Abstract
Protein fibers, such as wool and silk, play a vital role in the textile industry due to their softness, resilience, comfort, and biodegradability. However, their flammability, shrinkage, and limited UV-resistance prevent their wider use in clothing, technical textiles, and safety-conscious settings. Conventional chemical finishings can address these issues, but often compromise environmental and human safety. Recent advances have investigated eco-friendly alternatives such as biomass-derived compounds, nanoparticles, and multifunctional finishing strategies capable of imparting functional properties. Attaining multifunctionality while maintaining the intrinsic properties of protein fibers is still a significant problem, despite hopeful advancements. Most existing literature has addressed these functional properties individually, and there remains a lack of comprehensive analysis on their integration within protein-based textiles. In order to fill this gap, this article carefully examines recent advancements in eco-friendly finishing methods for protein textiles. Environmentally friendly methods, synergistic formulations, and nanomaterial-based innovations that enhance sustainability and safety are highlighted. It also draws attention to the research opportunities that lie ahead and the challenges of finding a balance between performance, durability, and environmental responsibility. Further, this review aims to compile available knowledge in order to inform the development of high-performance, versatile protein textiles that meet consumer desires, comply with legal requirements, and address the global demand for safer and more environmentally friendly textile solutions.
Keywords
Introduction
Protein fibers are natural textile fibers derived from animal sources that play a crucial role in the textile industry and contribute substantially to the global economy. 1 These fibers exhibit notable characteristics, including warmth, breathability, softness, resilience, and biodegradability. These properties make them ideal to use as household textiles, clothing, and technical applications. 2 They can take a variety of dye classes, including acid, metal complex, and reactive dyes. Furthermore, these fibers offer a wide range of functional advantages in high-performance and luxury applications. These fibers are classified as secretion (silk and similar) and keratin-based fibers. 3 Currently, the rate of fiber production for synthetic fibers is 65% of the total 116 million tons of fibers produced in 2022, cotton, 22%, and the rest totaled 13% including natural fibers, such as wool, silk and linen, and regenerated fibers, for example, rayons, lyocells, and acetates. This was doubled in 20 years from the production of about 60 million tons of fibers. 4 Although these fibers are used in flame retardancy with some modification, the use of especially synthetic fibers is challenging to degrade naturally, and they gradually generate micro- and nano-plastic particles that easily penetrate marine systems, as well as animal and human organs and blood. 5 Whereas natural fibers, such as protein fibers, are widely applicable and environmentally friendly. 6 However, they present certain challenges, most notably, their high flammability, shrinking, and less UV protection.7,8 This property raises significant safety concerns, particularly in environments where fire hazards and UV protection are prevalent. For example, silk offers significant advantages such as abundant resources and biodegradability. 9 However, silk is highly flammable and tends to burn rapidly when exposed to a fire source, accompanied by the release of thick smoke. These fibers have less UV protection ability and shrink during end use. 10 When protein fibers are ignited, they burn quickly, spread flames rapidly, and generate substantial smoke, making it difficult to extinguish. 11 In recent years, the increasing number of fires involving protein fibers has become a significant threat to human safety in addition to ultraviolet protection. 12 Mostly fire accidents occur in urban and industrial regions with high population density, infrastructure-challenged areas, as well as electrical fire accidents, upholstering furniture, bedding, and nightwear. 13 For example, the statistics of electrical fire accidents were recorded in China. According to the latest statistics, from January to August 2024, China experienced 660,000 fire accidents, resulting in 1324 fatalities, 1760 injuries, and direct property losses amounting to CNY 4.92 billion. Among these 208,000 fires were attributed to electrical causes, representing a 14.4% increase compared to the same period last year and accounting for 31.4% of the total number, making it the primary cause of fires. 14 Therefore, developing highly flame-resistant protein fiber with improved shrinkage and better UV protection is a critical area of scientific research with practical implications since the global market has an increasing need for flame-resistant materials. 15
There are several techniques to impart flame retardant onto textiles, which are broadly grouped into fiber-level and fabric-level methods. Flame retardant at the fiber level includes co-polymerization, blending and grafting during the production of fibers. 16 Whereas the fabric level (finishing method) is the application of flame-retardant chemicals after the fabric is made. Among these, the finishing method is particularly favored owing to its straightforward implementation, effectiveness, and ability to enhance the fabric properties. 17 There are phosphorus-based, halogen-based, and even protein-based, such as groups of flame retardants for textiles. Among the traditional and protein-based flame retardants, Casein, a milk protein rich in phosphorus groups, is commonly used as an eco-friendly flame retardant for protein fibers and cotton. 18 Besides protein-based flame retardants, traditional chemical flame retardants have been used for textiles based on phosphorus chemistry, like ammonium polyphosphate (APP), which are commonly used as intumescent flame retardants. 19 The traditional halogen-based flame retardants, like decabromodiphenyl ether (DecaBDE). 20 However, their use has diminished due to these substances’ release of cancer-causing dioxins and environmental concerns. Therefore, the pursuit of eco-friendly finishing solutions for protein fibers is aimed at enhancing the flame retardancy while minimizing environmental harm. 21 Various natural materials, such as biomass-based, protein-based, and inorganic materials, have been explored as potential eco-friendly flame-retardant alternatives. 22 But, reconciling the preservation of textile characteristics during finishing procedures with the effectiveness of flame retardancy is still a considerable difficulty. 23 Recent advancements in combined formulations have focused on multiple flame retardants to improve protein fiber efficiency while also providing extra thermal, dimensional, and mechanical qualities, which this paper critically examines.
Protein fibers, especially wool, have been valued for their softness, warmth, elasticity, and luxurious feel. However, in addition to flammability, they have the tendency to shrink and felt during washing and handling. 24 This is due to the scale surface structure of protein fibers and their sensitivity to moisture, heat, and agitation. This makes protein-based textiles have poor dimensional stability, limited use in mass production of apparel, unstable for technical textiles, and limited fashion applications. 25 Consequently, untreated wool garments often lose their original size and shape after laundering. Due to this sensitivity, protein-based textiles are less suitable for technical textiles for durability and resistance as well as for mass production of apparel, where consistent sizing and easy care are essential. 26 These drawbacks also reduce their flexibility in fashion applications, where frequent washing and structural integrity are required. 27 Therefore, shrink-resist treatments of protein fibers have garnered considerable scholarly interest. Conventional shrink-proof treatments commonly utilize potassium permanganate (KMnO4), which has proven effective against shrinkage. 28 However, these conventional methods raise environmental and health concerns. In response to these concerns, there is a growing focus on eco-friendly shrink-proof solutions for protein textiles. 29 These alternatives can be categorized into natural shrink-proof agents, biotechnology-based approaches, and nanomaterials. Integrating these alternatives into textiles offers a promising pathway for the development of durable, dimensionally stable, and environmentally friendly protein-based textiles. 30
In addition to shrinkage and flammability, protein fibers, especially silk, have poor resistance to UV. Prolonged exposure to sunlight weakens silk fibers significantly, causing them to lose strength, become brittle, and yellow over time. 31 This is partly due to the lack of strong, stable UV-absorbing chromophores in the natural protein structure. 32 UV absorbers can be categorized into two types: inorganic and organic. The commonly used organic absorbers include 2-hydroxyphenyl benzotriazoles, 2-hydroxy benzophenones, 2-hydroxy phenyl-S-triazines and chemical absorbers such as benzoic acid esters and hindered amines. Inorganic compounds such as titanium dioxide and ceramic materials in the UV region lies between 280 and 400 nm and reflect the visible and infrared rays. However, these substances release cancer-causing dioxins, and pollute the environment. 33 As a result, the development of eco-friendly finishing techniques for protein fibers aims to improve UV protection while minimizing environmental impact. For example, a study reported that wool fabric was dyed with the buckthorn plant. In this study, not only the dyeing ability but also the UV protection properties of buckthorn were investigated. 34 Another study shows the treatment of silk fabric with nanoparticles to improve its resistance to ultraviolet light and to wrinkles. A finishing method of coating the silk fabric surface with nano-silica (nano-SiO2) was used. The results show that the UV protective factor (UPF) value could reach a maximum of 84.52 after finishing in 10 g/L nano-SiO2 and 20 g/L silane coupling agent (KH570) solution at 80°C. 35
Moreover, the growing demand for multifunctional textiles reflects rising living standards and technological advancements, driving the need for fabrics that offer flame resistance, shrink resistance, and UV protection, especially within the home furnishings, automotive, and aerospace industries. This demand has led to increased interest in specialized functional agents and treatment methods for developing innovative fabrics with multifunctionality. 36 The fabrication of textiles with multifunctional activity typically involves either a two-step or a single-step approach. Although the two-step method allows for broad applicability, it may encounter challenges owing to the potential interactions between flame retardants, shrink-proof, and UV protective substances that could hinder functionality. 37 Conversely, single-step methods often use synthetic agents that combine multifunctional properties, albeit with complex designs and eco-friendly challenges. 38 Additionally, identifying optimal processes and functional materials remains a significant challenge in the field of multifunctional textile research. Numerous review articles have been published on flame-retardant, shrink-proof proof and UV protective fabrics. These reviews have covered various aspects, including textile materials, 39 functionalizing materials, different mechanisms, 40 recent advancements and sustainability, 41 durable flame- retardant finishing, and shrink proof, 23 UV protective agents, 42 finishing technologies, 43 phytic acid-based flame retardants, 44 Biocompatibility, 45 nanoparticles, 46 and chitosan based unprotective. 47
While the development of eco-friendly functionalizing materials for textiles has become a dynamic area of research, a comprehensive review addressing their use in achieving flame retardancy, shrink resistance, and UV protection has not yet been systematically compiled. Although various studies have examined these properties separately, a comprehensive overview is lacking from the current body of literature. This paper aims to fill this gap by providing a holistic overview of all three functionalities in the context of protein-based textiles. 48 In 2025 alone, devastating wildfires swept through California, Canada, and Israel. In California, January fires claimed 27 lives, destroyed over 12,000 structures, and resulted in more than 440 excess deaths linked to smoke exposure. Canada experienced its worst wildfire season on record, with over 10 million hectares of land burned. In Israel, wildfires in the Jerusalem Hills ravaged forests and forced the cancelation of major public events. These tragedies highlight the growing threats posed by climate change, weaknesses in existing infrastructure, and the urgent need for enhanced fire resilience and UV protection. 49 This review aims to bridge that gap by offering a critical synthesis of multifunctional textile treatments that integrate flame retardancy, shrink-proofing, and UV resistance, with a particular focus on eco-friendly and sustainable solutions. By doing so, it not only addresses a key deficiency in current research but also offers valuable insights for advancing safety and sustainability in modern textile applications.
Review methodology
To conduct this comprehensive review on sustainable strategies for enhancing flame retardancy, shrink resistance, and UV protection in protein-based fibers, an extensive and systematic literature search was undertaken across leading scientific databases, including PubMed, Scopus, Web of Science, Google Scholar, ScienceDirect, and Wiley Online Library. Carefully selected keywords and phrases such as “eco-friendly flame retardants,” “sustainable textile finishing,” “shrink-proofing,” “UV protection,” “green chemistry,” and “protein fibers” were employed to ensure a broad yet focused retrieval of relevant and high-quality publications. In the beginning, more than 800 references were identified through this systematic search. It was done using a methodical screening procedure with clearly defined inclusion and exclusion criteria. Peer-reviewed reviews, book chapters, conference proceedings, and articles that focused on eco-friendly protein fiber treatments were the only ones considered. To maintain scientific rigor, irrelevant studies, editorials, and non-peer-reviewed papers were excluded. The inclusion criteria focused on peer-reviewed articles, reviews, book chapters, and conference proceedings that specifically addressed green solutions for protein fibers. Non-peer-reviewed articles, editorials, opinion pieces, and publications unrelated to core topics were excluded to maintain the quality and relevance of the literature. Following the initial search, titles and abstracts of the identified references were screened to assess their relevance. Ultimately, 310 publications were selected for detailed analysis.
Data extraction focused on the types of green agents used (e.g. bio-based compounds, biodegradable polymers), treatment methodologies, performance outcomes (e.g. flame retardancy, UV-blocking, shrink resistance), and environmental assessments. This categorization enabled a structured analysis of trends, innovations, and gaps in existing research. Each study underwent a quality assessment that evaluated methodological rigor, including sample size, experimental design, and replicability of results. This assessment helped to determine the reliability of the findings and provide directions for future research. A comprehensive synthesis of the collected data was conducted to identify the common themes, innovations, and challenges within the field. The review concludes with a critical synthesis addressing environmental benefits, industrial relevance, and future research directions.
Overview of protein fibers
Protein fibers are natural fibers composed of long-chain polypeptides, primarily derived from animal sources. In the first half of the 20th century, scientific communities worldwide endeavored to diminish dependence on expensive and scarce animal fibers like wool and silk. Their efforts focused on developing regenerated protein fibers, including soy, zein, and casein, to provide comparable benefits to natural protein fibers, such as lustrous appearance, warmth, and a soft feel. 50 The structure of protein fiber is based on amino acids linked by peptide bonds, giving them unique physical, chemical, and esthetic properties. 51 Figure 1 shows the schematic illustration of the protein structure in silk–soy composite films before and after water annealing treatment. Wool, obtained from sheep and other animals, is known for its crimp, elasticity, warmth, and resilience. Its surface is covered with microscopic scales, which contribute to felting and shrinkage during washing. Silk, produced by silkworms, is valued for its luster, smooth texture, and high tensile strength, though it is more susceptible to abrasion than wool. 52 These fibers exhibit excellent dye affinity due to reactive amino and carboxyl groups in their structure, allowing for vibrant, long-lasting colors. 53 They are comfortable to wear, as they absorb moisture without feeling wet. The overview and classification of protein-based fiber is shown in Table 1. Moreover, the regenerated protein fibers, such as casein, soy, zein, collagen, or recombinant spider silk depend on denaturation, reassembly, and controlled spinning. Whereas, natural fibers such as silk, wool, and cashmere rely on clearly defined β-sheet or keratin coiled-coil structures. These structural variances explain variations in strength, elasticity, and processability. 54 Recent developments include improved keratin and collagen extraction, electrospinning techniques that enhance alignment and functionality, and the recombinant manufacture of high-molecular-weight spider-silk motifs. Nonetheless, issues with scalability, uniformity, and environmental effect still exist. 55

