Abstract
The identification of reproductive toxicants is a major scientific challenge for human health. Prenatal life is the most vulnerable and important time span of human development. For obvious ethical reasons, in vivo models cannot be used in human pregnancy, and animal models do not perfectly reflect human physiology. This review describes the in vitro test models representative of the human feto–maternal interface and the effects of environmental chemicals with estrogen-like activity, mainly bisphenol A and para-nonylphenol, with a particular emphasis on the effects at low, nontoxic doses similar to concentrations commonly detected in the population.
The Feto–Maternal Interface: A Complex Network of Endocrine, Paracrine, and Autocrine Factors
Human pregnancy is a complex and finely regulated process during which 2 genetically different organisms, the mother and the embryo/fetus, establish a very intimate contact. 1 –3 A huge number of molecules are secreted at the feto–maternal interface by both the mother and the embryo/fetus and act on both sides. 4 Thus, the mother and the embryo interact via specific tissues in a reciprocal exchange of molecules that act as communication signals.
Placentation in human pregnancy involves invasion of the blastocyst in maternal endometrium up to the spiral arteries. The trophoblast cells, which surround the blastocyst, first contact the uterine epithelium, then dislodge the endometrial epithelial cells, and invade the maternal endometrium. During its journey, the trophoblast follows 2 differentiation pathways—the villous and the extravillous ones (Figure 1). 5 In the villous pathway, the mononuclear cytotrophoblast cells forming the internal layer of villi fuse and form the multinucleated syncytiotrophoblast, which is the epithelial covering of the chorionic villi (floating villi). In the extravillous pathway, the cytotrophoblast cells move beyond the overlying syncytium and form multilayer cell columns of extravillous trophoblast that fix the villi to the maternal tissues (anchoring villi). These cells then move deeper into the maternal tissues up to the proximal third of the myometrium and endometrial spiral arteries (Figure 1).

Villous and extravillous trophoblast in human placentation.
Human endometrium has a key role for reproductive efficiency, and its remodeling takes place under the control of steroid hormones. 6,7 This tissue undergoes dramatic cyclic changes regarding cell proliferation, secretory functions, regression, and regeneration. Hormonal stimuli lead the maternal tissues to the formation of the decidua at the end of each cycle. If fertilization has occurred, the blastocyst reaches the uterine epithelium and implantation starts.
The correct signaling between the decidua and the fetal trophoblast is of paramount importance for blastocyst implantation and successful pregnancy (Figure 2). 2,7

The interplay of endocrine and paracrine mediators at the feto–maternal interface. hCG indicates human chorionic gonadotropin; GFs, growth factors; PGs, prostaglandins.
In the complex scenario of the feto–maternal interface, endocrine, paracrine, and autocrine factors must be taken into consideration. Hormones, such as estrogens, progesterone, and human chorionic gonadotropin (hCG) are the major players of the endocrine regulation that take place at the feto–maternal interface. Human chorionic gonadotropin, which is the hormone produced by the trophoblast in the very early stages of pregnancy, plays a key role in making sure that the endometrium is ready to receive the embryo implantation. 8 Those molecules which have an autocrine/paracrine action are important factors in the establishment and advancement of pregnancy. 9 Among these are cytokines including the interleukins (IL)s IL-1, IL-4, IL-5, IL-6, IL-8, and IL-10, the macrophage migration inhibitory factor (MIF), colony stimulating factors (CSFs) and the leukemia inhibiting factor, and different growth factors (GFs) such as the epidermal GF and the vascular endothelial GF. All these molecules are potent immunoregulators that play a key role in the feto–maternal tolerance to the semiallogeneic embryo. 10 These molecules are also mediators of cell proliferation/differentiation and apoptosis contributing to fetal growth and expansion in the maternal tissues. 11,12 Therefore, their presence at the feto–maternal interface may modulate the maternal immune response and contribute to the expansion of fetal tissues in the maternal uterus.
