Abstract
1,3-Dinitrobenzene (mDNB) is a widely used intermediate in commercial products and causes testicular injury. However, genotoxic effects upon low-level exposure are poorly understood. The present study evaluated the effects of very low-chronic doses of mDNB on sperm nuclear integrity. Male hamsters were treated with 1.5 mg/kg/d/4 wks (group A), 1.5 mg/kg/mDNB/d/week/4 weeks (group B), 1.0 mg/kg/mDNB/3 d/wk/4 wks (group C), or polyethylene glycol 600 (control). Nuclear integrity of distal cauda epididymal sperm was determined using the sperm chromatin structure assay and acridine orange staining (AOS). The germ cell nuclear integrity was assessed by the comet assay. Testicular histopathology was conducted to evaluate the sensitive stages. The comet assay revealed denatured nuclear DNA in group A (in diploid and polyploid cells from weeks 2-5); respectively at week 4 and weeks 3 to 4 in groups B and C. According to AOS, only group A animals exhibited denatured sperm DNA (weeks 1 and 3). The effective sperm count declined from weeks 1 to 6. Mean sperm DNA denaturation extent, percentage cells outside the main population, and standard deviation indicated altered sperm nuclear integrity in group A. Same animals exhibited progressive disruption of the Sertoli cells, while groups B and C exhibited damages on germ cells. The results suggest that mDNB affects sperm nuclear integrity at very low chronic doses targeting cell-specific testicular damage.
Keywords
Introduction
1,3-Dinitrobenzene (mDNB) is a common isomer of benzene and is a key intermediate used in the synthesis of industrial and commercial products including rubber, chemicals, pesticides, dyes, explosives, and plastics industries. 1 The US production of meta-dinitrobenzene has increased from 73 000 metric tons in 1960 to 1 390 000 metric tons by 2007. 2 Occupational exposure to nitrobenzene occurs via inhalation of the vapor or dermal contact with liquid or vapor. National exposure survey conducted by Fonger 3 estimated that about 5080 workers including 475 women were potentially exposed to nitrobenzene. Exposure to nitrobenzene is not limited to occupational exposure as the chemical was detected in air, hazardous waste sites, and in ground water in many parts of US cities. 3 Populations who live or work near weapon ammunition or manufacture plants get exposed to nitrobenzene. Occupational exposure and exposure by drinking contaminated water and food result in liver damage and blood disorders. 4
Although nitrobenzene is important for the chemical industry, its potential environmental and health risks are evident as a result of their abundant use. 1,3-Dinitrobenzene is a multitarget toxicant that affects both the nervous and reproductive systems. 5 Continuous exposure to mDNB causes neuropathology in the central nervous system, liver damage, platelet aggregation, and methemoglobinemia in humans. 6
A common effect of chronic exposure to mDNB is that it can inhibit metabolism via inhibition of pyruvate dehydrogenase complex. 7 Similarly, isomers of mDNB, 1,4-dinitrobenzne, were able to induce oxidative damages in the epididymis and testis leading to reproductive deficits. 6
Many alkylating agents can cause sterility in males by suppressing spermatogenesis and normal output, morphology, counts, and motility of spermatozoa. 8 These effects may occur due to abnormal structural changes in sperm nuclei. 9 Studies have shown that 1,3-DNB can impair fertility and sperm motility, 10 decrease synthesis in steroid hormone, and interfere with the cell cycle to inducing apoptosis. 11 This includes the spermatocyte cell population 12 in experimental animals. Rats exposed to high dose levels of mDNB exhibited altered pachytene spermatocyte-specific genes leading to decrease in the number of spermatocytes. 13
Since mDNB is frequently utilized as an intermediate chemical in many commercial products, inevitably mDNB can escape into the workplace or more insidiously into the general environment causing threat to male fertility. 14 Although it has been clearly demonstrated that a number of mDNB can adversely affect the male reproductive system of experimental animals, its genotoxic effects remain unclear at low dose levels and therefore in light of this, the present study investigated the effects of 3 low doses of reproductive toxicant, mDNB on hamster germ cell nuclear integrity using comet assay, acridine orange (AO) staining, and sperm chromatin structure assay.
Materials and Methods
Chemicals and Reagents
1,3-Dinitrobenzene, >99% pure; solutions of mDNB were prepared in 60% polyethylene glycol 600 (PEG 600). Test solutions were prepared just prior to use.
Treatment of Hamsters
Male hamsters (weight 30-40 g) were housed in an animal house (temperature: 24°C-25°C, photoperiod: 12 hours of day and 12 hours of dark and relative humidity: 55%-60%) with free access to water. The animal handling was in accordance with the institutional guidelines for the care and use of laboratory animals, (ACT, 1986), University of Sheffield, United Kingdom. Previously, we showed that low doses of mDNB led to marked reduction in sperm counts and severe decline in sperm fertilizing ability 10 and therefore, same doses were employed in the present study.
Group A, male hamsters (n = 6) were treated with 1 mL of 1.5 mg/kg mDNB/d for 4 weeks and animals were killed weekly following the final dose up to 6 weeks.
Group B, males (n = 6) were treated with 1 mL of 1.5 mg/kg mDNB/every Wednesday (d/wk) for 4 weeks. Based on previous results of fertility, 10 animals were killed on the third- and fourth-week posttreatment.
Group C, males (n = 6) were given 1 mL of 1.0 mg/kg mDNB every Monday, Wednesday, and Friday (3 d/wk) for 4 weeks. The animals were killed on the third and the fourth week following treatment.
