Abstract
Although bisphenol A (BPA) has been associated with impaired spermatogenesis, the mechanisms remain unclear. Tight junction occludin plays important roles in spermatogenesis. The objective of the present study was to explore the effects of BPA exposure in adolescent mice. Male mice were orally treated with low-dose (0.05 mg/kg/d), middle-dose (5.0 mg/kg/d), or high-dose (50 mg/kg/d) BPA in corn oil from postnatal day (PND) 35 to 65. Animals were killed on PND 65 and PND 125. On PND 65, the sperm count, sperm motility, and the expression of occludin showed a dose-related decline. On PND 125, the sperm count, sperm motility, and the expression of occludin were in recovery. However, there remained significant decreases in these parameters in the 50 mg/kg/d group on PND 125 compared with the control. The dose-related effects on the measured parameters and occludin expression suggest an early suppressive or damaging effect on the blood–testis barrier followed by recovery after dosing ceased. At a BPA dose of 50 mg/kg/d, recovery did not occur, suggesting that higher doses of BPA may cause irreversible damage to reproduction in male mice.
Introduction
Bisphenol A (BPA) is an endocrine disrupter and has generated significant concern due to its potentially negative health effects. Over 8 billion pounds are produced each year, and more than 100 tons are released into the atmosphere annually. Human exposure to BPA is increasing.
Exposure to BPA during pregnancy may impair fetal growth in human. 1 Men’s exposure to BPA may be associated with lower semen quality and increased DNA damage in sperm. 2 Bisphenol A can impair Sertoli cells 3 and spermatogenesis in rats. 4 As a result, BPA exposure has gained considerable attention from the scientific community and the public. As a protective measure, the US Environmental Protection Agency and the European Food Safety Agency have established a tolerable daily intake (TDI) of 50 μg/kg/d, applying an uncertainty factor of 100 to the no observed adverse effect level (NOAEL) of 5 mg/kg/d. 5
The blood–testis barrier (BTB) is an important ultrastructure in the seminiferous tubules of mammalian testes. The BTB consists of coexisting tight junctions (TJs), basal ectoplasmic specialization (ES), and gap junctions between Sertoli cells near the basement membrane. It segregates the seminiferous epithelium into the basal and the adluminal (apical) compartments, which allow post-meiotic germ cell development to take place in a unique microenvironment. This prevents the production of antispermatid antibodies, many of which are expressed transiently during spermiogenesis. 6 The BTB also restricts paracellular flow and/or transport of biomolecules, water, electrolytes, ions, and hormones across the barrier, as well as maintaining cell polarity in the epithelium. 6 Tight junctions are the only known example of occluding junctions composed of integral membrane proteins, namely occludins, claudins, and junctional adhesion molecules. 7 Occludin plays an indispensable role in TJ function in the testis, demonstrated by a targeted mutation in the occludin gene causing male sterility. 8 Previous research has shown that BPA can lead to a reduction in occludin, ZO-1, β-catenin, and androgen receptor. 9
The main aim of this study was to explore the effects of the current “safe” dose of BPA (TDI) and a higher dose of BPA on TJ protein occludin and semen quality. We attempted to clarify whether BPA is a reproductive toxicant using adolescent mice model and also demonstrate the possible underlying mechanism.
Materials and Methods
Animals
Male Kunming mice (35 days old) were purchased from the Chinese Food and Drug Administration and were housed under controlled temperature (23 °C ± 2 °C), lighting (12-hour light and 12-hour dark cycle), and relative humidity (55% ± 5%) conditions. As soybean-derived diets have the potential to influence the results of studies aimed at measuring endocrine-related effects, 10 a soy-free breeding diet and reverse osmosis water were provided ad libitum.
Study Design
Sixty-four 35-day-old male Kunming mice were divided into 1 vehicle control group and 3 treatment groups with 16 mice for each group. The treatment groups were given low-dose (0.05 mg/kg/d, TDI, 16 mice), middle-dose (5 mg/kg/d, NOAEL, 16 mice), or high-dose (50 mg/kg/d, 16 mice) BPA (Sigma Chemical). The BPA was dissolved in corn oil and given via oral route (10 mL/kg) during postnatal days (PNDs) 35 to 65. Food was suspended 12 hours before the end of the experiment, and water was supplied until the animals were euthanized. The mice were killed humanely with carbon dioxide. Half the animals for each group (32 mice) were killed on PND 65 and the other half for each group (32 mice) on PND 125. One testis from each rat was fixed in Bouin’s solution for histopathological evaluation, and the other testis was snap-frozen in liquid nitrogen and stored at −80 °C until used for Western blotting.
