Abstract
Background:
Exposure to high arsenic concentrations in drinking water has been associated with skin lesions. Our goal was to conduct a systematic review of studies on skin lesions and arsenic exposure, with emphasis on results at low level of exposure.
Methods:
We conducted a systematic review of studies reporting estimates of either prevalence or risk of skin lesions associated with exposure to more than 2 levels of arsenic in drinking water. We reviewed and abstracted the relevant results, with the aim of conducting a dose-response meta-analysis.
Results:
Nine studies of skin lesions were reviewed. Strong heterogeneity in the results did not meet the criteria for performing a meta-analysis. The relative risks for an increase of 10 μg/L arsenic in drinking water ranged from 1.002 to 1.140 (p-value of heterogeneity < 0.0001). Protection from bias and confounding was inadequate in most studies.
Conclusion:
Current studies are inadequate to conduct meta-analysis on dose-response relationship between exposure to arsenic in drinking water and skin lesions. Studies with complete exposure histories indicate skin lesions are associated with arsenic exposure in excess of 50 µg/L or higher.
Introduction
High-level exposure to arsenic in drinking water occurs in various regions of the world, including China, Mongolia, Taiwan, India (West Bengal), Bangladesh, Argentina, and Chile (Cohen et al., 2013). 1,2 Exposure is mainly from natural sources, although in some areas of Japan, Mexico, Thailand and other countries, industrial activities have resulted in elevated water levels. 3
Exposure to high levels of arsenic in drinking water has been associated with increased occurrence of skin lesions, including alteration of pigmentation and keratosis (Chen et al., 1997). 4 -6 Hyperpigmentation (melanosis) consists of diffuse dark-brown or blackish areas on the skin of the neck, trunk, or extremities, as well as oral mucosa, and diffuse or spotted pigmentation on the trunk and other parts of the skin. Hypopigmentation (leukomelanosis) is characterized by whitish patches, also referred to as raindrop pigmentation. Keratosis is characterized by bilateral thickening of the palms and soles, small protrusions on palms and soles, more rarely on the dorsum of the hands and feet.
It is not clear, however, whether exposure to low concentrations of arsenic in drinking water (i.e., levels below 100 or 150 µg/L) is also associated with skin lesions. The objective of the present study was to conduct a systematic review of epidemiology studies that assessed the dose-response relationship between exposure to arsenic in drinking water and skin lesions, and find whether there is an increased prevalence of skin lesions among subjects’ exposure to low concentrations of arsenic in drinking water. We therefore did not try to identify the model that best fits the results of each study, rather to consider whether a meta-analysis of linear, non-threshold regressions, similar to what is done by regulatory authorities, is applicable to the available studies.
Methods
A systematic review and meta-analysis of epidemiology studies (cohort, case-control, and cross sectional,) reporting association of exposure to arsenic with skin lesions, and which included multiple categories of exposure to arsenic in drinking water, were conducted according to the PRISMA and MOOSE guidelines. 7,8 A PRISMA checklist is included as Appendix 1. PubMed and Scopus databases were searched in January 2020 for studies providing results on incidence or prevalence of skin lesions among individuals exposed to arsenic in drinking water. In order to increase the sensitivity of the search we used broad strings that also included skin cancer, reported in Appendix 2. The study protocol is available from the authors. Each step of the systematic review was performed independently by 2 of the authors (PB, CB). Results of each step were compared between them, and disagreements were resolved by consensus. The lists of studies that were examined at each step of the selection process, are available from the authors.
Selection of Studies
A total of 3074 potentially relevant articles were identified. The titles and abstracts of the articles were reviewed by the 2 authors, and a shortlist of 30 articles was selected for full-text review based on the study selection criteria (Appendix 3). The lists of references of the articles selected for text review and one recent review 4 were also searched for additional studies.
Inclusion criteria of studies for the meta-analysis were (i) Design: cohort, case-control or cross-sectional design; (ii) Arsenic exposure: at least 2 exposure categories (reference category and one “exposed” category) of level of arsenic in drinking water; (iii) Outcome: either incidence or prevalence of skin lesions; (iv) Measures of occurrence: prevalence or cumulative incidence of skin lesions, either reported in the article, or sufficient data to calculate them; (v) Language: English, French, German, Spanish, Italian; (vi) Measures of association: either measures of association with the outcome of interest (prevalence odds ratio [POR], odds ratio [OR], or hazard ratio [HR], collectively indicated as relative risk [RR]), including 95% confidence intervals (CI), reported in the article, or sufficient data to calculate them.
Exclusion criteria included (i) Design: ecologic on non-comparative design; (ii) Exposure categories: less than 2 low levels of exposure; (iii) Measures of arsenic exposure: other than level of arsenic in drinking water (e.g., urinary level of arsenic metabolites); (iv) No measures of association and CIs, or sufficient data to calculate them; (v) Multiple reports: partial or total overlap with another report of the same study, with less extensive data (e.g., early follow-up of a cohort for which a subsequent report with longer follow-up was available).