In all instances, the control animals (n = 6/group) were gavaged at the same time with 60% PEG.
Germ Cell Preparation
The male hamsters were killed at different times after treatment by sodium pentobarbitone (J.M. Loveridge, Southampton, United Kingdom) given intraperitoneally. The left testis of each animal was carefully removed and washed with phosphate buffer saline (PBS) without Ca2+ and Mg2+ (GIBCO, Loughborough, UK). The testicular capsule was opened and the seminiferous tubules extracted in PBS and allowed to settle between 3 washings. After the final wash, tubules were covered with 20 mL of collagenase solution (1 mg/mL in PBS) and kept at 37°C. This tubule suspension was shaken for 15 to 20 minutes.
Comet Assay
The basic alkaline technique described by Singh et al 15 was used. A sandwich preparation was made using the resulting cell suspension. A sandwich preparation was made and lysed using cold lysing solution at 4°C for 30 minutes. Subsequently, the slides were subjected to electrophoresis. All of these steps were carried out in dim light to prevent additional DNA damages. 16 Upon neutralization, the slides were stained with ethidium bromide and observed under an epifluorescence microscope (BH-2; Olympus Ltd, Tokyo, Japan) equipped with a barrier filter of 590 nm. Photographed images were enlarged up to ×500 since the bigger the tail and head the more accurately the measurements could be made. Comet length (cm) and head diameters (cm) in a minimum of 25 randomly selected cells per slide were determined. Comets spaced too closely were avoided.
Acridine Orange Staining
Upon killing the animals at various times described in Materials and Methods section, the right distal cauda epididymides were taken to obtain sperm for DNA assessment. The method described by Liu and Baker 17 was adopted in the present study. Briefly, sperm smears were prepared on precleaned microscopic slides (Chance Propper Ltd, Warley, England) and stained with AO staining solution for 5 minutes. The percentage of sperm with normal DNA was determined by counting 500 sperms from each part of the cauda epididymis of each hamster under epifluorescence microscopy (BH-2, Olympus Ltd) with excitation of 490 nm.
Calculation of Effective Sperm Count
The total number of sperm in the distal cauda epididymis was recovered by carefully mincing the tissue to release all cells. The sperm count was determined using the improved Neubauer hemocytometer. Effective sperm count was calculated by multiplying the percentage of green fluorescence after AO staining by the total sperm count of the distal cauda epididymis.
Sperm Chromatin Structure Assay
The left distal cauda epididymides from each animal were removed, pooled, and placed in TNE buffer. The cell suspension was drawn and filtered through 160 µm pore size and 25 mm diameter nylon mesh (Millipore Corporation, Bedford, Massachusetts) and samples were sonicated with a Soniprep Sonicator 150 (Measuring Scientific Instruments Ltd, London, United Kingdom) for 30 seconds. Since the sonication step raises the temperature, the solution was cooled in an ice bath and sonicated for an additional 30 seconds. The resulting sonicate was centrifuged at 3000 rpm (GS-6R Beckman Centrifuge, Brea, California, USA) for 3 to 5 minutes and resuspended in TNE buffer to approximately 107 sperm heads/mL and held in ice. About 0.2 mL of sonicated suspension was mixed with 0.4 mL of chilled acid treatment solution and the cells stained with AO solution. The flow cytometry analysis was begun exactly 3 minutes after initiation of the procedure. The cells with intact DNA was sorted using a Coulter epics elite cell sorter (Beckman Coulter Ltd, Hialeah, Florida) equipped with ultra-sense optics and Lexel 100 mW (488 nm) for 30 minutes. 18 Approximately 5000 cells were measured at a rate of approximately 200 to 300 cells/sec. The instrument was validated prior to use.
In the SCSA, the extent of DNA denaturation (α1) was calculated from each sperm using List View Software (Phoenix Flow Systems, San Diego, CA, USA) and results are expressed as mean αt (Xαt), standard deviation of αt (SDαt), coefficient of variance of αt (CVαt), and percentage of cells outside the main population of αt (COMPαt).
Histopathology
Upon killing, testes of the males from groups A and control were excised on weeks 3 and 4, washed, and blotted and fixed in Bouin fixative. After the routine processing of tissues, 4 µm thick sections were made via microtomy and stained with hematoxylin and eosin for microscopic examination. 19
Statistical Analysis
Minitab software (Minitab Xtra Inc, State College, PA) was used for statistical analysis. An α level of .05 was used to determine significant levels in all the statistical analysis. For the AO staining, the results from each animal were pooled to increase the replicate number to 6 per group. Two-way analysis of variance was performed; and if there was a significant interaction between weeks and treatment, either unpaired t test or Turkey test was carried out. To establish correlation between fertility and green AO fluorescence or effective sperm counts, Pearson correlation coefficient test was carried out.
Results
Comet Assay
Group A (1.5 mg/kg mDNB/daily for 4 weeks)
The mean Comet tail length for control and test animals in group A is exhibited in Figure 1A. At this dose level, there was significant DNA damage from weeks 2 to 5. The highest DNA damage (as determined by Comet tail length and head diameter) was observed after 3 and 4 weeks of exposure (P < .001) when compared to the mean values of all weeks. DNA damage was increased from weeks 1 to 3. After 3 and 4 weeks of exposure, it reached a nadir followed by a gradual decrease. At weeks 1 and 5 of exposure, there was no significant damage compared to their controls.