Assessment of Spermatozoon Count, Motility, and Viability
Spermatozoa were collected from the caudal epididymis. Immediately after collection, samples of the epididymis were placed into a prewarmed Petri dish with modified Hank’s balanced salt solution for 10 minutes at 37°C. The solution was then gently filtered through nylon gauze and centrifuged for 5 minutes at 5009g. The deposit was resuspended in 1 mL of M199 medium (Abcam Chemical). Ten microliters of sperm suspension were uniformly placed into a warmed sheet glass, and the mean number of viable spermatozoa in at least 4 of the random squares was counted. The spermatozoa motility was evaluated with a spermatozoon motility analyzer (Weili Medical Treatment). Those with fast progressive motility (greater than 20 μm/s) were labeled as level A, and those with modest motility (less than 20 μm/s, greater than 5 μm/s) were marked as level B.
The sperm viability was assessed by eosin-nigrosin staining. Smears were prepared by mixing 1% eosin (0.01 g/mL), 10% nigrosin (0.1 g/mL), and the spermatozoon suspension on a microscope slide, which was allowed to air-dry at room temperature. The slide was then analyzed using a standard optical microscope, as previously described. 11 Sperms with a partial or complete pink were interpreted as dead and only unstained sperms were considered to be viable. The viability of spermatozoa was expressed as a percentage.
Immunohistochemistry
Expression and localization of occludin in the seminiferous epithelium were determined by immunohistochemical analysis. Cross-sections (3 μm) of paraffin-embedded testes were deparaffinized and rehydrated, and antigens were unmasked by heating in 10 mM sodium citrate solution. Sections were incubated with 3% H2O2 to block endogenous peroxidase activity. Primary antibodies used in this study were rabbit antioccludin (1:100; Abcam Inc, ab31721). Sections were incubated overnight in primary antibodies at 4 °C. After washing 5 times in phosphate-buffered saline, the sections were incubated with biotin-labeled goat antirabbit IgG (Beijing Zhongshan Biotechnology Co) for 1 hour at 37 °C. The negative controls were incubated with phosphate-buffered saline.
Western Blotting Analysis
The testes were homogenized in a 5-fold volume of ice-cold lysis buffer containing 50 mmol/L Tris–HCl, 150 mmol/L NaCl, 0.1% sodium dodecyl sulfate (SDS), 1% NP-40, 0.5% sodium deoxycholate, 1% EDTA-2Na, and 2% (vol/vol) protease inhibitor cocktail. Samples were schizolyzed for 30 minutes on ice with gentle rocking followed by centrifugation at 12 000 rpm for 5 minutes at 4 °C, with the supernatant then transferred to a new tube. Total protein was measured with a Bio-Rad Bradford kit, with 30 µg of total protein run on a 12% SDS polyacrylamide gel and transferred to a nitrocellulose membrane blocked with nonfat dry milk in Tris-buffered saline overnight at 4 °C. The membrane was then probed with a primary antibody, washed several times with Tween-20 (0.3%), and incubated with a horseradish peroxidase–conjugated secondary antibody. Finally, the membrane was washed with an enhanced chemiluminescence system. The primary antibodies used were occludin at a dilution of 1:200. A mouse monoclonal antibody against GAPDH (1:1000, TA-08, Beijing Zhongshan Biotechnology Co) was used as a loading control. The intensity of each target band was semiquantified using Quantity One imaging software (Bio-Rad Laboratories). Data were analyzed using the ratio of the densitometric intensity of the target bands and GAPDH bands on the same membrane.
Statistical Analysis
Data were entered into SPSS 20.0 software. The same group at different times was evaluated using repeated-measures analysis of variance. Pair-wise comparisons between the control group and treatment groups were calculated using the least significant difference test for multiple comparisons. We considered P < .05 as statistically significant.
