minutes. 5. The termination solution was added, the mixture was mixed thoroughly, and the OD values of each well were measured. Finally, the cytokine concentrations in the brain tissue homogenates were calculated via a standard curve.
2.13. Western Blot
Brain tissue proteins were extracted with RIPA lysis buffer, and the protein concentration was determined via the BCA method. Samples (20 µg) were subjected to SDS‒PAGE and transferred to PVDF membranes. The samples were incubated with 5% BSA at room temperature for 2 hours. The PVDF membrane was incubated overnight with rabbit antibodies against the following proteins: Bcl-2 (1:2000, ab182858, Abcam), Bax (1:10000, ab32503, Abcam), cleaved caspase-3 (1:4000, ab214430, Abcam), ZO-1 (1:4000, ab214430, Abcam), GFAP (1:10000, ab7260, Abcam), GBP2 (1:2000, PA5-87188, Thermo Fisher), PTX3 (1:1000, PA5-101097, Thermo Fisher), C3 (1:2000, ab200999, Abcam), Serping1 (1:2000, SAB5701229, Sigma‒Aldrich), S100A4 (1:1000, ab197896, Abcam), NF-κB (1:10000, ab32536, Abcam), p-NF-κB p65 (1:1000, ab76302, Abcam), and rabbit anti-β-actin (1:3000; ab8227; ABCAM primary antibody working solution. After three washes with TBST buffer, the membranes were incubated with HRP-labeled goat anti-rabbit IgG (1:3000; product code ab6721; Abcam) and secondary antibody working solution at room temperature for 2 hours. The PVDF membrane was then exposed to an enhanced chemiluminescence (ECL) chemiluminescent developer, developed on X-ray film, and analyzed for target protein expression. β-actin served as the internal reference, with protein levels from the sham or control groups normalized to calculate relative expression values.
2.14. Statistical Analysis
The severity of SAH bleeding and neurological function in the rats were assessed in a double-blind manner, with experimental data visualization and statistical analysis performed via GraphPad Prism 10 software. The data are presented as the means ± standard deviations (x±s). Multivariate analysis was conducted via one-way ANOVA, followed by Bonferroni post hoc analysis for multiple comparisons. Statistical significance was defined as a two-tailed P <0.05.
3. Results
3.1. DEX Improves Neurological Function and Attenuates Brain Edema
As shown in the figure, this study established an SAH mouse model. The mice were divided into four groups: the sham group, SAH+saline group, SAH+DEX group, and SAH+DEX+ATI group (Figure 1A). Garcia scores, beam balance tests, and brain water content measurements were conducted within 24 hours after SAH induction. Compared with the sham group, the SAH+saline group exhibited significant neurological deficits, with reduced scores in both the modified Garcia test and the beam balance test. Compared with saline administration, intraperitoneal DEX administration alleviated these neurological impairments. However, when the cells were treated with the α2 adrenergic receptor antagonist atipamezole (ATI), the neuroprotective effect of DEX diminished (Figure 1B and C). Brain edema measurements via the dry‒wet method and HE staining revealed increased brain water content in the saline group, indicating significant edema following SAH induction. Conversely, DEX reduced the brain water content in the DEX+SAH group, demonstrating that DEX improved cerebral edema. Notably, compared with the DEX+SAH group, ATI significantly enhanced edema in the brain (Figure 1D). HE staining revealed no significant inflammatory cell infiltration in the brain tissue of the sham group, with a clear and orderly arrangement of neuronal cells. In the SAH+saline group, the number of neuronal nuclei decreased, accompanied by a reduction in the cell count and disordered cellular arrangement. Compared with that in the SAH+saline group, the degree of neural cell damage to the brain tissue in the SAH+DEX group was significantly mitigated. Compared with the SAH+Saline group, the SAH+DEX group presented significant improvements in relatively intact cellular structures, whereas nuclear shrinkage, rupture, and dissolution were markedly reduced. After ATI administration, the neuroprotective effect of DEX on neuronal damage in mouse brain tissue was significantly weakened (Figure 1E). These results demonstrate that DEX effectively alleviated neurological deficits and cerebral edema in SAH mice, whereas ATI partially reversed the neuroprotective effects of DEX. Effects of DEX on neurological disorders in the SAH model
3.2. Effects of DEX on SAH-Mediated Neuronal Damage and BBB Permeability
Nissl and TUNEL staining revealed a significant increase in the number of apoptotic neurons in mouse brain tissue 24 hours after SAH compared with that in the sham group, with DEX treatment reducing the number of apoptotic neurons. Compared with the SAH+DEX group, the SAH+DEX+ATI group presented more apoptotic neurons (Figure 2A-D). Protein blot analysis of the apoptosis-related proteins Bcl-2, Bax, and Caspase-3 revealed significantly elevated Bax and Caspase3 levels in the SAH group, whereas the level of Bcl-2 decreased. DEX treatment significantly reversed these effects, whereas ATI treatment markedly counteracted the regulatory effect of DEX on apoptosis-related proteins (Figure 2E). Immunofluorescence revealed a lower intensity of positive staining for the tight junction protein ZO-1 in perivascular areas in the SAH group than in the sham group; DEX treatment increased ZO-1 positivity, whereas ATI treatment diminished the therapeutic effects of DEX (Figure 2F). BBB permeability assessment revealed reduced Evans blue extravasation in the SAH group following DEX treatment, with ATI alleviating this phenomenon (Figure 2G). Western blotting was used to detect the protein expression of ZO-1 and Occludin. The results revealed that the expression of ZO-1 and Occludin in SAH+saline mice was significantly lower than that in sham mice. After DEX treatment, the expression of ZO-1 and Occludin increased but was still lower than that in the Sham group. Compared with that in the SAH+DEX group, the expression of tight proteins in the SAH+DEX+ATI group was reduced (Figure 2H). Effects of DEX on neuronal damage and vascular permeability after SAH.
