Abstract
Purpose:
To compare image quality and radiation dose among different protocols in patients who underwent a 128-slice dual source computed tomography coronary angiography (DSCT-CTCA).
Methods:
Ninety patients were retrospectively grouped according to heart rate (HR): 26 patients (group A) with stable HR ≤60 bpm were acquired using high pitch spiral mode (FLASH); 48 patients (group B) with irregular HR ≤60 bpm or stable HR between 60 and 70 bpm using step and shoot mode; and 16 patients (group C) with irregular HR >60 bpm or stable HR ≥70 bpm by retrospective electrocardiogram pulsing acquisition. Signal to noise ratio (SNR) and contrast to noise ratio (CNR) were measured for the main vascular structures. Moreover, the dose-length product and the effective dose were assessed.
Results:
Both SNR and CNR were higher in group A compared to group C (18.27 ± 0.32 vs 11.22 ± 0.50 and 16.75 ± 0.32 vs 10.17 ± 0.50;
Conclusion:
The correct selection of a low-dose, HR-matched CTCA scan protocol with a DSCT scanner provides substantial reduction of radiation exposure and better SNR and CNR.
Keywords
Introduction
Computed tomography coronary angiography (CTCA) has emerged as the elective noninvasive imaging modality to rule out the presence of significant obstructive coronary artery disease (CAD); thanks to its high negative predictive value. 1 This imaging modality allows to characterize coronary plaques and assess the atherosclerotic burden and stenosis severity, hence playing a key role for patient risk stratification. 2 The handling of the acquired data allows a versatile postprocessing such as maximum intensity projections (MIPs), curved multiplanar reformations (c-MPRs), and 3D volume rendering (VR) that define the vessel course and its anatomical relationships with the surrounding structures. 3,4 However, the expanding application of CTCA has raised concerns about radiation exposure, since it has been widely demonstrated to be related to an eventual increased risk of carcinogenesis. 5,6 Several strategies have been developed to reduce patient radiation exposure, such as retrospective electrocardiogram (ECG) pulsing acquisition with tube current modulation, prospectively ECG-gated CTCA also called step and shoot mode (SAS), and high pitch spiral mode (FLASH). 7 Lowering of radiation dose is therefore a clinical priority that has to be balanced with the need to obtain diagnostic examinations according to the principle of dose optimization. Moreover, in order to have an adequate image quality, these low-dose acquisition methods require a low and stable heart rate (HR), which is not always possible to obtain in all patients. 8 -11 The purpose of this study was to compare image quality and radiation dose using different CTCA scan protocols with a second-generation dual source CT (DSCT) scanner in order to define the parameters for an efficient fine-tuning of a CTCA examination.
Materials and Methods
Study Population
The anonymized records of 90 patients who underwent a 128-slice DSCT-CTCA were retrospectively examined. Patients with history of percutaneous intervention and/or bypass surgery and persistent arrhythmias were excluded. Thirteen patients had typical chest pain, 32 patients had atypical chest pain, 23 patients had nonanginal symptoms, and 22 patients were asymptomatic. Different protocols (FLASH, SAS, and retrospective) were selected according to patient’s HR at presentation. Patients were divided into 3 groups: group A (FLASH) composed by 26 patients with stable HR ≤60 bpm; group B (SAS with acquisition during 66%-75% of RR [inter-beat] interval) composed of 48 patients with irregular HR ≤60 bpm or stable HR between 60 and 70 bpm; and group C (retrospective ECG pulsing modulation acquisition) composed of 16 patients with irregular HR >60 bpm or stable HR ≥70 bpm. Informed consent was obtained from all patients.
Computed Tomography Acquisition and Image Reconstruction
Imaging was performed with a DSCT system (Definition FLASH, Siemens Healthcare, Forchheim, Germany). All patients underwent a sequential scan before contrast administration for preliminary quantification of calcium score with the Agatston method (slice thickness: 3 mm, slice increment: 3 mm, a medium smooth reconstruction kernel (B36f)). Calcium score was evaluated with a dedicated software (CaScore; Siemens, Germany).
Therefore, CTCA was performed with intravenous contrast enhancement. Contrast medium was injected in the antecubital vein using a dual head power injector (Stellant; Medrad, Indianola, Pennsylvania). To synchronize the acquisition of the CTCA data set to the arterial enhancement, a “test bolus” protocol was used: 10 mL of contrast agent of iopromide (Ultravist 370, 370 mgI/mL, Bayer Schering Pharma, Berlin, Germany) were followed by 60 mL of saline solution, both at flow rates of 5 to 6 mL/s, and the time to peak enhancement in the aorta was measured using a series of transaxial scans acquired at 2-second increments, with the first image being acquired 15 seconds after the start of injection. During CTCA, all patients received a dose of 60 mL of contrast agent injected at a flow rate of 5 to 6 mL/s followed by 60 mL of saline solution. Image acquisition was started with a delay corresponding to the measured contrast transit time plus 5 seconds. For FLASH mode, pitch was between 3.2 and 3.4 (table feed: 46 cm/s). Image acquisition was prospectively triggered by patient ECG and started at 60% of the R–R interval. The total duration of data acquisition was dependent on the selected pitch value and the length of the scan volume and varied between 220 and 300 milliseconds. For SAS mode, the triggering phase was set at 66% to 75% of RR interval. For spiral mode, pitch was automatically adapted to HR, and a radiation exposure-modulation algorithm (ECG pulsing) was used to minimize the radiation dose outside the pulsing temporal window. Full exposure was set to 35% to 75% of the RR interval and decreased to 20% of the peak mAs outside this temporal window. The kV and mA were adapted to patient size by automatic exposure control. Reconstructed slice thickness was 0.6 mm, slice increment was 0.3 mm, a medium soft reconstruction kernel was used (B26f), and the field of view was 20 cm.
Subjective and Objective Image Quality
For analysis, image data sets were transferred to an off-line workstation (Multimodality Workplace; Siemens Healthcare). Both objective image quality and subjective image quality were assessed for all patients. Subjective image quality was independently evaluated on a per-vessel basis by 2 independent physicians with experience in cardiovascular imaging of more than 5 years. During the CT image interpretation session, the overall quality of axial slices, MPR, c-MPR, MIP and VR were assessed. A 4-point scale was used (1 excellent = absence of artifacts; 2 good = minimal artifacts, mild blurring or structure discontinuity but fully evaluable; 3 suboptimal = moderate artifacts and blurring or structure discontinuity; 4 not diagnostic = doubling or discontinuity in the course of the segment preventing diagnostic evaluation; Figure 1). 12 In case of disagreement between the observers, consensus was reached in a joint reading to determine the final image quality score. A per-patient image quality score was defined as the worst score found in any coronary artery for each patient.