Overview and classification of protein-based fiber (natural and regenerated).

These natural and regenerated fibers are sensitive to high heat, strong acids, and prolonged exposure to sunlight, which can cause degradation and dis-coloration. 74 Protein fibers are sustainable textile materials since they are renewable and biodegradable. They must be handled carefully to preserve their performance and look, though, because they are combustible. These fibers are valuable for clothing, furniture, and luxury fabrics all over the world because of their comfort, attractiveness, and functional qualities. 75
Flame-retardant finishing of protein-based textiles
Mechanism of flammability
The enhancement of safety and functionality in textile materials, particularly in protein fibers, is a crucial area of focus in materials science. Protein fibers, renowned for their exceptional blend of mechanical strength, esthetic appeal, and biocompatibility, are widely utilized in various applications. 76 However, they have a significant limitation, their inherent flammability, which can cause rapid ignition and burning, posing safety hazards and material damage. Although protein fibers have better resistance to flame than cellulosic fibers, due to the Sulfur and nitrogen content within their structure, they still cannot pass the vertical burning test. The untreated fabrics are highly combustible, posing a considerable fire hazard that requires the development and application of effective flame-retardant treatments. 77 For example, Figure 2. Show that the flame-retardant mechanism of P-containing flame retardants.

Flame-retardant mechanism of P-containing flame retardants. 78
Combustion is the exothermic oxidation of a material that releases heat and light. In contrast, pyrolysis describes the thermal decomposition of the peptide bonds in the absence of oxygen, leading to the formation of flammable gases, char, and tar. Protein-based textile fibers such as wool and silk exhibit a distinct mechanism of flammability due to their peptide-based structure. 79 When exposed to heat, they first lose moisture, which delays ignition. As the temperature rises (30°C–350°C), thermal decomposition of peptide bonds occurs, releasing gases such as ammonia, carbon dioxide, water vapor, and hydrogen sulfide.80,81 These decomposition products are largely non-flammable, which suppresses flame propagation. Simultaneously, aromatic amino acid residues promote crosslinking and char formation. At higher temperatures, limited volatile fragments ignite, but the fiber mostly degrades into a protective carbonaceous char. This char layer restricts further access of heat and oxygen to the underlying material. As a result, protein fibers burn with difficulty, often self-extinguish when the flame source is removed, and emit a characteristic burning hair odor. 82
Mechanism of action in flame retardants
Flame retardants effectively inhibit ignition, restrict heat release, and limit the spread of flame by releasing radicals, diluting flammable gasses, promoting char formation, freezing surfaces, and halting combustion processes. 83 Fire safety is greatly enhanced by their activation in response to heat sources and flames. They work well because of their inherent chemical properties as well as how they interact with fibers and polymers. In the solid, liquid, and gas phases, these substances can obstruct combustion through a variety of physical and chemical processes. This complex mechanism interaction usually involves many steps. 84 Understanding the full mechanism of flame retardants requires consideration of several physical phenomena that prevent burning. One primary physical method involves the formation of a protective layer, which significantly reduces the heat transfer and restricts the oxygen flow. 85 This barrier not only lowers the degradation rates of the polymer but also minimizes the production of pyrolysis gases, underscoring the fundamental principles of flame retardancy. Moreover, additional physical mechanisms include the cooling effects provided by various compounds such as phosphorus and silicon-containing substances, boric acid- based additives, inorganic borates, and low-melting glasses. 86 These materials effectively cool the substrate to temperatures below the ignition thresholds, thereby triggering endothermic processes, such as the decomposition of the additives. In conjunction with cooling, dilution plays a vital role in reducing the concentration of flammable gases in both condensed and gas phases. 87 This is achieved by the incorporation of inert substances or additives, including fillers, hydrates, char-promoting agents, and halogen-containing compounds. During decomposition, these substances release non-flammable gases, which dilute the fuel concentration in both phases, ultimately lowering the flammable gas levels below the ignition limits. 88 Figure 3 shows the combustion cycle of a typical textile material as it approaches fire.