3.3. DEX Mitigated SAH-Mediated Inflammation in Vivo
ELISA and RT‒PCR analyses of the levels of inflammatory cytokines, including IL-1β, IL-6, TNF-α, and HMGB1, in brain tissue revealed significantly elevated levels of these inflammatory factors after SAH. Following DEX treatment, homogenates from SAH mouse brains presented reduced levels of Il-1β, IL-6, TNF-α, and HMGB1. However, ATI treatment largely offset the anti-inflammatory effects of DEX on proinflammatory cytokines (Figure 3A-H). DEX alleviates neuroinflammation in SAH by regulating inflammatory cytokines. Effects of DEX on SAH-mediated neuroinflammation
3.4. DEX Inhibited SAH-Induced “A1” Astrocytic Activation
Compared with the sham surgery group, the SAH group presented significantly greater numbers of GFAP-positive cells (Figure 4A and B) and elevated GFAP expression levels in brain injury foci (Figure 4C). Additionally, the expression of both A1 polarization markers (C3, GBP2, and Serping 1) and A2 polarization markers (PTX3 and S100A10) increased in the SAH group (Figure 4D). Following DEX treatment, the number of GFAP-positive cells and the expression level of a GFAP-positive marker decreased, whereas the expression of A1 polarization markers (C3, GBP2, and Serping 1) decreased, and the expression of A2 polarization markers (PTX3 and S100A10) increased (Figure 4A-D). However, when ATI was administered, astrocyte