Curved multiplanar reformations (c-MPRs) of the right coronary artery illustrating the 4-point scale used for image quality. From left to right: (A) the image was given a score 1 because of absence of any artifacts; (B) the image was given a score 2 because of mild blurring; (C) the image was given a score 3 because of structure discontinuity and moderate blurring; and (D) the image was given a score 4 because of doubling and discontinuity in the course of the vessel preventing diagnostic evaluation.
Quantitative image quality was assessed evaluating objective parameters such as mean arterial attenuation, image noise, signal to noise ratio (SNR), and contrast to noise ratio (CNR). Mean arterial attenuation (in Hounsfield Units, HU) was measured by placing regions of interest (ROIs) in the ascending aorta at the pulmonary bifurcation and in the proximal segments of the right coronary artery, the left main artery, the left anterior descending artery, and the left circumflex artery. A circular ROI as large as possible was placed in the lumen of the target vessel taking care of not including vessel walls, calcifications, or plaques. 13 The attenuation values of 3 ROIs, 2 cm2 sized and placed in the air surrounding the patient, were averaged in order to calculate the background noise. 14 The mean attenuation of the central parts of pectoral muscles and the deep paraspinal muscles was measured on both sides, and values were averaged so that obtaining the muscle attenuation was useful for calculating the CNR. 15 Both SNR and CNR were calculated as follows using the above-mentioned parameters:

and

The overall SNR and CNR were finally computed averaging the respective values for vascular structures. A comprehensive workflow is reported in Figure 2.

