Abstract
Naproxen sodium is a nonsteroidal anti-inflammatory drug (NSAID) having antipyretic and analgesic properties, mainly used for the treatment of rheumatoid arthritis and osteoarthritis. Eryptosis is an alternative term used for suicidal erythrocyte death. In the current study, eryptotic effect of naproxen sodium characterized by membrane blebbing was investigated in erythrocytes after 48 hours of treatment with different concentrations (1-25 µM). The experimental work related to investigation of eryptosis was done by cell size measurement and confirmation of calcium role in the induction of membrane blebbing. As a possible mechanism of eryptosis, oxidative stress induced by naproxen sodium was determined by catalase, glutathione peroxidase, and superoxide dismutase activities. Similarly, hemolytic effect of naproxen sodium was also determined by hemolysis measurement. Results of our study illustrated that the therapeutic doses (10-25 µM) of naproxen sodium induce oxidative stress, confirmed by significant decrease in superoxide dismutase, catalase, and glutathione peroxidase activities that lead to the triggering of cell death by eryptosis and hemolysis.
Introduction
Naproxen sodium [(S)-6-methoxy-alpha-methyl-2-naphthaleneacetic acid sodium salt], a nonsteroidal anti-inflammatory drug, poses antipyretic and analgesic properties. 1 The mode of action of this drug include COX inhibition and blockage of prostaglandin synthesis. 2 Different inflammatory conditions, especially rheumatoid arthritis and osteoarthritis, are frequently treated with naproxen sodium. 3 Gastrointestinal toxicity, hepatotoxicity, nephrotoxicity, and jaundice are among the reported side effects of this drug. 4 Similarly, naproxen sodium also confirmed to induce oxidative stress by the production of reactive oxygen species. 5
Characteristics of eryptosis include shrinkage of cells, membrane blebbing, 6 and cell membrane scrambling which leads toward phosphatidylserine translocation in the membrane. 7 Splenic macrophages recognize, engulf, and decompose erythrocytes with exposed phosphatidylserine on outer leaflet. 8 Oxidative stress, osmotic shock, and energy-depleted environment may trigger Ca2+ [Ca2+]i-permeable cation channels that result in the stimulation of Ca+2 entry and subsequently different events of eryptosis. High cytosolic Ca2+ leads to Ca+2-sensitive K+ channels activation 9 that causes cell shrinkage due to KCl loss with water. 6 Similarly, membrane blebbing due to the breakdown of cytoskeleton and phosphatidylserine translocation in erythrocyte’s cell membrane is completely dependent on high calcium influx. 10 It is reported that stimulated eryptosis may lead to anemia that participates in pathophysiology of several clinical issues. 8
Different xenobiotic compounds were investigated and found to have strong eryptotic effect. 7,10 In the present work, therapeutic doses of naproxen sodium were used to determine their role in the induction of oxidative stress, eryptosis, and hemolysis in erythrocytes.
Methods
For experimental work, fresh and screened blood samples were obtained from different blood banks of Faisalabad. The work was conducted after the approval of directorate of graduate studies and IBC, University of Agriculture Faisalabad, Pakistan.
Leukocyte-depleted cells were prepared by following protocol explained by Fink et al. 11 Isolated erythrocytes were stored in separate micro-centrifuge tubes. In vitro incubations of erythrocytes were performed at a hematocrit of 0.4% in Ringer solution (pH 7.4) that contain (in mM) MgSO4 1, NaCl 125, KCl 5, glucose 5, CaCl21, N-2-hydroxyethylpiperazine-N-2-ethanesulfonic acid 32 at 37°C for 48 hours. 12 Isolated erythrocytes were then exposed to naproxen sodium (Sigma-Aldrich, St. Louis, Missouri, United States) at the indicated concentrations. 13
Oxidative Stress Measurement
To determine the oxidative stress in naproxen sodium–exposed erythrocytes, antioxidant enzyme (superoxide dismutase [SOD], catalase, and glutathione peroxidase) assays were performed.