Results
Spermatozoon Count, Viability Rate, and Motility
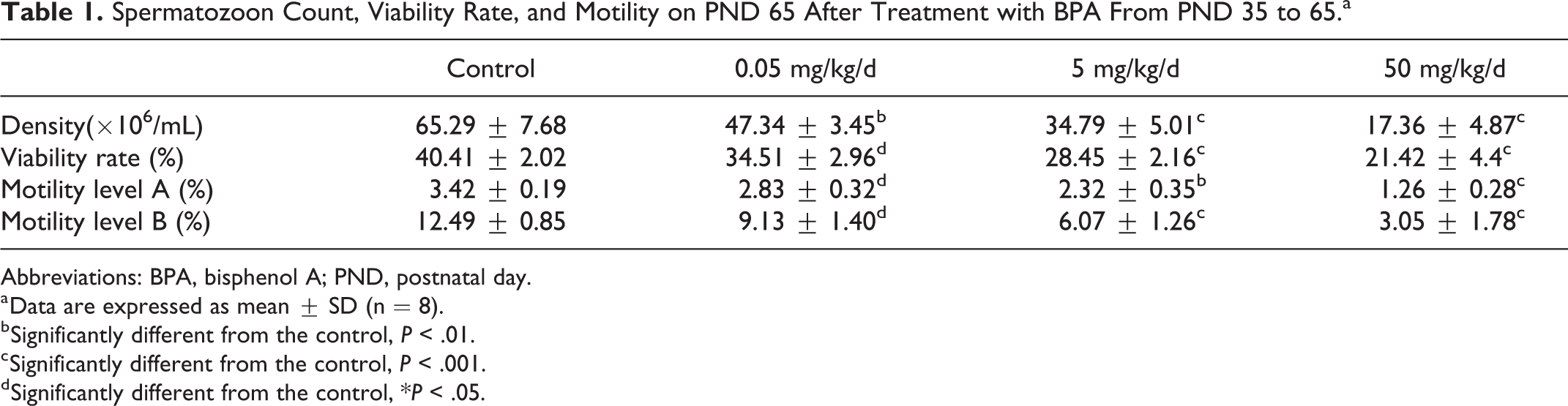
The semen quality of study mice is presented in Tables 1 and 2. There was a significant decline in the semen quality showed in the experimental groups compared with the control group.
Spermatozoon Count, Viability Rate, and Motility on PND 65 After Treatment with BPA From PND 35 to 65.a
Abbreviations: BPA, bisphenol A; PND, postnatal day.
a Data are expressed as mean ± SD (n = 8).
b Significantly different from the control, P < .01.
c Significantly different from the control, P < .001.
d Significantly different from the control, *P < .05.
Spermatozoon Count, Viability Rate, and Motility on PND 125 Following Dosing With BPA on PND 35 to 65.a

Abbreviations: BPA, bisphenol A; PND, postnatal day.
a Data are expressed as mean ± SD (n = 8).
b Significantly different from the control, P < .001.
c Significantly different from the control, P < .05.
d Significantly different from the control, P < .01.
Specifically, a significant effect of the BPA treatment on the sperm parameters (sperm count, sperm viability rate, and sperm motility) was found when BPA experimental groups were compared to that in the control group, showing a dose-related decrease on PND 65 (Table 1). All groups on PND 125, with the exception of the 50 mg/kg/d group, produced a significant recovery in spermatid number, viability rate, and motility, while the 50 mg/kg/d group still showed a significant decrease with respect to the control group (Table 2).
Immunohistochemistry
The specific cells involved in the localization of occludin was similar in the control and experimental groups; however, the intensity of localization differed significantly.
Occludin staining showed a BPA dose–response decrease on PND 65 when compared to that in the control group. Apart from the 50 mg/kg/d group, the immunoreactivity of occludin was in recovery on PND 125 (Figure 1). On PND 65, occludin staining became light colored or absent in the experimental groups, which was also present in the 50.0 mg/kg/d treatment group on PND 125 (Figure 1B, C, D, and H).