Line plots are showing the mean tail length (A) and mean head diameter (B) in germ cells of hamsters over a period of 6 weeks. Germ cells were taken from hamsters treated with either 1.5 mg/kg/mDNB/daily or PEG for 4 weeks. Error bars represent standard errors of mean values. *P < .05, **P < .01 and ***P < .001. mDNB, indicates 1,3-dinitrobenzene; PEG, polyethylene glycol.
The head diameter of the Comet was measured across the nuclear core (Figure 1B). In group A (1.5 mg/kg mDNB/daily), there is a significant stretching of the DNA from the core of the head of cells from treated males from weeks 2 to 6 (weeks 2, 5, 6; P < .05 and weeks 3, 4; P < .001). The effect is highly significant in weeks 3 and 4 when compared to mean values of each weeks (P < .01).
Group B (1.5 mg/kg mDNB/daily for 4 weeks)
The mean Comet tail length and head diameter of animals in group B (1.5 mg/kg/d) depicted a significant increase in DNA migration in week 4 (P < .001) but not at week 3 (Figure 2).

Bar charts of DNA migration patterns in individual cells of 6 animals from both controls (PEG) and treatment (1.5 mg/kg mDNB/d/wk for 4 weeks) under denaturation conditions in an electric field. Both the mean tail length (A) and mean head diameter (B) were measured. Means are presented with the standard error. **P < .01 and ***P < .001. mDNB indicates 1,3-dinitrobenzene; PEG, polyethylene glycol.
Group C (1.0 mg/kg mDNB/3 days per week)
In group C (1.0 mg/kg mDNB/3 d), the stretching of DNA from the nuclear core was notably increased (P < .001 and P < .01) at both 3 and 4 weeks of exposure compared to the control (Figure 3).

The levels of DNA strand breaks determined by Comet assay in germ cells of hamsters given 1.0 mg/kg mDNB/3 d/wk or PEG for 4 weeks. DNA damages were measured in terms of mean tail length (A) and mean head diameter (B). Error bars represent standard errors of mean values. **P < .01 and ***P < .001. mDNB indicates 1,3-dinitrobenzene; PEG, polyethylene glycol.
Effects on Nuclear Integrity
All the regions of sperm DNA was denatured in group A test dose level. The sperm nuclear DNA of distal cauda showed denaturation at weeks 1 (P < .01) and 3 (P < .01). The results are summarized in Table 1.
Sperms Were Stained With Acridine Orange in Hamsters Which Were Treated With PEG or 1.5 mg/kg of mDNB/Daily for 4 Weeks.a
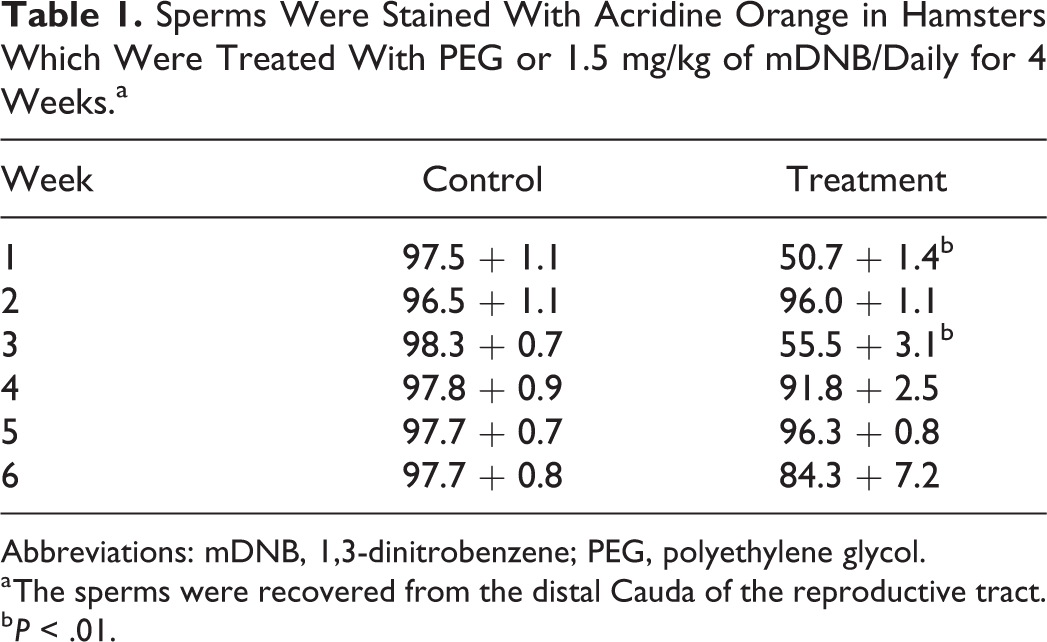
Abbreviations: mDNB, 1,3-dinitrobenzene; PEG, polyethylene glycol.
a The sperms were recovered from the distal Cauda of the reproductive tract.
b P < .01.
Both 1.5 mg/kg mDNB/d/wk for 4 weeks (group B) and 1.0 mg/ kg mDNB/3 d/wk for 4 weeks (group C) had no significant effects on sperm nuclear integrity. In fact, all the sperm samples exhibited a higher percentage of green AO fluorescence (Table 2).