Prostaglandins (PGs) also play a pivotal role in angiogenesis, mitogenesis, cell proliferation, and differentiation and exert an important role during the early stages of pregnancy. 7,13,14
Environmental Contaminants: A Threat for Human Reproductive Health
There are increasing data that support the adverse effects on human reproductive health caused by environmental contaminants, especially endocrine-disrupting chemicals. These chemicals are man-made or plant-derived compounds that can bind to the receptors of steroid hormones and thus impair hormone-driven physiological functions in female and male reproductive system. 15,16 Among these, diethylstilbestrol (DES), a nonsteroidal estrogenic compound, has been commercialized as therapeutic for reproductive disorders. Regrettably, only after its commercialization its deleterious effects became evident. Among industrial compounds is bisphenol A (BPA), a polymer that can be released by polycarbonated plastics and by the linings of metal cans that are used for food and beverages. 17,18 Consequently, BPA can be easily absorbed via the food chain, 19 and indeed it can be easily detected in the human body. With regard to pregnancy, levels of BPA have been detected in the placenta, fetal liver, in the blood, and in the follicular fluid in a range of 0.3-40 nmol/L. 18,20,21 Bisphenol A shares many similarities with the endogenous estrogens 22 and acts on estrogen-responsive organs by binding to estrogen receptor (ER) isoforms, ERα and ERβ. 23,24 The estrogenic activity of BPA has been demonstrated in Ishikawa cells, an endometrial cell line. 6 Bisphenol A also binds to the progesterone receptors (PRs) and thus exert antiprogestin activity. 25,26 Furthermore, BPA is able to mimic glucocorticoids, another class of steroid hormones fundamental at the feto–maternal interface. Bisphenol A is indeed able to bind with the mineralocorticoid and glucocorticoid receptors (GRs). 27,28 Interestingly, it has been demonstrated that environmental concentrations of BPA (10 nmol/L) increased the messenger RNA (mRNA) expression and enzymatic activity of the enzyme 11β-hydroxysteroid dehydrogenase type 1 in the adipose tissue and the visceral adipocytes isolated from children. 29 Furthermore, perinatal exposure to BPA resulted in a decreased expression of GR in the brain of female rats compared to controls. 30 This environmental chemical was listed as reference compound within the European ReProTect Programme because of its well-known reproductive toxicity. 31,32 Another environmental chemical derived from the manufacturing industry is para-nonylphenol (p-NP), an alkylphenol applied as a plasticizer and surfactant, known to have estrogenic activity since 1991. 33 Human exposure to p-NP may occur by cutaneous absorption, ingestion of contaminated food or water, and inhalation. 34,35 Estrogenic activity of p-NP has been known since 1991 33 and reported in a number of in vitro 36 and in vivo studies. 37 Interestingly, studies in rats showed that maternal exposure to p-NP resulted in an increase in uterine calbinding-D 9k (CaBP-9k) mRNA and protein expression in maternal and neonatal uteri, suggesting its potential transfer through the placenta. 38,39 CaBP-9k is a cytosolic calcium-binding protein expressed in various tissues (eg, intestine, uterus, and placenta) and a marker of estrogenic compounds exposure. 40
Since the number of man-made chemicals released in the environment have been growing exponentially in the last decades, 41 the assessment of the risks that derive from the exposure to chemicals is of paramount importance. 31
Risks Assessment of Intrauterine Exposure
Because of their ability to interfere with steroid hormones and their widespread distribution in everyday products, many of the chemicals in the environment represent a hazard to the reproductive system of adult women. Much greater concern arises when we take into account prenatal exposure to these bioactive compounds. Prenatal life is indeed the most critical phase of the life cycle, as it is the period in which organs and tissues are formed. 42
Already during the periimplantation period, the intrauterine environment is able to provoke epigenetical changes that will persist later in life. 43 Barker and coworkers initiated the investigation of the developmental origins of many diseases that appear in later life. 44 Then many researchers followed, revealing a dramatic scenario in which alterations in the intrauterine environment can result in pathologies and dysfunctions in adulthood. 45
For many decades, the human placenta has been considered a protective organ. It was assumed that the placenta interdicted the passage of harmful compounds to the fetus. However, the placenta has not been adapted to act as a barrier to the many substances that have been produced by man in the last decades. Therefore, the human placenta might not be able to prevent fetal exposure to these potentially dangerous compounds. Indeed, due to their high lipophilicity, many environmental estrogens are able to elude the placental barrier and potentially harm the fetus. 46 Moreover, the placenta is a highly sensitive tissue to environmental contaminants with estrogenic activity as it expresses both ERα and ERβ. 47 Since the placenta is the fundamental organ to maintain pregnancy and assure fetal development and growth, it seems to be extremely important to estimate the risk of exposure during in utero life by evaluating the effects of these contaminants on the placenta and the uterus. Animal models remain a solid and important shield to study human pregnancy establishment and development, but, unfortunately, they often fail to completely reproduce the complexity of human reproduction. Many efforts have been taken in order to shed light on the physiological, pathological, and toxicological aspects of human pregnancy, and in vitro models represent a valid help for researchers in this field.
