Abstract
Background:
Iatrogenic and traumatic sensory nerve injury at the level of the wrist can lead to debilitating neuroma. Targeted muscle reinnervation (TMR) is an effective treatment for the management of symptomatic neuromas. We investigate the use of the anterior interosseous nerve (AIN) as a recipient nerve for the treatment of iatrogenic neuromas. This case series describes 4 patients treated for neuromas of the lateral antebrachial cutaneous nerve (LABC), palmar cutaneous branch of median nerve (PCB), and radial sensory nerve (RSN).
Methods:
Four cases involved a symptomatic neuroma of the LABC, PCB, or RSN. These were treated with TMR, using the AIN motor branch to pronator quadratus. The neuromas were identified in all 4 cases and transected distally. The AIN was identified through a proximal extension of the exploratory incision and an end-to-end coaptation was performed to the proximal aspect of the donor nerve and the distal AIN.
Results:
All 4 patients underwent routine follow-up for a duration of 2 to 10 months, with a long-term follow-up from 25 to 49 months. At routine follow-up, all patients reported resolution of pain and symptoms and had a negative Tinel’s sign over their previous neuroma site. At the long-term follow-up, 2 patients reported recurrence of hyperesthesia, both to a lesser severity than before treatment. All 4 patients reported returning to work or routine and stated the TMR procedure improved their pain and symptoms.
Conclusions:
The motor branch of the distal AIN to pronator quadratus is a viable option as a TMR recipient for the management of symptomatic neuromas of the wrist level. Long-term follow-up shows reduction of reported pain and improvement of function.
Introduction
Traumatic and iatrogenic injuries to sensory nerves of the wrist can lead to debilitating neuromas. A neuroma is formed when damaged axons regrow in an uncontrolled manner resulting in a mass of axon fibers and neural tissue. 1 The commonest symptoms include a painful mass, paresthesia, hypesthesia, allodynia, and cold intolerance over the nerve distribution. 1 Iatrogenic injury can occur during wrist operations such as carpal tunnel release, excision of ganglia, thumb denervation, and trapeziectomy with or without ligament reconstruction. The sensory nerves at the wrist include the lateral antebrachial cutaneous nerve (LABC), palmar cutaneous branch of the median nerve (PCB), the radial sensory nerve (RSN), and the dorsal ulnar sensory nerve (USN).2 -6
Long-term success when treating neuromas is challenging with up to 27% of painful neuromas requiring further operations. 7 Various surgical techniques have been used to manage symptomatic neuromas at the wrist but have failed to reach unanimity.8 -12 Determining treatment depends on the location of injury and amount of scarring. If a distal nerve end is available and scar free, an interpositional nerve graft is preferred. In scarred wounds or those without a distal target, nerve grafting is unpredictable and may not be possible. Excision of the neuroma and burying the proximal nerve end into nearby tissues, such as muscle, can reduce scar formation and provide symptomatic relief but fails to address neuroma reformation. Without a distal nerve target of the injured nerve, novel treatments that inhibit reformation include targeted muscle reinnervation (TMR) and regenerative peripheral nerve interface (RPNI). One meta-analysis concluded that TMR was superior to neurectomy alone. 13
The use of the anterior interosseous nerve (AIN) motor branch to pronator quadratus (PQ) as a recipient nerve for TMR of sensory nerves at the wrist level has been shown as an option in cadavers. 14 Moreover, a few case studies have been published demonstrating it as a suitable technique with follow-up outcomes reported up to 13 months.15 -17 We present a case series exploring TMR using the AIN for iatrogenic neuromas of the LABC, PCB, and RSN, with follow-up of up to 4 years.
Methods and Materials
Case Series
We retrospectively compiled a series of isolated, iatrogenic painful neuroma cases who underwent TMR to the AIN motor branch of the median nerve from 2019 to 2022. These cases were completed by a single senior surgeon. All patients who underwent this technique were included with no exclusion criteria.
Patients were referred regarding wrist-level symptoms after iatrogenic injury. All 4 patients had debilitating pain after a wrist-level surgery affecting the use of their hand. Their symptoms included pain, allodynia, and a positive Tinel’s sign over the neuroma and previous surgical scar. All had confirmatory injections with local anesthetic, in which their symptoms resolved temporarily.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5). Informed consent was obtained from all patients for being included in the study.
