Abstract
Background:
This study was designed initially to explore the effect of chemoradiotherapy on patients diagnosed with head and neck cancer (HNC) with respect to the alteration of systematic immunity.
Methods:
We did a retrospective study enrolling patients received concurrent chemoradiotherapy (CCRT), with or without induction chemotherapy (IC). Blood tests were performed before IC, before and after CCRT. Flow cytometric analysis and turbidimetric inhibition immunoassay were used for detection.
Results:
A total number of 58 patients were included from April 1, 2018, to March 31, 2019. Levels of immunoglobulins (Ig), including IgA, IgG, and IgM, declined after 2 to 3 cycles of IC and CCRT, respectively. Serum level of total hemolytic complement (CH50) increased (P < .001) after IC, but kept stably post-CCRT. Natural killer (NK) cells decreased (P < .01) after IC and enhanced (P < .001) post-CCRT. The number of CD3+CD4+ T cells got increased (P < .01) after IC and decreased (P < .001) post-CCRT. Consistently, both IC and CCRT induced the increase in CD3+CD8+ T cells significantly (P < .001 vs P < .01).
Conclusion:
Both radiotherapy (RT) and chemotherapy (CT) induced dual effect of immune response. Concurrent chemoradiotherapy created an active immune response based on the effect induced by IC, suggesting that RT exerted a potential function on mobilizing immune system.
Keywords
Introduction
Despite therapeutic advancements, head and neck cancer (HNC) still poses a great threat to human health. 1,2 Global annual incidence of HNC is more than 710 000 cases, 3 accounting for approximate 336 000 cancer-related deaths worldwide each year. As a great part of HNC, head and neck squamous cell carcinoma (HNSCC) remains one of most troublesome common cancer worldwide and a 5-year survival rate is less than 50%. 4 Irradiation as one of main therapeutic tools in treating HNC plays a crucial role in local treatment. 5,6 Recently, immune checkpoint inhibitors (ICIs) have gradually sprung up in tumor treatments and the potential property of irradiation to induce or exaggerate antitumor immunity have attracted increasing attention from researchers. 7 However, the potential mechanism related to the amplifying effect created by the combination of radiotherapy (RT) and immune therapy remains unclear with respect to patients with HNC. Besides, the emergence of individual heterogeneity reminds us of the intervention of biomarkers as an indicator for prediction and prognosis of disease. 8 -10 Thus, we conduct our research to preliminarily explore the change of systematic immunity in patients with HNC upon chemoradiotherapy.
Notably, peripheral blood as an easily attained liquid has turned out to be an useful and practical substance to monitor immune response. 11,12 Consequently, we discover the shift of immune components upon induction chemotherapy (IC) and concurrent chemoradiotherapy (CCRT) in patients diagnosed with HNC, including the altered amount of natural killer (NK) cells, CD3+CD8+ T and CD3+CD4+ T cells, regulatory T cells (Tregs) as well as blood complements and several kinds of immunoglobulins (Ig). The changed pattern of immune system testifies the theory that RT enables to induce both suppressive and stimulative immune response. 13 Albeit adverse response RT does, we convince that RT boosts immune mobilization overall in accordance with the increase in NK cells and CD3+CD8+ T cells in patients’ peripheral blood. Significantly, diverse change of different immune-related components might provide us evidence to try to illustrate some phenomenon and guide us adjusting therapeutic schedules in time. Based on our study, we hope that we can find out an optimal therapeutic plan to benefit patients to the maximization and reduce the side effects unexpected to realize the full play of RT in tumor treatments in the future.
Materials and Methods
Patients
This retrospective study was performed in the Department of Radiation Oncology, Nanfang Hospital, Southern Medical University. All patients provided written informed consent before enrolment. Patients were eligible for enrolment if they met the following criteria: aged from 18 to 70 years; had histologically or cytologically confirmed nasopharyngeal carcinoma (NPC), or squamous cell carcinoma of the oral cavity, oropharynx, hypopharynx, or larynx; never received RT before the first diagnosis or recurrence; males or nonpregnant females; the Karnofsky Performance Scale score of general behavior state was ≥80; and agreed to join the study and signed an informed consent form voluntarily. Major exclusion criteria were an age of more than 70 years or less than 18 years; previous RT for recurrence; a history of malignant tumors, including basal cell carcinoma or squamous cell carcinoma and primary cervical carcinoma with adequate treatment; complication with immune disease; a long-term use of corticosteroids or other immunosuppressive drugs (within 1 year); and disruption of other serious disease that might pose a high risk or affect the compliance of the test. Case shedding standards included situations that patients with poor compliance affected the judgment of the results, patients went through severe adverse changes as well as patients quitted automatically.