In this report, we will mainly focus on BPA and p-NP as representative chemicals with estrogen-like activity. Special emphasis will be put on the effects at low doses that are relevant with or are lower than those found in the environment and in human tissues. In fact, it is important to underline that environmental chemicals with estrogenic activity can have different effects at different doses. 6 In particular, some of these compounds have their detrimental effects at very low concentrations while higher concentrations are not effective in the same way. 48 –50
In vitro Models of Human Placenta
Many efforts have been made to develop in vitro models for studies of the placenta. It is indeed important to emphasize that animal placenta does not model the human placenta and its physiology well. As human placenta is, in fact, an organ that varies from one species to another, therefore the use of animals could not be appropriate to study the mechanisms that take place during human pregnancy. Moreover, for obvious ethical reasons, studies on human placenta can be performed only in tissues and cells obtained from the organ after its delivery. Thus, in vitro models need to be developed to investigate placenta establishment and development in the maternal uterus as well as to identify factors influencing these physiological processes. These models mainly include trophoblast cells and cultures of placenta villous explants.
Freshly isolated cytotrophoblast cells can be obtained by enzymatic dissociation of villous placental tissue, followed by Percoll gradient separation. These cells are able to differentiate into multinucleated syncytiotrophoblast cells when cultured in complete medium. However, they do not proliferate in vitro and thus cannot be cultured for a long time. 51,52 For these reasons, many immortalized and carcinoma-derived cell lines (BeWo, JAr, JEG-3, and HTR-8/SVneo) have been set up to study selected aspects of the human placenta in vitro. 53 For example, the human trophoblast cell lines BeWo and HTR-8/SVneo cells are representative of specific differentiation pathways of trophoblast during placentation (Figure 3). In particular, the choriocarcinoma-derived BeWo cell line is representative of the villous pathway since it reveals most of the characteristics of the villous syncytiotrophoblast including cell fusion 54 and secretion of hormones such as the β-hCG. 55 The HTR-8/SVneo cells are representative of the invasive extravillous trophoblast, specifically of those cells which, after detaching from the chorionic villi, migrate to and infiltrate into the maternal decidua. These cells are originated from human first-trimester human placenta and in vitro immortalized by transfection with a complementary DNA construct that encodes the simian virus 40 large T antigen. 56 Based on the cells’ specific characteristics, any toxic effect observed on BeWo cells may forecast impairment of placenta growth, while any effect toward HTR-8/SVneo cells may contribute to an unsatisfactory blastocyst implantation and placentation in vivo.

Specific differentiation pathways of the BeWo and the HTR-8/SVneo trophoblast cells.
However, despite the high reproducibility of the experiments provided by these cell lines, the results obtained can be far from the in vivo situation. Furthermore, cell lines enable the study of a single cell type at a time, while the human placenta consists of many cell types. Some ex vivo models offer the opportunity to maintain all the main placental cell types in the same culture. 57,58
A suitable in vitro model more closely resembling the in vivo pattern of trophoblast differentiation is offered by primary cultures of placenta explants from fresh human placenta. 57 This model includes dissection of terminal villi from human placental tissues (Figure 4). A variety of culture conditions can be used to reproduce in utero environments at different times of gestation. Usually, explants from placental tissues at first-trimester pregnancy are cultured on a bed of Matrigel which the tissues adhere to and then expand, mimicking placenta establishment and its development in the maternal uterus. Explants from term placenta are usually cultured on the bottom of the well or as free-floating villi, hanging on a supporting device. This type of culture mainly reflects the syncytialization of the villous trophoblast. 57

Dissection and culture of placental explants. Representative villous explants culture from 9 weeks’ gestation. Fresh placenta is dissected and each villous fragment (15-20 mg wet weight) is placed in a 24-well culture plates (Ø 15.6 mm) previously coated with Matrigel. Villous pieces are then covered with a proper medium and cultured under different experimental conditions. Bar = 3 mm.