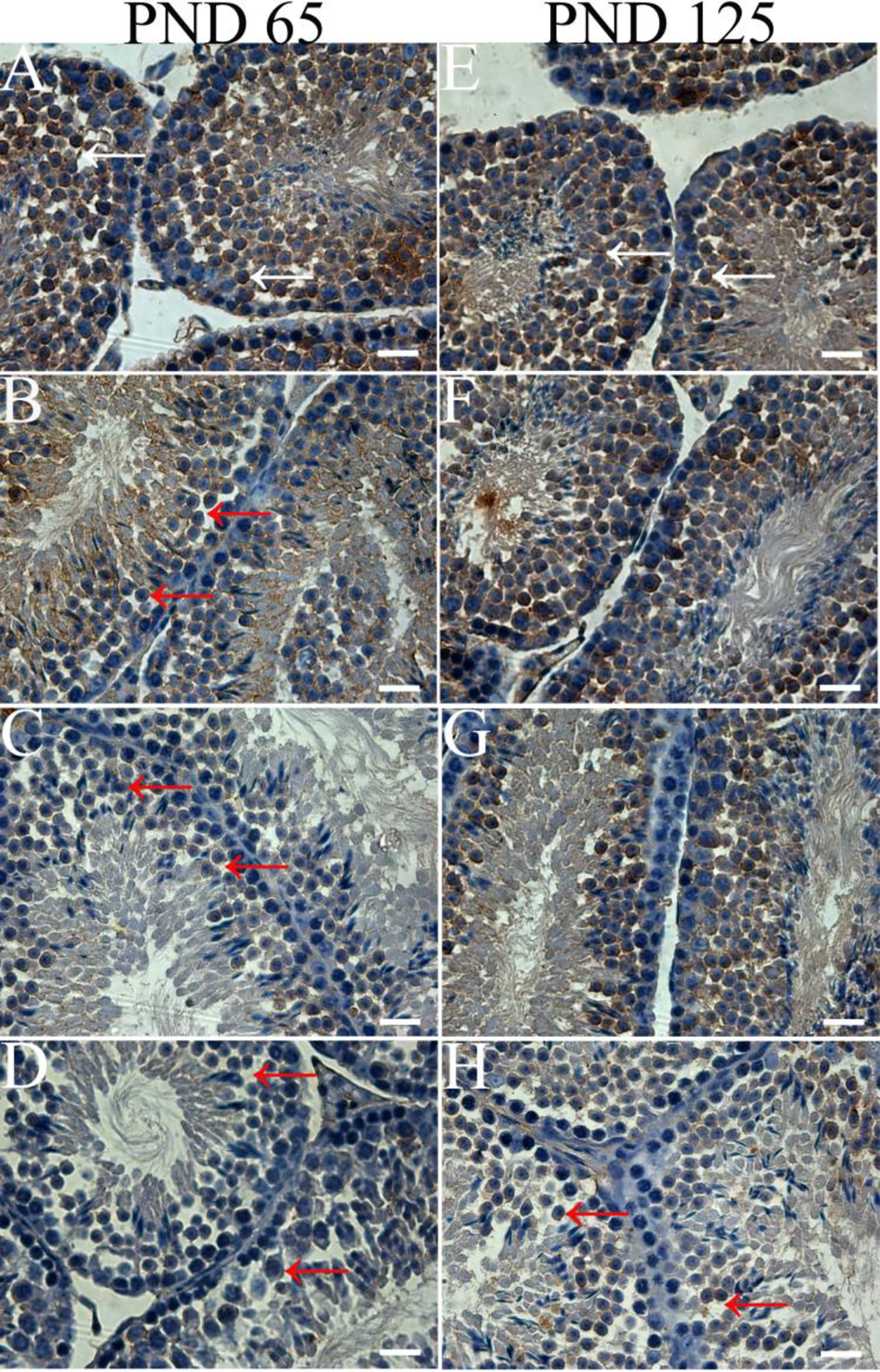
Occludin staining (white arrow) decreased with increasing bisphenol A (BPA) dosages compared with that of control on postnatal (PND) 65 (B, C, and D). Occludin staining still decreased in the 50.0 mg/kg/d treatment group compared with that of control on PND 125 (H). On PND 65, occludin staining became light colored or absent in the experimental groups, which was also present in the 50.0 mg/kg/d treatment group on PND 125 (red arrow). Seminiferous tubule cross-sections of testis of mouse (×630). Scale bars = 100 μm. Control (A and E): 0; low dose (B and F): 0.05 mg/kg/d; middle dose (C and G): 5 mg/kg/d; high-dose (D and H): 50 mg/kg/d.