activation intensified, with significant upregulation of “A” phenotype markers and downregulation of “A2” phenotype markers (Figure 4A-D). WB analysis revealed that the S100A4 and NF-κB signaling pathways were activated after SAH, and these changes were reversed by DEX treatment (Figure 4E). Importantly, ATI administration significantly reversed the inhibitory effects of DEX on S100A4 and NF-κB pathway activation (Figure 4E). Effects of DEX on astrocytic activation in the SAH model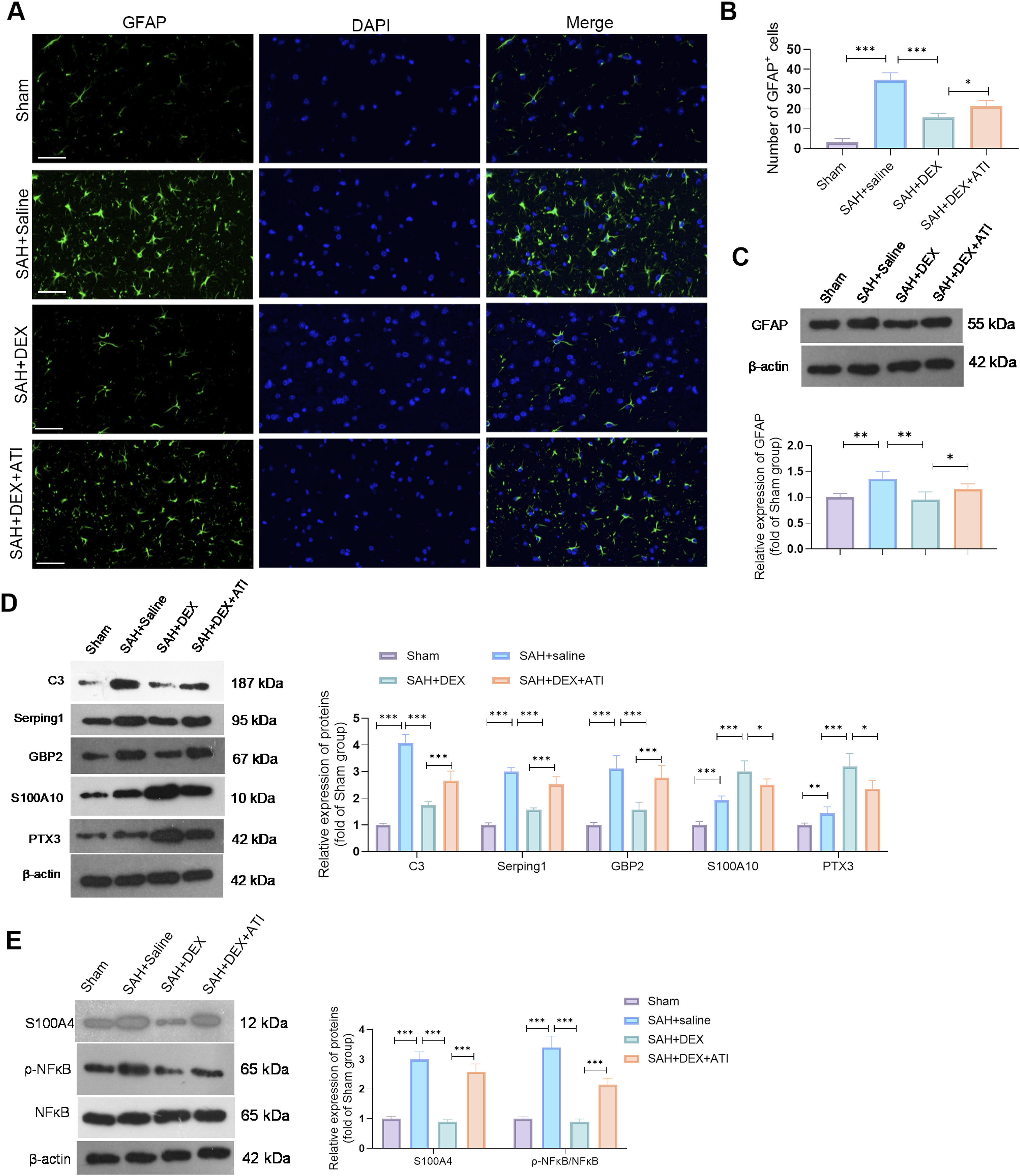
3.5. DEX Reversed S100A4-Mediated Astrocytic Reactivity in Vitro
To further validate the effects of DEX on astrocyte A1 polarization in vitro, we treated primary astrocytes with exogenous S100A4 for 24 hours to induce astrocyte activation. Immunofluorescence and Western blot analyses revealed that S100A4 treatment significantly increased the expression of GFAP and the “A1” markers C3, GBP2, and Serping 1 in astrocytes. After DEX treatment, the levels of the GFAP and “A1” markers decreased, whereas those of the “A2” markers (PTX3 and S100A10) significantly increased. The reversal of the inhibitory effects of DEX on the expression of GFAP and “A1” markers occurred after ATI supplementation (Figure 5A-C). Western blot analysis was used to detect S100A4 and NF-κB activation. Exposure to 80 ng/ml S100A4 significantly increased endogenous S100A4 and