Combustion cycle of a typical textile material. 89
Further, chemical reactions that occur in protein fibers like wool and silk that include sulfur and nitrogen also contribute to the effectiveness of flame retardants in disrupting the combustion processes. 90 In the gas/vapor phase, these agents reduce the concentration of free radicals, inhibit the flaming and exothermic reactions, and provide thermal feedback to the condensed phase. In the solid/condensed phase, flame retardants can diminish the production of volatile species, leading to the formation of carbonaceous char. 91 Halogenated flame retardants interact with -NH and -SH groups through radical scavenging, while phosphorus-based compounds react with -OH and -CONH- groups, promoting dehydration, crosslinking, char formation, and enhanced flame resistance. By stimulating the dehydration and crosslinking of peptide chains, phosphorus-based retardants break down to produce phosphoric or polyphosphoric acids, which increase char formation and lower flammable volatiles. During heating, nitrogen-based flame retardants produce inert gases (N2, NH3), which dilute combustible gases and oxygen in the combustion zone. Furthermore, the keratin or fibroin backbone is stabilized by metal salts like borates or sulfates, which encourage thermally stable residues.92,93 Together, these processes reduce heat release, inhibit the spread of radical chains, and greatly increase the protein fibers like wool and silk’s natural flame resistance. In the case of synthetic fibers, flame retardants facilitate polymer breakdown, allowing the material to flow away from the influence of the flame.94,95 Some of the main fire-retardant mechanisms of the bio-sourced FRs applied to textiles are shown in Table 2. In addition to clarifying the physics underlying fire resistance, an understanding of these processes emphasizes how crucial it is to choose the right flame retardants for various materials and applications.
Main fire-retardant mechanisms of the bio-sourced FRs applied to textiles. 96

Challenges with traditional flame retardants
Traditional flame retardants play a crucial role in enhancing the fire safety of textile fabrics, reducing hazards in clothing, upholstery, and industrial applications. These compounds function by delaying ignition, slowing combustion, or preventing flame spread, thereby minimizing fire risks. 97 Flame retardants are broadly divided into halogenated and non-halogenated types, each with distinct mechanisms. 98 Halogenated flame retardants, especially those containing bromine or chlorine, are highly effective as they act in the gas phase by disrupting radical chain reactions that sustain flames. Despite their efficiency, their use poses serious environmental and health challenges. 99 During combustion, halogenated compounds can generate toxic byproducts such as dioxins and furans, which are hazardous to human health. 100 Moreover, brominated flame retardants have been detected in remote regions and across ecosystems, indicating persistence, long-range transport, and bioaccumulation potential. Their presence in terrestrial and aquatic food chains raises concerns about ecological damage and long-term human health impacts. 101
Phosphorus-based flame retardants act primarily by promoting char formation, which creates a protective barrier that slows combustion. 102 They can be applied either as reactive systems or as additives in diverse sectors such as electronics, automotive, and construction materials. 103 While they significantly improve fire resistance, limitations remain, including variable effectiveness across substrates and potential impacts on mechanical properties. As a result, research is advancing toward phosphorus compounds that balance flame retardancy with reduced health and environmental risks. 104 Table 3 shows the summary of traditional flame-retardant technologies applied to textile materials, with specific emphasis on the mechanisms and performance of non-halogenated phosphorus-based flame-retardant treatments. Inorganic flame retardants, Particularly, metal hydroxides function by releasing water vapor during heating, which cools the substrate and dilutes flammable gases. Compared to halogenated compounds, halogen-free inorganic retardants are less toxic; metallic oxides further contribute by acting as both flame retardants and smoke suppressants, thereby enhancing overall fire safety. 105 Although metal oxide flame retardants offer some benefits, they present notable drawbacks. These compounds often require high loading levels to achieve adequate flame retardancy, which can negatively affect the properties of the protein fiber. 106 For example Guo et al. 107 reported that wool fabrics treated with a 9.9% weight add-on achieved a limit oxygen index (LOI) of 33.7%. Compared to the untreated control, the coated samples showed markedly enhanced fire safety, with the peak heat release rate and total smoke production reduced by 46.5% and 33.7%, respectively, during cone calorimetry testing. This high loading requirement may lead to decreased comfort, reduced breathability, and a stiffer hand feel for the treated fabrics. 108
Summary of phosphorus- based flame-retardant treatment of textile materials. 78

In light of the limitations associated with traditional flame-retardants, there is growing interest in alternative materials. Researchers are exploring compounds such as ammonium polyphosphate, pentaerythritol, melamine, metal salts, and organic-inorganic hybrid materials for their potential to enhance fire resistance while minimizing negative impacts. These emerging alternatives often combine the benefits of both organic and inorganic flame retardants, providing improved performance characteristics and a reduced environmental impact. Footprint. For example, sodium dodecyl sulfate was intercalated into the layers of Ca-Al hydrotalcite, resulting in a notable increase in the spacing between the layers. The organic macromolecule acrylic acid was successfully intercalated into the inorganic CaAl hydrotalcite layers by ion exchange method, resulting in the preparation of the organic-inorganic CaAl-AA-LDH new flame retardant.
109
Figure 4 Shows that the combined treatment of wool and silk fabric with Hydroxyl-phosphoramidate/BTCA,

Green flame-retardant solutions for protein-based textile
The green flame-retardant solutions for protein-based textiles can be categorized into two groups. The two primary categories of environmentally friendly flame-retardant methods that come from biological sources are plant-based and animal-based systems. 112 Phytic acid and lignin from seeds and grains, as well as polysaccharides like cellulose, starch, and alginate, are examples of plant-based flame retardants. These substances inhibit heat and smoke release, improve char formation, and form protective layers, making them viable halogen-free substitutes. Animal-based eco-friendly flame-retardant systems exploit natural biomolecules rich in nitrogen, phosphorus, or aromatic structures, which enhance char formation and suppress flammability, including chitosan, DNA, collagen, casein, and keratin. The Schematic representation of eco-friendly and bio-based approaches for flame-retardant functionalization of textiles is shown in Figure 5.
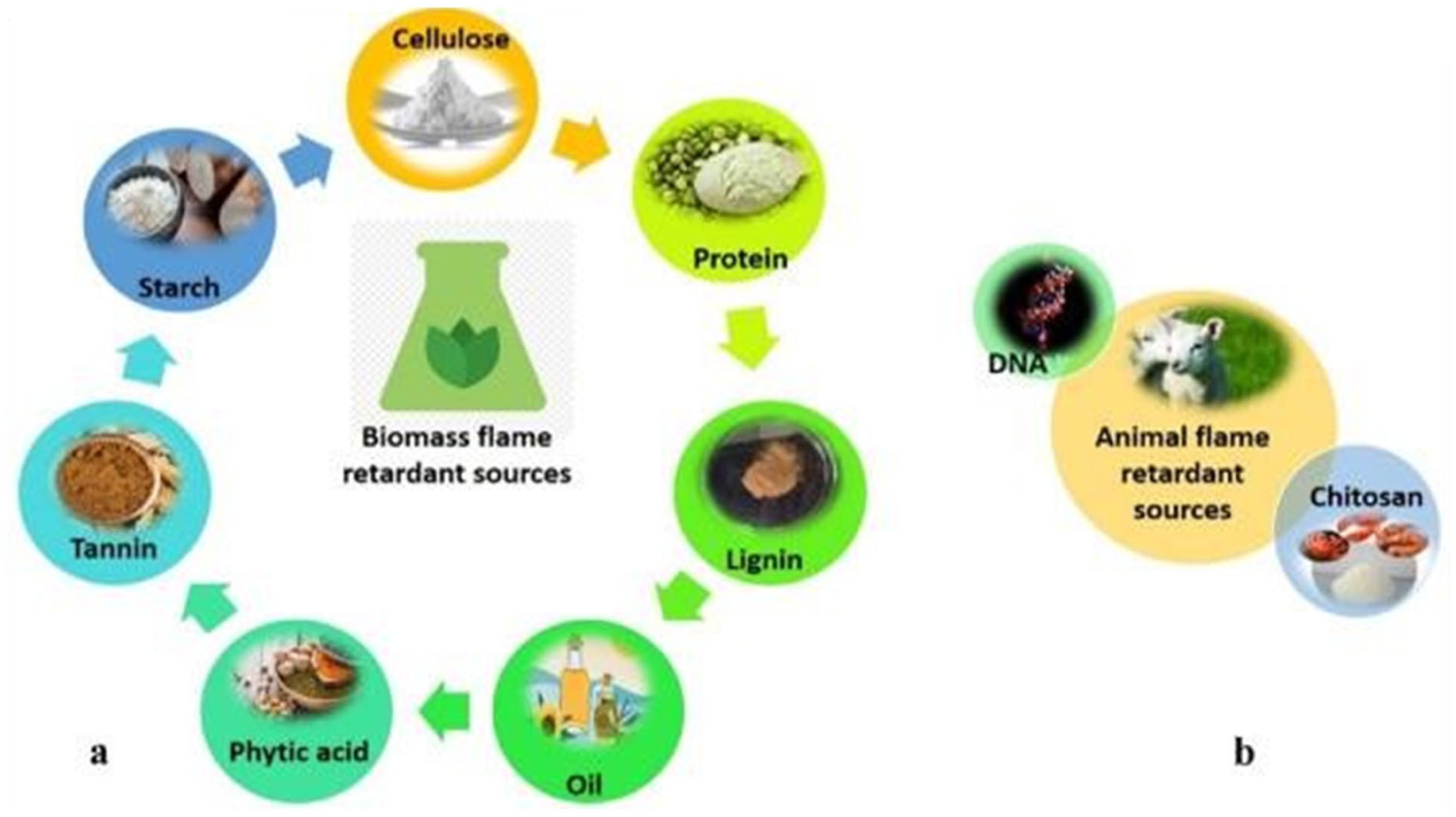
Schematic representation of bio-based approaches for flame-retardant functionalization of textiles. 113
Animal-based flame-retardant textiles (chitosan, DNA, and collagen)
Various proteins are flame retardants to protect textile materials. Fish DNA, casein, whey protein, chicken feathers, and plant-based biomacromolecules, such as spinach juice, banana juice, and coconut shell extract, are well known for their fire-resistant properties in cellulose and protein textiles. 114 The common types of animal-based flame retardants are chitosan and collagen, as well as DNA, which have all been extensively investigated as flame retardants for protein fiber, such as soybean, silk, and wool-based textiles. 78 For instance, on the fiber surface, chitosan creates a protective char coating that lowers flammability and smoke emission in addition to the inherent retardancy of protein fibers. However, chitosan coatings often exhibit limited flame-retardant effects, necessitating the use of composite materials such as layer-by-layer (LBL) systems or chemical modifications to create intumescent flame retardants that achieve the desired fire resistance.115,116 Therefore, the combined use of chitosan with other biopolymers and nano materials increases the retardant property of chitosan and other bio flame retardants.
These animal-based flame retardants may be used alone or combined. For example, biobased chitosan with good film-forming ability and polysaccharide structures that are prone to carbonize was used to develop a PANI/chitosan coating for silk fabric. 117 In the other study, a water-soluble polyelectrolyte complex (PEC) consisting of bio-resourced phytic acid and chitosan was fabricated at pH 1.3 and used to improve the flame retardancy of wool fabric by the repeated coating technique. 118 Further, silk fabric was treated with squid ink Edible squid ink proteoglycans as a multifunctional sustainable additive for silk coatings: Fire protection, UV shielding and as a colorant. Under heat, DNA functions as a natural intumescent, releasing non-flammable gases and creating a foamed protective barrier. By reducing fiber deterioration, collagen improves thermal stability. 119 These biopolymers preserve the environment while greatly enhancing flame resistance when applied to protect textile materials. Figure 6(a) and (b) illustrate the application of bio-sourced materials in textile treatments: Figure 6(a) shows the phytic acid/chitosan polyelectrolyte complex used as a flame-retardant treatment for wool fabric, while Figure 6(b) depicts silk fabric treated with squid inks.