Superoxide Dismutase
Superoxide dismutase’s activity was measured by following the protocol of Rana et al. 9 The reaction solution contained methionine 0.222 g in 15 mL H2O, NBT 0.015 g in 17.5 mL H2O, Triton-X 0.0375 mL in 17.5 mL H2O, riboflavin 0.0132 g in 17.5 mL H2O, and buffer 0.2 M.
Glutathione Peroxidase
Phosphate buffer (pH 5) 50 mM, guaiacol 20 mM, H2O2 40 mM, and enzyme extract 0.1 mL were added in reaction mixture by following the protocol of Ullah et al, 14 and activity was measured at 470 nm after every 20 seconds.
Catalase
Catalase activity was determined following the method described by Ullah et al. 14 Phosphate buffer (pH7) 50 mM, H2O2 5.9nM, and enzyme extract 0.1 mL were added in reaction mixture, and absorbance was read at 240 nm.
Cell Size Measurement
Changes in cell size were observed by mean cell volume (MCV) and forward scatter (FSC).
Mean cell volume
Mean cell volume was measured to determine the cell size of control and naproxen sodium–treated cells. Mean cell volume was checked by using automated hematology analyzer. 9
Forward scatter
After incubation, a 50-µL cell suspension was washed in Ringer solution containing 5 mM CaCl2, and the FSC of the exposed cells was determined on FACS Calibur (BD, Heidelberg, Germany). 12
Confirmation of Ca+2 Role
Amlodipine is a calcium channel blocker. 15 To confirm calcium role in triggering of eryptosis, erythrocytes were treated with naproxen sodium (1-25 µM) in the absence and presence of 10-µM amlodipine. The inhibition of eryptosis was confirmed by MCV measurement.
Hemolysis Measurement
After incubation, erythrocyte samples were centrifuged (3 minutes at 400 g at room temperature), and supernatant was collected to determine the percentage of hemolysis. Hemoglobin concentration was measured at 405 nm. The absorbance of the supernatant of erythrocytes lysed in dH2O was defined as 100% hemolysis. 16
Statistical Analysis
All the data are expressed as arithmetic means ± standard error of mean. Statistical analysis was made by using analysis of variance with Tukey test as posttest or t test, as appropriate. 10 *P < .05, **P < .01, and ***P < .001 indicates significant difference from the absence of naproxen sodium.
Results
All the results of our studies are demonstrated with original tables and figures. As reported previously, oxidation is a possible mechanism of eryptosis, so the possible oxidative effects of naproxen sodium on erythrocytes were measured by antioxidant enzyme activities in treated cells and control cells. The effect of naproxen sodium on the activities of studied enzymes including SOD, catalase, and glutathione peroxidase as means ± standard deviations (SD) are given in Table 1.
Effect of Naproxen Sodium on Superoxide Dismutase, Catalase, Glutathione Peroxidase, and Hemolysis in Erythrocytes.a
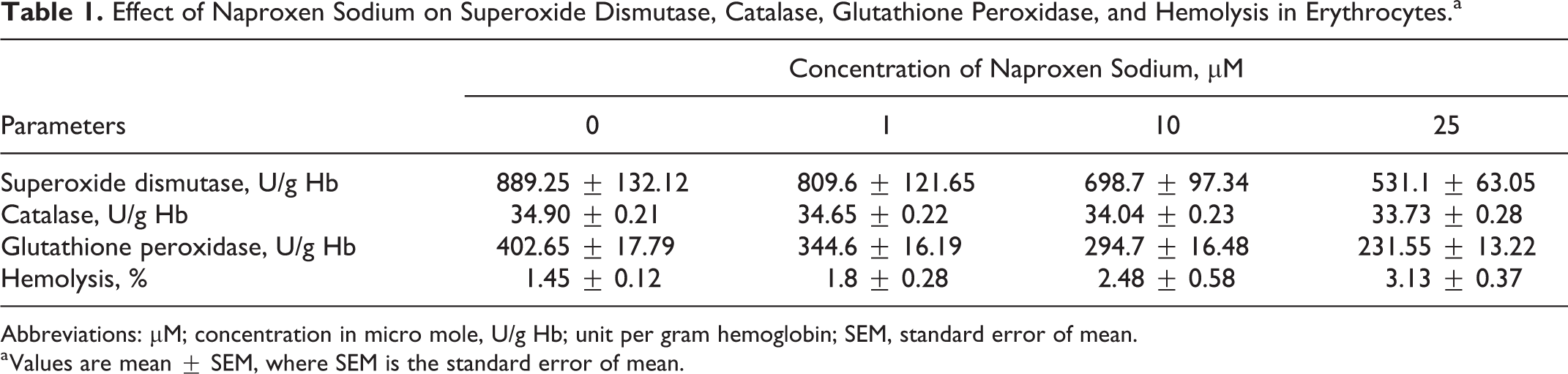
Abbreviations: µM; concentration in micro mole, U/g Hb; unit per gram hemoglobin; SEM, standard error of mean.