Objective image quality workflow. LM indicates left main coronary artery; LAD, left anterior descending coronary artery; CX, left circumflex coronary artery; RCA, right coronary artery; SNR, signal to noise ratio; CNR, contrast to noise ratio.
Radiation Dose
For each patient, the dose length product (DLP) was recorded from the automatically generated patient protocol. The effective radiation dose was calculated by multiplying the total DLP by a conversion factor for the chest (k = 0.017 mSv × mGy-1 × cm−1) as previously suggested and adopted in large trials as effective dose. 16
Statistical Analysis
Statistical analysis was conducted using R Core Team (version 3.03; Austria, Vienna) and a
Results
Clinical characteristics of patients (age, sex, body mass index, pretest probability, and calcium score) are listed and compared in Table 1: no significant difference was found among the 3 groups (
Clinical Patients Characteristics and Dosimetric Parameters.
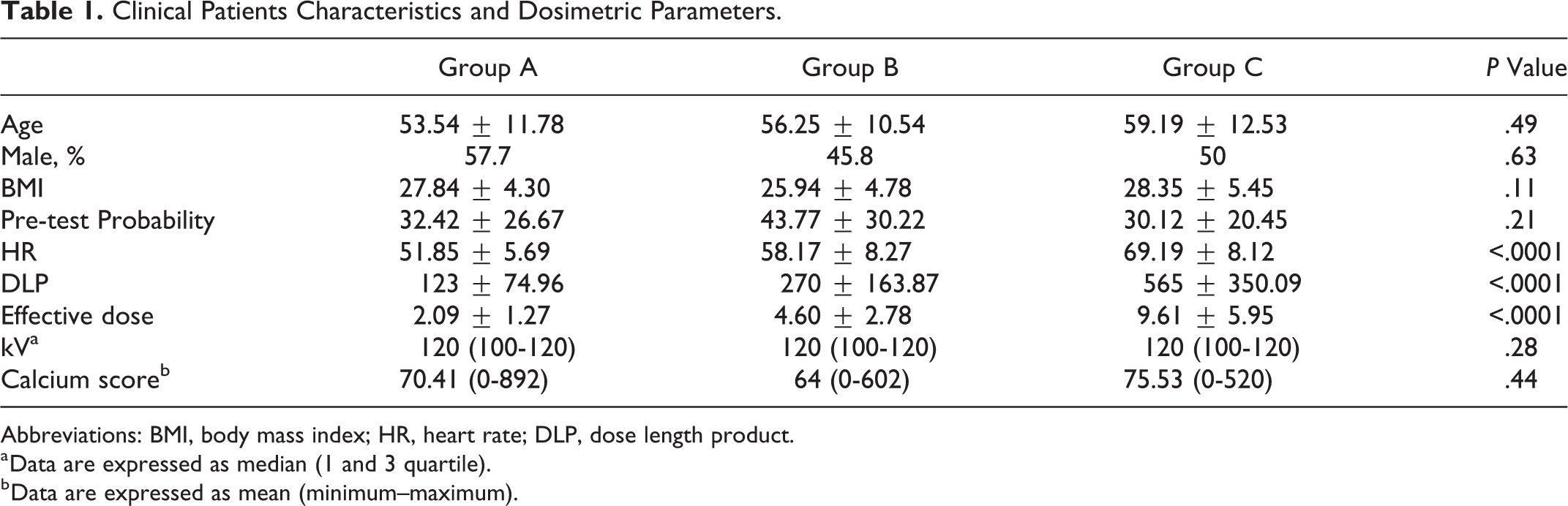
Abbreviations: BMI, body mass index; HR, heart rate; DLP, dose length product.
a Data are expressed as median (1 and 3 quartile).
b Data are expressed as mean (minimum–maximum).

(A) DLP, (B) HR, (C) SNR and CNR, and (D) subjective image quality in FLASH, SAS, and retrospective group. DLP indicates dose length product; HR, heart rate; SNR, signal to noise ratio; CNR, contrast to noise ratio; FLASH, high pitch mode; SAS, step and shoot mode.
Image Quality Assessment.

Abbreviations: SNR, signal to noise ratio; CNR, contrast to noise ratio.
a Data are expressed as median (1 and 3 quartile).
Multiple Comparisons.

Abbreviations: HR, heart rate; DLP, dose length product; SNR, signal to noise ratio; CNR, contrast to noise ratio; A, maximum intensity projections (MIP); B, curved multiplanar reformations (c-MPR)R; C, conventional angiography; d, 3D volume rendering images.
A representative case of a 57-year-old-woman is reported: the effective dose using FLASH scan was 2.38 mSv, the anatomical details are well depicted, and the examination revealed a mixed mostly soft plaque with significant stenosis (>50%) confirmed by further CA (Figure 4).