Sperms Were Taken From the Hamsters Treated With 0, 1.5 mg mDNB/kg/d/wk/4wks (Group B), and 1.0 mg mDNB/kg/3 d/wk/4wks (Group C).a
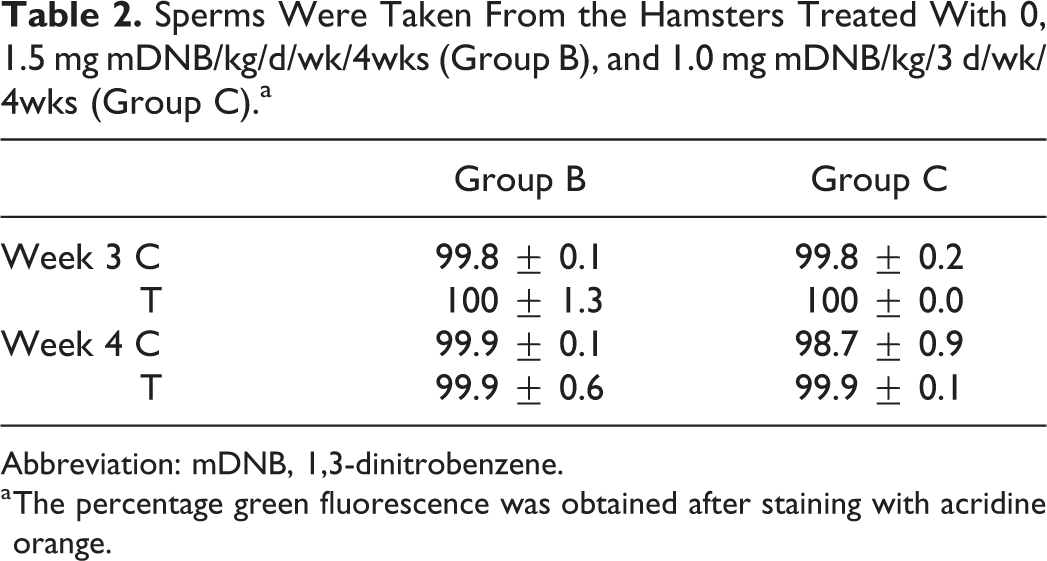
Abbreviation: mDNB, 1,3-dinitrobenzene.
a The percentage green fluorescence was obtained after staining with acridine orange.
Effective Sperm Count
In group A (1.5 mg/kg mDNB/daily), the effective sperm counts were reduced throughout the treatment period. However, the effects were highly marked in 3 and 4 weeks after exposure by reaching the effective sperm count to zero. The results are plotted in Figure 4.

The effective sperm counts of hamsters treated with 1.5 mg/kg of mDNB/daily for 4 weeks. The control animals were given PEG only. The means are presented with the standard error. *P < .05 and **P < .01. mDNB indicates 1,3-dinitrobenzene; PEG, polyethylene glycol.
At the 2 lower chronic doses of mDNB (groups B and C), no significant effects on effective sperm count at any point were observed.
Sperm Chromatin Structure Assay
In group A, mDNB significantly affected sperm chromatin structure in males at weeks 2 to 6 (Figure 5A–D). Xαt increased significantly at weeks 3 and 4 (P < .05 and P < .001 respectively), with a maximum increase occurring at week 4. The SDαt values are significantly different from controls from weeks 3 to 5 (P < .001, P < .001, P < .01, and P < .05 respectively) with a gradual recovery (Figure 6A). Increased COMPαt is significant from weeks 2 to 5 (P < .05 at week 2; P < .001 at weeks 3 and 4 and P < .01 at week 5). The CVαt showed a significant increase from weeks 2 to 6 (weeks 1, 5, 6; P < .001, week 3; P < .01 and week 4; P < .05). The greater response for Xαt, SDαt, and COMPαt occurred at weeks 3 and 4.

Comparison of data (mean ± SE) derived by measurements made with the flow cytometry on distal cauda (A-D) epididymal sperm. SCSA data include SDαt, COMP αt, Xαt, and CVαt. Sperm from 6 hamsters were used at each time point. *P < .05, **P < .01, and ***P < .001. COMP αt indicates cells outside the main population of αt; CVαt, coefficient of variance of αt; SDαt, standard deviation of αt.
