Abstract
Studies of pulmonary toxicity induced by oral exposure to n-hexane are very few, in contrast to those studying the exposure by inhalation. This research tackles the oral toxic effect of n-hexane solvent on the lungs after subchronic exposure of Wistar male rats at 300, 600, and 1200 mg/kg, respectively, each day for 8 weeks. The pneumotoxicity observed in this study was marked by an immune toxicity in the form of a significant increase in the levels of white blood cells, lymphocytes, granulocytes, and eosinophils, as well as a significant increase in relative and absolute lung weight in both groups treated at the doses of 600 and 1200 mg/kg. n-Hexane also resulted in a significant increase in serum total proteins and acid phosphatase in the 3 doses tested daily for 8 weeks. In addition, we found a significant increase in total protein and a decrease in glutathione at 600 and 1200 mg/kg, in the pulmonary homogenate. Furthermore, the rate of lipid peroxidation increased in the 3 doses tested. Histological findings revealed a pneumonia characterized by bronchopneumonia, fibronecrotic lesions, congestion, hemorrhage, type II pneumocyte hyperplasia, alveolar lesions, bronchial epithelium degradation, and inflammation.
Introduction
Today, the use of chemicals is essential for the development of our society because it contributes to the economic growth that many regions have witnessed. 1 However, exposure to these chemicals may have adverse effects on the health of living creatures. Among these chemicals we find the solvents, which are among the most frequented in our environment, few materials of everyday life do not carry traces. 2
Man is exposed in industries and laboratories to organic solvents such as n-hexane, by inhalation or dermal routes, usually. Still, it can be administered orally. It is quite worrying that residues of n-hexane have been found in vegetable oils. In North America, people are exposed to n-hexane in vegetable oils but at very low levels. 3
n-Hexane is a fully saturated aliphatic hydrocarbon with 6 carbons, 4 and it is widely used in food and chemical industry, particularly in the extraction of vegetable oils, synthetic rubbers, pharmaceuticals, and manufacture of various products (adhesives, paint, printing inks). 5 It is considered as a neurotoxic solvent which is manifested by peripheral neuropathy. 6 –8 The majority of the toxicity studies of n-hexane have been by inhalation (anticipated to be the principal route of human exposure) and have focused on neurotoxicity as the presumed principal toxicological effects. There appear to have been few oral studies, and the most commonly cited (Krasavage et al) 9 had information on only the nervous and reproductive systems.
However, n-hexane can also cause lung dysfunction at the physiological and biochemical levels. Previous and recent studies have both shown that the exposure to n-hexane causes damage to bronchioles and pulmonary alveoli. 10 Respiratory diseases have been found in humans exposed to n-hexane; a chronic obstructive pulmonary disease has been reported in humans who chronically inhaled n-hexane vapors 11 and also pneumonia with respiratory distress have been observed following acute exposure to n-hexane. 12 The purpose of this study was to evaluate respiratory changes involving changes in some oxidative stress markers and biochemical and pulmonary histological damage induced by the subchronic exposure to n-hexane.
Materials and Methods
Chemical Product
This article tackles the use of the commercial n-hexane with a purity of 95% (Sigma-Aldrich, St. Louis, Missouri). The acute toxicity of the solvent is relatively low (LD50 in rats: 28 700 mg/kg).
12
Chemicals used for the determination of biochemical markers: Trichloroacetic acid (TCA 20%), ether, chloroform, NaOH (0.1 N), Coomassie brilliant blue (CBB) G250, 0.67% thiobarbituric acid (TBA), 0.02 M EDTA, sulfosalicylic acid (SSA) 0.25%, Ellman reagent (DTNB), and Tris. Most of the products were purchased from Sigma-Aldrich, the rest were obtained from standard commercial suppliers. Chemicals used for the staining of histological sections: Alcian blue, hematoxylin solution, 2% aqueous ferric chloride, sodium thiosulfate, crocein scarlet-acid fuchsin, 5% acetic acid, and 5% aqueous phosphotungstic acid. The products were purchased from standard commercial suppliers.