p-NF-κB protein levels in astrocytes, with DEX directly inhibiting these markers. Notably, ATI treatment increased S100A4 and p-NF-κB levels (compared with those in the S100A4+DEX group) (Figure 5D). These results confirm that S100A4 induces A1-type astrocyte activation. Additionally, DEX treatment reduced A1-type astrocyte activation, and ATI partially counteracted the effects of DEX. Effects of DEX on S100A4-mediated astrocyte activation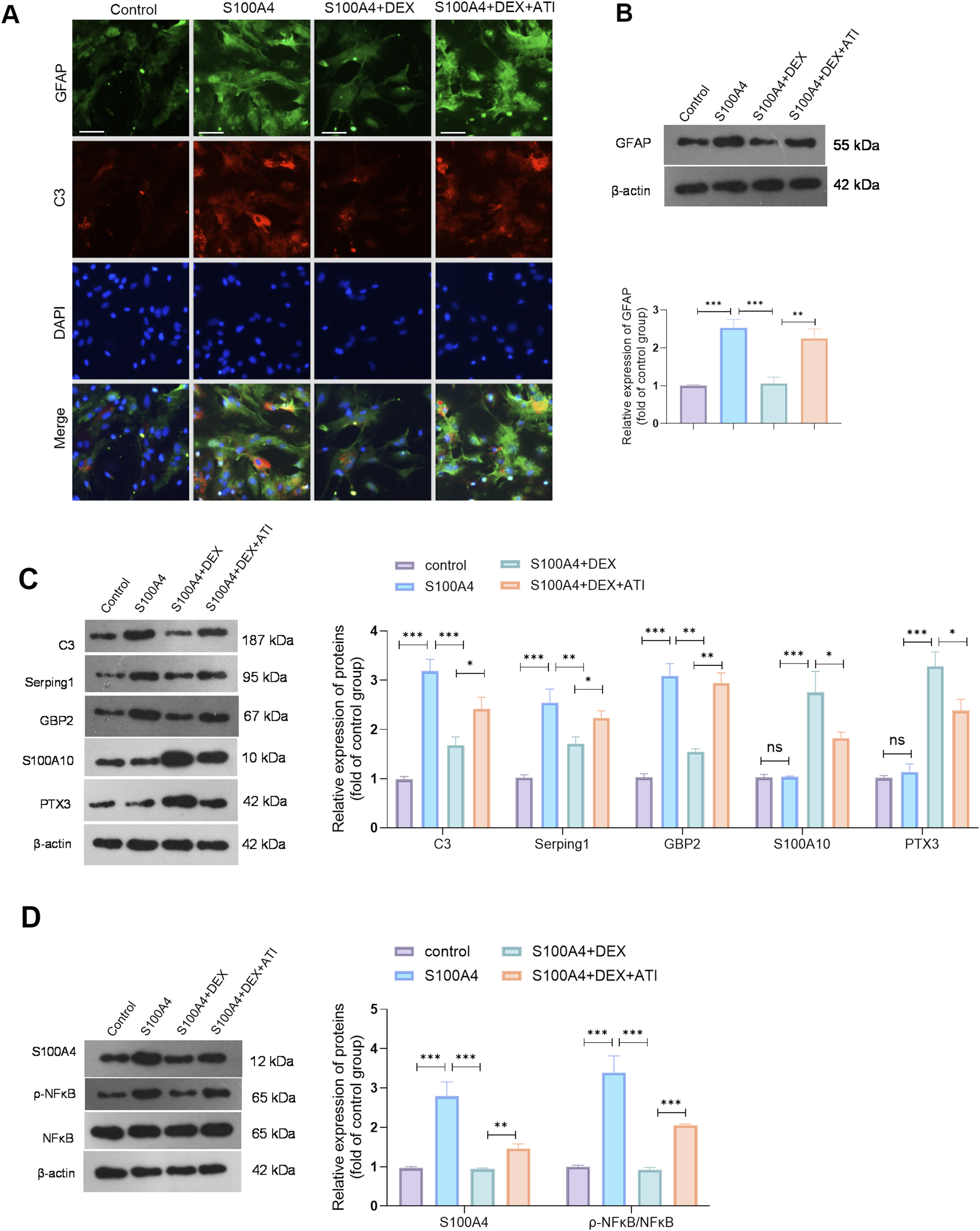
3.6. Effects of DEX on S100A4-Mediated Astrocytic Inflammation in Vitro
To investigate the effects of DEX on S100A4-mediated astrocyte inflammatory responses, ELISA and RT‒PCR were used to measure inflammatory factor release and mRNA levels, respectively. S100A4 treatment significantly increased the Il-1β, Il-6, TNF-α, and Hmgb1 levels in astrocyte supernatants, whereas DEX effectively suppressed these increases (Figure 6A-D). Notably, this inhibitory effect could be partially counteracted by ATI (Figure 6A-D). RT‒PCR further confirmed that S100A4 suppresses the mRNA levels of these inflammatory factors, whereas DEX counteracts this suppression. Addition of ATI increased the levels of proinflammatory factors (Figure 6E-H). These findings collectively indicate that DEX inhibits S100A4-mediated astrocyte inflammatory responses. Effects of DEX on S100A4-mediated inflammation in primary astrocytes.