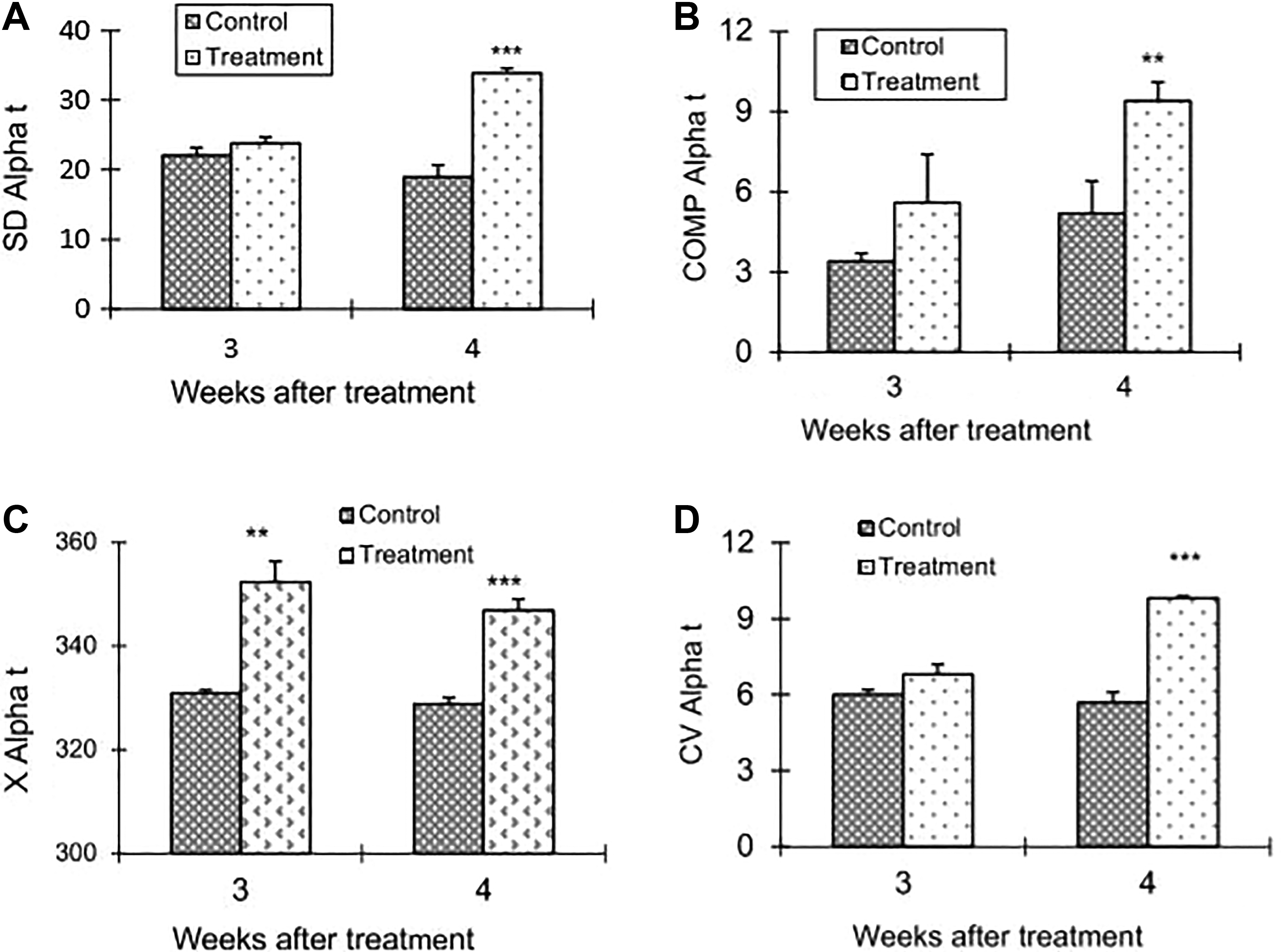
The changes in flow cytometry parameters of the sperm chromatin structure from group B (1.5 mg mDNB/kg/d/wk) males. The values of COMPαt, CVαt, and SDαt showed a significant (all P < .001) increase over the control only at week 4. In contrast, Xαt showed a significant difference from that of the control at both weeks 3 (P < .01) and 4 (P < .0001). mDNB indicates 1,3-dinitrobenzene. COMP αt indicates cells outside the main population of αt; CVαt, coefficient of variance of αt; SDαt, standard deviation of αt; Xαt, mean αt.
Figure 6A–D displays the changes in flow cytometry parameters of the sperm chromatin structure of group B. The values of COMPαt, CVαt, and SDαt showed a significant (all P < .001) increase over the control only at week 4. In contrast, Xαt showed a significant different from that of the control at both week 3 (P < .01) and 4 (P < .0001).
In group C, Xαt exhibited a significant (P < .05) increase in mDNB-treated males at both weeks. Similarly COMPαt values also differed significantly (P < .01) from the controls at both weeks. Both SDαt and CVαt showed a maximum increase at week 3 (P < .001) and a slight increase at week 4 (P < .05). Histograms are given in Figure 7A–D.