The flowchart for the selection of the studies included in the meta-analysis is shown in Figure 1.

Flow chart for the identification of articles included in the review.
Extraction of Data
The following information was extracted from each study: (i) study design; (ii) study settings (country, geographic area, period of enrollment and follow-up for cohorts; period of ascertainment of cases for cross-sectional and case-control studies); (iii) study population (number of cohort or survey members and of cases and non-cases; demographic characteristics); (iv) definition of skin lesions; (v) exposure variables and categories with mean or midpoint; (vi) number of cases and person-years (cohort studies), number cases and total number of subjects (cross-sectional studies), number of cases and controls (case-control studies) in each exposure category; (vii) prevalence, cumulative incidence, and RR with 95% CI, or sufficient data to calculate them; (viii) potential confounders included in the analysis; (ix) potential sources of bias.
The preferred exposure variable was average level of arsenic in drinking water (µg/L) over the whole lifetime. However, in several studies, information was available for arsenic level at the current residence only
We did not attempt to harmonize the definition of skin lesions; for studies reporting several types of lesions separately (e.g., hyperkeratosis and hyperpigmentation), we used the sum number of the total lesions, or the type with the highest prevalence, when the number of total lesions were not available.
We derived from previously developed scales 9,10 a quality score taking into account opportunity for bias from study design, assessment of arsenic exposure, incidence/prevalence of skin lesions, and confounding. Details on the score are reported in Table 1.
Characteristics of Studies of Included in the Review.

* Period of enrolment and follow-up (FU) for cohorts; period of ascertainment of cases for cross-sectional and case-control studies; NA, not available.
** I, individual-level; V, village-level; SD, subdistrict-level.
‡A, age; S, sex; Sm, tobacco smoking; D, type of dwelling; E, education; O, occupation; BMI, body mass index; W, wealth.
‡‡Components of score.
: Design: case-control or cohort +1; cross sectional 0.
Exposure assessment: current and past +1; current only 0; individual 0; less than individual -1.
Skin lesion assessment: validation (in part or full) +1; no validation 0; examination by physicians 0; examination by non-physicians -1.
Confounders: At least sex, age and tobacco smoking +1; less than that 0.
† Same population as Guha Mazumder et al. 11
Statistical Analysis
We considered both the measure of occurrence of skin lesions (incidence or prevalence) and the measure of association with arsenic exposure (RR and 95% CI). For several cross sectional studies, RR were reported based on nested case-control analyses, comprising all cases of skin lesions and a sample of non-cases.
Adjusted measures were used, if available in the original publications. If measures of occurrence or association were not reported in the publications, we calculated them from the available data, without adjusting for potential confounders. If results were reported only in strata (e.g., by sex), we combined them using a fixed effect meta-analysis. We fitted linear regression models to derive study-specific estimates of change in RR for an increase of 10 µg/L arsenic exposure. We used the STATA command glst. 21
We assessed inter-study heterogeneity of results using the Q and the I2 statistics, 22,23 to consider whether a meta-analysis was justified. Although there are no established guidelines on the amount of inter-study heterogeneity that can be accepted to perform a meta-analysis, many authors agree that a p-value of the Q test < 0.01 or a I2 > 70% are indicators of strong heterogeneity, preventing a meta-analysis. 24 Therefore, we decided to meta-analyze the results of the studies included in the review if at least one of these 2 criteria was satisfied (i.e., p > 0.01 or I2 < 70%).
We used the same approach to conduct a secondary analyses of results in categories with mid-level exposure up to 50, 100 and 125 µg/L arsenic in drinking water.
In addition, we assessed publication bias through visual inspection of the funnel plot, 25 and applying the test proposed by Egger and colleagues, 26 for which the STATA programs metafunnel and metabias were used. 27
Original data are available from the authors upon request.
Results
Ten articles, referring to 9 independent studies, were identified in the systematic review, which addressed the study selection criteria. Key characteristics of these studies, are provided in Table 1, together with quality scores. Eight of the 9 studies with a cross-sectional design, measured prevalence of skin lesions. Three of the 8 cross-sectional studies 12,14,16 included a nested case-control analysis. One additional study, 20 with a cohort design, measured prospectively lesion incidence. The results that were used to derive estimates of changes in RR of skin lesions are reported in Table 2. The studies are described below.
Results Included in the Dose-Response Analysis.

Ref., reference category; RR, relative risk; CI, confidence interval.
* Exposure level (µg/L arsenic in drinking water) corresponds to the mean, median, or midpoint of the exposure category and is the value used in the dose-response analysis.
‡Results included in the analysis restricted to exposure up to 125 µg/L arsenic.
† Results for keratosis.
††RR results for keratosis.
** Argos et al. 20 measured cumulative incidence instead of prevalence.
Numbers in italics were derived from the raw data reported in the publication.
