Abstract
In this study, we investigated the relationship between target of rapamycin (TOR) and H2O2-induced hormetic response in the budding yeast
Introduction
Based on its intensity, stress can be considered as either beneficial or harmful. 1 –5 Mild stress can stimulate organisms’ biological functions and result in an acquisition of their resistance to high doses of the same stressor (preadaptation) as well as other stressing factors (cross-protection or cross-adaptation). 1,6 –9 This phenomenon also known as hormesis is observed in a variety of organisms responding to a wide range of chemical, physical, and biological stressors. A hormetic response is usually limited to the 30% to 60% increase in biological function under mild stress conditions. 3
It was recently found that hydrogen peroxide (H2O2) plays a crucial role in the induction of hormesis. 6 Stimulation of the reproductive potential through hormesis-induced compounds, like H2O2, appears to be an effective approach to improve yeast survival and cross-adaptation to different stressors. 7,8 In turn, the physiological state of organism is critical in the mobilization of stress-responsive defenses. As reported earlier, fructose-grown yeast in the exponential phase (short-term model) exposed to H2O2 demonstrated higher survival than glucose-grown cells. 10 The phenomenon was explained by the protective role of fructose, in spite of its more potent ability to produce reactive oxygen and carbonyl species compared to glucose. 11 –13 Short-term application of fructose has been suggested to induce a mild carbonyl/oxidative stress stimulating defense mechanisms responsible for cell survival under lethal H2O2 stress: a reduced level of reactive oxygen species in fructose-grown cells after exposure to H2O2 was consistent with a broad peak of superoxide dismutase and catalase activation by H2O2. At the same time, cells grown on glucose demonstrated an increase in the level of reactive oxygen species and a sharp rise in the antioxidant enzyme activities followed by their rapid inactivation. 10 Unlike short-term model, in the stationary phase (long-term model), a higher level of carbonyl/oxidative stress markers has been detected, which was correlated with a higher aging and mortality rate of fructose- compared to glucose-grown yeast. 14
The specificity of stress response is also determined by the nature of the stressor and the variety of downstream effectors involved. 15,16 A hormesis suggests the existence of complex mechanisms which sense and respond to different kinds of stress. As one of the main signaling mechanisms, the target of rapamycin (TOR) pathway that regulates cell growth and metabolism is involved in cellular responses to many types of stress. 6,17 –20 In spite of the apparent and sometimes hotly debated contradictions between the hormesis and TOR hypothesis of aging and stress resistance, both are applied to explain some common molecular mechanisms. 21,22
Target of rapamycin promotes cell growth in response to nutrient availability. 23 Most studies on nutrient-mediated activation of TOR were focused on nitrogen sources, 20,24,25 but little attention was paid to carbohydrates. In addition, TOR, as a central controller of cell growth, may respond to different types of stress and play an important role under stressful conditions other than nutrient limitation. 24
Here we used TOR-deficient strains grown on glucose or fructose to study the effects of different concentrations of H2O2 on yeast. It was shown that H2O2 induced the hormetic dose–response in
Materials and Methods
Yeast Strains and Chemicals
The
Growth Conditions, Stress Induction, and Cell Extracts
Yeast cells were grown at 28°C with shaking at 175 rpm in a liquid medium containing 1% yeast extract, 2% peptone, and 2% glucose (YPD) for 24 hours. For experiments, the obtained cultures were diluted to about 75 × 106 cells/mL in the same medium. Aliquots of the main culture after 24 hours growth were exposed to different concentrations of H2O2 followed by their incubation at 28°C for 1 hour. Control cells were incubated under the same conditions but without H2O2. After incubation, cells from experimental or control cultures were collected by centrifugation (5 minutes, 8000
Reproductive Ability
Yeast reproductive ability was analyzed by plating in triplicate on YPD agar after proper dilution. The plates were incubated at 28°C for 3 days and the colony-forming units counted. 34 Reproductive ability was expressed as percentage of total amount of cells plating on YPD agar.