Discussion
In this study, we demonstrated that DEX alleviates post-SAH encephalopathy by inhibiting the S100A4-mediated A1-type polarization of astrocytes. The results revealed that DEX improved neurological deficits, cerebral edema, neuronal survival, and BBB integrity in SAH mice. DEX also suppressed neuronal apoptosis and reduced neuroinflammation. Mechanistic studies further revealed that its anti-inflammatory effects likely occur through inhibition of the polarization of the “A1” phenotype in astrocytes (Figure 7). All these effects were partially reversed by the α2-adrenergic receptor antagonist ATI, confirming the involvement of α2-adrenergic receptors. Mechanistic diagram of DEX-mediated neuroprotective effects in SAH
Astrocytes are the most abundant cell type in the CNS and perform multiple critical functions in the healthy brain while also participating in the pathological processes of numerous CNS disorders. Studies have indicated that within 6 hours after SAH, astrocytes in the ventral cortex undergo activation and transition from a quiescent state to an activated state with increased cell volume.8,34 GFAP, a commonly used marker of astrocyte activation, reflects the degree of astrocytic reactivity. 35 The results of this study confirmed that the number of GFAP-positive cells significantly increased in the brains of SAH group mice, as indicated by elevated GFAP protein expression levels (Figure 4A-C), indicating robust astrocyte activation following SAH. DEX treatment significantly reduced GFAP expression, and this effect was partially reversed by the α2-AR antagonist ATI, indicating that DEX suppresses astrocytic reactivity by activating α2-ARs. Activated astrocytes contribute to BBB disruption through multiple mechanisms, forming a core pathway in EBI-induced brain edema following SAH. First, the level of matrix metalloproteinase-9 (MMP-9) significantly increases post-SAH and plays a key role in BBB destruction through degradation of the BBB matrix. 36 Research by Feng D et al. revealed that the sharp increase in MMP-9 expression following SAH primarily originates from reactive astrocytes, whose secretion is regulated by the NDRG2-PPM1A signaling axis in astrocytes. 8 Beyond MMP-9-mediated degradation, the structural integrity of the BBB itself relies on astrocytic foot processes. These processes tightly envelop cerebral capillaries and support barrier function through “cell-to-cell interaction” pathways. 37 In this study, DEX treatment improved BBB leakage (Figure 2G) and upregulated the expression of the tight junction proteins ZO-1 and Occludin (Figure 2H), suggesting that DEX may indirectly protect BBB structure and function by inhibiting astrocyte activation.
Persistently activated astrocytes differentiate into distinct phenotypes (A1 and A2). A1 astrocytes release neurotoxic factors that exacerbate neuronal injury, whereas A2 astrocytes secrete neurotrophic factors that promote tissue repair. 38 Following SAH, astrocytes are activated into the neurotoxic “A1” subtype under the stimulation of damage-associated molecular patterns (DAMPs), such as HMGB1, S100B, and hemoglobin. This activation is accompanied by the upregulation of the activity of the JNK/STAT1 pathway, 39 STAT3 signaling pathway,40,41 and NF-κB signaling network, 10 leading to the release of proinflammatory factors such as TNF-α and IL-1β. These cytokines promote neuroinflammation, compromise blood‒brain barrier integrity, and cause neurotoxic substances in the bloodstream to leak into brain tissue. 42 Conversely, “A2”-polarized astrocytes release neurotrophic factors such as TGF-β and IL-10 along with anti-inflammatory agents, demonstrating neuroprotective effects and ameliorating cerebral microvascular damage. 43 In this study, we also observed a marked increase in the number of astrocytes within the brain injury regions of SAH mice, accompanied by elevated levels of inflammatory markers (IL-6, IL-1β, and TNF-α) and HMGB1. Although the expression of A2 phenotype markers slightly increased, that of A1 phenotype markers significantly increased. These findings indicate that A1 astrocytes represent a more pronounced pathological feature in early-stage SAH. Modulating astrocyte polarization represents a potential therapeutic strategy for SAH. For instance, Zhang et al. reported that targeted silencing of BRD4 in astrocytes significantly reduced H4K8la lactylation and exacerbated A1 polarization, suggesting that BRD4 is a potential therapeutic target for SAH. 40 Our study revealed that DEX treatment significantly suppressed the expression of A1 markers (C3, Serping1, and GBP2) while promoting the expression of A2 markers (PTX3 and S100A10) (Figure 4D), indicating that DEX shifts astrocyte polarization from the neurotoxic A1 phenotype toward the protective A2 phenotype. Future studies could further explore its regulatory targets.
