Abstract
The effect of remote ischemic preconditioning (RIPC) has been proposed that mediates the protective response in ischemia reperfusion injury (IRI) of various organs. In this study, we investigated the effect of RIPC in hepatic IRI, by assessing biomarker of oxidative stress and inflammatory cytokines. Moreover, we intended to demonstrate any such protective effect through nitric oxide (NO). Twenty-five rats were divided into the 5 groups: (1) Sham; (2) RIPC; (3) hepatic IRI; (4) RIPC + hepatic IRI; (5) C-PTIO, 2-(4-carboxyphenyl)-4,5dihydro-4,4,5,5-tetramethyl-1H-imidazolyl-1-oxy-3oxide, + RIPC + hepatic IRI. RIPC downregulated the level of aspartate aminotransferase (AST), alanine aminotransferase (ALT), histologic damage, and activity of Malondialdehyde (MDA). However, there was no significant reduction in the level of tumor necrosis factor-alpha (TNF-α) and nuclear factor kappa B (NF-κB). AST and ALT levels, and hepatic tissue morphology in the C-PTIO group showed a significant improvement compared to those of the RIPC + hepatic IRI group. The application of RIPC before hepatic ischemia downregulated the oxidative stress, not the inflammatory cytokines. Moreover, these protective effect of RIPC would be mediated through the activation of NO as well as anti-oxidant effect.
Introduction
Hepatic ischemia reperfusion (IR) is an important clinical issue that follows liver resection surgery, transplantation, and circulatory shock. IR injury occurred during liver transplantation is an inevitable event, which causes up to 10% of early transplant failure or rejection. 1 In addition, possible consequences of hepatic IR include secondary organ dysfunction (systemic complication) as well as liver failure, and subsequently increase patient morbidity and mortality. Therefore, many therapeutic strategies have been proposed to attenuate or prevent the hepatic IR injury, however, none have been completely successful.
Remote ischemic preconditioning (RIPC), whereby several transit periods of ischemia followed by reperfusion in one organ protects distant organs. 2,3 This therapeutic approach is less invasive and more clinically practical than local ischemic preconditioning (IP). IP is induced by brief ischemia and reperfusion before subsequent prolonged ischemia, which results in enhancing organ tolerance against the lethal IR injury. 4 Protective effect of IP against hepatic IR injury has been known, however its clinical application is limited because of detrimental effect of its invasive intervention. In this regard, application of RIPC, which allows the intervention to a remote organ from the liver, may have the superiority to the IP.
In this study, we hypothesized that RIPC can attenuate the production of oxidative stress induced by IR injury. In addition, we intended to demonstrate the effects of RIPC on hepatic IR injury in rats and prove the protective mechanism through nitric oxide (NO) in the effects of RIPC on hepatic IR injury.
Materials and Methods
Animals
Male Sprague-Dawley rats (290-310 g) were used in the experiment (Central Lab. Animal Inc., Seoul, Korea). All procedures were approved by the Yeungnam University College of Medicine Institutional Animal Care and Use Committee (YUMC-AEC2018-021), and were conducted in accordance with the institutional guidelines.
Experimental Protocol
Twenty-five rats were divided into the different 5 groups (n = 5 per each group) as follows: (1) sham operated (Sham); (2) remote ischemic preconditioning group (RIPC); (3) hepatic IR injury (IRI); (4) RIPC + hepatic IRI (RIPC + IRI); (5) 2-(4-carboxyphenyl)-4,5dihydro-4,4,5,5-tetramethyl-1H-imidazolyl-1-oxy-3oxide (C-PTIO) + RIPC + hepatic IRI (C-PTIO + RIPC + IRI).
Rats were anesthetized with ketamine and xylene. RIPC was induced through 3 cycles of ischemia/reperfusion, which was carried out by repeated occlusion/opening (5 min/5 min) of unilateral femoral vascular bundle at 30 min before hepatic ischemia. Vascular occlusion was induced by an automated cuff inflator of 250 mmHg. 2 mg/kg C-PTIO was administered 5 min prior to hepatic ischemia. 5
After a midline laparotomy was done, lateral and median lobes of liver were rendered ischemia during 30 min by clamping the pertinent portal triad branch using microvascular clamp (approximately 70% of liver) and then clamp was removed to induce reperfusion during 2 h. Blood was collected from the heart to minimize hemolysis during sampling and hepatic tissues were divided into 2 sections for molecular (snap-frozen in liquid nitrogen at -80°C) and histopathological (fixed in 10% formaldehyde and embedded in paraffin) analysis.