Plant-based flame retardants
Plant-derived biomass offers a sustainable and eco-friendly alternative to conventional halogenated flame retardants. Biomass-based compounds, such as lignin, starch, cellulose, tannins, proteins, and phytic acid, contain functional groups (e.g. hydroxyl, phosphate, nitrogen-containing moieties) that contribute to flame-retardant behavior. 120 These compounds promote char formation, limit heat release, and reduce the emission of toxic gases during combustion. Researchers are increasingly exploring these natural substances for their potential to create effective flame-retardant coating systems for textiles, particularly for protein-based textiles, owing to environmental, and health concerns associated with conventional flame retardants. 121 For example, Jiang et al. 122 developed a novel and eco-friendly flame-retardant silk fabric through the Maillard reaction between glucose and poly (glutamic acid) for treating silk fabric. The study elucidates the synthesis of Maillard reaction products (MRPs) and verifies their deposition onto the silk fabric surface by observing changes in surface morphology, functional groups, and charged characteristics. The flammability tests demonstrate that MRPs treated silk fabrics had a high limiting oxygen index of over 27% and a charred length of less than 12 cm, indicating effective flame retardancy. The reaction and the preparation process of are shown in Figure 7.

Illustration of: (a) Maillard reaction between Glc and amino-containing compounds and (b) the preparation process of MRPs treated silk fabric. 122
Starch, lignin, and cellulose-based flame retardants have been extensively studied to enhance the fire safety of cellulosic as well as protein-based textiles. Starch is a polymer that is synthesized by plants as energy storage. Starch can be hydrolyzed into simpler carbohydrates, also called dextrin, by acids or enzymes. For example, cyclodextrin is a component of the flame-retardant system obtained by degradation of starch by Bacillus amylobacter bacteria. Sorbitol can also be used in flame-retardant formulations. The fermentation of glucose and grape stock generates itaconic and tartaric acids, respectively. These two organic acids have been used as raw materials for the development of flame retardants. 123 Cellulose itself is not a flame retardant; it can be effectively used in conjunction with various flame retardants to improve the fire resistance of fabrics. These materials are particularly attractive owing to their renewable and biodegradable characteristics. Cellulose is a linear homopolymer composed of D-glucose units linked by a beta 1–4 glycosidic bond, used as flame retardancy, combined with other retardant agents and additives. The thermal decomposition of cellulose occurs in one main step between 300°C and 400°C, the higher mass loss rate being observed around 370°C. Below 300°C, cellulose undergoes dehydration reactions that give rise to a small mass loss. 124 At high temperature (800°C), a stable char representing 15 wt% of the initial mass is observed. This residue has a structure similar to that of lignin char. It should be added that the degradation pathway of cellulose may be modified depending on the heating conditions and the presence of other components in the surroundings. Thus, at a low heating rate, dehydration reactions are favored, and the char yield is enhanced. 125 Alginate-based flame-retardants have also gained attention owing to their environmental friendliness and effectiveness. These are typically incorporated phosphorus-, nitrogen-, and silicon-based compounds to enhance fire resistance. Thus, alginates could be considered as an anionic polysaccharide. 126 These flame-retardants typically incorporate phosphorus-, nitrogen-, and silicon-based compounds to enhance fire resistance. The mechanisms of alginate treatment of protein-based textiles involve both condensed and gas-phase actions. In the condensed phase, alginate forms a char layer on the protein fiber surface, which acts as a barrier to heat and oxygen transfer. Char formation is often enhanced by phosphorus-containing compounds that promote the dehydration and cross-linking of the fabrics. In the gas phase, nitrogen-containing compounds may release nonflammable gases that dilute combustible gases and reduce the heat release rate. Application methods for alginate-based flame retardants typically involve dip-coating or pad-dry-cure techniques. 127 In conclusion, lignin, alginate, starch, and cellulose are potential materials for the creation of natural flame-retardants for cotton fabrics. Because they are renewable and biodegradable, these materials not only improve fire safety but also support environmental objectives. To maximize their efficacy and resolve any potential disadvantages, more research in this field is necessary to guarantee a safer and more environmentally friendly future for textile applications.
Phytic acid, a flame retardant widely present in plant tissues and contains six phosphate groups. Due to its high phosphorus content and capability to interact with protein fibers by means of ionic bonding, PA has a promising flame-retardant application for protein-based textiles. 128 It is readily available and can be extracted from various plant sources, including oil seeds, beans, and cereal grains. unique structure, high phosphorus content (28 wt% relative to its molecular weight), and its ability to adhere effectively to cellulose, proteins, and a variety of fabrics, including cotton, wool, nylon, silk, and poly (lactic acid) nonwoven materials. 129 Furthermore, PA and its salts are recognized as effective partners for positively charged counterparts in layer-by-layer (LbL) assemblies, a technique widely used to create flame-retardant fabrics. It also facilitates dehydration reactions and supports carbonization. 130 Figure 8 illustrates the adsorption and flame-retardant properties of bio-based phytic acid on wool fabric

Schematic representation of bio-based phytic acid-treated wool fabric. 131
Another important benefit of using phytic acid (PA) in flame-retardant fabrics is its ability to form a protective barrier before the untreated material begins to decompose. However, when applied to cotton fabric, PA alone shows limitations because its acidic nature can damage cellulose, reducing fabric strength. 132 For this reason, PA is often combined with other compounds, leading to improved flame-retardant performance and durability in cotton textiles. In contrast, PA can be used effectively on protein-based fibers such as wool or silk, where it does not cause structural degradation, making it a valuable standalone flame-retardant agent for these materials. 133
Plant-based extracts are the other group of flame retardants, becoming more and more popular as environmentally benign and sustainable flame retardants, mostly because of their ability to create stable char layers and emit non-flammable gases as they burn, which increases fire resistance. Some plant extracts and waste products, such as false banana, spinach juice and pomegranate rind extract, contain phosphorus and positive metal ions, which can serve as flame-retardant agents for cellulosic, lignocellulosic, and protein fiber-based textiles. 134 For example, Silk fabrics were treated through a simple, facile method using bio-based tannic acid, sophora flower extract, and metal salts. The synergistic modification imparted durable flame retardancy and UV resistance while maintaining fabric performance, offering a sustainable and facile approach for functionalizing natural textiles. 135 Recent advancements in utilizing plant-originated materials as flame-retardant additives for textiles and exploring the latest developments in bio-based flame-retardant additives are shown in Figure 9, which provides insights into the total number of publications (both review and research papers) that are published and listed in the Scopus database.

Scopus distribution of research and review publications on flame-retardant textiles derived from polysaccharides and metabolites. 136
The flame-retardant action of plant extracts mainly relies on their ability to promote dehydration and carbonization, leading to the formation of a thermally stable char layer on textile surfaces. This barrier limits oxygen access and heat transfer, slowing combustion. 137 Tannin-based bio-macromolecules, rich in phenolic hydroxyl groups, can interact with protein fibers such as wool and silk through hydrogen bonding and complexation, enhancing char yield. 138 These natural compounds also catalyze crosslinking within the polymer matrix, improving thermal stability. While plant-derived flame retardants offer a sustainable alternative to synthetic ones, challenges remain in durability, leaching resistance, and washing fastness for industrial applications.
Recently, biomass-based flame retardants have focused on synergistically combining natural compounds to enhance fire resistance while maintaining eco-friendliness. 139 Biomass resources such as starch, lignin, phytic acid, tannins, and plant extracts are increasingly studied for their ability to form protective char layers, release non-toxic gases, and inhibit heat transfer. Synergistic formulations, such as lignin with phytic acid or starch with tannins, improve thermal stability, and reduce flammability more effectively than single components. For example, Mohapi et al. 140 studied that PA was used together with PER and 1,2,3,4 butanetetracarboxylic acid (BTCA) to synthesize a reactive and phosphorus-containing FR agent, namely hexa-pentaerythritol phytate hexa-butane tetra carboxylic acid ester (HPPHBTCA), which was expected to be grafted on wool fiber by the formation of ester covalent bonds using the pad-dry-cure finishing process, because the esterification reaction between HPPHBTCA and wool fiber would take place at high curing temperatures. The combinations leverage complementary properties, carbon-rich materials promote char formation, while phosphorus- or nitrogen-containing compounds enhance intumescence, creating sustainable, non-toxic, and efficient flame-retardant systems for greener materials.
Summary of natural-based flame retardants
Natural-based fire retardants have emerged as a promising and eco-friendly solution in fire safety and materials science. Extracted from renewable resources such as cellulose, starch, lignin, phytic acid, tannins, and plant-based compounds, these materials utilize their inherent polysaccharide and phenolic structures to impart flame resistance. Their primary mode of action occurs in the condensed phase, where they promote char formation when exposed to heat. 141 The resulting carbon-rich char layer serves as a durable protective barrier, effectively insulating the substrate, reducing heat release, and slowing the escape of volatile gases that sustain combustion. This mechanism is typically associated with dehydration reactions and aromatic stabilization, which enhance the integrity of the char and extend its protective function. 116 As a result, natural flame retardants are particularly attractive for applications in textiles, polymers, and coatings where long-term thermal stability and environmental safety are critical. Table 4 shows the summary table for natural-based flame retardants. In addition to their condensed-phase activities, many biomass-derived flame-retardants also function synergistically in the gas phase, further enhancing fire resistance. During thermal degradation, they can release inert or non-combustible gases that dilute flammable volatiles and oxygen, thereby weakening the combustion environment. 142 Among natural-based flame-retardants, especially those containing phosphorus or phenolic groups, also act as radical scavengers, capturing highly reactive free radicals responsible for flame propagation. This dual mechanism-char formation coupled with gas-phase interference makes natural-based flame retardants highly effective compared to traditional synthetic additives. By leveraging these synergistic effects, bio-based systems provide a sustainable, non-toxic, and efficient strategy for advancing fire safety while aligning with the growing demand for environmentally responsible materials. 143
Summary of natural-based flame retardants.