Treatment
In all cases, given the terminal nature of the nerves at the wrist level, there was no distal target for nerve grafting and TMR was deemed the most appropriate treatment. The neuromas were identified and transected at their distal extent. The AIN motor branch to PQ was identified through a proximal extension of the exploratory incision, just proximal to the leading edge of PQ. The recipient AIN branch was transected as proximal as necessary to reach the donor nerve without tension. Using microscopic magnification, an end-to-end coaptation was performed with the proximal aspect of the donor sensory nerve to the proximal end of the AIN motor branch to PQ. Postoperative follow-up was then carried out routinely, and the patients were contacted again for long-term follow-up of up to 4 years later.
Results
Four patients were included in this study, 3 females and 1 male. The age range was 28 to 67 years. The average duration from operation to discharge from routine postoperative care was 5 months, and they were contacted for long-term follow-up between 25 and 49 months. All patients’ neuromas were caused by iatrogenic injury. Two patients developed neuroma following volar wrist ganglion excision, both resulting in neuromas of the LABC nerve. One patient had neuroma of the PCB following trapeziectomy and ligament reconstruction with tendon interposition (LRTI). One patient sustained trauma from a table saw requiring repair of the trapezium, extensor pollicis longus, extensor pollicis brevis, abductor pollicis longus, radial artery, and developed a neuroma of the RSN. All patients had symptoms of paresthesia and allodynia into the respected nerve distributions. All patients had a positive Tinel’s sign over their neuroma site (Table 1).
Patient Demographics, Neuroma Etiology, and Symptoms.
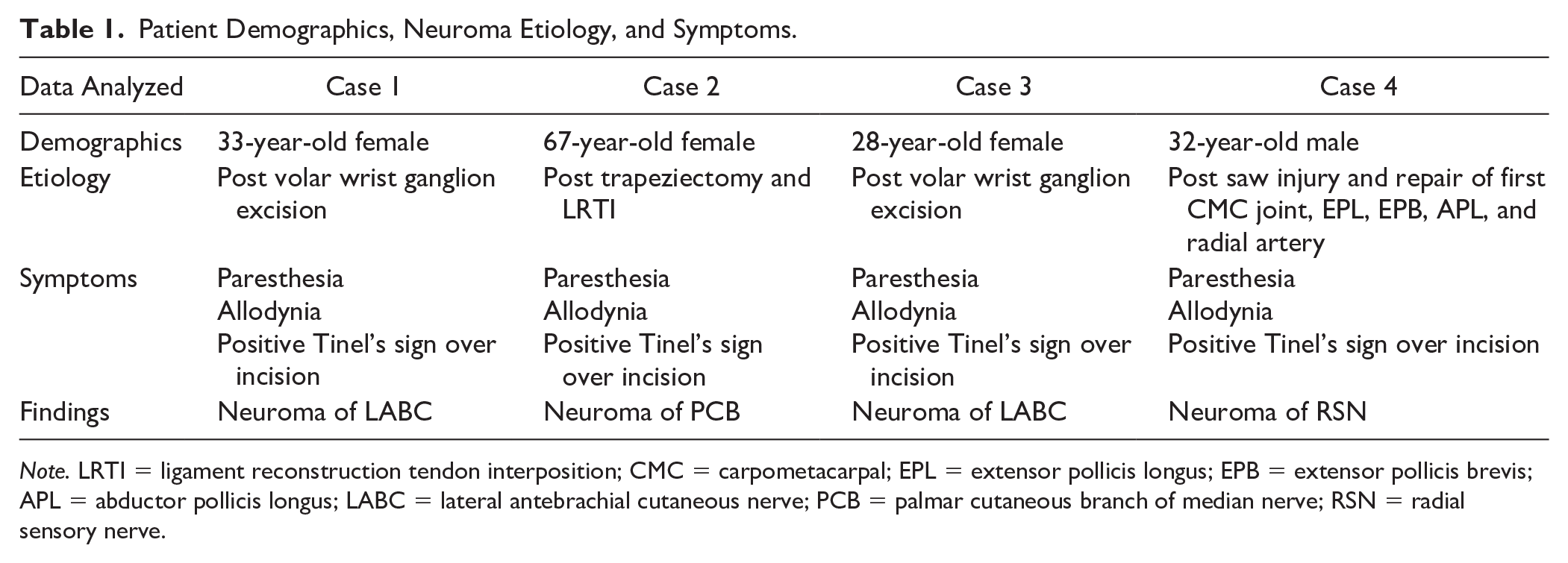
Note. LRTI = ligament reconstruction tendon interposition; CMC = carpometacarpal; EPL = extensor pollicis longus; EPB = extensor pollicis brevis; APL = abductor pollicis longus; LABC = lateral antebrachial cutaneous nerve; PCB = palmar cutaneous branch of median nerve; RSN = radial sensory nerve.