Prevalence Studies
Guha Mazumder et al. 11 conducted a cross-sectional study in 1995-1996 in 2 areas within a district in West Bengal, India. One area included 25 villages with high level of arsenic and the second—32 villages with low levels of arsenic in drinking water. 7,818 individuals were included in the study out of a total population of 150,457, and water arsenic levels were obtained for 7,683 of them. In the high exposure area, households were randomly selected with the goal of recruiting 50 to 150 participants in each village. In the low exposure area, sampling was restricted to villages with more than 100 houses and residents of every fourth house were invited to participate. Each participant was questioned by field workers about their sources of drinking water, current diet and water intake, medical symptoms, height and weight and other variables. A general medical examination was administered by a physician, including an inspection for arsenic-related skin lesions. Keratosis was defined as diffuse bilateral thickening of palms or soles with or without nodules; diagnosis of hyperpigmentation comprised areas of mottled dark brown pigmentation bilaterally distributed on the trunk. Field workers were not blind to the exposure status of the villages. Water samples were collected from private and public tube-wells used for drinking and cooking purposes by each recruited household. Arsenic levels from 644 tube-wells were analyzed by flow-injection hydride generation atomic absorption spectrophotometry. Arsenic concentrations ranged from below detection to 3400 µg/L. A total of 4,093 women and 3,590 men were included in the study. The prevalence of keratosis in women was 1.2%, that of hyperpigmentation was 3.1%; corresponding values in men were 3.0% and 6.5%. The prevalence of both types of lesion was low in subjects younger than 20, but there was no trend in the prevalence above that age. The results on keratosis according to arsenic concentration are reported in Table 2, the association between hyperpigmentation and arsenic exposure was weaker than that of keratosis. For both types of lesions, the prevalence of lesions given the same exposure was higher in men than in women, and it was higher in women with poor nutritional status compared to other women, while this difference was not observed in men.
Limitations of this study include lack of blindness to exposure assessment, lack of historical exposure, and lack of information about potential confounders (e.g., tobacco smoking, occupation UV light exposure), that may explain the higher prevalence of lesions in men compared to women.
Haque et al. 12 conducted a case-control analysis of 4,185 subjects out of the 7,683 participants in the study by Guha Mazumder et al. 11 For this study, they selected 265 individuals with lesions, whose drinking water level of arsenic was below 500 µg/L. Of the 265 individuals with skin lesions, 174 had hyperpigmentation, 15—keratosis and 76 both types of lesions. Controls were individuals who matched the cases, selected from the same population whose drinking water level of arsenic was below 500 µg/L, and the only difference from the cases was that they were without skin lesions. Of the 530 selected cases and controls, 192 cases and 213 controls were included in the study.
Cases and controls were visited in their homes in 1998 and later in 2000. Study subjects were interviewed using a questionnaire which included information on residential history, past water sources at home and work, current and past (5 years) fluid consumption, smoking and socioeconomic status. They also underwent a full medical examination, with full attention to skin lesions. Sometime in the middle of the project, photographs of the suspected lesions were taken for the most affected skin areas. Pictures were subsequently reviewed by experts and classified as definitely, probably, possibly or not related to arsenic. Lesions of participants without pictures were classified as having a skin lesion if the interviewer recorded on the questionnaire that the dermal changes were of a type related to arsenic. Water samples were taken from sources used by cases and controls at least 6 months in the previous 20 years, if still existed. However, many wells were closed for several reasons, including arsenic contamination. Of the 192 interviewed cases, 72 no longer had skin lesions. Between the 1995-1996 cross sectional study and this study, most of subjects consumed water with arsenic concentrations below 50 µg/L. Of the 213 controls, 25 were found to have lesions during the interview. Their water consumption history revealed past exposure to high arsenic levels (up to 253 µg/L).
In this study, water arsenic measurements were available for 94 (49%) cases and 102 (48%) controls. All cases with confirmed skin lesions and complete water arsenic information had a peak arsenic level higher than 100 µg/L. This represents one of the most informative results in this set of studies since it is based on a subset of subjects with good information on both exposure and outcome, and the authors concluded that arsenic concentrations above 100 µg/L are necessary to cause sufficient toxic skin effects. The results of the analysis RR for increasing categories of average arsenic exposure, based on the original classification of cases and controls irrespective of validation, are reported in Table 2. This is the only study that included a validation of the data on prevalence of skin lesions; the study shows no evidence of an increase in risk below 100 µg/L peak arsenic level in drinking water, in the most informative group (confirmed cases with full data on drinking water arsenic data): cases/controls were 0/22 in the category below 50 µg/L, and 0/12 in the category 50-99 µg/L. This study addressed some of the limitations of the original study by Guha Mazumder et al. 11 since it included a re-assessment of skin lesions and an assessment of arsenic concentration from all sources used in the previous 20 years. Limitations include the facts that complete data were available only for a subset of cases and controls. Photographs of lesions were taken only for part of the study subjects, participants without pictures were classified as having a skin lesion if the interviewer recorded on the questionnaire that the dermal changes were of a type related to arsenic, and several wells used in the past by cases and controls were closed for several reasons, including due to arsenic contamination. Furthermore, 72 cases with skin lesions in 1995-1996 no longer had lesions at later visits: it is not clear whether this indicates that arsenic-associated skin lesions are reversible and disappear when exposure is removed. Despite these potential limitations, this study was assigned the highest quality score (Table 1).