Enzymatic Activity Measurements
The parameters were measured spectrophotometrically with a Spekol 211 spectrophotometer (Carl Zeiss, Germany) and CΦ-46 (ЛOMO, USSR).
The activity of GR and G6PDH was measured by following the consumption or production of nicotinamide adenine dinucleotide phosphate reduced form (NADPH), respectively, at 340 nm. The extinction coefficient for the coenzyme of 6.22 mmol/L−1·cm−1 was used for calculations. 33
The activity of GLO1 was measured by following the formation of S-D-lactoylglutathione at 240 nm. The extinction coefficient for S-D-lactoylglutathione of 3.1 mmol/L−1·cm−1 was used for calculations. 35
One unit of the activity of the enzymes was defined as the amount of supernatant protein that produces or utilizes 1 μmol of product or substrate per minute. The activities were measured at 25°C and expressed per milligram of soluble protein in supernatant.
Protein Concentration Measurement and Statistical Analysis
Protein concentration was determined by the Coomassie brilliant blue G-250 dye-binding method
36
with bovine serum albumin as the standard. Experimental data are expressed as the mean value of 3 to 7 independent experiments ± the standard error of the mean, and statistical testing was carried out used Student
Results
Hormetic Effect of H2O2 Depends on TOR and Type of Carbohydrate in Yeast Growth Medium
According to the data on the vital effect of reducing monosaccharides, glucose and fructose have various influences on cell survival under stress conditions.
12,37
–41
Recently, we have found that glucose- and fructose-supplemented growth differently affected
Figure 1 demonstrates the influence of different concentrations of H2O2 on the yeast reproductive ability. The biphasic dose–response dependence, characterized by low-concentration stimulation and high-concentration inhibition of yeast colony growth, has been observed in all cases, except fructose-grown
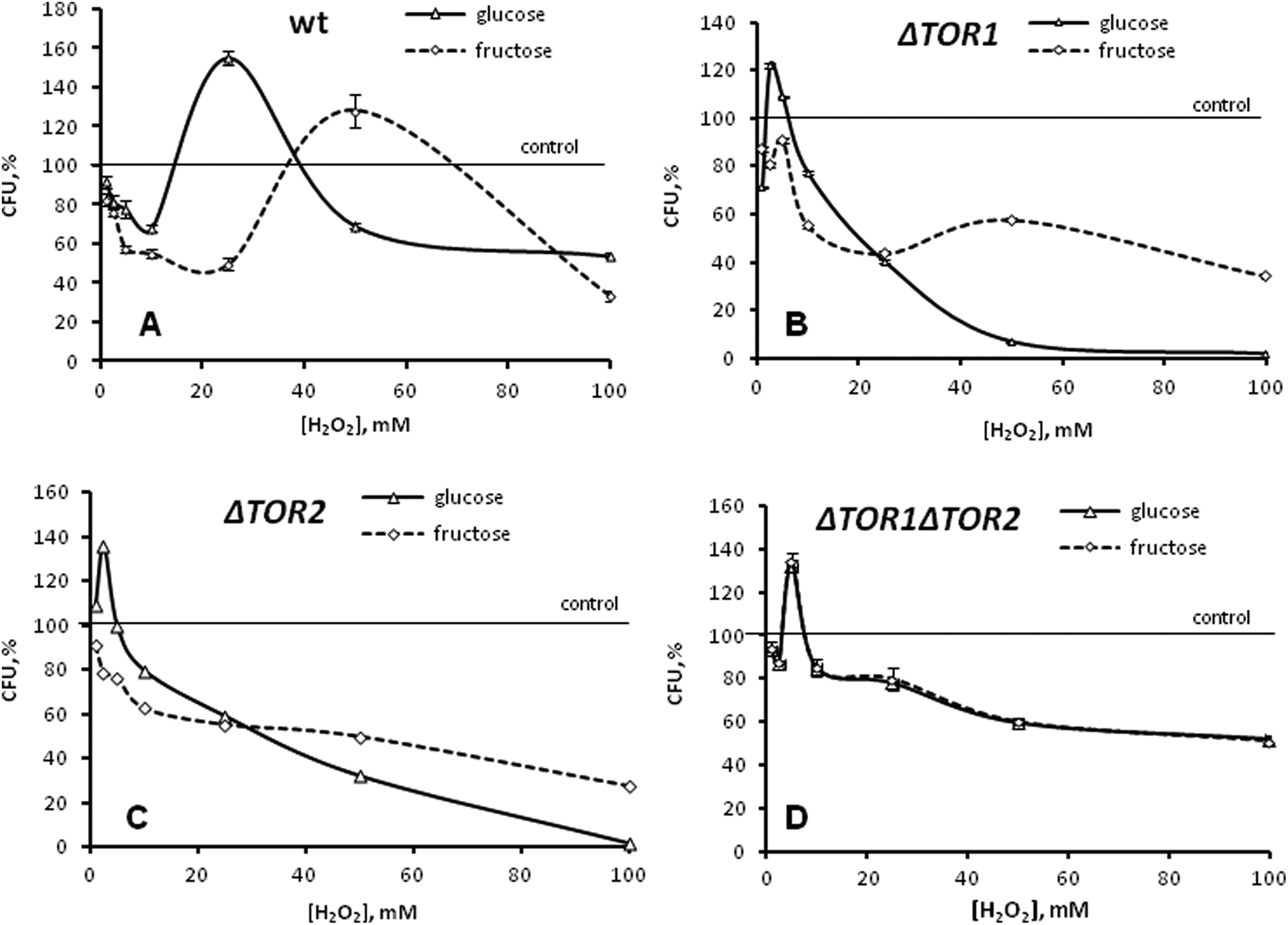
Effect of hydrogen peroxide on reproductive ability of
Figure 1B and C presents the reproductive ability of the
Exposure of the
Glutathione Reductase is Involved in Hormetic Effect of H2O2
Next we examined the potential role of GR, G6PDH, and GLO1 in H2O2-induced hormesis using glucose- and fructose-grown
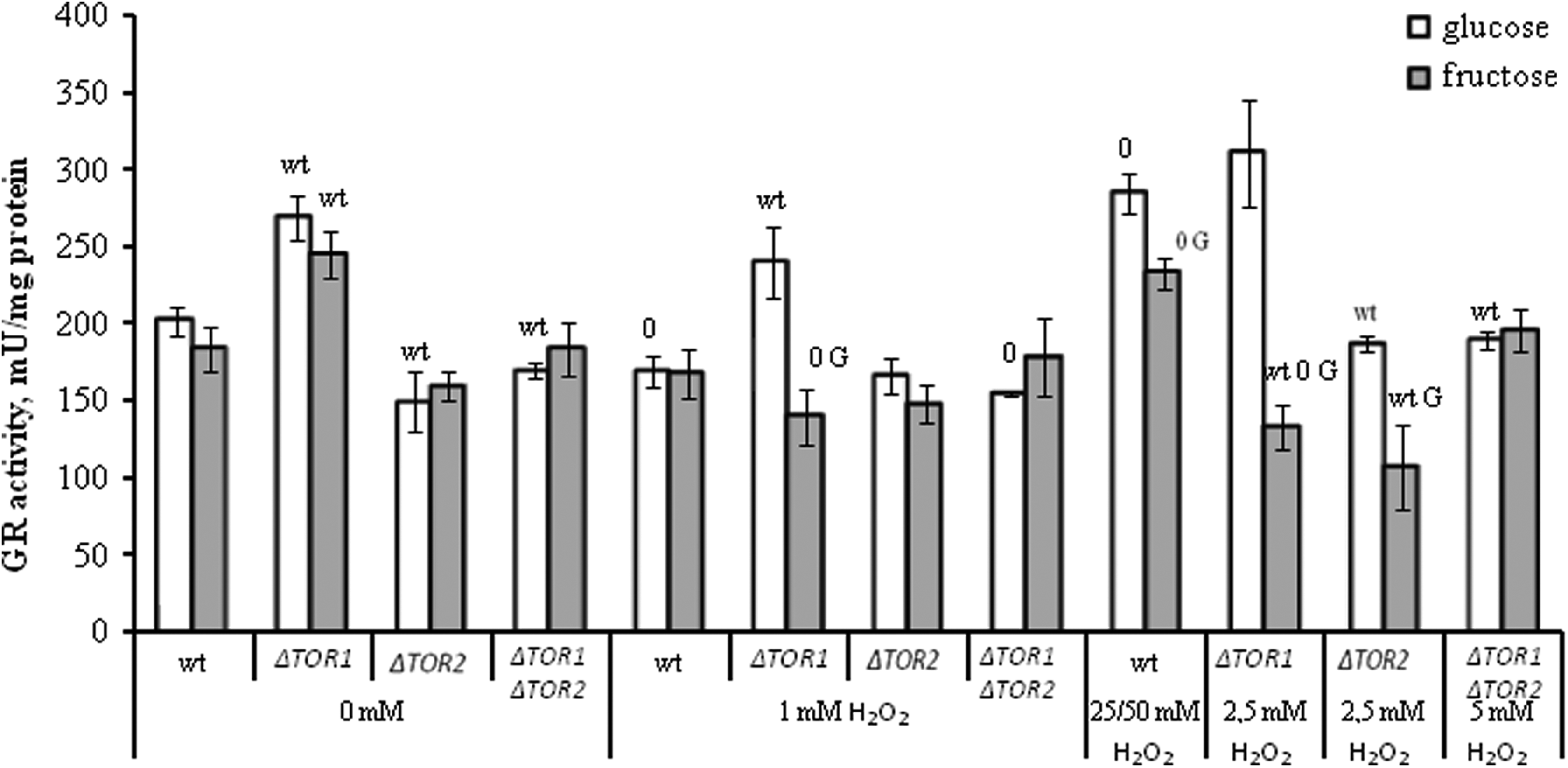
The activity of glutathione reductase in wild-type and target of rapamycin (TOR)-deficient
In general, the activity of GR was not changed or somewhat decreased during incubation of the yeast strains with as little as 1 mmol/L H2O2 compared to the control untreated cells. At the same time, the hormetic concentrations of H2O2 (25 mmol/L and 50 mmol/L H2O2 for glucose- and fructose-grown wild type, respectively) increased GR activity by about 1.4-fold in the parental strain. Hormetic concentration of H2O2 for glucose-grown cells lacking
When compared the activities of GR in the parental and mutant strains, it should be noted that under control conditions the parameter was higher by 1.3-fold in the
Comparing the 2 studied cell types (glucose- and fructose-grown), no significant difference was found between them under control conditions (without H2O2) and at 1 mmol/L H2O2 in all cases, except the
Interestingly, H2O2 affected both the yeast colony growth (Figure 1) and the GR activity (Figure 2) in a similar way under all experimental conditions used in this study that suggested the involvement of GR in hormetic effect of H2O2.
Mutation in Either TOR1 or TOR2 Genes Causes Decline in the Activity of Glucose-6-Phosphate Dehydrogenase
Like GR, G6PDH is involved in the stabilization of the pool of reduced glutathione, important antioxidant and antiglycation agent in the cell.