Western Blotting Analysis
The protein levels of occludin in mice testes following administration of oral doses of BPA were investigated by immunoblotting (Figure 2A and B). Image analysis results of the experimental groups revealed a significant decrease in the expression of occludin compared with that in the control group on PND 65 (P < .01). On PND 125, the expression of occludin in the 50.0 mg/kg/d group was also lower compared with that of the control group (P < .01). The same experimental groups between PND 65 and PND 125 were significantly different (P < .01); however, the control group between PND 65 and PND 125 showed no significant differences (Figure 2C).
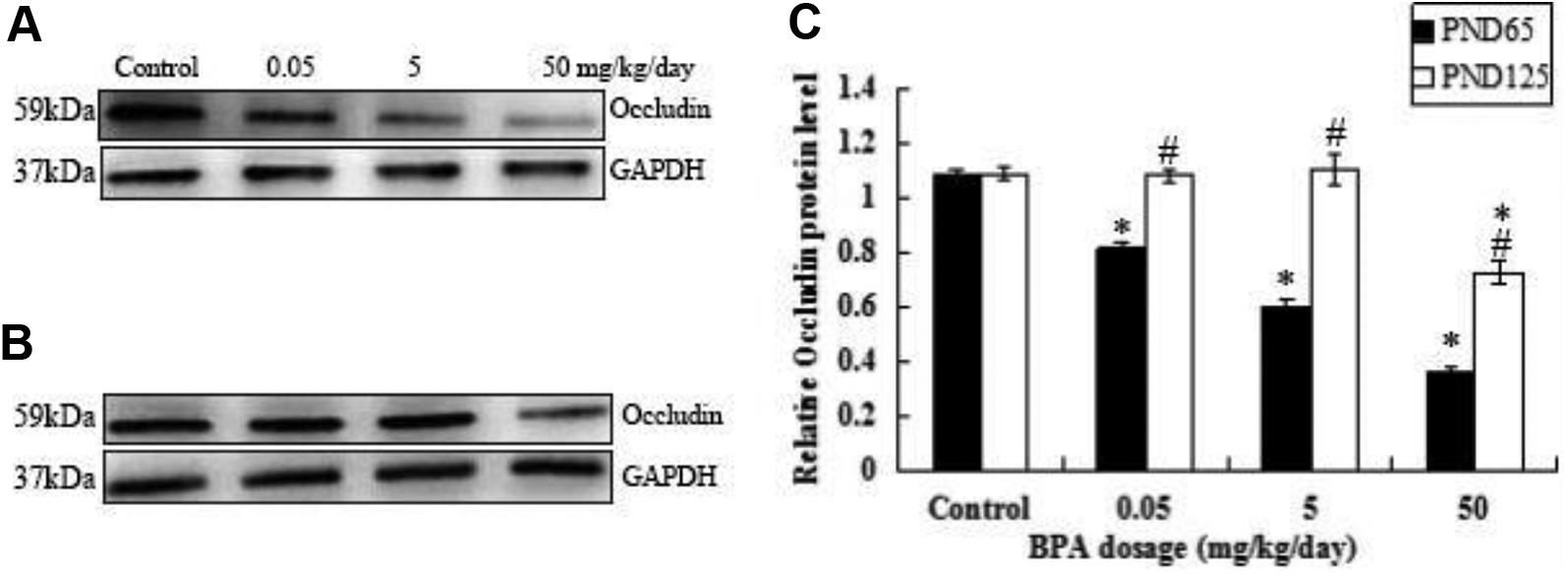
Bisphenol A (BPA) exposure decreased the expression levels of occludin. The protein levels of occludin in whole testis homogenates from postnatal (PND) 65 (A) and from PND 125 (B) were determined by Western blot analysis in response to BPA exposure. GAPDH served as the loading control. Quantified relative protein level between PND 65 and PND 125 (C). *Significant differences between the treatments and the control. #Significant differences between PND 65 and PND 125 in same treatments (*P < .01, # P < .01).