Tondel et al. 13 studied the population of 4 villages in Bangladesh for which there were existing measurement reports for arsenic concentrations in drinking water. The concentrations ranged from non-detected to 2040 µg/L. The study was restricted to subjects who lived in the same village and used the same well throughout their lifetime. A total of 1,481 individuals older than 30 years was included in the study, of whom 430 (29%) had skin lesions, including pigmentation changes of unexposed body surfaces and keratosis, especially on the palms of the hands and the soles of the feet. The prevalence of skin lesions was higher in men than in women for comparable levels of arsenic exposure, which can be explained by factors such as occupational exposure to UV and higher consumption of drinking water. The POR for an increase of 1000 µg/L was found to be 1.55 (95% CI 1.23 -1.91) in men and 1.42 (95% CI 1.04 -1.90) in women. The authors state that there were too few subjects in lower level categories to calculate the lowest arsenic concentration leading to skin lesions. indicating a threshold. The results of this study are summarized in Table 2 including prevalence of skin lesions for different levels of arsenic. The main limitations of this study are limited exposure assessment and lack of adjustment for potential confounders, including tobacco smoking and UV light exposure, which might explain the apparent higher prevalence of skin lesions in men compared to women.
Rahman et al. 14 screened 166,934 residents in Matlab, Bangladesh, during 2002-2003, and experts confirmed 504 cases with arsenic-associated skin lesions. These subjects were included in a case-control analysis in which 1836 controls were randomly selected from the same area. Skin lesions were identified in 6 of the controls. These subjects were included in the group of potential cases, leaving 1,830 controls. Water sources used by cases and controls since 1970 were recorded at the interview and checked against results of household economic surveys conducted in 1974, 1982 and 1996 in the study area, which contained information on sources of drinking water. Arsenic levels were measured in all wells of the study area at the time of case ascertainment and these results were used to derive an individual history of arsenic exposure, as well as average and cumulative exposure indices. Surface water sources were assigned a concentration of 0 μg/L. For non-functioning wells (32% of the total), the average concentrations of functioning wells in the same village were used, despite the fact that they could have been closed because of high arsenic contamination. Cases were statistically significantly of an older age, higher education and higher household asset score than controls; age and education were controlled for in the analysis, but details on the adjustment were not provided. Males were found to have higher risk of developing skin lesions than females, despite the fact that their estimated arsenic exposure was not higher than that of women: among men, the average arsenic concentration in drinking water was 200 μg/L in cases and 143 μg/L in controls; among women, these values were 211 μg/L and 155 μg/L. Corresponding values for cumulative arsenic exposure in men were 6,059 μg/L-years in cases and 3,067 μg/L-years in controls. Respectively, the exposure in women were 6,323 μg/L-years in cases and 3,464 μg/L-years in controls. The RR for average and cumulative exposure were reported for men and women separately and we combined them using a fixed-effect meta-analysis (Table 2). The study has a number of limitations: It is unclear whether the controls were selected during the same time-period as the cases, since cases were apparently selected from a survey covering the whole study area, and controls were examined for skin lesions in a different setting.Potential confounders, including tobacco smoking and UV light exposure, were not accounted for, and might explain the apparent higher prevalence of skin lesions in men compared to women, for comparable arsenic exposure levels. The use of arsenic levels at baseline to reconstruct past exposure is a further limitation of this study.
Guo et al. 15 conducted a cross-sectional study in 1 village in Wuyuan county of Inner Mongolia, China. A total of 589 inhabitants participated in the study (89% of all residents). Study subjects were interviewed on their residential history, sociodemographic conditions, sources of drinking water at each residence, duration of well use, amount of water consumed daily, occupation, tobacco smoking, and alcohol drinking. They were also examined by a dermatologist in order to provide a diagnosis of skin lesions (keratosis and keratosis with pigment disorder). The analysis was restricted to 448 subjects whose age was 18 years or older. Water samples were collected from 106 household wells used in the village and analyzed for arsenic concentration (it is not specified how many study subjects used these wells). Of the 448 study participants, 227 were diagnosed with skin lesions and 221 did not have effects. The cases were older and more frequently males (suggesting a possible effect of sun exposure from employment or higher consumption of drinking water) and smokers compared to subjects without skin lesions. The average arsenic level in drinking water was 277 (SD 239) μg/L in subject with skin lesions and 207 (SD 195) μg/L in subjects without lesions. There were 35 subjects with keratosis and 5 subjects with pigment disorders who were exposed to less than 50 μg/L arsenic. POR, adjusted for age, sex and smoking, were reported separately for keratosis and pigment disorders, results for keratosis are summarized in Table 2. No dose-response relationship was observed in this study. Limitations of this study include the very high prevalence of skin lesions (51% of study subjects), the small number of wells with arsenic data, the lack of consideration of the use of water sources other than the wells included in the analysis, and the potential for residual confounding.