28,42
Under stress conditions, GR and G6PDH were found to demonstrate a strong relationship,
33,44
however in the present study the enzymes showed different behaviors in the yeast treated with H2O2 (Figures 2 and 3). Unlike GR, the activity of G6PDH was not affected by H2O2 in neither wild type nor

The activity of glucose-6-phosphate in wild type and target of rapamycin (TOR)-deficient
Comparison of G6PDH activities in the parental and mutant strains clearly indicated that the parental strain had the highest value, while both the single knockouts had the lowest values (3-4-fold lower than wild type), independent of the experimental conditions. The double knockout and wild type did not differ significantly from each other in their G6PDH activities under control conditions, however the
When compared the 2 studied cell groups (glucose- and fructose-grown), no significant difference was observed between them in all strains and under all experimental conditions used in this study.
Mutation in Either TOR1 or TOR2 Genes Activates Glyoxalase 1
Glyoxalase 1 is functionally associated with GR and G6PDH via reduced glutathione, since the latter is used by GLO1 to detoxify the reactive carbonyl metabolites produced during carbohydrate metabolism.
27,28
Figure 4 shows that H2O2 did not change GLO1 activity under any experimental conditions used in all strains studied, except the
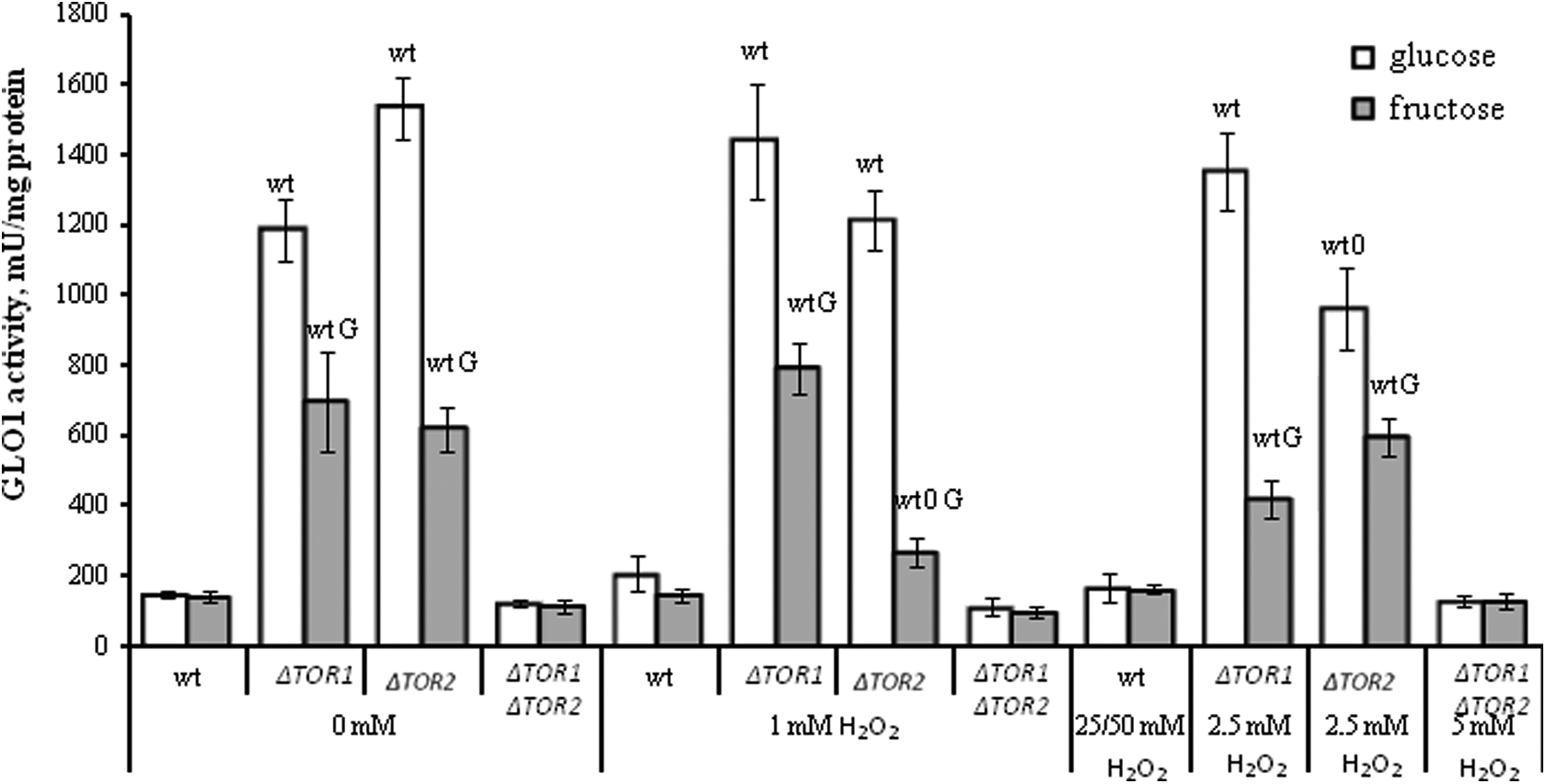
The activity of glyoxalase 1 in wild-type and target of rapamycin (TOR)-deficient
Unlike G6PDH, GLO1 activity in the parental strain demonstrated the lowest value, while both the single knockouts had the highest parameters (2- to 11-fold higher than that in the wild type), independent of the experimental conditions. Like G6PDH, GLO1 activities did not differ significantly in both the wild type and the double knockout of all experimental groups of cells.
Comparing the 2 studied cell groups (glucose- and fructose-grown), no significant difference was observed between them in the parental and double mutant strains, independent of H2O2 concentrations used. At the same time, both the single mutants demonstrated GLO1 activity higher by 1.6- to 3.2-fold in glucose- than fructose-supplemented medium at all H2O2 concentrations used.
Discussion
Recent studies strongly support the notion that H2O2 plays a dual role in biological systems.
6,21,45,46
Its effects can be considered as either beneficial or harmful, because at high concentrations H2O2 causes a stress, when oxidative damage to cell structures occurs, whereas at low concentrations it is a part of many cellular signaling systems. At low concentrations, H2O2 also plays a crucial role in the induction of hormesis.