Animal
This experiment was carried out on 35 adult Wistar male rats (the average body weight of the rats per batch is 300 ± 18 g) from Pasteur Institute in Algeria. The rats were maintained in a controlled environment in the facility under the following conditions: temperature of 25°C ± 2°C, humidity of 57%, and a clear dark photoperiod of 8 hours/12hours. They were fed with water ad libitum and food in the form of 20 g croquettes made of wheat, maize, and barley for each rat, which was purchased from the agro-food complex (El-Kseur, Béjaia).
Experimental Design
Intending to study the effect of n-hexane on lung functioning, rats were divided into 5 groups, 7 rats per each (n = 7). The 3 treated groups received n-hexane dissolved in 0.5 mL of cottonseed oil at doses of 300, 600, and 1200 mg/kg per gavage daily for 8 weeks. After the experimental period, all rats were killed by decapitation. Blood was collected and the lung of each animal was removed, weighed, and stored immediately in formalin (C = 10%).
Control: exposure to 1 mL of water. Positive control: exposure to 0.5 mL of cottonseed oil.
13
Group 1: exposure to 300 mg/kg n-hexane dissolved in 05 mL of cottonseed oil. Group 2: exposure to 600 mg/kg n-hexane dissolved in 05 mL of cottonseed oil. Group 3: exposure to 1200 mg/kg n-hexane dissolved in 05 mL of cottonseed oil.
Studied Parameters
Complete blood count
Blood was collected in EDTA tubes to determine the number of white blood cells (WBCs), lymphocytes (LYM), granulocytes (GRA), and eosinophils (EOSs) using the hematology analyzer Abacus 4.
Acid phosphatase in serum
The serum was collected after centrifugation at 3000 rpm for 10 minutes and stored at −20°C until the biochemical parameters analysis (acid phosphatase and total proteins). The activity of acid phosphatase was determined in the serum according to the colorimetric method. 14 The estimation of this enzyme was carried out using a Spinreact reagent kit.
Total serum and pulmonary proteins
Total serum proteins were assayed using the colorimetric biuret method,
15
using a Spinreact reagent kit. The total amount of pulmonary protein was measured using bovine serum albumin as a standard, in order to determine proteins in unknown samples, according to the Bradford method in 1976,
16
which consists of adding 100 mL of lung homogenate or that of the standard range to 5 mL of the preparative reagent (CBB“G 250” + ethanol + phosphoric acid). The absorbance reading is performed at a wavelength of 595 nm at 6405 UV/vis spectrophotometer.
Pulmonary glutathione
The glutathione (GSH) assay was carried out at the lung level according to the method of Weckbecker and Cory in 1988 17 and expressed in μM/mg of proteins. A lung tissue of 200 mg was homogenized in 8 mL of 0.02 M EDTA solution, and was, next, mixed using 0.25% SSA. After centrifugation at 1000 rpm for 5 minutes, 25 μL of DTNB and 1 mL of Tris-EDTA were added. The absorbance was measured at 412 nm.
Pulmonary malondialdehyde
The concentration of malondialdehyde (MDA), which is the indicator of lipid peroxidation, was determined in the lung by Draper and Hadley’s method in 1990, 18 which is based on the colorimetric measurement of the reaction between TBA and MDA. A 0.5 mL aliquot of lung supernatant was homogenized in TCA and centrifuged at 5000 rpm for 10 minutes at 4°C. The supernatant was then incubated for 30 minutes in TBA (0.67%)/TCA (20%) at 95°C. The absorbance of the reaction mix was recorded at 532 nm. The amount of MDA in the lung is obtained using a standard curve made with 1,1′,3,3′-tetraethoxypropane.