Patients’ Treatment
The tumor target volume (gross tumor volume [GTV]) was the volume of tumor tissue visible on imaging (such as tumor-enhanced computed tomography or enhanced magnetic resonance imaging) and endoscopy (such as fiberoptic nasopharyngoscope or fiberoptic laryngoscope), including primary tumor (GTVp) and cervical metastatic lymph node (GTVnd). The clinical target volume (clinical target volume [CTV]) CTV1 was a high-risk area, CTV2 was a preventive exposure area. Both GTVp and GTVnd extended 3 mm as planned target volume (planning target volume [PTV]) PTVp and PTVnd, CTV1 and CTV2 expanded 3 mm to PTV1 and PTV2, respectively. All patients were treated with intensity-modulated radiation therapy with a total dose of 60 to 70 Gy for PTVp and PTVnd, 64 Gy for PTV1, and 54 Gy for PTV2, 5 times a week for 30 to 33 times in all. Docetaxel with carboplatin/paclitaxel with cisplatin/paclitaxel with carboplatin/docetaxel with cisplatin regiments were used for IC for 2 to 3 cycles within 1 to 2 months before RT. Concurrent chemotherapy consisted of cisplatin (100 mg/m2) given in weeks 1, 4, and 7 during RT, or carboplatin (AUC 5) given in days 1, 22 and 43 during RT.
Preparation and Detection of Peripheral Blood Samples
Five milliliters of venous blood samples was taken from each patient with HNC before and/or after CCRT or IC within 1 to 3 days, respectively. Turbidimetric inhibition immunoassay was used to detect the level of immunoglobulins including IgG, IgA, and IgM as well as the activity of complement 3 (C3), C4, and CH50. The subsets of immune cells including CD3+CD4+, CD3+CD8+, CD4+CD45RO+, CD4+CD45RA+ T cells, and NK cells as well as Tregs in peripheral blood collected were detected by flow cytometry using fluorescence-labeled antihuman antibodies (Millipore, Massachusetts, USA). Ethylenediaminetetraacetic acid blood was used for flow cytometric analysis immediately, and the serum was stored at −80°C for use. All detections were performed within 7 days before and/or after CCRT or IC, respectively.
Statistical Analysis
The data were processed by IBM SPSS version 20.0 statistical software. All data were presented as mean ± standard error (x ± s), and two-tail unpaired Student’s t-test was used for analysis. A P value of .05 was considered statistically significant (*P < .05, **P < .01, ***P < .001). The significance level was α = 5%. Charts were made using GraphPad Prism version 7.0 software.
Results
The Shift of Systematic Immunity Reflected in Peripheral Blood From Patients With HNC Upon CCRT
A total number of 58 patients had accepted local RT concurrent with chemotherapy. Demographics and clinical characteristics of patients involved in this study were summarized in Table 1. Albeit the level of C4 got enhanced, there was no significant change regarding to the activity of C3 and CH50. Meanwhile, the level of immunoglobulins including IgA, IgG, and IgM declined. Although the number of CD3+CD4+ T cells tended to decrease, cytotoxic CD3+CD8+ T cells as well as NK cells augmented significantly at the same time. Additionally, the proportion of CD4+CD45RO+ T cells kept stable and Tregs increased lightly. And the number of naive CD4+CD45RA+ T cells was weakened simultaneously (Figure 1A-L).
Demographics and Clinical Characteristics of Patients.

Abbreviations: CCRT, concurrent chemoradiotherapy; IC, induction chemotherapy.