6,7,21,47
The potential effect of H2O2 depends not only on its concentration but also on the physiological state of the cell. Using the model organism
The experiment mentioned earlier has been conducted with an exponential culture of
The findings of the present study demonstrate that both the cell types studied (glucose- and fructose-grown) respond differently to exogenous H2O2 (Figure 1A). Although in both cases we observe typical biphasic concentration–response curve, exhibiting a hormetic effect of H2O2, H2O2 triggers the hormetic response in the 2 cell groups at different concentrations. While the reproductive ability of cells grown on glucose reached a maximum value after yeast incubation with 25 mmol/L H2O2, in fructose-grown cells the peak hormetic response shifted to a higher concentration of 50 mmol/L H2O2. This is consistent with our previous suggestion on the ability of fructose to induce a mild carbonyl/oxidative stress stimulating cellular defensive mechanisms responsible for cell survival under more severe stress. 10,41 The last mechanism can be posited from the in vitro and in vivo studies reporting that fructose is a more potent glycoxidation agent, than glucose, and therefore capable of producing greater amounts of reactive species. 11 –14
The oxidative stress response in
An increasing number of studies have reported a lack of correlation between gene expression at different levels of cellular organization. 50,51,54 –56 The weak elevation in antioxidant enzyme activities in yeast treated with sublethal concentrations of H2O2 10,33,48,57 did not correspond to reported earlier high level of synthesis of the respective enzyme molecules. 26,58 Exposure of microorganisms to low hormetic concentrations of H2O2 leads to the acquisition of cellular resistance to a subsequent lethal stress. 7,8,59 We may suppose a rapid reorganization of gene expression and accumulation of nonactive synthesized de novo stress-protectant molecules in yeast cells exposed to low hormetic doses of H2O2, since a sudden increase in the antioxidant activities could dramatically disturb the intracellular redox homeostasis leading to other kinds of stress, for instance reductive stress. 42 Nevertheless, such accumulation provides the cells with the capability to respond quickly and survive consequent lethal challenge via rapid posttranslational activation of synthesized de novo nonactive proteins.