Histological examinations
Lung samples were immersed in the liquid paraffin, after being fixed in 10% formalin and liquid paraffin solidified afterward. Histological sections of 3 µm were achieved and then stained according to the method of Russell-Movat in 1972 19 ; pentachrome stain (specific histochemical) obtained by a mixture of 5 dyes was used as well for the same purpose. The histological reading was done using a Leica optical microscope and a Hamamatsu Nanozoomer 2.0 HT scanner which permitted the identification of different structures and abnormalities present in the lung.
Statistical Study
The results obtained are expressed as the average plus or minus the standard deviation (average ± standard deviation) and were subjected to a 1-way analysis of variance followed by a Tukey test for multiple comparisons. GraphPad Prism software (V.7) was used. Significant differences were established at *P ≤ .05, **P ≤ .01, and ***P ≤ .001.
Results
General Appearance
Hair loss, nosebleed, shortness of breath, loss of appetite, and death of 2 rats (immediately after gavage) were observed in rats exposed to 1200 mg/kg n-hexane.
Food Consumption
In rats exposed to n-hexane at 600 and 1200 mg/kg, food intake decreased significantly compared to controls (*P ≤ .05 and **P ≤ .01), respectively (Table 1).
Initial and Final Body Weight of Rats, Food Consumption, Absolute, and Relative Lungs After Treatment With N-Hexane Compared to Controls.a
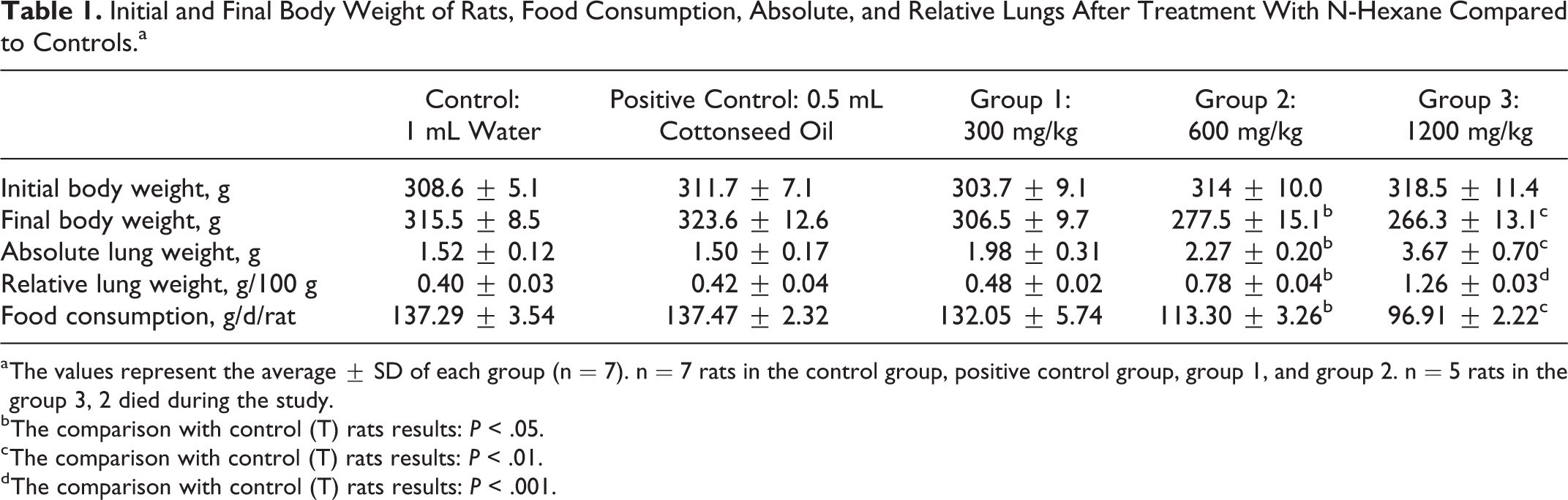
a The values represent the average ± SD of each group (n = 7). n = 7 rats in the control group, positive control group, group 1, and group 2. n = 5 rats in the group 3, 2 died during the study.
b The comparison with control (T) rats results: P < .05.
c The comparison with control (T) rats results: P < .01.
d The comparison with control (T) rats results: P < .001.