Inorganic Flame-retardant
Inorganic flame retardants are materials such as nanoclays, silica, metal nanoparticles, carbon nanotubes, and graphene that enhance textile and polymer fire resistance by forming heat-resistant barriers, promoting char formation, reducing smoke, and improving thermal stability and durability. 147 For example, nano-kaolinite was used in both pad-batch and exhaust techniques to provide a flame-retardant finish to lightweight wool fabric. The results show that thermal stability, char formation, flammability, and fire resistance were all improved by the treatment, indicating long-lasting and efficient protection appropriate for high-performance and safety-critical textile applications. 150
Silica (SiO2) and other silicate-based nanoparticles are widely used in flame-retardant textiles due to their high thermal stability and barrier-forming ability. These nanoparticles form ceramic-like protective layers on the surface of fibers when exposed to heat, slowing heat and oxygen transfer and enhancing char formation. 151 They also improve mechanical strength and reduce smoke and toxic gas emissions. Silica and silicates can be incorporated as coatings, fillers, or part of hybrid flame-retardant systems, offering an eco-friendly and efficient way to improve the fire performance of textiles. 147 For example, a flame-retardant silk fabric was developed using a nano-silica sol prepared via the sol-gel method. To enhance flame resistance, boric acid, urea, cyanoguanidine, melamine cyanurate (MCA), 1-hydroxyethane-1,1-diphosphonic acid (HEDP), and 6H-dibenz (C, E; 1,2) oxaphosphorin-6-oxide (DOPO) were incorporated into the silica sol. These additives were integrated through both physical doping and chemical bonding, improving the fabric’s durability and flame- retardant performance. The vertical test for the treated and untreated silk fabric is shown in Figure 10.

Vertical burning images of the original silk and the treated silk fabrics using modified silica sol flame retardant systems on silk fabric. 137
The other class of inorganic flame-retardants for protein fibers are the inorganic nanoparticles. Among these, nano clays have been demonstrated to outperform carbon nanotubes in improving the fire properties of intumescent formulations. When incorporated into polymers, nano clays enhance the formation of in-tumescent chars, which act as protective barriers during combustion. They act as flame retardants by forming a heat-resistant barrier on textile surfaces, reinforcing the char layer, and diluting flammable gases when intercalated with water or other volatiles.152,153 Nano clays often work synergistically with other flame-retardants, such as phosphorus or nitrogen-based compounds. Their advantages include thermal stability, abundance, enhanced mechanical strength, and reduced smoke and toxic emissions. They are commonly applied in coatings, polymeric textiles, composites, and upholstery. 154 Whereas carbon nanotubes (CNTs) are multifunctional, providing improvements in mechanical strength, electrical conductivity, and thermal stability, and are effective at low concentrations. 155
Graphene and graphene oxide consist of single or a few layers of sp 2 -hybridized carbon atoms arranged in two-dimensional sheets. Their flame-retardant action is based on creating tortuous pathways that slow heat and gas transfer, enhancing char formation and barrier strength. 156 Graphene can also be functionalized to act synergistically with other flame-retardant additives like phosphorus, nitrogen, or metal nanoparticles. 157 Figure 11 illustrates the mechanism of flame retardancy in textiles. Advantages of Graphene and graphene oxide flame-retardant finishing include improved mechanical strength, thermal stability, and electrical conductivity, with thin layers providing effective flame retardancy at low additive concentrations. 158

Schematic representation of graphene-based flame retardants for textiles. 159
Metal-based nanoparticles, including silver (Ag), copper (Cu), zinc oxide (ZnO), titanium dioxide (TiO2), and iron oxide (Fe2O3), have attracted great attention in textile applications due to their multifunctional properties. 160 In flame-retardant textiles, these nanoparticles primarily act through condensed-phase mechanisms, where they facilitate char formation, enhance thermal stability, and catalyze carbonization. By strengthening the char layer, they effectively hinder heat and mass transfer, thereby reducing flammability. In addition, Ag, Cu, and CuO NPs nanoparticles impart antimicrobial activity, 161 while ZnO and TiO2 nanoparticles provide ultraviolet (UV) protection, making them highly versatile . Their major advantages include long-lasting functionality, low additive concentrations, and excellent compatibility with polymer matrices. For example, wool fabric was treated with Metal NPs, viz., Ag, ZnO, TiO2.
At the molecular level, metal nanoparticles influence polymer degradation pathways by restricting chain mobility and scavenging reactive free radicals. They enhance thermal conductivity, suppress bubble formation, and reduce heat release by lowering local oxygen concentration through redox reactions. 162 Recently, Nano metal oxide coatings on woven fabrics have gained significant attention because they provide comparable or superior flame-retardant performance to pure metal nanoparticles, while being more cost-effective and scalable for industrial applications. 163 The flame retardant of a fabric, however, strongly depends on the type of agent used and the method of application. Each method carries unique advantages and limitations, influencing overall performance and durability. A comparative summary of selected methods, along with their benefits and drawbacks, is presented in Table 5.
Advantages and disadvantages of flame-retardant application methods. 164

Recently, Nano metal oxide coatings on woven fabrics have gained significant attention because they provide comparable or superior flame-retardant performance to pure metal nanoparticles, while being more cost- effective and scalable for industrial applications. The flame retardant of a fabric, however, strongly depends on the type of agent used and the method of application. Each method carries unique advantages and limitations, influencing overall performance and durability. 165
Shrink-proof finishing of protein fibers
Protein fibers such as wool, cashmere, and mohair require shrink-resistant finishing because their surface scales can interlock during washing, leading to felting and permanent shrinkage. 166 There are finishing solutions for such problems. The most common solutions are chlorination, which partially degrades the scales, and polymer coatings, which smooth and mask these processes that enable the production of machine-washable “Super wash” wool. 167 In contrast, silk has a naturally smooth filament surface except for the relaxation shrinkage. To stabilize it, manufacturers often apply resin finishes that help maintain fabric dimensions. 168 These treatments are crucial for modern textile care, allowing traditionally delicate, hand-wash-only luxury fibers to be transformed into practical, durable materials suitable for everyday wear without sacrificing their natural softness, luster, and warmth. 169
Mechanism of shrinkage in protein fibers
The main cause of the shrinkage of protein fibers, especially wool, is their distinct scale morphology, which makes felting easier when wet and mechanically disturbed. Heat causes structural changes in proteins, which enhance contraction and shrinkage. These fibers have the characteristics of fiber morphology that influence their shrinkage behavior. Wool fibers consist of three main components: the outer cuticle, the cortex, and sometimes a central medulla. 170 The cuticle comprises overlapping flattened cells called scales, arranged longitudinally. These scales have rough surfaces, and their arrangement can be coronal, reticulate, or coronal-reticulate in different wool types. The scale structure enables short fibers to cling and spin into threads but is also responsible for the felting and shrinkage behavior. The scales tend to interlock during washing, leading to mechanical entanglement and shrinkage. The cortex of wool has two types of cells (ortho and para-cortical) with differing chemical compositions affecting the fiber’s properties. 171 Agitation in the presence of water causes the overlapping scales on wool fibers to open and interlock tightly, forming felt. This mechanical entanglement results in fiber matting and dimensional shrinkage of the fabric or garment. Further, moisture plays a critical role in shrinking protein fibers because wet fibers have swollen fiber components that allow scale edges to open more easily, promoting felting more. 172 Figure 12 shows the mechanism of felting shrinkage of wool.