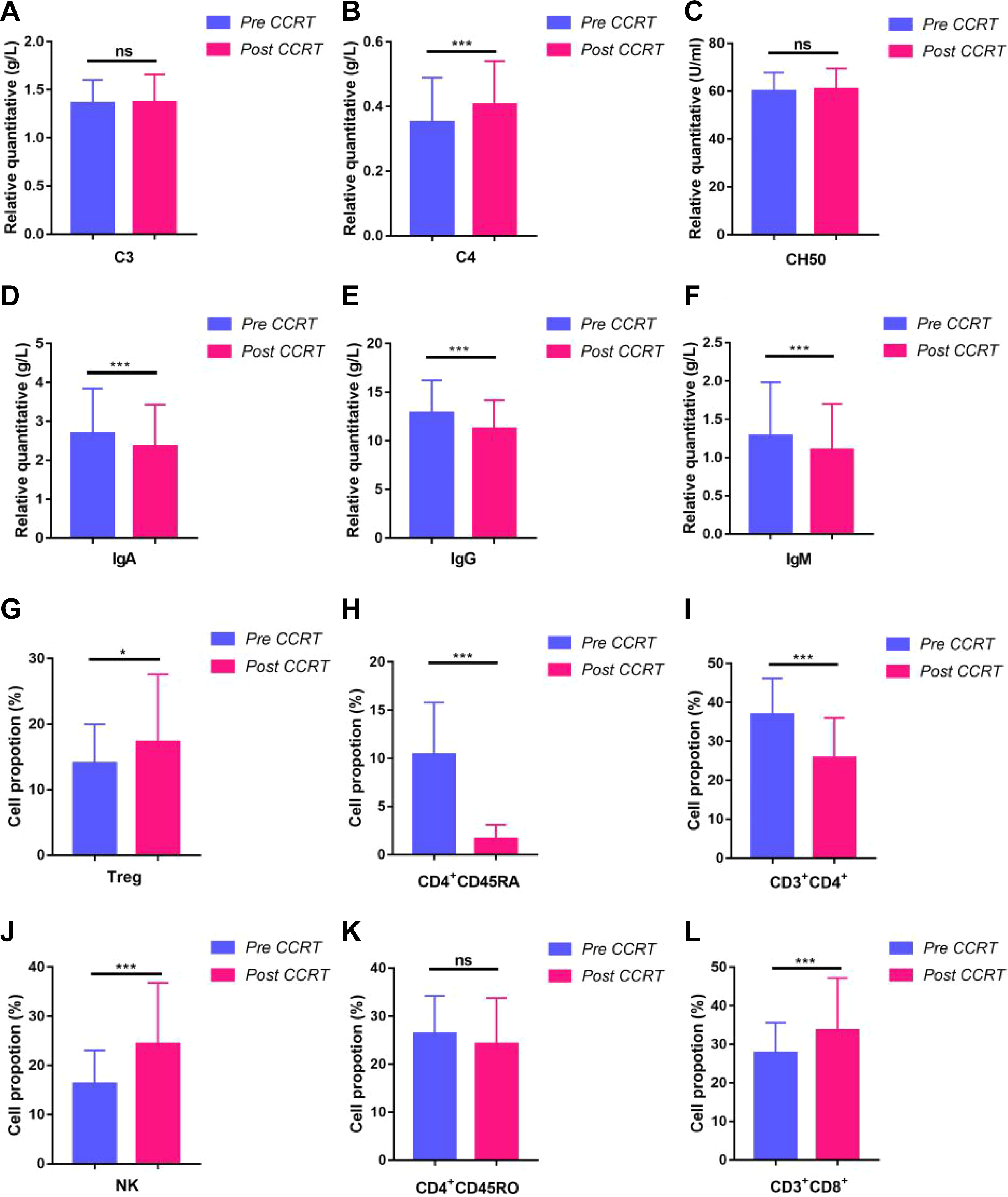
The altered profile of systematic immunity in peripheral blood in patients with HNC upon CCRT.*P < .05, **P < .01, and ***P < .001. CCRT indicates concurrent chemoradiotherapy; HNC, head and neck cancer.