a Values are mean ± SEM, where SEM is the standard error of mean.
Figure 1 illustrates that the 48-hour exposure of erythrocytes to naproxen sodium (1-25 µM) resulted in mild but significant decrease in activities of SOD at 25 µM of naproxen sodium with respect to control cells. Figure 2 is showing the significantly moderate reduction in catalase activities after 48-hour exposure of human erythrocytes to 25-µM naproxen sodium in comparison to the untreated cells. Similarly, Figure 3 is depicting highly significant decrease in the activity of glutathione peroxidase in erythrocytes at the exposure of 10- and 25-µM concentration of naproxen sodium.

Effect of naproxen sodium on superoxide dismutase activities (U/g Hb) in erythrocytes. Arithmetic means ± standard error of mean (SEM; n = 20) of the erythrocytes exposed for 48 hours to Ringer solution without (white bar) or with (black bars) 1- to 25-µM naproxen sodium. *P < .05 indicates significant difference from the absence of naproxen sodium (analysis of variance [ANOVA]).

Effect of naproxen sodium on catalase activities (U/g Hb) in erythrocytes. Arithmetic means ± standard error of mean (SEM; n = 20) of the erythrocytes exposed for 48 hours to Ringer solution without (white bar) or with (black bars) 1-25 μM naproxen sodium. **(P < .01) indicate significantly different from the corresponding control value (analysis of variance [ANOVA]).

Effect of naproxen sodium on glutathione peroxidase activities (U/g Hb) in erythrocytes. Arithmetic means ± standard error of mean (SEM; n = 20) of the erythrocytes exposed for 48 hours to Ringer solution without (white bar) or with (black bars) 1-25 μM naproxen sodium. ***(P < .001) indicates significant difference from the absence of naproxen sodium (analysis of variance [ANOVA]).
For the confirmation of eryptotic effects of naproxen sodium, MCV and FSC were measured. The results in Figure 4A demonstrates that the 48-hour exposure of erythrocytes with naproxen sodium (1-25 µM) resulted in apparent increase of erythrocytes MCV, which may be due to membrane blebbing. Additional confirmation of naproxen sodium–induced membrane blebbing was done by FSC measurement of erythrocytes, shown in Figure 4B. Further investigations were done for the confirmation of calcium role in induction of membrane blebbing, so the naproxen sodium–exposed cells were subsequently treated with calcium channel inhibitor. Figure 5 shows the cell size measurement of erythrocytes after 48-hour exposure to naproxen sodium (25 µM) in the absence and presence of calcium channel inhibitor amlodipine. In the presence of 10-µM amlodipine, a significant decrease in cell size in comparison to the cells treated in the absence of amlodipine was observed which might be due to inhibition of calcium entry.

(A) Effect of naproxen sodium on erythrocytes mean cell volume (fL). Arithmetic means ± standard error of mean (SEM; n = 8) of the erythrocytes exposed for 48 hours to Ringer solution without (white bar) or with (black bars) 25 μM naproxen sodium. *** (P value < .0001) considered extremely significant difference from the corresponding control value (t test). (B) The effect of naproxen sodium on erythrocyte forward scatter. Original histogram of the forward scatter of erythrocytes following exposure for 48 hours to Ringer solution with (gray area) and without (black line) of the 25-µM naproxen sodium.