McDonald et al. 16 reported results on prevalence of skin lesions, as a function of measured arsenic concentrations in wells used by subjects, as reported by the Bangladesh National Hydrochemical Survey, in a case-control study that was conducted by a nongovernmental organization. The case-control study was initially based on 176 pairs, but finally reduced to 155 pairs of women living in 53 villages from 12 sub-districts in 3 areas of Bangladesh in which 113 wells were tested for arsenic concentration. Results were reported according to average arsenic level by sub-district.
A total of 0.4% of women from sub-districts with an average arsenic concentration below 1 μg/L were diagnosed with skin lesions. In comparison, 0.34% in women from sub-districts with an exposure up to 10 μg/L were reported with skin lesions. Results are reported in Table 2. Limitations of this study include the lack of skin lesions validation, lack of individual- or village-level exposure information and of information on historical exposure, the fact that field workers were not blind of village exposure status, the unknown proportion of sources of drinking water used by study subjects that was covered by the wells included in the analysis, and the lack of consideration of potential confounders. This study was assigned the lowest quality score (Table 1).
Lamm et al. 17 conducted a cross-sectional study in 3 villages in the Huhhot region of Inner Mongolia (China). A total of 3179 subjects (51% men) participated in the study (98.5% of those eligible).The authors obtained well-use histories of participants, measured arsenic concentrations of 184 out of the 187 wells used by the study subject, and included a large number of participants. They estimated both maximum arsenic concentration exposure experienced as well as cumulative arsenic exposure Well with concentration below the detection limit of 10 μg/L (24% of the total) were set at 5 μg/L. Hyperkeratoses, and dyspigmentation (hyper- or hypo-pigmentation of the trunk), as well as skin cancers, were diagnosed by local physicians as related to arsenic exposure according to pre-defined criteria. Diagnoses were independently reviewed clinically and histologically by an expert from the Department of Dermatology of the University of Texas. The average arsenic concentration was 97 μg/L (sd 230 μg/L), the maximum level was 2000 μg/L; subjects were classified according to their highest arsenic exposure. Non-malignant lesions were diagnosed in 199 subjects (6.3%), of whom 121 had keratosis and 174 had dyspigmentation (94 subjects had both types of lesions). Eight subjects were diagnosed with skin cancer. The crude prevalence and RR of skin lesions for different categories of arsenic concentration are shown in Table 2.
Limitations of this study include the lack of information about occupational and other additional sources of arsenic exposures, the lack of validation of diagnosis of skin lesions, and the lack of adjustment for potential confounders.
Xia et al. 18 conducted a survey in 2004 in a village from Inner Mongolia, China. Interviews were performed in each household, eliciting information on demographic characteristics, occupation, smoking, alcohol drinking, residential history including water sources, and prevalence of chronic conditions and of arsenic-associated skin lesions (hyperpigmentation, hyperkeratosis or depigmentation). One medical member of the interview team assessed the presence of the skin lesions by visual examination in over 90% of the cases. Water samples were collected from the household well, shared well, or community well used by each household, and were analyzed for arsenic concentration. The study included 3,284 households with 12,334 individuals, of whom 11,416 (93%) had complete data and were therefore included in the analysis. Average arsenic level was 37.9 μg/L; a total of 622 subjects (also indicated as 632 in thevpublication) had skin lesions (5.4%). The prevalence of skin lesions was associated with female sex, low education, alcohol drinking, farm working, and use of community well. The age-adjusted POR for arsenic exposure from drinking water are shown in Table 2.
Limitations of the study include the lack of verification of diagnosis of skin lesions, the lack of consideration of historical exposure, and the fact that associations between skin lesions and sociodemographic and lifestyle factors in this study were not consistent with those observed in other studies. Given the large proportion of study subjects with low arsenic exposure, it is important to notice that limited exposure misclassification would have a greater impact at low exposure (because subsequent exposure categories are narrower) than at high exposure.
Fatmi et al. 19 studied residents of 110 households in 2 areas of Khairpur district in Sindh, Pakistan, in which individuals were found to be exposed to more than 50 μg/L arsenic in drinking water. Out of 610 individuals who were interviewed in 2008-2009, 534 were included in the final analysis. The presence of arsenic-related skin lesions (hyperpigmentation of a sun-unexposed part of the body thickening of the skin of palms and soles) was determined by field workers. Diagnoses of a subset of cases were validated by examination of the digital images of skin lesions and by random visits to the cases’ houses. Data on education, smoking and body mass index were collected. The average exposure level of study subjects was not reported but was estimated to be 257 μg/L from the categorical results reported in the article. The overall prevalence of skin lesions was 13.5% (72 cases), and was higher in women, younger subjects, and smokers. The unadjusted POR, for arsenic exposure, derived from the raw data reported in the publication, are reported in Table 2.