At the time of being discharged from routine postoperative care, all patients had resolution of their neuropathic pain and a negative Tinel’s sign where it was previously positive.
The findings at long-term follow-up are as follows: The first patient had the longest follow-up at 49 months and reported resolution of neuropathic pain and full return of function. They had a negative Tinel’s sign over the previous neuroma site and had full wrist range of motion (ROM). The second patient had long-term follow-up at 45 months and reported no pain for several months following the surgery, followed by recurrence of hyperesthesia in the nerve distribution distal to the neuroma, but reported to a lesser severity. They were unable to quantify the pain using a pain scale. They had full wrist ROM. The third patient had long-term follow-up at 35 months. They had a subsequent ulnar nerve decompression using an AIN graft. They reported resolution of all neuropathic pain, had a negative Tinel’s sign over their previous neuroma site, and had full wrist ROM. The fourth patient had long-term follow-up at 25 months and reported full resolution of neuropathic pain at rest but five-out-of-ten severity intermittent hyperesthesia with heavy use of the hand. Despite the intermittent discomfort, they reported returning to work from a previous leave attributed to the neuroma. They had restricted wrist ROM secondary to their original traumatic injury (Table 2).
Patient Outcomes.
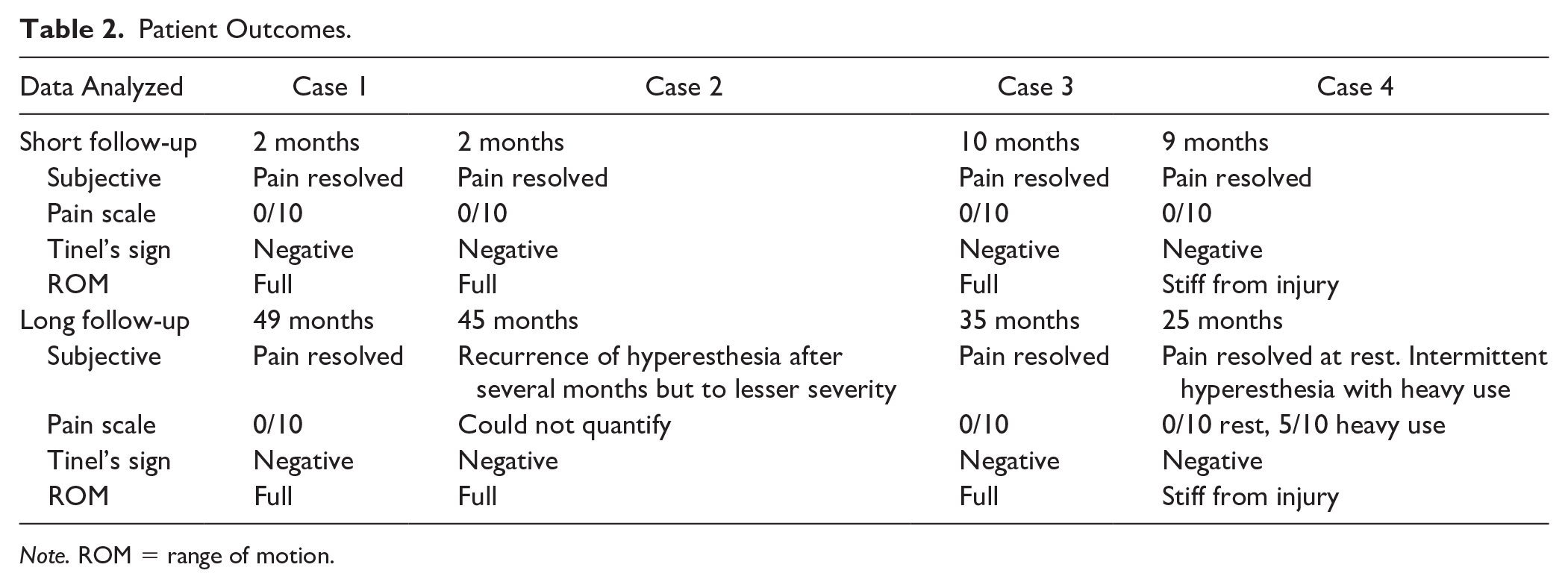
Note. ROM = range of motion.