Body Weight and Absolute and Relative Lung Weight of Rats
The body weight of rats decreased in a significant way in the group exposed to n-hexane at 600 mg/kg and was highly significant in the 1200 mg/kg group compared to the control and positive control group. A significant increase in absolute and relative lung weight of the n-hexane groups at 600 and 1200 mg/kg compared to the control and control group positive was observed (*P ≤ .05 and ***P≤ .001), respectively (Table 1).
Hematological Studies
The results obtained regarding WBCs showed a significant increase at *P ≤ .05, **P ≤ .01, and ***P ≤ .001 in the 3 groups treated with 300, 600, and 1200 mg/kg, respectively, compared to the control and the positive control group. For LYM, GRA, and EOS also recorded a significant increase, but at **P ≤ .01 and ***P ≤ .001 in all groups treated with n-hexane, respectively compared to positive control and control as demonstrated in Table 2.
Hematologic Changes in the Groups Exposed to N-Hexane Compared to Controls.a
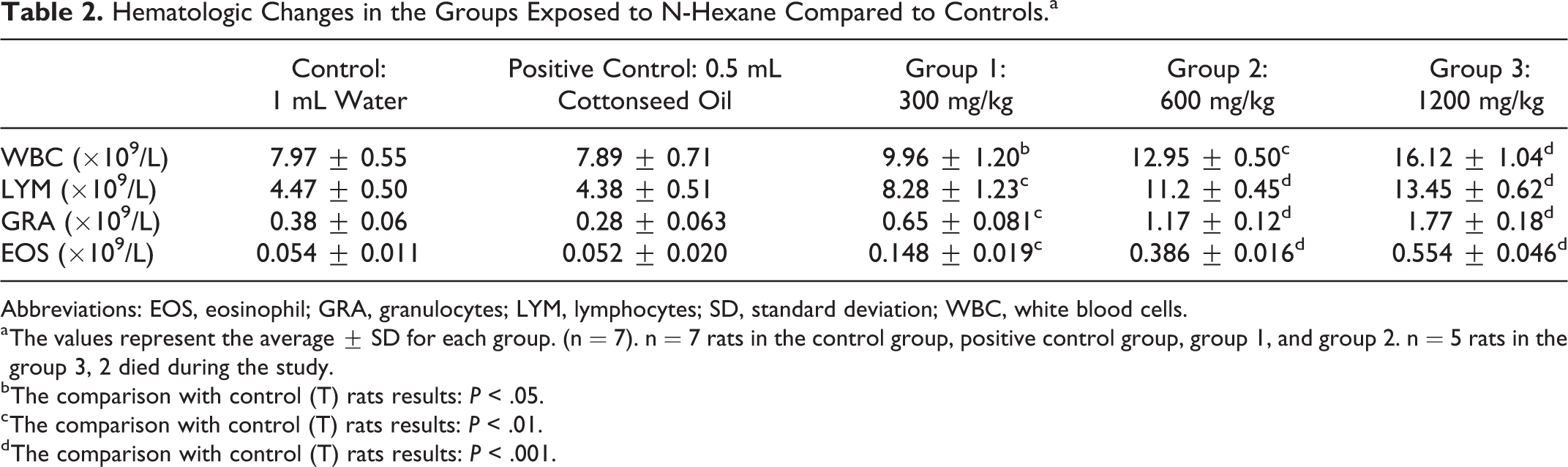
Abbreviations: EOS, eosinophil; GRA, granulocytes; LYM, lymphocytes; SD, standard deviation; WBC, white blood cells.
a The values represent the average ± SD for each group. (n = 7). n = 7 rats in the control group, positive control group, group 1, and group 2. n = 5 rats in the group 3, 2 died during the study.
b The comparison with control (T) rats results: P < .05.
c The comparison with control (T) rats results: P < .01.
d The comparison with control (T) rats results: P < .001.