The model of placental explants presents several advantages: First, unlike in the one with isolated cells, this model preserves the topology of intact villi including all the main fetal cell types in the chorionic villi, for example, the syncytio- and the cytotrophoblast, the villous stromal, and endothelial cells, thus preserving each mechanism of paracrine regulation. Second, this model mimics the trophoblast functions more realistically, including production and release of secretory components, proliferation, growth, and differentiation. Third, placental explants can be set up with tissues from early and term placenta, making it possible to compare the effects of substances both at early and term pregnancy. The main disadvantage of this model is its inability to distinguish among the functional roles of the various cell types present in this organ. Overall, the model of placental explants allows to study all aspects of placental biochemistry and molecular biology characterizing the physiological processes during the organ development, pharmacology, toxicology, and disease processes.
Other in vitro models of human placenta have been set up to study placental transfer. 58 Some of these models exemplify the transfer across a single cell layer while others retain all the components of the placental barrier, the syncytiotrophoblast, the cytotrophoblast, the basal membrane, and the endothelial layer. 32 Among the last type of models, the placental perfusion technique can be found. Thanks to the particular anatomy of the human placenta which consists of many circulatory units (cotyledons; Figure 5), a single cotyledon can be isolated and perfused. 59
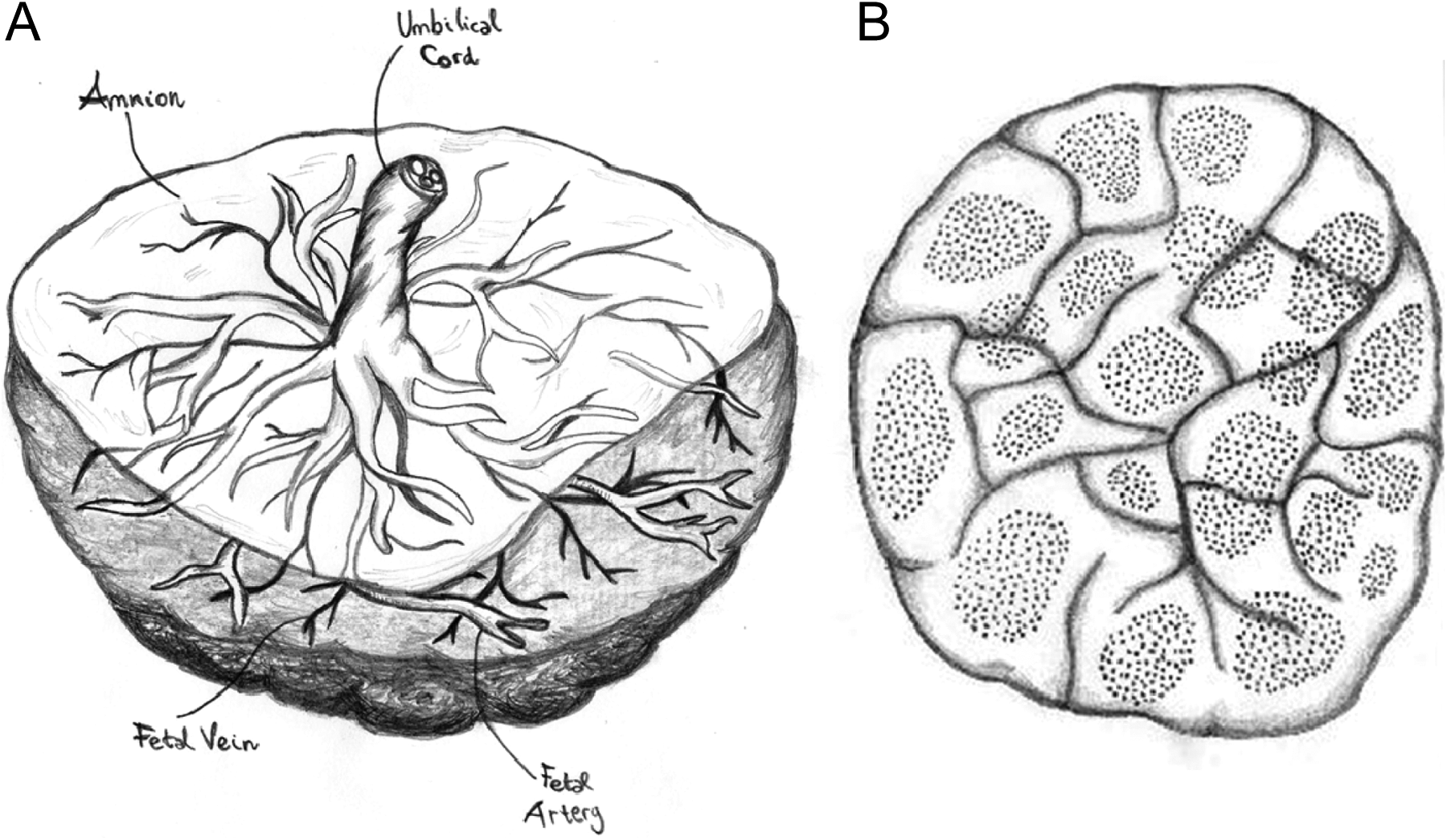
Anatomy of human placenta. A, Fetal side: the amnion, umbilical cord, and fetal vessels are visible. B, Maternal side: the cotyledons and their circulatory units are visible. Images by Chiara Mannelli.
Potential Biomarkers in Human Placenta