Discussion
Targeted muscle reinnervation is an operation that has been used in the treatment of painful neuromas. Targeted muscle reinnervation appears to give the transected nerve a more reliable pathway to regrow, thereby limiting the chance for unwanted symptoms. 18 Neuroma formation at the wrist level is often a result of iatrogenic injury from other wrist surgery. Our results are comparable to all previous case reports using the AIN with coaptation to the PCB and USN.15-17 With a longer duration of follow-up, we still see a significant improvement of the preoperative neuropathic pain. Three of our 4 patients had 0/10 pain at rest, with 1 patient describing some pain in the wrist with heavy use. No patients had return of their resting scar pain or significant pain with palpation over their injured nerve distribution. Two patients did have a positive Tinel’s sign over the site of the previous painful neuroma, but this did not cause pain in day-to-day function. All patients reported drastic improvement in neuropathic pain and return of function years after their surgery.
Alternatives to using the AIN have also been reported. Daugherty et al 19 reported the treatment of a neuroma of the RSN using an end-to-side TMR of a motor branch of the posterior interosseous nerve. In 2022, Cox et al 20 treated a neuroma of RSN with TMR to the motor branch of extensor carpi radialis brevis.
The AIN motor branch to PQ offers a suitable and convenient donor site at the wrist. First, donor site morbidity is low because the pronating action of PQ is small compared with its counterpart, pronator teres. 21 No patients reported any deficits in wrist pronation or supination with the use of AIN as a donor for this procedure. Furthermore, this has been well documented in the use of AIN motor branch to PQ to aid in recovery of intrinsic weakness following ulnar neuropathy or high ulnar nerve injury. 22 Second, the proximity of AIN motor branch to PQ offers an easily accessible and efficient donor nerve for wrist-level symptomatic neuromas (Figure 1). This is typically reached in a single incision and completed with a tension-free coaptation. Finally, the size match of sensory nerves at the wrist level and AIN motor branch to PQ is favorable. Although the consequence of nerve mismatch in TMR is not well understood, in theory, those nerves with similar size matches would give better results. Calotta and Dellon 4 reported their approach to neuromas at the wrist level and cited mismatch as a possible complication. We found the donor and recipient nerves to be of adequate size match (Figure 2).
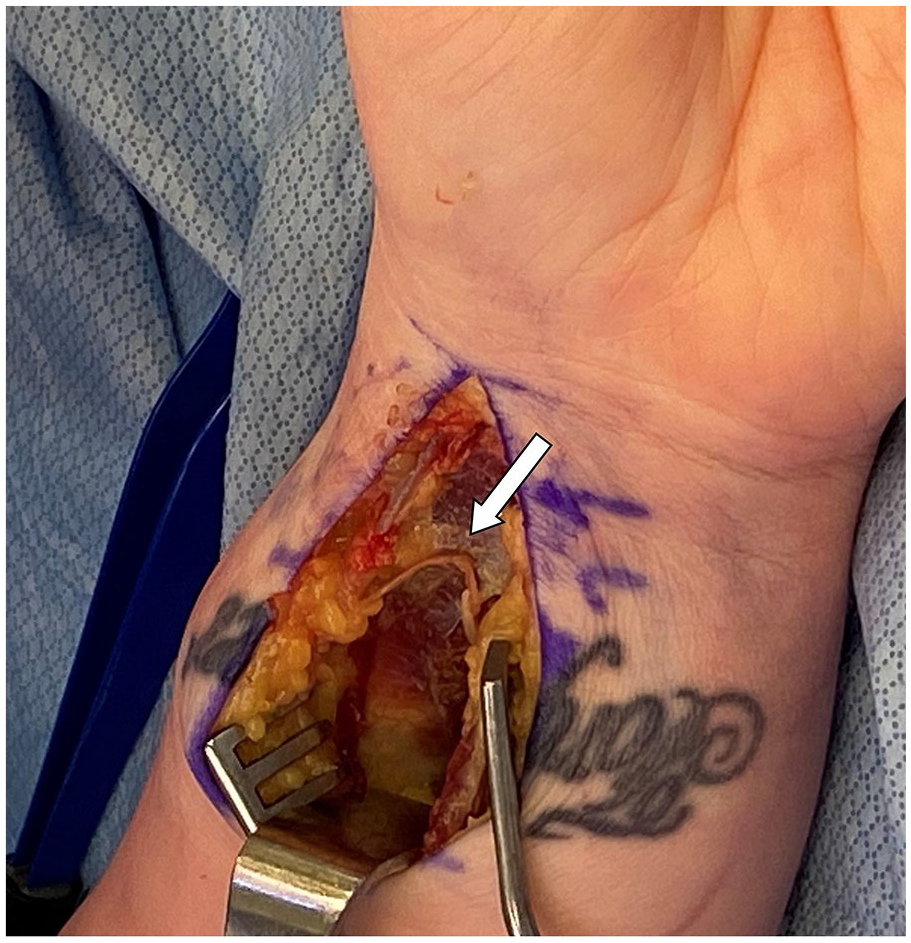
Coaptation of the lateral antebrachial cutaneous nerve and anterior interosseous nerve motor branch to pronator quadratus (arrow).