Biochemical Serum Markers
Serum levels of acid phosphatase and total proteins increased significantly in rats belonging to the 3 groups exposed to n-hexane, compared to the other 2 groups: positive control and control (*P ≤ .05, **P ≤ .01, and ***P ≤ .001; Table 3).
The Serum Level of Some Biochemical Parameters of the Groups Treated With N-Hexane Compared to Control.a

a The values represent the average ± SD for each group. (n = 7). n = 7 rats in the control group, positive control group, group 1, and group 2. n = 5 rats in the group 3, 2 died during the study.
b The comparison with control (T) rats results: P < .01.
c The comparison with control (T) rats results: P < .001.
d The comparison with control (T) rats results: P < .05.
Variation in Total Protein, Pulmonary MDA, and GSH
Rats treated with n-hexane at 600 and 1200 mg/kg showed a statistically significant increase (**P ≤ .01 and ***P ≤ .001) at the total protein levels in the pulmonary homogenate. The evaluation of the effect of n-hexane on lipid peroxidation in the lung revealed a significant increase as well (*P ≤ .05, **P ≤ .01, and ***P ≤ .001) in the 3 groups treated with 300, 600, and 1200 mg/kg, respectively, compared to the control and the positive control groups. Thus, the treatment involving n-hexane at 600 and 1200 mg/kg resulted in a significant decrease (*P ≤ .05 and **P ≤ .01) in the level of pulmonary GSH compared to the control and the positive control groups (see Table 4).
Total Proteins, MDA, and Pulmonary GSH Observed in Rats Treated With N-Hexane Compared to Control Groups.a

Abbreviations: GSH, glutathione; MDA, malondialdehyde; SD, standard deviation.
a The values represent the average ± SD for each group. (n = 7). n = 7 rats in the control group, positive control group, group, 1 and group 2. n = 5 rats in the group 3, 2 died during the study.
b The comparison with control (T) rats results: P < .01.
c The comparison with control (T) rats results: P < .001.
d The comparison with control (T) rats results: P < .05.
Histological Evaluation