In addition to neuroinflammation, neuronal injury is also a core pathological mechanism of SAH. Studies indicate that neuronal apoptosis is a key driver of early EBI following SAH. Targeting key genes that regulate neuronal apoptosis can improve neurological damage both in vivo and in vitro after SAH. 44 For example, Zhang et al. reported that puerarin significantly alleviated neurological dysfunction and cerebral edema in SAH mice, as evidenced by an increased Bcl-2/Bax ratio and reduced cleaved caspase-3 expression, thereby decreasing neuronal apoptosis. 45 Our study similarly revealed that DEX significantly modulates key apoptotic pathways. The results demonstrated that DEX treatment upregulated the expression of the antiapoptotic protein Bcl-2 in the hippocampus of SAH mice while downregulating the expression of the proapoptotic proteins Bax and cleaved caspase-3 (Figure 2E). These findings suggest that DEX exerts neuroprotective effects partly by inhibiting mitochondrial-mediated apoptosis, which is consistent with the results of previous studies demonstrating the antiapoptotic properties of DEX in various brain injury models.32,46 In this study, DEX increased the expression of apoptosis-related markers while simultaneously suppressing S100A4 expression. These findings suggest that S100A4 may participate in the regulation of apoptotic pathways or that the antiapoptotic effects of DEX may share synergistic mechanisms with its anti-inflammatory effects. Future studies could further explore the direct molecular link between S100A4 and mitochondrial apoptotic pathways, as well as whether DEX exerts its antiapoptotic effects through both S100A4-dependent and S100A4-independent pathways.
As a highly selective agonist, DEX targets the α2-adrenergic receptor. In various acute brain injury models, DEX has been shown to have significant neuroprotective effects.47,48 In SAH, DEX has been demonstrated to reduce neuroinflammation and oxidative stress, inhibit neuronal apoptosis, protect BBB integrity, maintain hemostatic–anticoagulant balance, and prevent vasospasm. 47 These protective effects are increasingly attributed to direct modulation of astrocyte function. For example, in neonatal hypoxic-ischemic brain injury (HIBD), DEX treatment significantly reduced the proportion of neurotoxic A1 astrocytes while increasing the number of protective A2 astrocytes. 49 DEX inhibits astrocyte apoptosis in vitro by blocking the JAK2/STAT3 signaling pathway and downregulating JMJD3 expression. It also counteracts glucose deprivation-induced astrocyte injury through autophagy activation and TSC2/mTOR signaling pathway regulation. 50 This study, for the first time, demonstrated that DEX treatment significantly inhibited A1 astrocyte aggregation in SAH models, suppressed proinflammatory cytokine release from these cells, and promoted the transformation of “A2” astrocytes, thereby improving neuroinflammation and injury.
To further confirm the neuroprotective effects of DEX in SAH mice through α2-adrenergic receptors, we administered an antagonist of ATI, an α2 adrenergic receptor agonist, to both mice and primary cultured astrocytes in this study. ATI largely reversed the DEX-mediated improvements in neurological function, BBB integrity, and neuronal survival, as well as the inhibition of A1 astrocyte polarization and S100A4/NF-κB signaling (Figures 1-4). This reversal by ATI provides direct pharmacological evidence that DEX exerts its multifaceted neuroprotective effects through α2-AR activation. Previous research has confirmed that ATI can counteract the protective effects of DEX in various CNS disorders, including septic encephalopathy,18,51 pentylene tetrazole-induced seizures, 52 and hypoxia-induced neurological damage. 53 In an LPS-induced septic model, DEX treatment increased astrocyte pyroptosis to suppress the release of IL-1β and IL-18, thereby reducing neuronal injury. However, ATI significantly reversed the protective effects of DEX. 54 In summary, the results of the present study demonstrate for the first time that DEX attenuates SAH-induced EBI by activating α2-adrenergic receptors to regulate astrocyte polarization.