Cell size measurement of naproxen sodium–exposed erythrocytes before and after calcium channel inhibition. Arithmetic means ± standard error of mean (SEM; n = 8) of the erythrocytes exposed for 48 hours to ringer solution without (white bar) or with (black bars) 25-µM naproxen sodium ***(P < .001) shows significant cell size difference from control. ### (P < .001) shows significant difference in cell volume between treated cells in the absence and presence of amlodipine (10 μM) (analysis of variance [ANOVA]).
The necrotic effect of naproxen sodium on erythrocytes was investigated by hemolysis measurement. Significant increase in hemolysis percentage after 48-hour incubation of erythrocytes with naproxen sodium was observed when compared to the percentage of hemolysis of control cells as given in Table 1 and shown in Figure 6.

Effect of naproxen sodium on erythrocytes hemolysis %. Arithmetic means ± standard error of mean (SEM; n = 6) of the erythrocytes exposed for 48 hours to Ringer solution without (white bar) or with (black bars) 25-μM naproxen sodium. *(P < .05) indicates significant difference from the absence of naproxen sodium (analysis of variance [ANOVA]).
Discussion
This study was designed to explore the effect of naproxen sodium on antioxidant’s enzymatic activities, erythrocyte’s size, confirmation of calcium role in the induction of suicidal death of erythrocytes, and hemolytic activity. The naproxen sodium doses used in the study were lower than concentrations used to treat nucleated cells. 17
Antioxidants are the biological substances with capability of scavenging the free radicals such as reactive oxygen species produced during oxidation process that may lead to oxidative stress. 18 -20 Superoxide dismutase catalyzes the dismutation of O2 free radicals, and high ROS production may lead to lowering of SOD level and disturb mitochondrial functions. 21 Catalase is a major antioxidant enzyme that decomposes H2O2 into H2O and O2. 22 Hydrogen peroxide’s accumulation may result in decreased level of catalase, and its overproduction showed a protective effect against oxidants in the cells. 23 Similarly, glutathione peroxidase in mitochondrial cell membrane prevents the accumulation of oxidized lipids and decomposes hydrogen peroxide into water. 24 Our experiments related to enzyme activities clearly indicate the oxidative stress in naproxen sodium–treated cells as SOD, catalase, and glutathione peroxidase activities reduced. Previous studies related to oxidative effects of naproxen sodium depicts that the lowering of the antioxidants enzyme activities due to overproduction of oxidants in the cell is the reflection of oxidation. 25
Erythrocyte membrane blebbing, that is, swelling or protrusions, is a reported marker of eryptosis. 26 Membrane blebbing in the cell is due to the activation of calcium-dependent cysteine endopeptidase calpain, which is involved in the breakdown of the erythrocyte’s cytoskeleton. 27 The happening of oxidation and membrane blebbing in naproxen sodium–treated cells confirming its eryptotic effect.
It is reported that the intracellular Ca+2 has key role in the triggering of oxidative stress-induced eryptosis. 9 For the confirmation of its role in the stimulation of membrane blebbing by naproxen sodium, calcium channel blocker amlodipine (10 µM) was used. Nonselective cation channels are triggered by oxidative stress. 10 Amlodipine nonselectively inhibits cation channels and stops the Ca+2 entry in to the cell. 9 By removing intracellular and extracellular Ca+2, similar effects would be observed in previous studies. 28
Disposing of defective erythrocytes before hemolysis is an important physiological role of eryptosis. 29 Hemoglobin is released through hemolyzed erythrocytes that may be filtered through kidney or precipitate in the lumen of renal tubules. 30 The release of erythrocytes content, especially hemoglobin during hemolysis, resulted in less nitric oxide bioavailability and promotes serious clinical issues, including systemic vasoconstriction, vasomotor instability, and endothelial dysfunction. 31
Conclusion
It is concluded that the used therapeutic doses (10-25 µM) of naproxen sodium may trigger the erythrocyte death rate by increased eryptosis and hemolysis due to the induction of oxidative stress and subsequent calcium influx.
Footnotes
Authors’ Note
Sajida Ilyas and Kashif Jilani contributed equally and thus shares first authorship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