Histological examination of the lungs of the rats exposed to n-hexane by gavage for 8 weeks has shown some changes and some lung lesions, which are described according to their severity (minimal, moderate, severe; Table 5). Their distribution was also characterized accordingly (focal, multifocal, diffuse), as well as their location (see Figure 1).
The Frequency and Severity of the Lung Changes, After Treatment With N-Hexane Compared to Controls.
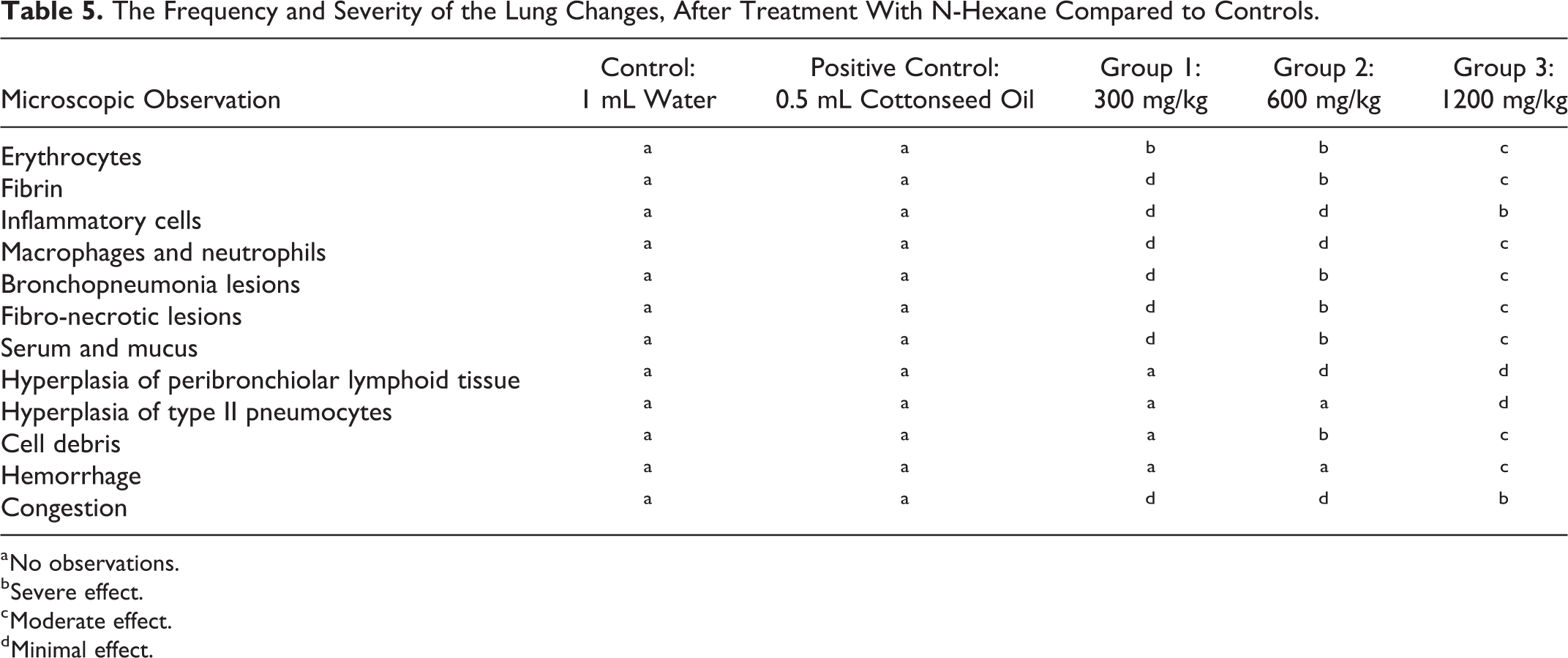
a No observations.
b Severe effect.
c Moderate effect.
d Minimal effect.

Photograph of histological lung sections of the Wistar male rats of the 3 groups treated with n-hexane compared to the control group and the positive control group. A, Control group. A*, Positive control group—normal histological appearance and architecture of alveoli and bronchioles. B, Group treated using n-hexane at the dose (300 mg/kg) which showed the presence of foci of erythrocytes filled with a small amount of fibrin (arrows); inflammatory cells (circles). C, Group treated with n-hexane at dose (600 mg/kg) showed necrosis of the bronchiolar epithelium. Their lumen contains necrotic debris and fibrin; hyperplasia of the peribronchiolar lymphoid tissue (square); fiber-filled alveoli and erythrocytes (arrows); inflammatory cells including macrophages and neutrophils (circles). D, Group treated with n-hexane at the dose (1200 mg/kg) revealed a highly dilated lung, filled with cell debris, fibrin, and erythrocytes (arrows); the bronchial epithelium was eroded (chevron); bronchioloalveolar lesions containing numerous macrophages and neutrophils (triangle). Coloring Movat pentachrome, magnification ×4, ×10, and ×20. AV indicates alveolus; BE, bronchiolar epithelium; BR, bronchiole; BV, blood vessel.
Histologically, all lung alterations observed in the rats treated with n-hexane at 300 mg/kg group (B) were defined by the presence of lesions compatible with acute interstitial pneumonia. They are light to moderate in severity, multifocal in distribution, and are characterized by more or less coalescent foci of alveoli filled with erythrocytes, a small amount of fibrin, and little serum. However, very few inflammatory cells are present; they are limited to some intra-alveolar macrophages and neutrophils.