Limitations of this study include the lack of consideration of historical exposures, the lack of validation of diagnosis of skin lesion, the lack of adjustment for potential confounders, including age and sex, and the lack of blind status of field workers with respect to arsenic exposure.
Incidence Studies
Argos et al. 20 reported the only prospective study on incidence of skin lesions and exposure to arsenic in drinking water. The study was conducted in Araihazar, Bangladesh. Between 2000 and 2002, 11,746 married individuals aged 18–75 years and residing in the study area for at least 5 years were enrolled in the study. Study physicians blinded to arsenic exposure of study subjects conducted in-person interviews and clinical skin examination. Study subjects were contacted every 2 years for a follow-up telephone interview and a skin examination until 2009. At baseline there were 714 subjects with prevalent skin lesions (6.2%); after excluding these subjects and those with no skin examination at baseline or at the first follow-up, the analysis was restricted to 10,182 individuals. Subjects indicated at baseline the well they used as their main source of drinking water; arsenic concentrations were measured in all 5,966 wells in the study area. The average arsenic level, derived from the categorical results reported in the manuscript, was 91.0 μg/L. Skin lesions included hyperpigmentation, hypopigmentation, and keratosis. Potential confounders included sex, age, education, smoking status and body mass index. Overall, there were 866 incident cases of skin lesion, with a cumulative incidence of 8.5% over an average 7 years of follow-up (9.6% loss to follow-up). Incidence of skin lesions was associated with old age, male gender, low education, tobacco smoking, and low body mass index. The adjusted RR for arsenic exposure in drinking water are reported in Table 2. In an analysis based on arsenic concentration in urine, the incidence of skin lesions was associated with high exposure at baseline; however, neither an increase or a decrease in exposure at follow-up influenced the risk determined from baseline exposure. Limitations of the study include the lack of validation of diagnosis of skin lesion and lack of historical information on arsenic level in wells used before enrollment in the study.
Synthesis of Evidence
The results of the dose-response analysis of RR are reported in Table 3. There was strong evidence of heterogeneity between the studies (p < 0.001, I2 = 95.8%): since neither of 2 criteria for heterogeneity were satisfied, we did not perform a meta-analysis.
Results of Study-Specific Dose-Response Analysis.

RR, relative risk of skin lesion for an increase of 10 µg/L arsenic in drinking water.
CI, confidence interval.
The exclusion of individual studies did not reduce the heterogeneity of results: the value of the I2 statistic consistently remained larger than 93% (detailed results available from the authors). The assessment of the funnel plot (Figure 2) suggested the presence of publication bias, which was supported by the results of the Egger test (p = 0.10).
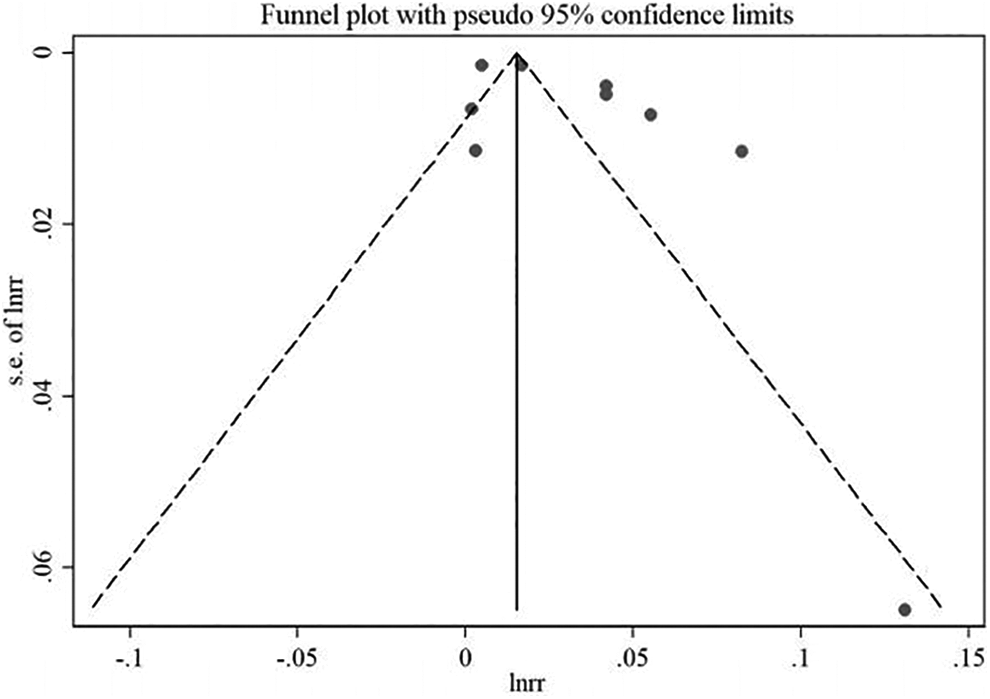
Funnel plot of dose-response results.