S100A4 (also known as fibroblast-specific protein 1 (Fsp1)) belongs to the S100 calcium-binding protein family. As Dampers (DAMPs), the extracellular form of S100A4 functions through various receptors, including Toll-like receptor 4 (TLR4) and RAGE, triggering signaling cascades involving downstream mediators. This promotes extracellular matrix deposition and myofibroblast generation while maintaining the sustained activation of myofibroblasts. 55 Notably, S100A4 is expressed in both the CNS and the peripheral nervous system (PNS) and is present in glial cells, astrocytes, and neurons. 56 The role of S100A4 in neuroinflammation is environmentally dependent. On the one hand, multiple studies have reported the protective effects of S100A4 under specific conditions. For example, serum and middle cerebral artery occlusion (MCAO) mouse brain tissue samples from ischemic stroke patients presented significantly elevated S100A4 expression. S100A4 knockout exacerbates neurological deficits, blood‒brain barrier disruption, and oxidative stress in MCAO mice. Conversely, the upregulation of S100A4 expression alleviates these pathological changes by activating the PI3K/Akt/Nrf2 signaling cascade. 57 Research by Pankratova et al. revealed that S100A4 can bind to ErbB receptors and promote neural survival in neurodegenerative disease models. 58 These findings suggest that under specific conditions, S100A4 may have neuroprotective effects. A growing body of evidence supports a predominantly proinflammatory role for S100A4 in CNS pathology. In an experimental autoimmune encephalomyelitis (EAE) mouse model, S100A4 knockout mice presented significantly reduced inflammatory cell infiltration and cytokine expression in their spinal cord and brain. Conversely, exogenous or endogenous supplementation with S100A4 can activate the TLR4/NF-κB signaling pathway to promote proinflammatory responses in microglia. 59 In rheumatoid arthritis mouse models, S100A4 decreases occludin expression levels, thereby enhancing the paracellular permeability of brain microvascular endothelial cells (MBECs). 60 In amyotrophic lateral sclerosis models, elevated S100A4 expression in spinal cord astrocytes and microglia was observed. Its transcriptional inhibitor clonidinium chloride suppresses the NADPH oxidase 2, mTOR, and NF-κB pathways, thereby improving cytoskeletal disorganization and phagocytic function. 61 These findings suggest that elevated S100A4 levels can exacerbate neurovascular damage and neuroinflammation. Recent studies have demonstrated that S100A4 activates the TLR4/NF-κB signaling pathway to promote neurotoxic astrocyte activation, whereas niclosamide significantly improves postoperative pathological pain, 31 indicating that S100A4 drives A1-type astrocyte polarization. Consistent with these proinflammatory findings, we observed significantly elevated S100A4 expression in the brain lesions of SAH model mice. In vitro experiments demonstrated that exogenous S100A4 induced marked A1-type astrocyte polarization, whereas DEX treatment significantly attenuated this effect and suppressed proinflammatory cytokine release. ATI reversed DEX-mediated modulation, suggesting that DEX acts through an α2-AR-dependent mechanism. These findings position S100A4 as a key driver of neurotoxic astrocytic polarization in SAH and identify DEX as a potential therapeutic target for this pathway.
Clinical studies have confirmed that cerebrospinal fluid GFAP levels are significantly elevated in patients with SAH and that early GFAP peaks are closely associated with poor patient outcomes. 62 Therefore, targeting astrocytic reactivity may hold translational potential for improving patient prognosis. From a translational medicine perspective, DEX is already widely used for sedation in intensive care units, providing a foundation for its potential application in SAH patients. Previous clinical studies have explored DEX in SAH patients, primarily focusing on its sedative and hemodynamic effects, and have suggested potential benefits in reducing vasospasm and delirium (NCT01664520, NCT06352593). However, the timing of administration is a critical consideration. In our experimental model, DEX was administered 30 minutes prior to SAH induction, representing a prophylactic strategy. In clinical practice, SAH is unpredictable, and treatment typically begins after hemorrhage. Whether DEX administered after SAH can achieve similar neuroprotective effects remains unclear. In the Egyptian trial (NCT06352593), DEX was administered intraoperatively during endovascular intervention (0.5 μg/kg for 10 min followed by 0.2–0.6 μg/kg/h), representing a clinically feasible time window shortly after hemorrhage. Whether DEX administered at this or later time points achieves neuroprotective effects similar to those observed in our prophylactic model remains unclear. The optimal therapeutic window for DEX to exert its glial-modulating and anti-inflammatory effects in SAH requires further investigation in future clinical trials, taking into account its pharmacokinetic profile and the dynamic nature of early brain injury. Another important consideration for clinical translation is the dose dependency of the effects of DEX. Studies have shown that 30–50 μg DEX as an adjuvant to local anesthetics is the optimal perineural dose for prolonging analgesia without increasing adverse events. 63 A recent randomized controlled trial in elderly surgical patients demonstrated that the neuroprotective effects of DEX are dose dependent, with 0.8 μg/kg more effectively reducing postoperative delirium and inflammatory cytokine levels than lower doses (0.2 and 0.5 μg/kg). 64 These studies underscore the importance of dose optimization for the therapeutic effects of DEX. In the present study, we used a single dose of DEX (50 μg/kg in mice or 1 μM in cells), which was selected on the basis of previous SAH studies.19,25 However, the optimal dose for achieving maximal glial modulation while minimizing sedative side effects in SAH patients remains to be determined. Future clinical trials should explore dose-ranging strategies to identify the most effective and safe dose for this patient population (Future Perspectives).