As for group (C) which was treated with n-hexane at the dose of 600 mg/kg, moderate multifocal bronchopneumonia and fibronecrotic lesions were observed. In the affected areas, bronchioles exhibit necrosis of the bronchiolar epithelium and their lumen contains fibrin and necrotic debris. A slight hyperplasia of the peribronchiolar lymphoid tissue was also noted. The alveoli are affected and filled with an exudate composed of serum, fibrin, erythrocytes, some necrotic cells, and few but varied inflammatory cells such as macrophages and neutrophils.
The lung damage observed in the rats belonging to group (D) and exposed to n-hexane at the dose of 1200 mg/kg is similar to that reported for group (C). However, this is more extensive in distribution and more marked in severity, dilated and filled with cell debris, fibrin, and erythrocytes, all corresponding to abundant fibronecrotic exudate and hemorrhagic exudate. Pulmonary lobe sections present diffuse and relatively severe bronchopneumonia lesions characterized by a bronchioloalveolar exudates, containing numerous macrophages and neutrophils as well as a non-negligible amount of serum and mucus. A slight multifocal hyperplasia of type II pneumocytes is also observed. Mild to moderate diffuse congestion is also observed. The lesions are consistent with subacute to chronic necrotic bronchopneumonia.
Discussion
In this study, pneumotoxic, hematological, and biochemical effects were reported after oral subchronic exposure of Wistar male rats to n-hexane. Groups exposed to the 2 high doses of n-hexane showed a significant decrease in body weight with an increase in absolute and relative lung weight. Our results are consistent with the results obtained by Adedara et al in 2014 20 and those of Linder et al in 1992 21 who reported decreased body weight after exposure to n-hexane in drinking water and gavage, respectively. This is probably due to the loss of appetite caused by the neurotoxic effect of n-hexane. Sympathetic neurons that innervate internal organs and control their activity, such as intestinal motility and salivation, are affected by neurotoxins. 22
In addition, Dunnick in 1991 23 found that the exposure of mice by inhalation to n-hexane causes an increase in relative weight of the lungs. The increase in relative weight represents inflammation and swelling of the organ. 24,25 The shortness of breath observed in the rats treated with n-hexane leads to an increase in the respiratory rate in order to increase the oxygen supply, which proves that n-hexane caused a breathing difficulty. The same thing was noticed by Lungarella et al in 1984, 10 after the exposure of rabbits to n-hexane by inhalation for 24 weeks. n-hexane causes a deconcentration of alveolar air and a marked decrease in oxygen levels. 26
White blood cells, LYM, GRA, and EOSs are defense cells in the body against foreign bodies. The increase in WBC observed in our work is consistent with the results of Akpan et al in 2014, 27 who reported that the exposure of mice to gasoline for 60 days caused leukocytosis. Levels of these cells were not increased in the Dunnick 1991’s 23 study on n-hexane in mice by inhalation, and no hematologic differences were reported by Cavender et al in 1984 28 after exposure of male and female rats to n-hexane by inhalation. We also noted an increase in the number of LYM, granulocytes, and eosinophils. However, there was no significant change in LYM and eosinophils in the results obtained by Ashmawy et al in 2007, 29 who subcutaneously exposed albino rats to 5 mg/kg of n-hexane induced in olive oil in 6 /7 days for 8 weeks.
The increase in WBCs denotes the triggering of the immune system against xenobiotics 27 which can be due to certain inflammatory syndromes, because the WBCs play an important role in the activation of the inflammatory response. 30 According to the study conducted by Tell et al in 1985, 31 pulmonary inflammation causes an increase in WBCs; this has been reported in several Norwegian smoking adolescents. Lymphocytes are a type of WBC, which is distinguished by 2 lineages: T cells and B cells. The entry of a foreign molecule stimulates LYM. T cells participate in the cell-mediated immune response by destroying foreign or abnormal cells. B cells are specialized in the production of antibodies against foreign molecules. 32
The results of this research showing an increase in acid phosphatase, total protein in the serum and in the lung homogenate, come in concordance with the results obtained by Sahu et al in 1982, 33 who recorded the same after the exposure of Sprague Dawley male rats to n-hexane by inhalation. Although Sahu et al in 1982 33 did report elevations in total proteins and acid phosphatase in bronchial lavage following n-hexane exposure, the overall levels seemed lower than those reported in the present study, and statistical analysis was not conducted.