Biochemical Analysis of Enzyme Activity
After centrifugation of blood samples, the serum supernatant was collected. Serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were assayed with a standard autonomic biochemistry analyzer (Vitros 250, Johnson and Johnson, USA).
Oxidative Analysis of Hepatic Tissue
Hepatic tissues were centrifuged at 3000 rpm for 15 min. Malondialdehyde (MDA), a measure of lipid peroxidation, was assayed spectrophotometrically in the supernatant using thiobarbituric acid reactive substances (TBARS). 6 The absorbance of complex formed by thiobarbituric acid and MDA was read at a 535 nm wavelength and the value was expressed as nmol/mg of protein weight.
Western Blot of Hepatic Tissue
Hepatic tissues were crushed in RIPA lysis buffer and heated. After homogenation, the samples were centrifuged at 12,500 rpm for 20 min and the extracted protein from supernatants was quantified using the Bradford assay. After mixing with a loading buffer solution (60 mM Tris-HCl, 25% glycerol, 2% SDS, 14.4 mM 2-mercaptoethanol, and 0.1% bromophenol blue) and gel electrophoresis was done in a 10% SDS polyacrylamide gel. Electroblotting was performed onto a nitrocellulose membrane (Whatman GmbH, Dassel, Germany). Primary antibodies were diluted in Tris-buffered saline containing 0.1% Tween-20 (TBS-T) and incubated for 2 h at room temperature. Following washes of membrane, it was incubated with tumor necrosis factor-alpha (TNF-α) and nuclear factor kappa B (NF-κB) (diluted 1:1000; Santa Cruz, CA, USA) antibodies and β-actin (diluted 1:5000; Cell Signaling, Beverly, MA) overnight at 4°C. Membrane was then incubated with secondary antibody (mouse horseradish peroxidase-conjugated antibody; diluted 1:2000) and enhanced chemiluminescence substrate kit (Advansta, California, USA). After exposing onto medical x-ray film and the band density was measured using NIH Image J 1.47v software.
Histological Examination
Hepatic tissues with 5 µm thickness were performed with hematoxylin and eosin staining after fixing in 10% buffered formalin and embedded in paraffin. Histopathological evaluation was graded according to scoring system of 0 to 3 in a blinded fashion; sinusoidal congestion, cytoplasmic vacuolation, and hepatocyte necrosis. 7
Statistical Analysis
Data were presented as means ± SEM of 5 rats per group. Paired Student's t-test and 1-way ANOVA followed by Bonferroni post-hoc test were performed to compare values between 2 dependent groups and among multiple groups, respectively. All analysis were conducted with SPSS version 23.0 (SPSS, Chicago, Illinois, USA). Statistical differences were considered significant at p < 0.05.
Results
Hepatic IR injury induced a significant increase in the AST and ALT levels compared to the sham and RIPC group (P < 0.05). The rats in the RIPC + IRI group showed lower AST and ALT levels than those of IRI alone group (P < 0.05). However, AST and ALT levels in the C-PTIO + RIPC + IRI group showed a significant rise compared to those of the RIPC + IRI group (P < 0.05) (Figure 1). To determine the oxidative stress, liver MDA activity, cellular lipid peroxidation marker, was investigated. When compared with sham and RIPC group, MDA level was significantly increased after hepatic IR. MDA level was reduced in the RIPC + IRI group in comparison with IRI alone group (P < 0.05). MDA levels in the C-PTIO + RIPC + IRI group did not show a significant difference compared to those of the RIPC + IRI group (Figure 2). To determine the inflammatory response, we investigated the TNF-α and NF-κB. Hepatic IR injury caused a significant increase in level of TNF-α and NF-κB, respectively, as compared with sham and RIPC group (P < 0.05). There was no significant reduction in the level of TNF-α and NF-κB in the RIPC treatment group after IRI in comparison with IRI alone group. Moreover, the difference in the level of TNF-α and NF-κB between RIPC + IRI group and C-PTIO + RIPC + IRI group was not detected (Figure 3). In the histological analysis, hepatic injury was evaluated by sinusoidal congestion, ballooning degeneration, and hepatocyte necrosis. For the sham and RIPC group, hepatic injury was not observed. However, rats in the hepatic IRI group exhibited severe sinusoidal congestion, cytoplasmic vacuolation, and necrosis compared with rat in the sham and RIPC group. Rats in the RIPC + IRI group showed much less histologic injury than those of hepatic IR group (P < 0.05). A significant histological injury was observed in the C-PTIO + RIPC + IRI group compared to the hepatic tissue morphology observed in the RIPC + IRI group (P < 0.05) (Figure 4).