The secondary analyses of results in categories with mid-level exposure up to 50, 100 and 125 µg/L arsenic were limited to few data points; in particular the analysis of categories with mid-level exposure up to 125 µg/L was restricted to 7 studies (Table 2 and showed high heterogeneity between study-specific dose-response results (p < 0.001, I2 = 79.8%; Table 3).
Discussion
Our systematic review of studies on prevalence and incidence of skin lesions and exposure to arsenic in drinking water identified 9 studies that responded to the selection criteria. All but one study were of cross-sectional design; only 1 study included a validation of the outcome. The results of these studies were highly heterogeneous, both across the whole ranges of exposure, as well as in an analysis restricted to subjects classified in categories with mid-level exposure up to 125 µg/L arsenic. As an example, the prevalence of skin lesions in the category of exposure 0-50 μg/L ranged from 0.1% in the study by Guha Mazumder et al. 11 to 34.2% in the study by Guo et al. 15 There was a 70-fold difference in the increased risk between the lowest (1.002 [Guo et al. 15 ]) and the highest (1.140 [McDonald et al. 16 ]) estimated RR for the same increase in arsenic in drinking water (10 μg/L). The heterogeneity was not attributable to any individual study. Since the indices of inter-study heterogeneity did not reach the predefined thresholds, a dose-response meta-analysis was not performed: this decision was further supported by the suggestion of publication bias in the available results.
The strong heterogeneity in results detected between the reviewed studies can originate from (i) selection bias in the recruited study populations, (ii) exposure measurement error, including determination of arsenic levels in the drinking water, difficulty in determining historical exposures, lack of individual exposure and ignoring possible additional sources of exposure, such as food and occupation (iii) outcome measurement error (e.g., lack of validation of diagnosis of skin lesions), (iv) residual confounding and differential effect modification (e.g., tobacco smoking and chewing, nutritional factors, occupational exposures), and (v) random error from low statistical power.
In some of the studies [e.g., McDonald et al. 16 ; Fatmi et al. 19 ] participating subjects were selected according to their arsenic exposure, which may have resulted in selection bias. Participation rate was reported in several studies, and was consistently very high (in the range 88-94%): although one cannot exclude that selection bias occurred also in the studies for which information on participation was not available, the data at hand do not support the hypothesis of a role of participation bias role in determining the patterns of results and their heterogeneity.
Errors in the measurement of arsenic exposure can have multiple origins. Except for 2 studies, 12,14 only wells used by study subjects at the time of the investigation were tested. The lack of historical exposure data, which was a limitation of most studies, would result in bias of undetermined direction if duration of residence varied between cases and non-cases as no data are available to test this hypothesis. Furthermore, even if duration of stay at current residence was comparable between cases and non-cases, restriction of the measurement of exposure to the current residence might result in overestimate of the dose-response relationship if exposure levels declined over time. Limited information is available to address this issue. Haque et al. 12 reported results for 213 non-cases with exposure data from current residence and for a subset of 89 non-cases with exposure data from their whole residential history and observed no differences in the exposure distribution of the subset compared to the whole population. On the other hand, the same authors compared exposure data collected in 1995-1996 and in 1998-2000 for the cases included in their study: 6/192 cases in 1995-1996 had exposure below 50 μg/L vs. 2/120 cases in 1998-2000 (p = 0.43). Furthermore, differences in the metric used to measure the effect of arsenic exposure (average over lifetime vs. current) is another potential source of heterogeneity of the results, since it is plausible that the use of current exposure entails a greater degree of misclassification than that of cumulative exposure. In particular, 6 studies reported and analyzed on current exposure level, 2 on peak exposure, 12,17 1 on baseline exposure, 20 and 1 on time-weighted average exposure. 14 Two studies also analyzed cumulative exposure. 14,17 Lack of past exposure is an important limitation of most available studies, and likely contributed to the heterogeneity of results, both across the whole exposure range, and at low levels of exposure.
An additional reason for heterogeneity between the results of individual studies is the fact that different approaches have been used to test water samples and measure arsenic content. While this issue would not invalidate the results of individual studies, it inhibits comparison between studies. Water sources were mostly measured at the place of residence, and other sources of drinking water, such as those at workplaces, were not taken into account. It is unclear whether these potential sources of exposure misclassification would have operated differentially between cases and controls, and it is difficult to assess the direction of the potential resulting bias.