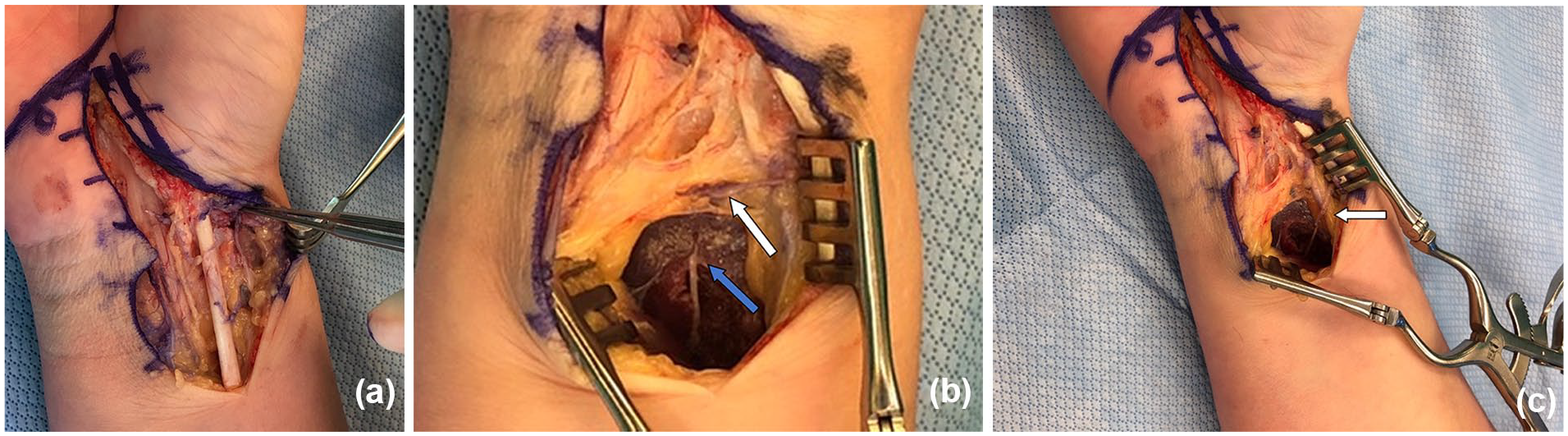
Case 3, treatment of LABC neuroma with TMR using AIN motor branch to PQ.
A potential limitation of this operation is the loss of AIN as a potential donor for end-to-end or end-to-side nerve transfer in high ulnar nerve injury. As it happens, the patient from case 2 eventually presented with symptoms of ulnar neuropathy. They underwent successful end-to-side transfer using the proximal end of AIN motor branch to PQ with recovery of 4+/5 British Modified Research Council scoring at 12 months post operation, which would suggest the proximal AIN motor fascicles were still able to be used as a donor motor nerve for end-to-side or end-to-end coaptation after the distal fascicle is used for a TMR procedure.
Interestingly, another technique has recently been described, combining TMR and RPNI, called TMRpni. Regenerative peripheral nerve interface is a technique where a transected sensory or mixed nerve is implanted into an autologous, denervated free muscle graft. 23 Given that PQ is left without innervation, it would offer a perfect donor as a free muscle graft for this technique should its long-term results prove to be superior to TMR alone. A combination TMRpni is possible using the PQ as a donor muscle, although this was not completed in this case series.
Limitations
Limitation of this study include the small number of patients included, which makes it difficult to draw definitive conclusions on the effectiveness of TMR compared with more traditional methods. This was a patient sample of consecutive patients presenting to a single surgeon’s practice, and all of those who underwent this technique were included. More rigorous comparative studies are needed to this end. This study was retrospective in nature, and in all cases, patients were undergoing a primary procedure with exploration of a potential neuroma as a secondary procedure. With the retrospective nature of the study, patient-reported outcome measurements, standardized pain scales, and isometric testing of strength postoperatively were not conducted. In a prospective, comparative study, these investigations would offer more objective information.
Conclusions
Despite numerous proposed operative techniques, a gold standard treatment for painful neuroma remains elusive. This 4-patient case series reports the use of AIN as a recipient nerve for TMR. This study includes long-term follow-up up to 4 years, showing low donor site morbidity and significant improvement of pain and function postoperatively. All patients made a return to work and routine. Further research is required with larger sample sizes and comparative study to other treatment options to clarify the best treatment.
Footnotes
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Statement of Informed Consent
Informed consent was obtained from all patients included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