Serum AST and ALT level in the experimental groups. Sham group; RIPC group, remote ischemic preconditioning; IRI group, hepatic ischemia (30 min) followed by reperfusion (2 h); RIPC + IRI group, RIPC was induced at 30 min before hepatic ischemia; C-PTIO + RIPC + IRI group, 2 mg/kg C-PTIO was administered 5 min prior to hepatic ischemia. *p < 0.05, vs. the sham group; † p < 0.05 vs. the RIPC group; ‡ p < 0.05 vs. the IRI group; § p < 0.05, vs. the RIPC + IRI group. Data are expressed as mean ± SEM (n = 5 rats/group).

Hepatic tissue MDA level in the experimental groups. Sham group; RIPC group, remote ischemic preconditioning; IRI group, hepatic ischemia (30 min) followed by reperfusion (2 h); RIPC + IRI group, RIPC was induced at 30 min before hepatic ischemia; C-PTIO + RIPC + IRI group, 2 mg/kg C-PTIO was administered 5 min prior to hepatic ischemia. *p < 0.05, vs. the sham group; † p < 0.05 vs. the RIPC group; ‡ p < 0.05 vs. the IRI group. Data are expressed as mean ± SEM (n = 5 rats/group).

Hepatic tissue TNF-α and NF-κB level in the experimental groups. Sham group; RIPC group, remote ischemic preconditioning; IRI group, hepatic ischemia (30 min) followed by reperfusion (2 h); RIPC + IRI group, RIPC was induced at 30 min before hepatic ischemia; C-PTIO + RIPC + IRI group, 2 mg/kg C-PTIO was administered 5 min prior to hepatic ischemia. *p < 0.05, vs. the sham group; † p < 0.05 vs. the RIPC group. Data are expressed as mean ± SEM (n = 5 rats/group).
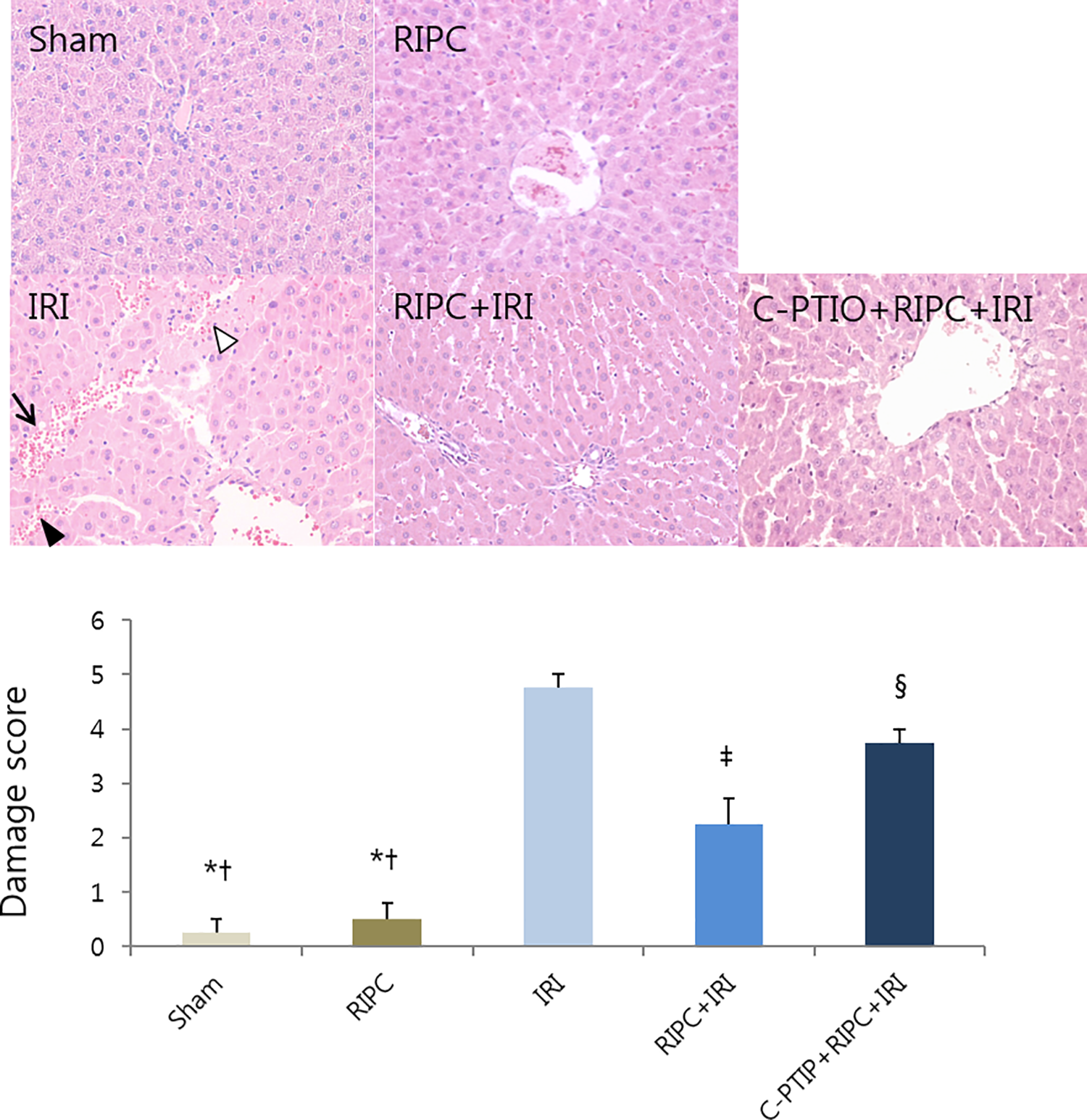
Histopathological photographs and damage score of hepatic tissue in the experimental groups. Periodic acid Schiff (PAS) stain, sinusoidal congestion (black arrowhead), cytoplasmic vacuolization (arrow), liver necrosis (white arrowhead), magnification 200X. Sham group; RIPC group, remote ischemic preconditioning; IRI group, hepatic ischemia (30 min) followed by reperfusion (2 h); RIPC + IRI group, RIPC was induced at 30 min before hepatic ischemia; C-PTIO + RIPC + IRI group, 2 mg/kg C-PTIO was administered 5 min prior to hepatic ischemia. *p < 0.05, vs. the sham group; † p < 0.05 vs. the RIPC group; ‡ p < 0.05 vs. the IRI group; § p < 0.05, vs. the RIPC + IRI group. Data are expressed as mean ± SEM (n = 5 rats/group).
Discussion
This study showed that RIPC improved liver function and histopathology. RIPC reduced MDA, while it had no effect on the TNF-α and NF-κB. These results suggested that oxidative stress was related to RIPC protection, while inflammatory cytokines was not to relevant to RIPC protection during hepatic IRI (30 min ischemia and 2 h reperfusion). In addition, these protective effect of RIPC would be mediated through the activation of NO, which was verified by the offset of functional marker (AST, ALT) and an improvement in hepatic tissue morphology in the C-PTIO treatment group.