The human placenta is an exquisite tool to study the effect of environmental chemicals on human reproduction, due to the key role of this organ in maintaining pregnancy and providing fetal health. Toxicity of chemicals needs to be tested in order to identify concentrations suitable for evaluating potentially deleterious effects in the functional processes of the placenta. Indeed, it is important to separate the effects of toxic doses from the ones that nontoxic doses might have on a biological system. To do so, in vitro models can be tested for cell viability and tissue damage in a wide range of chemical concentrations. All the immortalized and carcinoma-derived cell lines are easily cultured and propagated in vitro, and they are generally used for large-scale screening of many chemicals and/or the effects of their concentrations.
Once the curve of toxicity is obtained, functional studies can be conducted using nontoxic doses. Furthermore, in order to make these studies closer to the in vivo situation, concentrations of chemicals similar to those detected in the human body should be privileged. As reported in the literature, BPA levels in human pregnancy ranged from 0.3 and 40 nmol/L in adult and fetal serum, follicular and amniotic fluid, and placenta. 18,20,21 The p-NP levels ranged from 0.1 nmol/L in cord blood and 1 nmol/L in maternal plasma and blood. 60 –62
Among the trophoblast cell types, BeWo and HTR-8/SVneo cells are suitable to detect the specific effects of chemicals with estrogenic activity in human placenta since both of them express ERs. 63,64 Moreover, given their different differentiation pathways, these cells can be monitored for different biomarkers. Specifically, BeWo cells can be tested for the secretion of β-hCG and tissue expression of cleaved caspase-3, specific markers of endocrine activity of the syncytiotrophoblast and its apoptotic shedding, respectively. 62,65 The HTR-8/SVneo cells can be monitored by assaying the passage of these cells through a layer of collagen (cell migration) or matrigel (cell invasion) as well as by the release of metalloprotease (MMP)-2 and MMP-9, the most studied MMPs for trophoblast invasion. 66 The use of 2 different cell lines representative of the main differentiation pathways in human placenta provides information about the effect of chemicals in the whole process of placentation.