Comparison Between IC and CCRT in Terms of the Effect on Systematic Immunity
Aside from the simple analysis of the altered systematic immunity upon CCRT, we also conducted horizontal comparison on patients treated with IC and CCRT with 3 blood examinations individually. In brief, patients got 3 blood detection prior to IC and CCRT secondarily, and post-CCRT for the last time. Therefore, we could evaluate the immune effects created by RT approximately. Twenty-four persons were involved. At first, the level of immunoglobulins including IgG, IgA, and IgM reduced significantly regardless of treatments (Figure 2D-F). However, altered levels of C3, C4, and CH50 reflecting inflammatory response partially indicated that IC induced an intensive reaction while CT plus RT failed to boost the further improvement of C3 and CH50 on the whole (Figure 2A-C). Furthermore, it’s encouraging to find out that CCRT tended to stimulate immune system actively according to the indicated shifts of immune-associated cells (Figure 2G-L). The number of CD3+CD4+ T cells decreased upon CCRT, yet the proportion of CD3+CD8+ T cells got a great promotion following combined therapies compared to CT alone. Besides, the number of NK cells also elevated greatly after CCRT while it turned to decrease upon IC. Notwithstanding the proportion of CD4+CD45RO+ T cells elevated especially following IC, the percentage of CD4+CD45RA+ T cells increased significantly while it declined in patients receiving CCRT treatment.
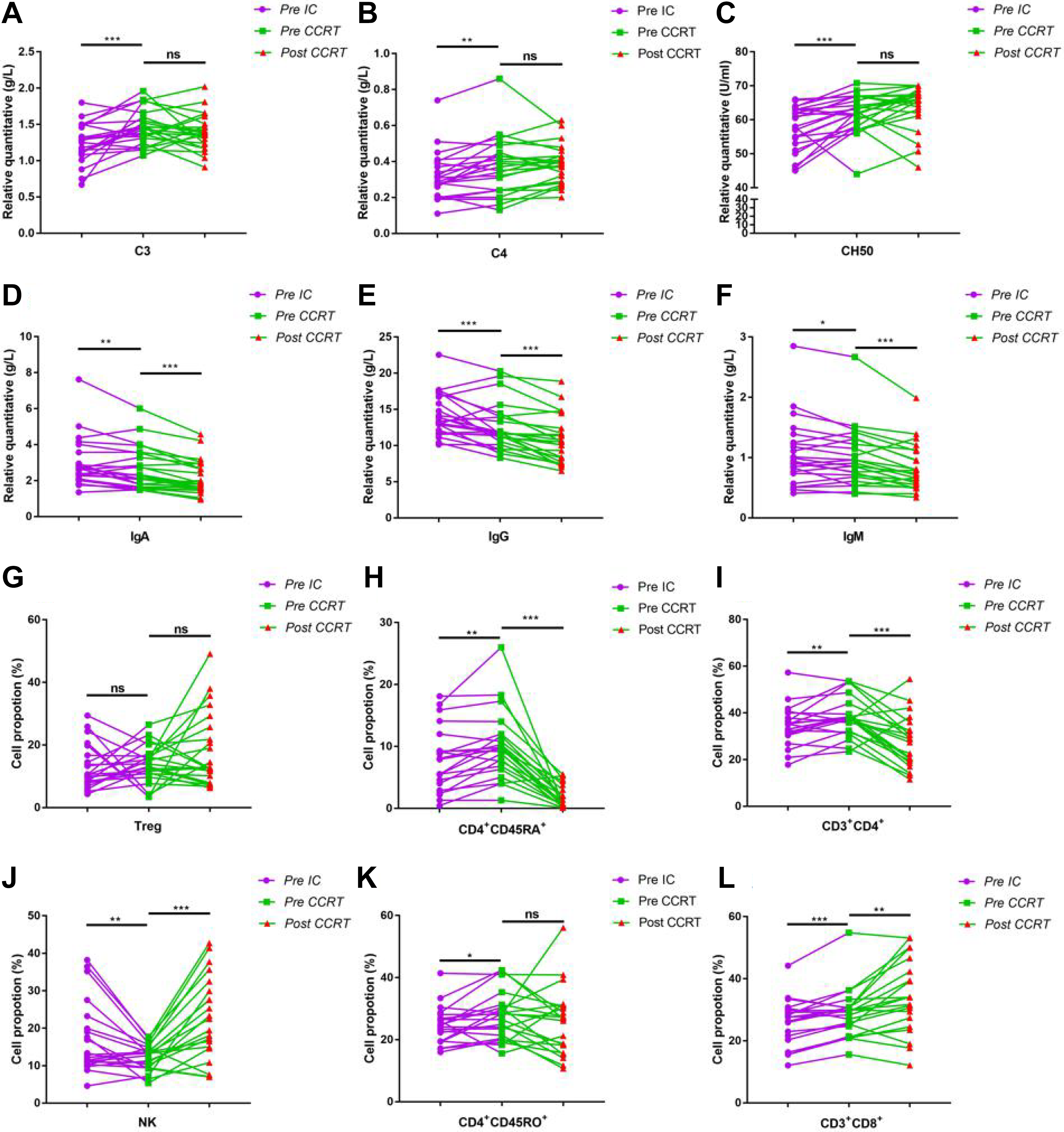
Comparison of altered systematic immunity in peripheral blood in patients with HNC between IC and CCRT. *P < .05, **P < .01, and ***P < .001. CCRT indicates concurrent chemoradiotherapy; HNC, head and neck cancer; IC, induction chemotherapy.