Discussion
Earlier research reported that neonatal exposure to BPA (100 or 500 μg/d) affected the expression pattern of estrogen receptors in reproductive tissue and might lead to fertility impairment in adult rats. 12 Recent research documented that neonatal exposure to BPA (2.4 μg/kg) in male rats caused impairment in the hypothalamus–pituitary–testicular axis, decreased sperm count and motility, and ultimately led to subfertility during adulthood. 13 Administration of BPA (20 or 40 μg/kg) also hampered spermatogenesis and the subsequent development of offspring in rats. 14 Early life oral exposure to BPA at levels well below the current NOAEL of 5 mg/kg/d has been found to alter sex-specific hypothalamic morphology in rats. 14 These findings are consistent with mounting evidence that prenatal and neonatal exposure in rodents to BSA, even at the current safe dose level (<50 μg/kg/d) accepted by the US Food and Drug Administration and the US Environmental Protection Agency and the European Food Safety Authority, 5 is linked to changes in sexual differentiation, defects in male and female reproductive tracts, meiotic abnormalities in fetal oocytes, complications during pregnancy, and increases in malignancies in adult animals. 15 -17 Adolescence is a key point for gonadal development, endocrine hormone synthesis, and metabolic regulation. Therefore, we selected pubertal male mice as the research model to study the effects on their fertility to exposure at “safe” dose and higher dose of BPA.
Occludin plays an indispensable role in TJ function in the testis, demonstrated by the targeted mutation in the occludin gene causing male sterility. 8 Sertoli cells are the primary cellular targets for toxicants. 18 Our results indicated that Sertoli cell junctional protein occludin was vulnerable and a target of BPA action. This result was consistent with the reported findings of de Freitas et al 9 that BPA decreased the expression of Sertoli cell junctional protein occludin. Diesel exhaust particles have been found to induce the dissociation of occludin and ZO1 in alveolar epithelial cells, 19 and 17β-estradiol has been found to decrease the expression of occludin and ZO-2. 20 In this study, the sperm parameters (sperm count, sperm viability rate, and sperm motility) and the expression of occludin were significantly decreased in the experimental groups compared with the control on PND-65. Studies have identified that BPA exposure could cause TJs to become incomplete and less distinct in mice testis. 21 In the present study, on PND-65, occludin staining became light colored or absent in the experimental groups, which was also detected in the 50 mg/kg/d group on PND 125. Therefore, even at the “safe” dose of BPA, TJs were impaired and the BTB was compromised and the spermatogenesis was impaired, which led to a reduction in sperm count, viability rate, and motility. After normal feeding for 2 months, they had recovered in the low-dose and middle-dose groups. In the high-dose group, however, they had not returned to normal. This suggests that not only the expression of occludin could be restored gradually, but spermatogenesis can also regain slowly. Nevertheless, this did not occur in the high-dose group during the experimental period. This might be related to damaged Sertoli cells or other components not yet showing recovery. The related evidence and results presented here imply that further studies are necessary.
The results of this investigation have shown that exposure of pubertal mice to the “safe” level of BPA still decreased the expression of occludin and destroyed the integrity of TJs, which could impair fertility by damaging effect on the BTB. However, these effects were apparently reversible. To our knowledge, this is the first study that has shown that there is recovery from exposure to the “safe” level of BPA (50 µg/kg/d) in pubertal mice. Nevertheless, recovery did not occur at a higher BPA dose (50 mg/kg/d), indicating irreversible structural damage in the male reproductive parameters. Further work is warranted to investigate this possibility to test the effect of higher BPA concentrations (5-50 mg/kg/d). A previous study showed the decrease in occludin protein during BPA exposure (15 µg/L) in male adult rare minnow, Gobiocypris rarus, exposed for 7 to 35 days shows a slowing trend. 22 However, in the present study, there was a BPA dose-related decrease in the expression of occludin. Similarly, Li et al 23 showed that in vitro using the primary cell culture of immature rats, BPA caused a dose-dependent reduction in several junction proteins, including occludin, and ZO-1, and basal ES proteins. Pritchett et al showed the hepatic capacity for BPA glucuronidation was in the order of humans > rats > mice. 24 Species-specific differences raise concerns about the extrapolation of data from rodent studies to human risk assessment. 25 The “safe” level of BPA may not be harmful to humans. To accurately assess the dangers of BPA on human reproductive toxicity, it is important to explore and establish a model of BPA evaluation that can apply to reproductive toxicology in humans in vivo.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was supported in part by Young Foundation(Scheme No. 1347) of The Affiliated Hospital of Qingdao University and prospective protection study of reproductive function on high-risk male infertility crowd of Qingdao people’s livelihood project (Scheme No. 13-1-3-22-nsh).