In the last decade, several studies of BPA and p-NP in in vitro models of human placenta have been conducted. Studies in BeWo and HTR-8/SVneo cells showed that these chemicals caused a decrease in cell viability in the concentration range of 1 µmol/L and 1 mmol/L. Surprisingly, low concentrations (pmol/L-nmol/L), not affecting cell viability, impaired functional markers of placentation. More specifically, both chemicals, p-NP and BPA, appeared to affect syncytialization of trophoblast as they both altered hCG secretion and cell apoptosis in BeWo cells. 32,47 Interestingly, the effect of estrogen-like chemicals, at low nontoxic concentrations, resulted in a change in hCG release with an hormetic or biphasic behavior. 67 The chemicals studied were indeed stimulating or inhibiting hCG release depending on their concentration. 67 More recent evidence in HTR-8/SVneo cells showed that cell migration and invasion were also reduced by BPA and p-NP. 50 For each chemical, the activity was higher at lower concentrations with a maximum activity between 0.1 and 10 pmol/L. Coculture studies of HTR-8/SVneo with human umbilical cord endothelial cells (HUVECs) revealed that trophoblast/endothelial interaction was significantly reduced by p-NP at 10 pmol/L.
All together, the above-mentioned studies on trophoblast cells indicate that low concentrations of the environmental chemicals BPA and p-NP are able to affect the main differentiation pathways of trophoblast, the villous, and extravillous ones.
It is noteworthy that effective concentrations of p-NP in hCG secretion were lower in BeWo cells (0.1-1 pmol/L) than in placental explants (1 nmol/L). 47,48 This discrepancy can be explained by a different pharmacokinetic behavior in the 2 different models probably due to a more complex organization in a tissue culture with respect to a cell monolayer.
Placental explants give the chance to monitor, with a closer look into the physiological situation, trophoblast differentiation both into syncytiotrophoblast and extravillous trophoblast. This model is also informative about the endocrine secretory activity of placenta (ie, secretion of hCG, placental lactogen, cytokines, GFs, and other paracrine/autocrine molecules produced by the placenta). 47,48 In particular, cytokines such as granulocyte macrophage-CSF (GM-CSF), interferon γ (IFN-γ), IL-1β, IL-4, and IL-10 were all increased by pmol/L to nmol/L p-NP with a maximum effect at 10 pmol/L, which was statistically significant for GM-CSF and IL-10. 47,48 Bisphenol A (1 nmol/L) was increasing the secretion of the cytokine MIF while BPA levels ranging from 0.2 to 2 nmol/L, increased tumor necrosis factor-α gene expression, and protein secretion. 49,68
Perfusion experiments on human placenta are a wonderful tool to investigate the transfer of substances. This model showed the transfer of BPA across the placenta. 32 Furthermore, placental perfusion can be used to study the placental metabolism of many chemicals and drugs, the role of transporters as well as the effects of acute toxicity in placenta (for detailed review, see Myllynen and Vähäkangas, 58 and Myllynen et al studies 69 ).
The effects of representative substances such as BPA and p-NP, in the scenario of environmental contaminants, raise great concern for the environmental risk to the health of the fetus. Even though they do not cause termination of pregnancy, low concentrations of these substances could produce anomalies in the functional processes of the placenta, leading to possible disorders and/or abnormalities later on in pregnancy. For example, higher levels of cytokines such as MIF in the maternal serum and increased levels of hCG in the second trimester of pregnancy, have been associated with preeclampsia, a serious syndrome of human pregnancy. 70 –73
In vitro Models of Human Endometrium
The human endometrium is a fertility-determining tissue. It consists of various cell types. Epithelial cells and glandular-epithelial cells are located in the lining of the uterine lumen. Stromal, endothelial, and immune cells are mainly located in endometrial stroma. The cell types that reside in the endometrium result to be more sensitive to steroid hormones compared to the cell types of other tissues. 31,74 This raises concern about the possible deleterious effects of hormone-mimicking compounds on the cycling and pregnant human endometrium. 16,75
Primary cells from human endometrium can be easily cultured and are able to proliferate in vitro. 76 These primary cultures of endometrial cells respond to hormonal stimuli but, unfortunately, tend to dedifferentiate after several passages and therefore cannot be cultured for long periods. 77,78 Immortalized and carcinoma-derived cell lines, such as Ishikawa cells (derived from epithelial endometrial cells) and St-T1b (derived from stromal endometrial cells), are also available. 6,78,79 These cell lines are easily cultured and maintained for long periods, but they often lose responsiveness to hormonal stimuli and this can lead to misleading results. In vitro models of human endometrial epithelium are used to study blastocyst attachment and generally to focus on the very first steps of pregnancy establishment. Cell models of endometrial stroma are a powerful tool to study trophoblast invasion in the maternal uterus. Furthermore, such models are often used to study endometrial pathologies such as endometriosis. 80 Endothelial stromal cells can also be used to investigate the physiological and pathological mechanisms that take place in the human endometrium. Unfortunately, primary endothelial cells from human endometrium tend to lose their hormonal responsiveness after few passages. 74 Nevertheless, endothelial cells of uterine origin are far more sensitive to hormonal stimuli than endothelial cells from other tissues, that is, HUVECs, and thus tissue-specificity should be always considered when selecting an in vitro model. 74 Models of tissue explants from human endometrium have been also used. 81 –83 These can be obtained from the decidual fragments remaining in the human placenta after elective termination of pregnancy or after delivery at term.