Discussion
As reported, most of HNC are squamous cell carcinomas, 1 and NPC occurs with high incidence particularly in Southern China. 14 Despite of steady progress and technical promotion achieved in irradiated treatment, the 5-year survival rate has subtly increased in patients with advanced HNSCC. 15 According to newly published global cancer statistic in the year of 2018, HNC led to more than 336 000 cancer-related deaths worldwide per year. Fortunately, increasing experimental data from multiple cancer models have suggested that ionizing radiation was the primary source to amplify the antitumor immune responses. 16 For one thing, RT is considered to induce suppressive immune response; reasons for this are varied, including the decreased expression of co-stimulatory markers CD80 and CD86 on immature Dendritic cells (DCs), 17 the induction of indicated passive chemokines and cytokines 18 -22 as well as the increased expression of programmed cell death 1 (PD-1) and its ligand PD-L1 on relative cells after radiation. 23 In addition, the destruction of tumor vascular and the later recruitment or local differentiation of M2-like macrophages to tumor bed resulting from hypoxia, 24,25 and the recruitment of myeloid-derived suppressor cells (MDSCs) into tumor surroundings were observed postradiation treatment. 26 On the other hand, accumulating evidence has suggested that ionizing radiation is able to create active immune response. Consistently, multiple factors are involved in this process, such as the secretion of type I interferon (IFN) that enhances the immune function of DCs, 27,28 the upregulated expression of major histocompatibility complex I class molecules on tumor cells, 29,30 and the release of damage-associated molecular pattern that promotes the maturation and activation of DCs upon ionizing radiation. 31,32 Furthermore, the activation of innate immune system including the intensive cytotoxic and elevated secretion of IFN-γ by NK cells, 33,34 as well as the increased tumor-infiltrating cytotoxic T cells along with lower intratumoral levels of MDSCs were noticed. 35 Moreover, the reconstitution of tumor vascular to improve T cells trafficking in and out of tumor bed 36,37 and the reduction of Tregs 38 -40 together with the polarization of tumor-associated macrophage toward type I 41 upon radiation were demonstrated. Likewise, conventional CT also boosts the activation of immune system via the attenuation of Tregs, restoration of T and NK cells effector function, and so on. 42 -44
Recently, as immunotherapy has been implemented in cancer treatment with impressive successes, immune monitoring is gradually of great importance. Compared to detecting tissues, peripheral blood as one body fluid has turned out to be a practical and convenient medium that very well integrates the significance of systemic informative value and is easy to acquire. 45,46 Excitingly, the correlation between peripheral blood cell populations and clinical benefit was reported. 11,47 For instance, high numbers of CD8+ T cells correlated with better clinical benefit. 11,48 Hence, it’s promising to detect peripheral blood alteration to attain useful information for treatments.