Mechanism of felting shrinkage of wool. 167
A combination of structural and physical changes brought on by washing and drying causes the silk fiber to shrink. Silk absorbs water and swells when in contact with moisture, which causes its densely packed structure to relax. The hydrogen connections that typically hold protein chains together are weakened by this swelling, which increases the fibers’ vulnerability to distortion. 173 Heat speeds up molecular motion, which results in a partial rearrangement of the protein structure, while mechanical agitation further relaxes the fiber alignment. When combined, these effects cause the fiber’s initial dimensions to decrease, and causes the fabric to visibly compress.Therefore, silk’s protein-based structure and susceptibility to environmental stressors substantially influence its shrinkage.
Conventional shrink-proof finishing methods
The traditional and commercially used shrink-resist treatments can be classified into two groups, namely (i) chlorine-based treatments, and (ii) non-chlorine/AOX-free processes. 158 Although shrink-proof is widely applied for wool, protein-like silk is also treated with finishing chemicals to make it dimensionally stable. Chlorine treatments for wool fibers chemically modify or remove outer scales by oxidizing and etching the surface. This reduces the barbed scale edges responsible for felting shrinkage. Typically, chlorine gas or sodium hypochlorite is applied, followed by neutralization and resin coating. Chlorination is highly effective in making wool machine washable, but it can weaken fiber strength and handle. It also produces environmentally hazardous chlorinated byproducts, posing challenges for effluent disposal and sustainability. 174
Resin-based finishes such as polyamide-epichlorohydrin (PAE) and polyurethane are applied to wool fibers to impart shrink resistance. These resins form thin, continuous polymer films that lock down scales and restrict their movement, preventing felting. Polyamide-epichlorohydrin resins provide wet strength and dimensional stability, while polyurethane offers flexibility. 175 Film formation occurs through cross-linking and curing on the fiber surface. Resin finishes improve shrink resistance without drastically affecting fabric softness but rely on chemical processing. For example, hercosett resin was synthesized and applied to wool fiber to impart shrinkage resistance. The mechanism of synthesis is that adipic acid is reacted with diethylenetriamine to form poly (diethyleneiminoadipamide) through condensation polymerization. This polydiethyleneiminoadipamide is again reacted with epichlorohydrin to form poly (chloro-hydroxypropyldiethylene adi-pamide ammonium chloride), which is marketed as hercosett 125 resin. 176 In the presence of an alkali, the chlorohydroxypropyl groups of this quaternary ammonium polyamide are converted into epoxy groups, forming poly (epoxypropyldiethylene adipamide ammonium chloride) that can react with carboxyl and hydroxyl groups available in wool fiber surface. 175
Thermoplastic polymer coatings create a flexible, protective layer over fibers or fabrics, preventing scale movement and moisture-induced swelling. They exhibit excellent durability due to their ability to soften and re-harden without chemical degradation. 177 Various polymeric materials are employed in textile finishing, encompassing polymers like silicones, fluoropolymers, polyurethanes, and beyond. 157 For example, polyurethane highly used for coating in the textile industry, among other polymers, because of its superior flexibility, weather resistance, and wear resistance. Several textile properties, including shrink-proofing, wear comfort, tensile strength, tear strength, wrinkle resistance, water contact angle, antibacterial properties, color fastness, etc., can be significantly enhanced by polyurethane coating. These coatings maintain fabric dimensional stability over prolonged use, reducing shrinkage and wear, making them suitable for structural and textile applications requiring long-term performance. 178
Protein fibers can also be blended with other fibers to improve dimensional stability and prevent shrinking in textiles. Because of their inherent structure and ability to absorb moisture, protein fibers like wool and silk have a tendency to shrink, causing swelling, and contraction when washed or exposed to heat. 179 The cloth as a whole becomes stronger, more elastic, and has a lower propensity for moisture when combined with synthetic fibers like polyester or nylon. For example, low-stress mechanical properties of wool-cotton blended khadi fabrics were studied by Abou-Taleb and El-Sayed 180 and the effect of different blend proportions of wool fibers and yarn count on the fabric properties was investigated. The results show that the wool-dominant fabric has higher hygral expansion in the weft direction than the warp direction. This blend preserves softness and breathability while reducing felting and shrinking. Chemical treatments can also be used to enhance fiber attachment and compatibility, ensuring the long-term shrink resistance of blended fabrics. This method strikes a balance between fabric performance and usefulness. 181 Although the traditional methods improve the shrinkage of protein fibers, the green method is the best to preserve fabric damage and reduce environmental hazards.
Eco-friendly and innovative approaches
Fibers such as wool the developed casein, fish protein fibers, and soy protein fiber have the tendency to shrink and are treated with eco-friendly finishing agents such as enzyme, biopolymers, and Nano finishing agents. 182 Wool is a scaly-surfaced natural protein fiber that provides warmth and durability but shrinks a lot when felt. Early artificial protein fibers were casein, which is made from milk protein, and fish protein fibers; they look like wool but are weaker, shrink less, and lose strength when wet. 183 Better washability and less shrinking are provided by soy protein fiber, which is smoother and more stable. 184 Silk, on the other hand, is a continuous filament fabric valued for its strength, elegance, and brilliance. Cultivated mulberry silk, wild tussah silk, muga, and Eri are among the varieties; they all have low shrinkage but vary in texture, color, and shine. 185
Enzymatic treatments
Determined that enzymes have played a significant role in enhancing the chemical processes now employed in textile wet processing. Enzyme applications in industrial processing necessitate the exact acquisition of enzymes with certain properties, which will motivate scientists to investigate novel approaches to mitigate their fragility. 186 Enzymes such as proteases, cellulases, laccases, and transglutaminases have been used in wool ant felting finishing. Proteases are used in wool finishing procedures, primarily in anti-felting treatments. Keratinases are a particular class of proteases, because of the disulfide linkages, keratin, and the protein found in wool, is difficult to break down. The wool protein chain is hydrolyzed by keratinases. These enzymes are classified as serine or metalloproteases by the reaction center. In addition to keratinases, an oxidoreductase called disulfide reductase (EC 1.8.1.8) is required for the transformation of keratin. Actually, the redox enzyme catalyzes the breakdown of the -S-S-bonds, which results in -SH groups and initiates the keratin degradation process. The chain’s stereochemistry is altered by this alteration, allowing Keratinase to enter and hydrolyze the amino acid chain either internally (endo) or at the N/C terminal (exo). 187 Figure 13 eco-friendly enzymatic shrink-proofing of wool fiber surfaces. Thus, to carefully treat wool, enzymes-more especially, proteases are utilized. They smooth and soften the scales by partially digesting the outer cuticle. By keeping the threads from sticking together, this lessens shrinkage without causing harm to the inner core. Eco-friendly, enzyme treatment maintains the natural feel, softness, and elasticity of protein fibers, especially for wool.

Rapid enzymatic shrink-proofing effect on wool fiber morphology. 188
Plasma treatments
Plasma treatments are dubbed as a sustainable, shrink-resistant treatment as they do not produce any effluent. Plasma is the fourth state of matter, as matter in that state has properties dissimilar to its solid, liquid, or gaseous state. It is a mixture of partially ionized gas that is generated by an electrical discharge. When gas molecules are excited by a high voltage discharge, they dissociate into negatively charged electrons and positively charged ions, producing plasma. 189 Wool fabric shrink resistance has only been studied with non-monomeric gases (air, oxygen, argon, etc.), even though monomeric gases, such as organosilanes and fluorocarbons, have been studied for hydrophobic and oleophobic coating of textiles. Instead of only affecting the fabric’s surface, plasma treatment also affects the scales of individual fibers. One can categorize the plasma generators into two groups: atmospheric pressure plasma and low-pressure plasma. The ability of both kinds of plasma treatments to prevent shrinkage in wool was examined. Because the treatment may be done in a continuous mode, the atmospheric plasma procedure is superior to low-pressure plasma. This group includes barrier discharge plasma systems and corona systems. 190 Furan is eco-friendly, energy-saving, non-deteriorative process for the fabrication of machine-washable wool tops (WTs) was proposed. WTs were exposed to atmospheric air or an argon plasma treatment at different powers for various durations, followed by coating with a protein biopolymer, namely keratin, using the pad-dry-cure technique. The felting resistance of the finished WTs was evaluated, and the results revealed that the finished sample has an enhanced felting resistance to the extent of machine-washable wool (no felt ball is formed), whereas the untreated sample forms a felt ball with a diameter of 2.166 cm after undergoing the felting test. The treatment process is shown in Figure 14.

Treatment of wool tops with plasma/keratin system.
Anti-felting finishing and low-temperature dyeing of wool by using plant protease were studied extensively. Plant protease acts by selectively breaking down keratin in the scale layer of wool fibers through a controlled hydrolysis mechanism. This process gradually peels away the outer surface layers of the wool scales, causing minimal damage to the inner layers and the cuticle microfibrillar complex (CMC) layer. As a result, the enzyme treatment effectively reduces felting shrinkage while preserving the overall fiber strength and integrity, enabling improved dye uptake at lower temperatures. 192
Biopolymer-based coatings
Renewable resources are used to make biopolymers, which can offer more environmentally friendly substitutes for conventional synthetic polymers. The primary source of cellulosic and protein fibers used as raw materials for textile goods has traditionally been biopolymers. However, because biopolymers are biodegradable, have a low carbon impact, and work well with natural fibers, the textile industry has recently shown a growing interest in using them for new purposes. 193 Table 6 illustrates the polymeric resin used for Shrink-resist treatments of wool fabric.
Shrink-resist treatments based on covering the inter-scale spaces with a polymeric resin and their shrink-resist performance. 158

Biopolymer-based coatings have been researched as effective and sustainable treatments for shrink resistance of wool and regenerated protein fibers. Studies show that biopolymers such as chitosan, gum arabic, and wheat starch can be used as coatings to reduce wool fabric shrinkage significantly (to less than 4%) without adversely affecting its original physical and mechanical properties, as shown in Figure 15. These coatings work by forming a protective layer on the fiber surface, enhancing dimensional stability while maintaining fabric strength and appearance. 194 Mondal et al. 195 studies the effect of biopolymer-treated wool fabric composed of gum arabic, chitosan, and wheat starch to improve its dimensional stability. The result shows that a good area shrinkage for the unmodified fabric was measured as 16.4%, the combined treatment showed only 3.6%. A reduced yellowness of the modified fabric was also found, which may be an advantage compared to conventional chlorine bleaching.