More recently, three-dimensional (3D) models of human endometrium have been established and are gaining increasing interest since these models are closer to the in vivo situation. Indeed, endometrial cells show different features when grown in 3D matrices compared to cell monolayers. 76,77,84
Potential Biomarkers in Human Endometrium
Among the available in vitro models of human endometrium, cultures of endometrial cell lines are the most commonly used in reproductive toxicology. As reported above about the studies of placenta, toxicity tests are needed to provide guidance for the range of concentrations suitable for functional testing in human endometrial physiology. In order to achieve this, viability and cell proliferation assays, together with assays of tissue damage (ie, such as lactate dehydrogenase measurement), are recommended. Cell lines, such as Ishikawa cells, are a powerful tool for screening the effects of environmental chemicals on the human endometrium. 6,85,86 Using these cells, Schaefer and co-workers 6 provided very interesting data on the effects that DES, BPA, and p-NP can have on endometrial receptivity, which is a key step for establishment of pregnancy. What is more, these authors investigated the effects of these substances in a wide range of concentrations, thus unraveling the dose–response effects of these chemicals on the endometrium. 6 Transfected Ishikawa cells also provided interesting insights into the estrogen-like activity of many environmental pollutants, such as DES, BPA, genistein, and o-p′-dichlorodiphenyltrichloroethane by monitoring the effect of these chemicals on the activation of estrogen response elements. 86
In vitro models of endometrial epithelial cells provide important insights into the effects of exogenous compounds on the binding surface that allows blastocyst attachment. Indeed, the expression of important surface molecules, such as integrins and osteopontin, after exposure to environmental chemicals, could represent a very good marker in order to assess the effect of chemicals on uterine preparation to blastocyst attachment. Many studies proved that the expression of such molecules is hormone sensitive and can be altered by exogenous hormone-mimicking compounds in vivo. 87 –89
On the other hand, endometrial stromal cells are useful to study the intimate mechanisms that prepare the uterus for a gestation and that could be disrupted by hormone-mimicking compounds. Primary cultures of endometrial stromal cells proved to be an effective tool to study the potential deleterious effects of BPA. 49,90,91 For this purpose, the expression of hormone receptors, that is, ERs, PRs, hCG/luteinizing hormone receptor, together with the secretion of paracrine/autocrine factors (ie, cytokines, PGs, and GFs) can be considered a powerful approach to assess the effect of environmental chemicals on the stromal compartment of the endometrium. Many authors investigated the effects of dietary relevant, low doses of phytoestrogens, such as genistein and daidzein, on endometrial fibroblasts by assessing the ability of these compounds to inhibit aromatase activity or cell proliferation. 92,93
Cultures of endometrial endothelial cells are useful to study the specific angiogenetic processes that take place in the uterine environment. The potentially negative effects of BPA or genistein on the endometrium have been tested on primary cultures of endothelial cells by checking cell viability, cell proliferation, and angiogenic activity via the tube-formation assay. 94,95
Furthermore, endometrial coculture systems have been used to investigate the effects of phytoestrogens on the important interactions between epithelial and stromal cells, such as ER activation and cell proliferation. 96 Endometrial explants are a natural coculture system of endometrial cells, which could be used for reproductive toxicology studies, even if they are, at the moment, applied mostly for physiological studies. 97,98
Coculture Models of Feto–Maternal Interface
Coculture models including cells and/or tissues representative of both the fetus and the maternal counterpart have been developed. 76,81 –83 Many of these focus on the mechanisms of blastocyst implantation. Usually, human blastocyst is mimicked by spheroids of placental cells 76,77 that are allowed to attach to monolayer cultures of endometrial cells or 3D matrices.