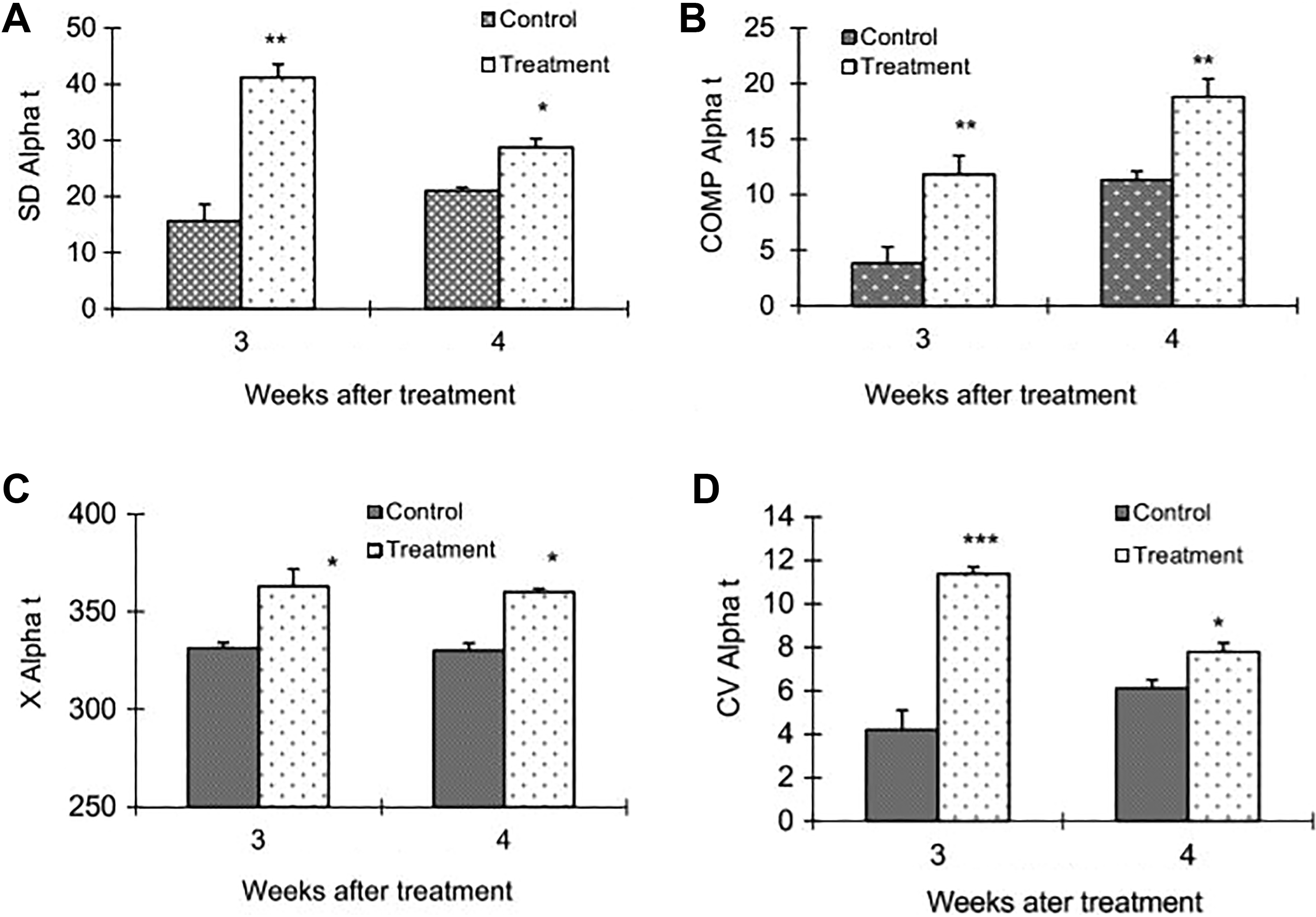
SDαt, COMPαt, Xαt, and CVαt of distal cauda (A-D) epididymal sperm at weeks 3 and 4 from hamsters exposed either to 0 or 1.0 mg mDNB/kg/3 d/wk for 4 weeks. *P < .05, **P < .01, and ***P < .001. COMP αt indicates cells outside the main population of αt; CVαt, coefficient of variance of αt; mDNB, 1,3-dinitrobenzene; SDαt, standard deviation of αt; Xαt, mean αt.
Histopathology
Testes sections from control males displayed consistently good histological preservation indicating that the fixation method and tissue processing were optimal. Hence, any changes in testis histology represented the effects of treatment and not artefacts of histological processing. A series of representative micrographs are shown in Figure 8A–D with increasing order of magnification. The germinal epithelium is well preserved with all the cell types present. There are no vacuoles in the epithelium and the lumens of the tubules do not contain sloughed immature cell types.
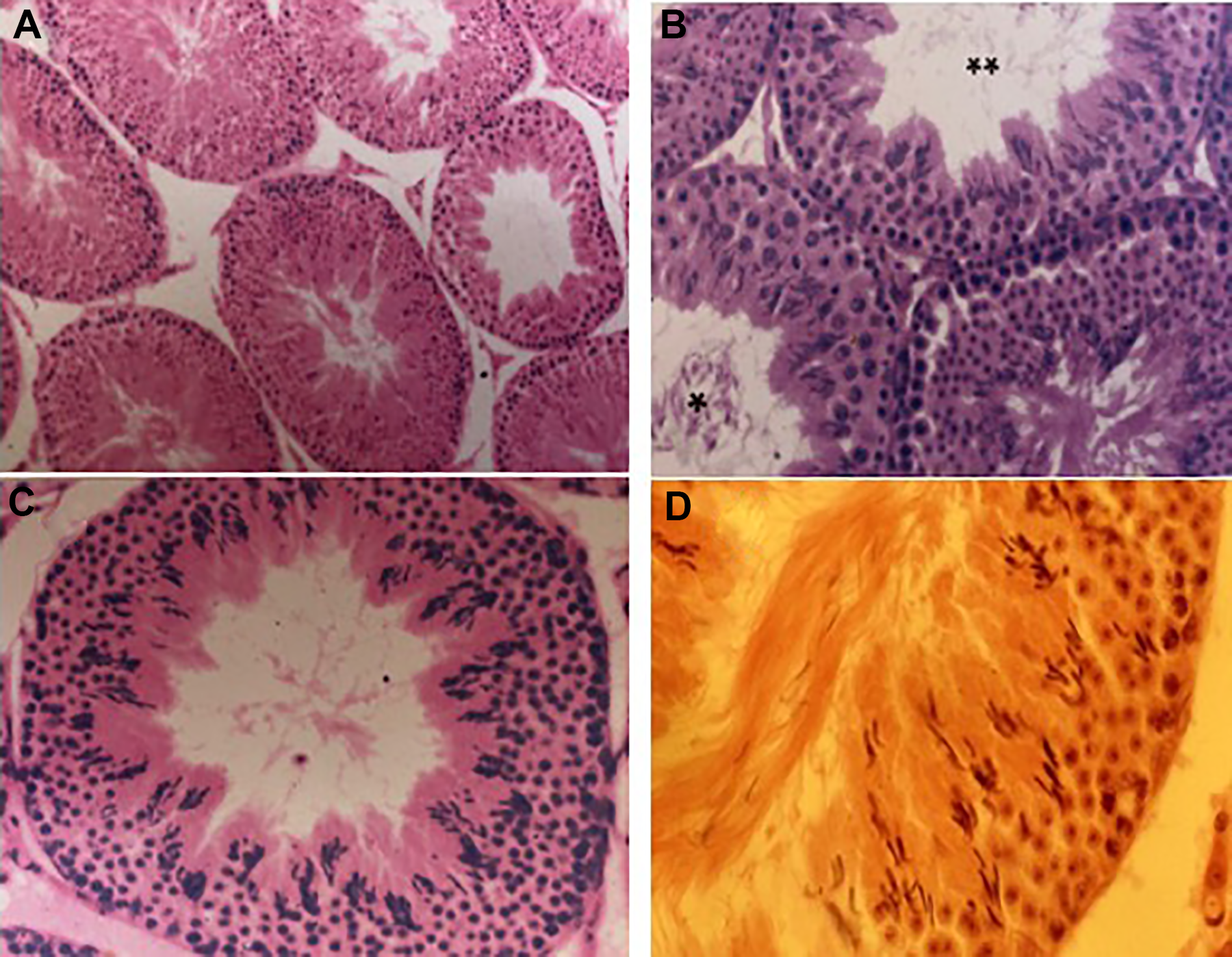
Micrographs of testes sections from control males at different magnifications. Tubules in a variety of stages (A) ×120; stage XII tubule (B) ×240; stage XIII tubule (C) ×320, and (D) ×400. The testes of control males displayed normal spermatogenesis throughout the experiment. * indicates stage IX tubules and ** indicates stage XII tubule.
The chronic low dose of mDNB had a severe effect on testis histology. Animals given 1.0 mg/kg of mDNB showed testes disruption of the seminiferous epithelium resulting in the sloughing of initially spermatocytes and then spermatids which were then presented in the tubule lumen (Figure 9A). Animals given 1.5 mg/kg mDNB/3 d per wk showed essentially the same results after toxic exposure. In group A or animals given 1.5 mg/kg mDNB/daily for 4 weeks exhibited progressive disruption of the Sertoli cells such that the seminiferous epithelium exhibited a ragged appearance by weeks 3 and 4, and very few testicular sperms were present. Further, it was observed that several cell types sloughed from the epithelium (Figure 9B). At the end of the observation period, the testis was not recovered fully and the occasional tubule section showed signs of damage. Similar results were observed with other treated doses.