Manipulation of the reproductive potential through hormesis-stimulating compounds, such as H2O2, appears to be an effective approach to improve yeast survival and cross-adaptation to different stresses.
7,8
As one of the main signaling mechanisms, the TOR pathway by regulating cell growth and metabolism is involved in cellular response to many types of stress.
6,17
–20
In spite of the apparent and sometimes hotly debated contradictions between the hormesis and TOR theories, both are applied to explain some common mechanisms of aging as well as stress resistance.
21,22
Our study demonstrates the involvement of the Tor1 and Tor2 proteins in the hormetic effect of H2O2 (Figure 1). The mutations in either
Since for the double mutant (Figure 1D) the peak hormetic response is found in the both studied cell groups (glucose- and fructose-grown) at somewhat higher concentration (5 mmol/L H2O2) than for the respective single mutants (2.5 mmol/L H2O2), a simultaneous loss of both
General mechanisms of yeast adaptation to stress and hormetic response to H2O2 can be suggested to involve enzymes such as GR, G6PDH, and GLO1. Glutathione reductase plays a crucial role in maintaining the proper range of such important low-weight antioxidant agents as reduced glutathione. The thiol group of reduced glutathione can directly or in concert with other antioxidants reduce and detoxify reactive oxygen species. 28,65 Glucose-6-phosphate dehydrogenase produces the reduced equivalents NADPH, which are used by GR to reduce oxidized glutathione, and therefore G6PDH also plays a significant role in cell protection against H2O2 as well as other reactive species. Besides its antioxidant role, reduced glutathione is an important part of antiglycation system that eliminates reactive carbonyl metabolites, which continuously produced during metabolism of carbohydrates. 13,27,43 Especially, utilization of fructose can lead to high generation of reactive metabolites. 