Other coculture models allow the study of a wider spectrum of interactions at the feto–maternal interface. 81 –83 Such approaches mainly focus on the invasion of human placenta inside the maternal decidua. To mimic this situation, ex vivo explants of human placenta and decidua have been utilized. 81,82,99 These coculture models are a powerful tool as they include all the main cell types involved at the feto–maternal interface. They nevertheless require synchronization of primary cultures from tissues which are sometimes difficult to obtain. In order to overcome these limitations, Mannelli and co-workers 49 recently applied an in vitro system that could be helpful to study the molecular interactions at the feto–maternal interface even in laboratories that do not have availability of fresh tissues. Indeed, in their article, the authors presented an in vitro model in which explants of chorionic villi were exposed to an endometrial stromal cell-conditioned medium collected in another laboratory. 49 Such encouraging approach could widen the possibilities of different research groups across the world, and many similar efforts have been taken in the last years. Indeed, Huppertz and co-workers 100 developed the method of cryogenic preservation of placental explants in order to overcome the paucity of this tissue in other laboratories.
Potential Biomarkers in Coculture Models of Feto–Maternal Interface
Coculture models of feto–maternal interface could represent a useful end point of toxicological studies, in which the effects of chemicals are tested under more complex and physiological conditions. These models have been largely used to investigate physiological processes of pregnancy 76,81 –83 while only little has been reported for toxicological studies. Using the model of endometrial stromal cell-conditioned medium and placental explants, Mannelli and co-workers 49 demonstrated that the maternal compartment is able to protect the placenta from the effects of BPA. The study indeed showed that direct exposure of placental explant cultures with very low BPA concentrations (0.5-1 nmol/L) triggered the secretion of MIF and β-hCG and that these effects were abolished/diminished in the placental cultures exposed to cell-conditioned medium from endometrial stromal cells pretreated with BPA. Although the exact mechanism/s of maternal protection are not known, the data highlight the importance of in vitro models reproducing the complex interactions between the mother and the fetus to verify the effect of environmental chemicals in pregnancy. What is more, the choice to use very low concentrations of BPA revealed how concentrations in the range of 1 nmol/L are able to trigger a significant physiological unbalance in reproductive tissues. 49
Concluding Remarks
Although not fully respecting the in vivo situation, in vitro models are informative about the effect of chemicals at the maternal–fetal interface and can give fruitful insights about the possible correlations between environmental pollutants and reproductive disorders. However, given the dynamic and complex mechanisms responsible for blastocyst implantation and placenta development in the maternal uterus, it is difficult to classify chemically induced alterations as adverse or nonadverse effects. One can only state that chemicals that are widely distributed in the environment and present in daily used products such as BPA and p-NP have the potential to interfere with the physiological processes of preparation of pregnancy and placentation. What raises great concern is that chemical concentrations lower than those causing cell death can alter fundamental biological processes such as uterine receptivity and placenta development. This might indicate that maternal contamination with these types of chemicals, at concentrations that do not cause termination of pregnancy, may cause dysfunction in pregnancy and fetal development. Moreover, as prenatal life is a critical period of life in which the body is formed and develops, any abnormality in its development will have negative consequences for adult life. This review aimed to give an exhaustive overview of all the in vitro models available and of their current use. Our hope is that more complex models will be applied to studies of environmental toxicology, and that the use of low concentrations of chemicals in toxicological tests will become of common use. Indeed, due to the limitations of in vitro models, it is important to prepare an experimental design that is as close as possible to the in vivo situation. The different dose–response effects and the tissue-specific effects of chemicals should be always considered as well. 6,91
In conclusion, the data reported here raise great concern for the environmental risk to pregnancy, indicating the need to develop more accurate tests for the protection of both the prenatal life and the development of the fetus.
Footnotes
Acknowledgments
This study has been supported by NSC Grant Preludium 2013/09/N/NZ5/03062 and from Funds at the Department of Life Sciences of the University of Siena, Siena (Italy).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