Testis section showing sloughing off of spermatocytes to the lumen in males given 1.0 mg/kg mDNB/daily/4 wks after the third week (A). Note disrupted Sertoli cells (indicated by an arrow) with sloughing of spermatocytes and spermatids; magnification ×240. Testis section from male given 1.5 mg/kg/mDNB/3d/wk of mDNB after 3 weeks (B); Several cell types have sloughed from the epithelium (indicated by an arrow) and note the ragged appearance of the epithelium (indicated by 2 arrows); magnification ×320. Testis section from males given 1.5 mg/kg mDNB/d/4 wks (C). Sertoli cell disruption is also evident (indicated by an arrow). Seminiferous epithelium is depleted of germ cells in later stage of development and the lumens are devoid of sperm (indicated by arrows). Magnification ×320. mDNB indicates 1,3-dinitrobenzene.
Discussion
The Comet assay is a relatively inexpensive visual method used for the detection of DNA damages in both somatic 20 and germ cells. 21 The size and number of chromosomes are irrelevant and the assay is especially sensitive to measure single-strand breaks of individual cells. 14 In the present study, the Comet and sperm chromatin structure assays were used to evaluate the DNA damage of germ cells induced by short-term in vivo exposure of low chronic doses to a widely used toxicant. Differences in results obtain from test and control animals can be clearly assigned to determine any toxic effects of the toxicant rather than the procedures.
Germ cells from treated males in group A exhibited significant DNA damage from 2 to 6 weeks as measured by Comet assay with highest damage occurring at weeks 3 and 4. At week 6, the damage was too low to be determined by the tail length. According to Nandakumar et al, 22 the head diameter is able to detect very low levels of DNA damages. This is an evident for the significant difference observed at week 6 only with the head diameter. However, the experimental time period was not long enough to determine the recovery period with the tested dose. In group B, there was no significant effect of mDNB on DNA damage at week 3. This is evident for both parameters. But in contrast, at week 4 there was significant genetic damage indicated by both the tail length and the head diameter. In group C, the effects of treatment were detected in weeks 3 and 4. Both migration and stretching of DNA from across the nuclear core increased DNA damage. Therefore, it is clear that even at very low dose levels and very short exposure time period, the effects are more significant.