RIPC, whereby several transit periods of ischemia followed by reperfusion in one organ protects distant organ. 2,3 This therapeutic approach is clinically practical because of non-invasively application, by inflating and deflating a cuff placed on the either extremity, and the application of various time points: 12 – 24 h before of target organ ischemia (delayed RIPC) or immediately before of target organ ischemia (RIPC) or onset of target organ ischemia (remote ischemic perconditioning), or onset of target organ reperfusion (remote ischemic postconditioning). 8 The components of RIPC can be divided into 3 parts: signal generation, signal transfer to target organ, and organ protection. Though the exact mechanisms underlying RIPC induced organ protection remain unclear, neural factors (autonomic ganglions, bradykinin receptors, and adenosine receptors), humoral factors (opioids, adenosine, and bradykinin), and systemic responses including anti-inflammatory and anti-apoptotic are thought be mediate the protective effects of organs or tissues. 8 In other words, triggering and signal transduction via humoral/blood and neuronal transmission have been proposed, and these factors act partially overlapped, not mutually exclusive.
Oxidative stress and inflammatory response play a central role in the pathognomonic mechanism of hepatic IR injury, these can lead to lipid peroxidation of cell membrane, DNA damage, collectively lead to cell necrosis or apoptosis, and finally result in the loss of cell viability. 9 During reperfusion period, activated machrophages release the extracellular ROS, which are the initiators of reperfusion injury. Whereas neutrophils are the key cells in the late phase of IR injury showing damaged endothelials and further release of inflammatory cytokines. 10 Moreover, adaptive responses of different phase have evidences in the direct IPC 11,12 or RIPC 13,14 : early phase begins soon after reperfusion and short lived of 3 to 4 h, whereas late phase starts 12 to 24 h later. 11,12 In this study, the protective effects of RIPC were associated with attenuation of oxidative stress showing decreased MDA levels, verifying oxidative free radicals, and which seems to play an important role in the various mechanisms of inducing ischemic tolerance by RIPC. However, TNF-α and NF-κB had no effects on the RIPC against hepatic IR injury. Here, the activation of TNF-α by NF-κB can lead to inflammatory damage in the IRI. 15,16 NF-κB is known to prototypic transcription factor in inflammatory response and cell differentiation 15 and TNF-α has potent inflammatory and cytotoxic effects. 16 This result might be explained one possible reason; different reperfusion time during IR injury, in this study, we analysed the initial phase of reperfusion injury (2 h reperfusion time), not the late phase of it. Further studies about longer reperfusion time might be needed for better understanding the pathophysiology of hepatic IR injury.