As demonstrated, our study suggests that both CT and RT enabled to affect systematic immunity, which is consistent with previous research. 13 Moreover, we find out that patients diagnosed with HNC had an active immune response following proper treatment, especially CCRT with an accumulating number of 30 to 33 times in normal 2 Gy fraction. Compared with the decreased tendency upon single IC treatment, the proportion of NK cells attained an accelerated promotion after CCRT. In spite of the decline in CD3+CD4+ T cells, the number of CD3+CD8+ T cells and CD4+CD45RA+ T cells got greatly enhanced following CCRT in turn, suggesting that RT exactly had the potential to stimulate immune system and offset the passive effects caused by CT alone. Regardless of results mentioned above, the stable level of CH50 proved that RT did not enhance the side effects similar to that resulting from CT alone. Overall, our study provided evidence to support the fact that CCRT enabled to create an active immune response based on single CT treatment, emphasizing the importance of RT in mobilizing immune system and coordinating immune reaction.
So far, it’s well recognized that whether RT plays an active or passive role during the process of treatment depends largely on the given does, the model of fraction as well as the intrinsic character of indicated tumor. For instance, it’s observed that mice received a hypofractionated regimen (8 Gy × 3) revealed an abscopal effect, which was not observed in the group treated with a single dose of 20 Gy. 49 Moreover, a single RT dose of 15 Gy promoted Tregs infiltration, of which the potentially passive effect was attenuated when the same dose was given in 2 doses of 7.5 Gy. 50 Furthermore, diverse tumor perhaps require different patterns of radiation to induce active immune response. Based on previous evidence, mouse 4T1 breast carcinomas model exhibited increased infiltration of CD8+ T cells upon radiation with 2 fractions of 12 Gy in the context of CTLA4 blockade. 51 However, a similar infiltration by CD8+ T cells along with a reduced intratumoral level of MDSCs was observed in mice-bearing colorectal carcinomas treated with a single RT dose of 30 Gy. 35
Remarkably, it’s reported that low-dose RT enabled to attenuate inflammatory reaction, 52 which seems consistent with our result that 2 doses RT plus CT failed to promote the level of CH50 compared to pure CT treatment to some extent. Based on the phenomenon especially that CD3+CD8+ T cells and NK cells got increased, it seems that CCRT improves the immune response further more than IC. However, we cannot ignore the fact that the proportion of CD3+CD4+ T cells turned to reduce greatly upon CCRT, indicating that the dose or fraction of RT and some details remained to be improved to minimize the unexpected results without affecting the beneficial outcome.
In brief, we deeply convince that RT has a great potential to stimulate immune system to execute antitumor function than its passive immune response overall. Meanwhile, it seems that CCRT amplified the therapeutic response than IC alone. Given the facts that previous evidence originated most from animal models or preclinical tests and most of clinical trials and researches are underway, 53 additional studies are needed to systematically investigate the mechanism how diverse exposed doses and schedules determine the outcome of RT-induced immune response. Recently, it’s demonstrated that the change of functional immune T cells within tumor predicting clinical benefit was consistent with corresponding alteration in peripheral blood, 11 suggesting the promising use of blood examinations as an immune monitoring tool to predict prognosis in the future. To date, no research is conducted in terms of the altered immune system upon RT in patients with HNC, which provides us another evidence to support the view that RT boosts the mobilization of systemic immune. Notably, as a large number of clinical trial grow regarding the joint action of RT plus ICIs, 54 it will further illustrate the possibility to use RT as an immunostimulant in cancer treatments in the future.
Footnotes
Authors’ Note
Weiqiang Huang and Yao Fan contributed equally to this study.
Acknowledgments
The authors thank Tianjin Key Laboratory of Radiation Medicine and Molecular Nuclear Medicine, Tianjin, China, for providing experimental platform.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (nos. 81602685, 81672992); Clinical Research Startup Program of Southern Medical University by High-level University Construction Funding of Guangdong Provincial Department of Education (LC2019ZD008); Clinical Research Startup Program of Southern Medical University by High-level University Construction Funding of Guangdong Provincial Department of Education (2018CR021); Health & Medical Collaborative Innovation Project of Guangzhou City, China (201803040003); the Natural Science Foundation of Guangdong Province (no. 2017A030313486); Guangzhou Science and Technology Plan Project (201707010025); and CAMS Innovation Fund for Medical Sciences (CIFMS, 2016-I2M-1-017, and 2017-I2M-B&R-13).