Schematic representation of biomaterial-based shrink-resistant treatment of wool fabric. 158
Nano-finishing techniques
Textile industries started working on specialty nano finishes in bulk production. Metal NPs, viz., Ag, ZnO, TiO2, are being used for various functional finishes in addition to biomass-based nanoparticles. Figure 16 shows the proposed mechanism of wool surface acylation and attachment of TiO2 nanoparticles to the wool fabric. The finishing of textile goods ultimately depends on the end uses. The apparel quality fabric needs a softer finish, whereas an industrial fabric may need a fire-retardant finish. 195 The use of nanotechnology has opened up the textile finishing area to numerous possibilities for achieving multifunctional properties in natural textiles. 196 In comparison with conventional chemical, physical, and physicochemical modifications, nanotechnology is considered a futuristic approach for improvement in the performance of textiles. For example, nano-chitosan derived from the chitin component of the shells of crustaceans was used in wool fabric modification. It holds promise for the replacement of some synthetic polymers having desirable properties that consist of nontoxicity, biocompatibility, biodegradability, antimicrobial activity, and chemical reactivity. It had been used mainly for shrink resistance, dye-ability, and antimicrobial effect in wool finishing treatments. 197 Another research using protease-enzymatic wool was treated with citric acid and Fe3O4 nanoparticles. The result shows that Fe3O4 nanoparticle/citric acid treatment on the protease enzymatic wool improved tensile strength and elongation, along with the higher alkali resistance and controllable hydrophilicity/hydrophobicity. 198 In the other study, silver nanoparticles have been used to functionalize wool fabric. The treatment was durable up to 20 wash cycles, resulting in better color and surface functional properties, which can indirectly affect shrink-resistance. 199

The proposed mechanism of wool surface acylation and attachment of TiO2 nanoparticles to the wool fiber surface. 200
Summary on shrink-proofing protein fibers
Eco-friendly shrink-proof finishing techniques are being used more and more on protein-based fibers like collagen, keratin, and elastin, soybean and regenerated casein, even for silk fiber, since it has a relaxing shrinkage, and are no longer just for wool.201,202 Similar to wool, these fibers are prone to shrinking and creasing, thus, preventative treatments are necessary to preserve their delicate structure and improve usability and durability. Sustainable methods aim to reduce their negative effects on the environment while enhancing the longevity and performance of fibers. Enzymatic treatments provide an eco-friendly alternative. Protease enzymes can selectively hydrolyze the surface scale layers of protein fibers, thereby minimizing the felting shrinkage responsible for fabric deformation. 188 For regenerated protein fibers such as casein and soybean, carefully controlled enzymatic hydrolysis reduces shrinkage without significant loss of tensile strength. The effectiveness of enzymatic methods is further improved when combined with nanoparticles or natural polymers like chitosan, creating multifunctional finishes with both protective and antimicrobial effects.Whereas the plasma treatment offers an additional sustainable option, as it is a solvent-free, dry process that alters fiber surface chemistry to increase hydrophilicity or hydrophobicity. This enhances shrink resistance and can be seamlessly integrated with nanoparticle coatings or enzymatic finishes for greater efficiency. 203 Nanoparticles such as nano-chitosan and titanium dioxide are widely used to enhance surface properties, antimicrobial activity, and dyeability, in addition to shrink resistance. This process promotes stronger adhesion of nanoparticles to fibers, resulting in improved cohesion and shrink resistance. 204 In general, synergistic approaches combining nanoparticles, biopolymer coatings, enzyme treatments, and plasma technologies are necessary for environmentally friendly shrink-proof protein fiber finishing. These methods promote the shift to more sustainable textile production by lowering dependency on traditional chlorination, maintaining fiber quality, and adding functionality.
UV-protective finishing solution for protein fibers
There is a progressive increase in UV radiation on human skin caused by the depletion of the ozone layer in the Earth’s atmosphere. As long-term exposure to UV light can result in a series of negative health effects, such as acceleration of skin aging, photo dermatosis, erythema, and even skin cancer. Developing textiles with UV protection functionality has been widely researched up to now. 205 Fabrics has been specifically processed to protect personnel from risks brought on by extreme environmental changes, hazardous working circumstances, or enemy activity The phrase “personal protective” refers to a broad range of items, such as good visibility clothing for employees near traffic, flame-proof protective gear for racing drivers, flame and heat-resistant turnout suits for firefighters, gas, liquid, and dust-proof outfits for chemical industry workers, chain-mail gloves for food vendors, conveyor-belt garments for the forest management workforce, safety goggles and hard-toe boots for construction workers, welders’ UV protective gloves and goggles, and hearing protection. Simply put, protective clothing is anything that a person wears to put distance between themselves and danger, allowing them to remain nearby while being less likely to be injured. Utilizing protective clothing and accessories in addition to avoiding the sun is considered sun protection. Reduced sun exposure, sunblock use, and wearing protective clothing are the three main defenses against the harmful effects of UV rays.206,207
Mechanism of UV-protection
The mechanism of UV protection involves three main actions: absorption, reflection, and scattering of harmful ultraviolet rays. 208 These absorbers may be organic or inorganic from different sources. The functionality of natural origin-based fibers can provide minimal protection against UV exposure; however, surface modification of fabrics can help to achieve better outcomes. Plant-derived organic substances like flavonoids, tannins, and lignin absorb UV rays and neutralize them by transforming them into innocuous energy. 209 Additionally, animal-derived proteins that bind UV-absorbing compounds, such as keratin and chitosan from crustacean shells, can improve protection. 210 Inorganic substances, including metal nanoparticles such as zinc oxide and titanium dioxide, are good UV ray reflectors, and scatters. 211 When grafted or applied to textiles, these substances form a multipurpose barrier that lowers UV ray penetration while preserving the comfort and longevity of the fabric. For example, Zinc oxide nanoparticles offer textiles efficient UV protection via a variety of methods. It has a broad band gap of around 3.37 eV, effectively absorb UV light by stimulating electrons and blocking damaging rays from reaching the skin. 212 This is especially true in the UVA and UVB wavelengths below 380 nm. 213 Their addition to textile fabric greatly raises the ultraviolet protection factor (UPF); increased ZnO loading results in UPF values which guarantee better blocking of UVA and UVB rays. The schematics of the photocatalytic mechanism of ZnO NPs are shown in Figure 17. Furthermore, ZnO produces reactive oxygen species (ROS) through photocatalytic activity when exposed to UV light, which has antibacterial and self-cleaning properties while preserving fabric integrity. 214

Schematic presentation of ZnO’s photocatalytic mechanism.
Challenges with traditional UV-blockers
Conventional UV-blockers are essential for preventing UV radiation damage to textiles, particularly delicate ones. These substances, which include mineral oxides, benzotriazoles, and benzophenones, scatter or absorb dangerous radiation. 215 They improve longevity and maintain the esthetic attributes of textiles across applications by reducing yellowing and fiber deterioration. The mechanisms of action are achieved by using aromatic–carbonyl conjugation to absorb UV light, benzophenones achieve excited states and release energy as light or heat, while they can also occasionally generate reactive oxygen species. Using excited-state intramolecular proton transfer (ESIPT), benzotriazoles absorb widely and release energy. They cycle effectively and are photostable, transforming dangerous UV rays into innocuous thermal energy. 216 Protein fibers with a high sensitivity to UV light include silk (fibroin) and wool (keratin). Long-term UV exposure causes fiber weakening, discoloration, and a loss of natural. Because sunshine and other external factors can readily erode these fragile fibers’ beauty and durability, handling them carefully is necessary to protect them. 217 For example, the photo yellowing and poor UV-protection ability of silk, wool and nylon fabric was improved through the anti-UV finishing with UV-absorbers. This study is concerned with the adsorption properties of a water-soluble benzotriazole UV-absorber on silk, wool and nylon. It was found that the adsorption kinetics of the UV absorber on the three fibers followed the pseudo-second-order kinetic model, and the rate constant indicated a faster adsorption rate for the UV absorber on silk than those for wool and nylon. 218
Conventional UV blockers like benzophenones and benzotriazoles have long offered reliable protection, but there are issues with their possible negative effects on health and environmental persistence. 219 This has sparked research into safer substitutes, especially inorganic UV absorbers that provide broad-spectrum protection with low toxicity, including titanium dioxide and zinc oxide. Natural UV protection is also provided by plant sources such as flavonoids, phenolic acids, and cyclosporine-like amino acids. 220 Inorganic filters, hybrid nanomaterials, and encapsulating techniques are among the alternatives that try to lessen environmental and health dangers. While inorganic compounds offer non-toxic alternatives, they are occasionally constrained by esthetic or material compatibility concerns. Organic-based retardants are nevertheless useful for striking a balance between performance and stability. A strong desire to develop solutions that preserve protective performance while improving environmental safety and human health compatibility is seen in the continuous development of new UV stabilizers. 221
Green UV-protection solutions for protein-based textiles
Eco-friendly UV protection approaches, including naturally based and inorganic for protein-based textiles are becoming increasingly important owing to the aforementioned environmental and health concerns associated with benzotriazoles and benzophenones. The escalating global threat of ultraviolet (UV) radiation is manifested through multifaceted damage pathways, including cutaneous carcinogenesis, photodegradation of organic substrates, marine ecosystem destabilization, and infrastructure weathering. These urgent challenges have catalyzed sustained interdisciplinary efforts toward advanced UV-shielding technologies spanning biomedical, environmental, and industrial domains. Current material arsenals include melanin, lignin, tannin, polydopamine, zinc oxide, and titanium dioxide, etc. 222 In particular, UV protection products derived from plants, animals, and inorganic materials are essential to developing practical defenses against dangerous radiation. This section examines the advantages of eco-friendly UV protection techniques in improving performance, sustainability, and safety.
Animal-based UV-protection solution
Natural substitutes for synthetic filters that are harmless to the environment and effective are UV-protecting chemicals derived from animals. Mycosporine-like amino acids (MAAs), which are produced by marine creatures like fish, corals, and sea urchins, are photostable and biodegradable despite their significant absorption of UV-A and UV-B rays. 223 Fish eggs and some marine species contain gadusol, anothersignificant substance that serves as a natural sunscreen to shield delicate embryos from UV rays. 224 Furthermore, melanin, which is found in animal tissues, absorbs damaging UV rays and releases them as heat. Sustainable, bio-based UV protection solutions with lower dangers to ecosystems and human health are inspired by these natural processes. 225 Andrady et al. 226 studied shows that the treatment of chitosan combined with bio-mordants like Rubia cordifolia dye improved the ultraviolet protection factor (UPF) of silk fabric remarkably. The result showed excellent UV resistance with improved washing and light fastness, alongside antimicrobial activity against bacteria like E. coli and S. aureus.
Further, to improve the flame retardancy and UV protection of silk fabric, a new and environmentally friendly coating that combines polyaniline (PANI) and chitosan was created. Due to PANI’s potent UV-absorbing qualities, this novel coating provides exceptional UV shielding, yielding a high Ultraviolet Protection Factor (UPF) of more than 50. By working in concert, the polyaniline/chitosan composite offers long-lasting protection without sacrificing the inherent properties of the fabric. This method provides an efficient and environmentally responsible way to create sophisticated protective silk fabrics that may be used in a variety of high-performance applications. 111 Effective UV protection for silk textiles is achieved through the layer- by-layer self-assembly production of oxybenzone-grafted chitosan biomass coatings, as seen in Figure 18. By utilizing the UV-absorbing oxybenzone that has been grafted onto chitosan, this novel coating technique improves UV protection. The silk fabric’s UV resistance is greatly increased by the biodegradable and biocompatible coating, which qualifies it for protective textile applications. 227