11,12,14 For instance, reduced glutathione is consumed in the reaction catalyzed by GLO1, the fundamental function of which is the metabolism of reactive carbonyl species to less reactive products. 27,28 In general, the enzymes mentioned previously are important in the metabolism of carbohydrates as well as H2O2, 13,27,42,43 however little is known about their role in hormetic response as well as functional connection with TOR.
Since GR is a member of H2O2 stimulon in
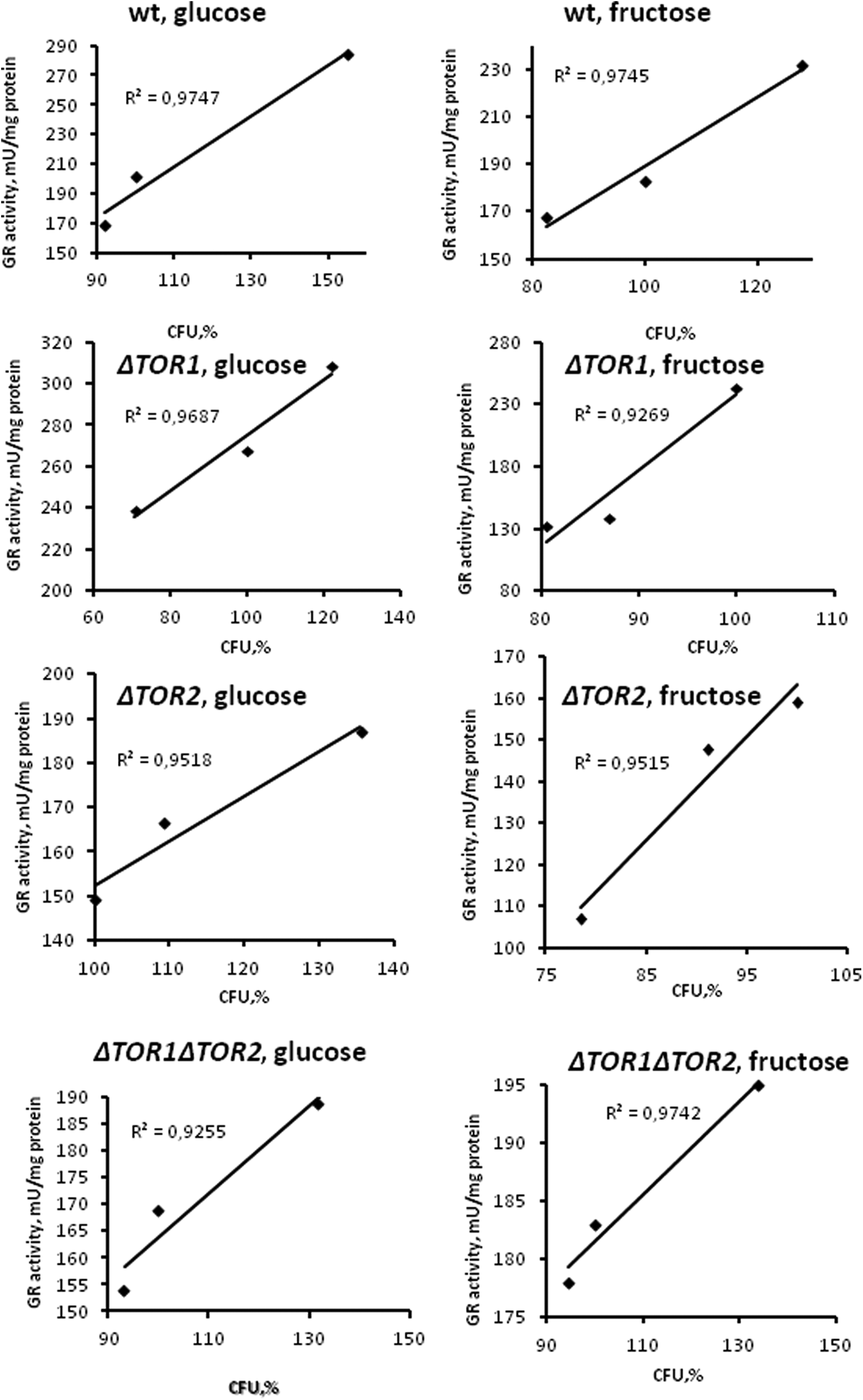
Correlation analysis of data obtained with wild type and target of rapamycin (TOR)-deficient
Activity of G6PDH, another member of H2O2-stimulon in
In spite of the functional relationship between GR, G6PDH, and GLO1, the last one demonstrates a pattern of the activity (Figure 4) quite different from GR (Figure 2) and G6PDH (Figure 3). Moreover, when GLO1 activity was plotted against the activity of G6PDH, a strong negative correlation between the parameters can be seen (Figure 6). Unlike G6PDH, GLO1 activity in both

Correlation analysis of data obtained with wild type and target of rapamycin (TOR)-deficient
In summary (Figure 7), several interesting conclusions can be drawn: (1) H2O2-induced hormetic response in

Relationship between hydrogen peroxide (H2O2)-induced hormetic response in
Footnotes
Acknowledgments
The authors are grateful to Prof Michael Hall for providing the yeast strains and Dr Liudmyla Lozinska for critical reading of the manuscript. H.S. would like to express sincere gratitude and appreciation to Hmeleva Humanitarian-Investment Project and Mr Vladyslav Kyrychenko for support and providing creative work atmosphere, in which the manuscript has been prepared. Two anonymous referees are gratefully acknowledged for their highly professional and helpful work improving the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