This study has several limitations. First, although we demonstrated that S100A4 is involved in astrocyte polarization and that DEX can inhibit its expression, the direct molecular link between α2A-AR activation and S100A4 downregulation remains to be elucidated. Second, this study focused on early-stage brain injury (24 hours after SAH). The long-term effects of DEX on neurological recovery and whether the shift in polarization persists require further investigation. Third, the experimental subjects in this study were limited to 8-week-old male C57BL/6 mice. The well-established sex dimorphism in inflammatory responses following stroke and SAH leads to differences in infarct size and recovery. 65 In particular, estrogen has been shown to exert a protective effect in animal models by influencing astrocyte reactivity and neuroinflammatory pathways 66 ; however, whether the S100A4-mediated mechanism identified in this study applies equally to females remains unclear. Future studies should include both sexes to determine whether the neuroprotective effects of DEX and the underlying S100A4-mediated mechanism exhibit sex-dependent characteristics. Fourth, this study did not conduct a power analysis for sample size calculation. Although the sample size used was sufficient to detect statistically significant differences, the lack of a power analysis may increase the risk of error.
Future Perspectives
In addition to the neuroprotective effects of DEX on S100A4, the neuroprotective effects of DEX may depend on other signaling pathways in glial cells. There is well-established crosstalk between astrocytes and microglia in the pathophysiology of SAH. 42 Previous studies have demonstrated that DEX promotes microglial M2 polarization and alleviates brain injury after cerebral ischemia‒reperfusion by inhibiting the activation of the JNK and p38 MAPK pathways. 67 Therefore, the potential effects of DEX on microglial polarization in SAH, as well as the interaction between these two glial cell types, represent important avenues for future investigations. Furthermore, recent studies have highlighted the role of other S100 family members, such as S100B and S100A8/A9, in neuroinflammation in the central nervous system. 68 Notably, S100B is highly expressed in astrocytes and has been identified as a potential biomarker for SAH. 69 Whether DEX regulates the expression or release of S100B warrants investigation. The S100A8/A9 heterodimer, a potent activator of TLR4, may represent another potential target of the anti-inflammatory effects of DEX. 70 Future research should focus on whether DEX differentially affects distinct astrocyte subpopulations and how it modulates complex intercellular communication networks within the neurovascular unit. At the molecular level, further research is needed to elucidate the precise mechanism by which DEX modulates S100A4 signaling. Specifically, whether DEX directly inhibits S100A4 expression, interferes with its binding to receptors such as TLR4 or RAGE, or acts through a combination of both mechanisms remains to be determined. Experimental approaches such as surface plasmon resonance, competitive binding assays, and transcriptional reporter systems are essential for elucidating these possibilities. Determining whether DEX acts at the transcriptional level or posttranslational level or by directly interfering with S100A4-receptor binding is essential for understanding its full therapeutic potential in SAH. Studies utilizing conditional knockout models (e.g., astrocyte-specific S100A4 knockout mice), longer observation periods, and region-specific interventions will be essential to fully elucidate the spectrum of the effects of DEX on glial modulation and its therapeutic potential in SAH, including whether the inhibition of A1 polarization is transient or sustained. Ultimately, well-designed clinical trials incorporating dose-ranging strategies and sex-specific analyses will be necessary to translate these preclinical findings into clinical practice.
Conclusion
In conclusion, this study revealed that DEX has a significant neuroprotective effect in the early stage of SAH, including reducing neurological deficits, reducing neuronal damage, improving BBB leakage, and inhibiting SAH-mediated neuroinflammation and “A1” astrocyte polarization. Mechanistically, DEX inhibited the expression of S100A4 and suppressed S100A4-mediated ‘A1’ astrocyte polarization and NF-κB signaling pathway activation in an α2-AR-dependent manner. DEX is a potential strategy for improving early brain injury after SAH.
Supplemental Material
Supplemental Material - Dexmedetomidine Improves BBB and Neuronal Damage in Subarachnoid Hemorrhage by Repressing S100A4-Mediated Astrocytic Reactivity
Supplemental Material for Dexmedetomidine Improves BBB and Neuronal Damage in Subarachnoid Hemorrhage by Repressing S100A4-Mediated Astrocytic Reactivity by Jiayang Yin, Qiaomin Xu, Kuan Lu, Jinchao Wu, Weiwei Cai in Dose-Response
Footnotes
Ethical Considerations
All experimental procedures and animal care were approved by the Ethics Committee of Wenzhou Medical University Lishui Hospital (Approval number: LL-22-5043). They were based upon the Chinese National Institute of Guiding Principles for the Care and Use of Laboratory Animals.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