The animals were treated for 4 weeks at all the dose levels. Furthermore, only the diploid and polyploid cells were considered in the present study. These could be either diploid spermatogonia or polyploid spermatocytes. 23 The primary target of mDNB is believed to be Sertoli cells, 10 which is evident in the present study. Due to destruction of Sertoli cells, they lose their protective ability for the developing testicular germ cells. Sonee et al 24 observed that acute oral dose levels of mDNB can cause alterations in the chromatin structure of pachytene spermatocytes (polyploid cells) in rat models. DNA damage time also suggests the toxic effect of mDNB on pachytene spermatocytes. In groups B and C, germ cells would be spermatogonia at the cessation time of the treatment. Therefore, it is clear that mDNB can also affect spermatogonia populations. Since the Sertoli cells are important to maintain germ cell integrity, 25 any damage to these cells could lead to DNA damage in the polyploid spermatogonia cells. Thus, mDNB may not only act as a reproductive toxicant but also act as a genotoxicant. The effects of low chronic doses of mDNB are significantly higher than the highest test dose level. This was probably due to changes in the dosing protocols. Animals dosed repeatedly with 1.5 mg/kg mDNB showed defects in sperm DNA from weeks 3 to 5. These sperms would have been epididymal sperms, round spermatids, pachytene spermatocytes, and spermatogonia at the end of the treatment period. 26 With the highest dose of mDNB round spermatids, pachytene spermatocytes and spermatogonia would have been affected.
According to Shamsi et al, 27 AO staining the high incidence of green fluorescing sperm indicates a high rate of fertile cells. It has been reported that when the proportion of human sperm displaying green AO florescence was <50% in the ejaculate, the sample showed a decreased ability to fertilize oocytes in vitro. 1 Tejada et al, 28 observed that fertile men exhibited an effective sperm count of ≥50 million/mL while infertile men had lower values. Our observations in the hamsters contradict these findings as no such relationships observed. The results are more in keeping with the studies of Liu and Baker 17 who found no significant relationship between the green AO fluorescence and sperm functional capacity. One of the important observations made in the present study was that irrespective of dose level, maximum genetic damage was observed at 4 weeks after the treatment. This would indicate that low chronic doses of mDNB cause an increase in cell nuclear disruption up to week 4 after exposure with only a gradual recovery thereafter.
In general, the findings obtained with the SCSA corroborated the AO staining results. According to the present results, the increase in the amount of single-strand DNA breaks began in distal cauda sperm by week 2 and steadily rose to a maximum by weeks 3 and 4. Subsequently, the level began to decrease after about week 5 reaching the control levels at week 6. This is reflected in the increase in SDαt, CVαt, and COMPαt values from weeks 2 to 5. At week 3 and 4, even Xαt was affected corresponding to the maximum single-strand breaks. At weeks 3 and 4, the sperms that were in the cauda with abnormal chromatin structure would have been spermatogonia stem cells at the beginning of the exposure and at the round spermatid and pachytene spermatocyte stages at the end of the exposure. This indicated effects on early spermatogenesis stages. 29 The late spermatid stage may be susceptible to toxicants due to the fact that transitional proteins in the sperm nucleus allow exposure of the DNA. 30–31 But the results of the present study indicate DNA damages prior to late spermatid stage at all the dose levels. This is more likely due to effects found earlier when germ cells are undergoing active DNA synthesis. 32
At all the dose levels, the maximum strand breaks were observed at weeks 3 and 4 when the cells were spermatogonia stem cells at the initiation of exposure. Usually, there is sufficient time for DNA repair before cells enter the mitotic phase of the cycle at which time either the damages would be repaired or expressed as DNA strand breaks or base mismatches. 33 These damaged or improperly repaired DNA strands may lead to improper chromatin packaging in the mature sperm thereby increasing its susceptibility to acid-induced partial denaturation. Studies conducted with mDNB showed that both occupational and environmental exposures are relatively high leaving populations at great health risk.
Chronic doses of mDNB had a profound effect and it was apparent at highest dose that there was systemic toxicity. The chronic low dose of the compound caused considerable sloughing of germ cells and general disruption of the seminiferous epithelium, which displayed a ragged appearance presumably because Sertoli cells were also being exfoliated. Since Sertoli cells “nurture” germ cells at all stages of development, there loss will have a major impact on the number of sperm being produced. Based on the experimental results, it could be proposed that prolonged chronic treatment led to a depletion of spermatids within the tubules after 3 to 4 weeks. The effects of the testicular toxicant mDNB on the hamster testis were similar to those reported for studies in other rodents, principally the rat. In both cases, pachytene spermatocytes were the most sensitive cell type even when the initial target cell for the compounds may be different. 34 –36
It can be concluded that mDNB affects sperm nuclear integrity in vivo. It is clear that low chronic doses of mDNB alter DNA structure of diploid and polyploidy germ cells thus altering the nuclear integrity of the cauda epididymal spermatozoa leading to impaired fertility. Although the results suggest that mDNB can alter the sperm nucleus, it remains unclear whether the sperms are able to fertilize an egg when damaged. Alternatively, damaged sperm nuclei could be microinjected into oocytes to examine developmental potential. The results of the present study also confirm that continuous exposure to mDNB is a major concern for humans especially resulting in mutagenic effects, which could affect future generations.
Footnotes
Author’ Note
LDC Peiris designed, conducted, analyzed data, and wrote the paper, P Chathu analyzed data and helped in writing; D.D.B.D. Perera analyzed data, wrote and edited the final draft; HDM Moore designed and supervised the project.
Acknowledgments
The authors would like to acknowledge Mr Ian Morton for his help with flow cytometry and to Mr David for embedding the testis tissue sections.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Commonwealth Commission, United Kingdom.