Errors and differences in the assessment of skin lesions is likely to represent an important potential source of bias and heterogeneity of results across studies. Diagnosis of skin lesion was done by different professional personnel across the studies, who, in most instances, were not blind to the exposure status of the subjects; only 2 studies included a blind review as part of the diagnoses of skin lesions. 12,14 The heterogeneity of prevalence across studies for the same estimated level of exposure is hardly explained by potential effect modifiers, such as tobacco smoking, sun exposure and diet, since it was present even when studies with different characteristics were excluded. It likely reflects the approach used to measure skin lesions. Misclassification of skin lesion can be particularly relevant when assessors (interviewers, health workers or physicians) were not blind of the exposure status of study subjects.
Potential confounders were seldom adjusted for in the available studies (Table 1). However, when an adjustment for multiple confounders was performed, no substantial changes in the RR were reported. 12,20 Notably, no studies accounted for the potential confounding effect of exposure to sunlight. Random error remains an additional possible source of heterogeneity, in particular for studies with a small number of cases (e.g., [Fatmi et al., 2013]).
Our systematic review of studies that addressed the dose-response relationship between estimated level of arsenic in drinking water and occurrence of skin lesions was limited by the small number of relevant studies, the cross-sectional design for most of them, and several important potential biases. The strong heterogeneity between study-specific results and the suggestion of publication bias, that prevented us from performing a meta-analysis, strongly cautions their interpretation. It is worth noting that in one of the 2 studiesthat included both an assessment of exposure at multiple times and a diagnostic validation, and was assigned the best quality score in our review, 12 no cases with skin lesions experienced exposure below 100 µg/L arsenic exposure. Several studies supported the conclusion of no increase in skin prevalence up to at least 50 µg/L arsenic exposure, 16,17,20 while only 2 papers detected an association also below this level, 14,18 although a complete exposure history is lacking in both studies. The lack of increased prevalence of skin lesions at low level of exposure is consistent with those of an ecologic study conducted in the early 1980s in 6 villages of West Bengal, India, in which no patients with skin lesions used tubewells with arsenic concentration lower than 200 µg/L, 28 and those of a study from Iran, in which no increased prevalence was detected below 1 g of total estimated arsenic intake, corresponding to 70 µg/L. 29
Our analysis was restricted to a linear, non-threshold model, and did not address other dose-response relations. While non-linear models have been proposed to explain the association between arsenic exposure and prevalence of skin lesions, 5 we selected the linear model because it represents a more conservative approach, and it is adopted by regulatory authorities such as EPA for carcinogenic effects. 30 Furthermore, we based on analysis on average (lifetime) exposure, despite the fact that other aspects of exposure such as duration peaks are additional important components of arsenic toxicity. 5,31 -33 This choice was justified by the limited number of studies that reported valid results on indices of arsenic exposure other than average drinking water level. Overall, our analysis suggests that linear dose-response modeling, coupled with use of average arsenic concentration, lack of complete exposure history, inaccuracy in skin lesion diagnosis as related to arsenic, and lack of consideration to heterogeneity of results can result in false positive dose-response at low doses.
In conclusion, currently available data are not sufficient for conducting meta-analysis with the purpose of determination of causal association between low exposure to arsenic and skin lesions. More high-quality studies with prospective design, protection from bias in the assessment of both arsenic exposure and diagnosis of skin lesions would be necessary before firm conclusions can be drawn on the dose-response relationship. Studies with complete exposure histories indicate skin lesions are associated with arsenic exposure in excess of 50 µg/L and higher.
Supplemental Material
Supplemental Material, appendix_1 - Dose-Response Analysis of Exposure to Arsenic in Drinking Water and Risk of Skin Lesions: A Systematic Review of the Literature
Supplemental Material, appendix_1 for Dose-Response Analysis of Exposure to Arsenic in Drinking Water and Risk of Skin Lesions: A Systematic Review of the Literature by Paolo Boffetta, Carlotta Zunarelli and Claire Borron in Dose-Response
Supplemental Material
Supplemental Material, arsenic_skin_lesion_dose_resp_appendix_2 - Dose-Response Analysis of Exposure to Arsenic in Drinking Water and Risk of Skin Lesions: A Systematic Review of the Literature
Supplemental Material, arsenic_skin_lesion_dose_resp_appendix_2 for Dose-Response Analysis of Exposure to Arsenic in Drinking Water and Risk of Skin Lesions: A Systematic Review of the Literature by Paolo Boffetta, Carlotta Zunarelli and Claire Borron in Dose-Response
Supplemental Material
Supplemental Material, arsenic_skin_lesion_dose_resp_appendix_3_rev1 - Dose-Response Analysis of Exposure to Arsenic in Drinking Water and Risk of Skin Lesions: A Systematic Review of the Literature
Supplemental Material, arsenic_skin_lesion_dose_resp_appendix_3_rev1 for Dose-Response Analysis of Exposure to Arsenic in Drinking Water and Risk of Skin Lesions: A Systematic Review of the Literature by Paolo Boffetta, Carlotta Zunarelli and Claire Borron in Dose-Response
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PB was involved as expert for the plaintiff in litigation involving arsenic carcinogenicity.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was partially supported by the Arsenic Science Task Force.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