Computed tomography coronary angiography (CTCA) performed with high-pith spiral scan (FLASH) protocol in 57-year-old woman with hypertension and hyperlipidemia with atypical chest pain and family history of CAD. Prescan heart rate was 51 bpm, low pretest probability (8%), calcium score according Agatston method 134.1, and estimated radiation dose (ED) 2.38 mSv. A, MIP. B, c-MPR. C, Conventional angiography. D, Three-dimensional volume rendering images. The exam revealed (white arrows) a mixed mostly soft plaque which determines a significative stenosis (>50%) at the ostium of the right coronary artery. Further conventional coronary angiography, followed by percutaneous transluminal coronary angioplasty (PTCA), confirmed this result.
Discussion
The development and spread of DSCT equipment has introduced new outlooks in cardiovascular imaging, exploiting the potential of 2 X-ray tubes working simultaneously at same or different energies. 17 The CTCA has been confirmed as a robust clinical tool for diagnosis and noninvasive assessment of CAD 1 and, more generally, coronary pathologies. 3,4 Several strategies have been developed to reduce radiation exposure during cardiac examination adjusting acquisition parameters (scan range, geometry of scanning, tube voltage, tube current modulation, pitch, and slice thickness) and using postprocessing filters able to reduce noise while preserving image quality. 18,19
Another exciting development that offers great promise to further increase CT potential is dual energy CT (DECT) that can be performed by 3 different technologies: dual-source DECT, single-source DECT, and detector-based spectral CT. Dual source DECT utilizes 2 X-ray tubes operating at different kV (low and high) and 2 detectors to obtain simultaneous DE acquisition and data processing. Single-source DECT uses a single X-ray tube that rapidly alternates between low and high energies (fast-switching) and a single detector that quickly registers information from both energies. In detector-based spectral CT, a single X-ray tube with full-dose modulation capabilities is paired with a detector made of 2 layers (sandwich detector) that simultaneously detects 2 energy levels. 20 The DECT offers the possibility to obtain virtual unenhanced images (VUEs), virtual monochromatic images at different energy levels, and material density images iodine (-water). In detail, VUE provides an unenhanced series from an enhanced acquisition thus saving dose exposure. Moreover, VUE images have been demonstrated to have lower image noise when compared to true enhanced images, and, especially in smaller patients, rapid switching DECT showed lower radiation dose when compared to single-energy CT scans, even if misconceptions about the radiation dose from 2 simultaneous X-ray projections and questions regarding the accuracy of additional DECT reconstructions have limited the widespread clinical implementation of this technology. 21
As far as DSCT is concerned, the most widespread dose-saving protocols (retrospective acquisition with ECG-gated tube current modulation, SAS mode, and FLASH mode) carry advantages and drawbacks.
Indeed, in retrospective ECG-gated CTCA, radiation dose has been considered very high 22,23 but, on the other hand, it is useful to assess cardiac function, since data acquisition covers all phases of the cardiac cycle; in addition, the flexibility and reliability of phase reconstruction is the highest when compared to other strategies. 24 The SAS mode is characterized by turning on the X-ray tube only at a predefined time point of the cardiac cycle, usually in mid-to-end diastole, while keeping the patient table stationary. This has resulted in a significant radiation dose reduction from over 20 mSv to around 2 mSv by confining the CT scan to the smallest possible window at only 1 distinct mid-diastolic phase of the cardiac cycle, while the X-ray tube is turned off during the rest of the cycle. 25,26 Nevertheless, it requires low and regular HR and provides only anatomical data not allowing left and right ventricle functional analysis. 23
Another low-dose technique that has been introduced for cardiac CTCA with the advent of second-generation DSCT equipped with two 128-slice acquisition detectors is FLASH mode. 27 This protocol is based on data acquisition prospectively triggered with the ECG, but, unlike SAS, data are acquired in a spiral mode while the table runs with a very high pitch of 3.2 to 3.4. 28 When FLASH mode is used, the entire heart can be scanned within one single cardiac cycle, usually during mid-to-end diastole with a dose for CTCA reduced at 1 mSv or below. 12,29 Our results showed a significant reduction in radiation dose in FLASH mode compared to SAS and, especially, compared to retrospective protocol.
Although literature data 30,31 demonstrated an image quality similar between retrospective protocol and SAS when using FLASH mode in patients with high HR without cardiac arrhythmia and with image acquisition time set at 20% to 30% of the R-R interval, in our preacquisition HR setting (<60 bpm), a prospectively triggered acquisition was started at 60% of the R-R interval. However, with these acquisition parameters, image quality is maintained only when the HR does not change during the CTCA. 10,32 The use of our scan protocol recommendations to achieve optimal image quality at lowest dose, while maintaining diagnostic image quality on the whole relevant epicardial coronary tree, can substantially decrease the radiation dose in CTCA. In addition, these results emphasize the radiation dose reduction potential of HR-lowering medication (eg, β-blockers and ivabradine) 33,34 prior to CTCA in patients with HR >65 bpm, which can consistently reduce radiation exposure. Their action is however not only indicated for HR reduction but it is also aimed to its stabilization during the acquisition. 35 Aggressive administration of HR-lowering medication before scanning may be considered in young patients to induce an HR <55 bpm, which allows to use a FLASH protocol that is associated with a very low radiation dose.
Our study has some limitations; we included only patients in sinus rhythm, although there is evidence on the possibility to use the FLASH acquisition in patients with atrial fibrillation. 36,37 Another limitation is the relatively small number of patients in particular for retrospective acquisitions. Finally, the results of this study are valid only for a 128-slice DSCT scanner and cannot be extended to other CT systems characterized by higher effective temporal resolution.
In conclusion, the correct selection of a low-dose CTCA scan protocol according to HR with use of a 128-slice DSCT scanner results in a significantly reduced radiation dose and higher SNR and CNR. The major challenge is to obtain optimal image quality and an as-low-as-reasonably-achievable radiation exposure.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