Cell damage caused by chemicals can release biochemical molecules for defense purposes. Proteins contain thiol groups (-CH2-SH) which have a defensive role in the organism. 34 In the lung, acid phosphatase is present in alveolar cells of type II. 35,36
According to Sahu et al in 1982, 33 the increase in total protein and acid phosphatase in the n-hexane groups, compared to the control group, suggests that n-hexane caused lung damage, mainly pneumocytes II, which interferes with the production and storage of lung-maintaining substances. The administration of n-hexane provoked an increase in lipid peroxidation, monitored by peroxidation products (MDA), and a decrease in GSH. Our results correlate with those of Ezzat et al in 2011 37 who studied the toxic effect of gasoline on the respiratory antioxidant defense system after acute and chronic exposure, demonstrating an increase in lipid peroxidation with a decrease in GSH.
Glutathione acts as an essential defense against extra and intracellular oxidative stress that causes cellular damage. In the lung, GSH protects the epithelial cells and eliminates pulmonary inflammation. 38 In some pulmonary diseases (such as inflammatory diseases, cystic fibrosis, acute respiratory distress syndrome, and asthma), GSH levels decline. 37 The cause behind the decrease of GSH in the lung is its consumption for defensive reasons against the components of the gasoline. 39 On the other hand, lipid peroxidation is the origin of membrane lipid oxidation. The cell membrane is rich in lipids and peroxidation gives rise to functioning anomalies. 40 This increase in MDA indicates that n-hexane has altered the tissues by increasing oxidative stress. 20
Histologically, n-hexane caused lung damage in the rats exposed to the 3 doses (300, 600, and 1200 mg/kg), respectively. Histological changes in the lung are signified, in this study, by interstitial pneumonia, bronchopneumonia, fibronecrotic lesions, congestion, hemorrhage, type II pneumocyte hyperplasia, alveolar lesions, bronchial epithelial erosion, and pulmonary inflammation, including macrophage and neutrophil infiltration which are more detailed in “Results” section. Similar effects were observed by Parasuraman et al in 2014, 41 who demonstrated that the intraperitoneal exposure of Sprague Dawley rats to petroleum ether (250 and 500 mg/kg intraperitoneally) resulted in interstitial pneumonia perceived as fibrosis and pulmonary inflammation, including neutrophils and LYM.
A histopathological study of the lungs carried out by Ashmawy et al in 2007 29 after subcutaneous and subchronic exposure of albino rats to 5 mg/kg of n-hexane demonstrated that the latter caused lung damage. This damage is manifested by pulmonary congestion and hemorrhage with the presence of macrophages. On the other hand, exposure to n-hexane induces degradation of the bronchiolar epithelium, hyperplasia of type II pneumocytes, and alveolar lesions. 42 Morphological alteration of the lung is due to the direct effect of n-hexane on the lung which is one of its targeted organs. 29 Pulmonary changes caused by n-hexane are a consequence of pulmonary lipid peroxidation and inflammatory response. 43 In addition, the pneumocyte II hyperplasia, observed in rats treated with n-hexane at the highest dose (1200 mg/kg), is the result of the damage of the pneumocytes I, which are considered as the mother cells of pneumocytes II. 44,45
Footnotes
Acknowledgments
Foremost, we would like to thank God for giving us the strength to undertake this research study. The authors thank the members of the animal ecophysiology research group, Pr Cherif Abdennour, Rym Bencheikh, Imene Dellal, and Dr Abdallah Metai, as well as all the colleagues of the laboratory for their support and help. They also like to express their gratitude to the doctoral student Mohamed Khelifi from Tunisia and Mr Yassad Abdeldjouad.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