Studies in RIPC have demonstrated the role of NO. 17 -20 NO has an important role in initiating and maintaining preconditioning. Especially, in the liver, NO mediates preconditioning through activation of adenosine A2 receptor and inhibition of endothelin, 21,22 and subsequently has a protective effect on the microcirculation. Most of these studies assessed the activities of NO with respect to NO synthase; endogenous NOS (eNOS) and inducible NOS (iNOS). 23 The early phase of preconditioning is through activation of eNOS and the late phase of it mediated by transcription of iNOS. 20,24 Shear stress by RIPC acts strong physiologic stimuli of eNOS, which generates circulating NO metabolites (nitrite) and subsequently these factors modulate microcirculation at the remote organs with leading to remote organ protection. 25 Abu-Amara et al. showed that eNOS ablation abrogated the hepatoprotection with RIPC on the liver damage. 26 They identified NO dependent pathway by hindlimb IR mediated the hepatoprotection during the early phase of hepatic IRI. 26 Moreover, Rassaf et al. showed that nitrite was generated by shear stress during reactive hyperemia of RIPC, which is converted to bioactive NO by myoglobin, then NO confer the organ protective effects in the human and mouse heart with IR injury. 25 They implied that eNOS derived nitrite signal by RIPC contributed to cardioprotection during myocardial IR injury. 25 In this study, we used C-PTIO, NO scavenger, to neutralize the activation of NO. Liver function (AST and ALT) as well as hepatic tissue morphology were worsen in the C-PTIO administration group compared with RIPC group against hepatic IRI, which indirectly suggested the protective effect of RIPC dependent NO. In other words, through pharmacological scavenging, nitrite generation from NO abolished hepatoprotective effects from RIPC maneuver.
This study has some limitations. First, we conducted the experiments with the one reperfusion time for 2 h. As mentioned above, hepatic IRI has 2 distinct phase of liver injury. The early phase is characterized by oxidative stress which mostly activated by Kupffer cell, whereas late phase is an inflammatory disorder mediated by neutrophil. Therefore, for the better understanding the role of oxidants and inflammatory cytokines, further experiments in a time-dependent manner might be needed. Second, to verify the hepatoprotective effect of RIPC mediated NO production, we administered C-PTIO prior to RIPC and IRI. However direct NO measurement through nitrite, which are thought to be an index of NO production, might have the stronger power to prove the NO-dependent pathway on the hepatoprotective effect of RIPC. Finally, we applied the RIPC to a unilateral lower limb. By the some other studies, bilateral RIPC was more effective than unilateral RIPC. 27 Although our results showed the hepatoprotective effect of RIPC in rats, bilateral RIPC application might be have more clinical relevance and benefit results.
In conclusion, RIPC exerted protective effects against hepatic IRI, as indicated by reduction of oxidative stress. Also, hepatoprotective effect of RIPC would be mediated through the activation of NO, which is verified by C-PTIO, NO scavenger. This therapeutic approach, RIPC maneuver, is easy and convenient to apply clinically, therefore, application of RIPC to patients undergoing procedures complicated by IRI could elicite better outcomes.
Footnotes
Author Contribution
Eun Kyung Choi helped design the study, conduct of the study, analyze the data, and write the manuscript. Hoon Jung participated in designing the experiments and conduct of the study. Sungmin Jeon helped the study. Jung A. Lim analyzed the data and prepare the manuscript. Jungwon Lee analyzed the data and prepare the manuscript. Hyunjee Kim analyzed the data and prepare the manuscript. Seong Wook Hong prepare the experiment and manuscript. Min Hye Jang participated in pathologic analysis. Dong Gun Lim helped design the study and writing the manuscript. Kyung Hwa Kwak participated in designing the experiments and writing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Biomedical Research Institute grant, Kyungpook National University Hospital (2016).