Oxybenzone-grafted chitosan biomass coatings for UV-protective silk fabrics. 227
Plant-based UV-protection solution
Natural UV protection is offered by a variety of plants, particularly for protein fibers like silk and wool. Flavonoids that absorb ultraviolet light and function as antioxidants include apigenin, kaempferol, rutin, and quercetin. 228 Turmeric’s curcumin adds color while absorbing UV-B rays. Aloe and madder root anthraquinones can protect against UV rays. Gallic and ellagic acids are examples of phenolic acids that are beneficial. Extracts high in polyphenols from walnut hulls, grape seeds, and green or black tea provide UV protection; they are frequently used with natural binders or mordants for longevity. 29 Plant-based UV protection solutions for silk and wool textiles often use natural colors and plant extracts that contain bioactivesubstances that can absorb or block UV rays. Numerous studies demonstrate the efficacy of plant extracts and dyes that offer further benefits, including antibacterial activity in addition to UV protection. 229 A recent study investigated the enhancement of antibacterial and UV protection properties in blended wool/acrylic and silk fabrics by dyeing them with Mimusops elengi leaf extract, both with and without metal salt mordants. 230 Ghazal et al. 231 have done research concerning natural dye extraction from the leaves of eucalyptus and the application of this dye for wool fabric dyeing using two padding techniques, namely the pad-batch and pad-dry techniques, under different conditions. It was observed that with an increase in the dye concentration, the ultraviolet (UV) protection factor (UPF) values ranged between very good and excellent for wool fabric. Table 7 shows the sustainable dyeing of wool and silk with Conocarpus erectus L. leaf extract for the development of functional textiles. The results indicate an increase in ultraviolet (UV) protection, suggesting the effectiveness of the plant-based treatment.
UPF assessment of dyed and undyed wool and silk samples. 33

Further, plant extracts work in combination with other agents to improve the UV protection of wool and silk textiles. They can interact with bioactive chemicals in plant extracts to improve dye fixing and greatly increase UV protection in addition to their own UV protection. 232 Mixing nanoparticles like zinc oxide or silver with plant extracts offers antibacterial properties and improved UV blocking by combining chemical and physical UV absorption mechanisms. 233 When natural polymers like chitosan are combined with extracts, bioactive, biodegradable coatings are produced that increase durability and UV resistance. In textile finishing applications, these combinations improve color fastness, sustainability, and usefulness. 234
Inorganic UV-protection solution
Inorganic UV blockers are considered the best as they have no toxicity and are chemically stable under long exposure of high temperature and UV. These include clay and metal nanoparticles that are effectively incorporated into protein fibers to enhance their structural, functional, and protective properties. Clay nanoparticles, such as montmorillonite or kaolinite, improve thermal stability, mechanical strength, and barrier performance by creating a tortuous path within the fibrous matrix, while also aiding in moisture regulation. 235 Metal nanoparticles, including silver, zinc oxide, and titanium dioxide, impart antimicrobial, UV-protective, and catalytic functionalities, making the fibers suitable for biomedical, textile, and protective applications. 236
As very effective UV filters, metal nanoparticles such as ZnO and TiO2 nanoparticles are the main ingredients of inorganic UV-protection solutions for protein fibers. In order to protect the fibers and, when applied to textiles, the wearer’s skin, these nanoparticles scatter and absorb damaging UVA and UVB radiation. 237 The natural characteristics of the fibers are not substantially changed by application techniques, including dip-coating, surface immobilization, or in situ synthesis, which enable uniform nanoparticle integration. 238 The synergy of both organic-inorganic systems can offer versatility beyond UV protection, such as flame-retardant, photochromic, and long-lasting qualities. Crucially, these treatments preserve protein fibers’ mechanical integrity, biocompatibility, and biodegradability. For example, a study was done on TiO2-coated silk fabric with controllable UV-protection properties was successfully fabricated via atomic layer deposition (ALD). The result show that better luster, bending properties, crease recovery, good mechanical properties, air permeability, and better UV properties were obtained. 239 Another study was done on bio-interfacial material, tannic acid, to enhance the π interaction for better reusability of the nanostructured zinc oxide (ZnO) modified highly-improved-UV-protective natural textiles, which ultraviolet protection factor (UPF) maintained 309 despite 50 washing cycles as shown in Figure 19.

Tannic interfacial linkage within ZnO-loaded fabrics. 240
Summary of natural-based UV-protection finishing solutions
Protein fibers can degrade, lose strength, and fade in color due to their heightened sensitivity to UV radiation, and their inherent UV protection is low. 241 Using natural substances derived from plants and other biological sources reduces the environmental impact of synthetic UV stabilizers and provides a sustainable alternative. Animal and plant-based UV-protection finishing solutions for protein fibers provide an effective means to safeguard users from ultraviolet damage. Protein fibers are naturally vulnerable to UV radiation, which can degrade their molecular structure, resulting in weakened fabric strength and fading colors. 156 To combat this, finishing agents derived from both animal and plant sources are being developed and offer eco-friendly alternatives to synthetic UV stabilizers. Plant-based solutions typically include extracts rich in polyphenols, flavonoids, and tannins, found in botanicals such as green tea, grape seed, and pomegranate. These natural compounds absorb UV rays and neutralize free radicals generated by UV exposure, thus preventing photo degradation. 242
Animal-based UV protectants, on the other hand, may include chitosan, chitin and collagen derivatives, which improve moisture retention and fiber flexibility while creating protective barriers on the fiber surface. 213 Ensuring equal coverage and long-lasting UV protection requires combining these natural ingredients with appropriate application methods, such as padding or spraying. The softness, breathability, and biodegradability of protein fibers can be solved by these solutions, which also offer efficient protection against damaging solar radiation. By using UV treatments derived from plants and animals, the textile industry moves closer to eco-friendly methods that prolong the life of natural fiber goods and cut downon chemical waste. This harmonious integration of textile technology and nature-inspired science supports a more sustainable future for preserving premium fabrics. Employing green methods to modify protein-based textiles for UV protection is particularly vital in regions with high sun exposure. Figure 20 illustrates the difference in protective performance between untreated fabrics and those enhanced with eco-friendly UV-protective coatings

A schematic diagram showing the protective action of fabric with and without protective coating. 243
Eco-friendly finishing solutions with multi-functionalities
Multi-functionality approaches
The textile industry has developed cutting-edge, environmentally friendly finishing techniques that combine wrinkle resistance, UV protection, and flame retardancy. Combining these features into one treatment is a significant advancement that improves fabric performance, safety, and durability while adhering to environmental regulations. Such multipurpose finishes provide complete solutions for contemporary clothing and home textiles. 244 A variety of techniques that blend various treatments, materials, and structures to impart numerous desirable features simultaneously are used to give textiles multi-functionality. Surface functionalization is a popular technique that involves applying nanocoatings like graphene oxide, zinc oxide, titanium dioxide, or silver to textiles to give them antibacterial, UV, and self-cleaning qualities. 238 The approach involves the incorporation of functional additives like micro or nanocapsules, which can release fragrances, skin-care agents, or temperature-regulating phase change materials. These can be combined with antimicrobial agents such as silver nanoparticles, chitosan, or quaternary ammonium compounds to deliver multiple benefits simultaneously. 245 For example, a green facile nano-finishing route was developed to impart high antibacterial efficacy, UV-protection, self-cleaning and anti-wrinkle functions to cotton/wool and viscose/wool blends using TiO2, and/or Ag-NPs, as active ingredients, along with citric acid and/or succinic acid/SHP as ester-crosslinking/fixing systems. The result obtained demonstrated that outstanding, durable functional properties were achieved. 246
At the fiber level, modification during production allows permanent functionality to be built into textiles. Fiber blending, such as mixing cotton with wool or silk with cotton fibers, merges natural comfort with strength, conductivity, or protective properties. 247 Bi-component fibers are designed with dual properties, such as one side being hydrophobic and the other hydrophilic, enabling improved moisture transport and thermal control. Structural design also plays an important role in multifunctionality; layered composites combine water-repellent outer layers with moisture-wicking inner surfaces and antimicrobial cores, while advanced knitting or weaving patterns allow localized breathability, elasticity, or reinforcement. 248 In conclusion, using single plant extracts that have antimicrobial, UV protection, and antioxidant qualities; animal-based materials like chitosan that have antimicrobial, antistatic, and dye-fixing qualities; and inorganic agents like ZnO and TiO2 nanoparticles that have antibacterial and UV shielding properties, textiles can become multifunctional. Synergistically combining these materials results in fabrics that are high-performance, long-lasting, and sustainable.
Plant and animal-based finishing solutions with multi-functionalities
Plant-based extracts and essential oils have become viable and efficient substitutes for protein-based textile finishing, providing exceptional versatility. They can provide water-repellent, UV-protective, antioxidant, flame-retardant and wrinkle-free qualities. 249 These natural substances can improve textile performance and lessen dependency on synthetic chemicals. By combining several uses into one treatment, they raise fabric safety, comfort, and durability without sacrificing environmental norms. Because of their low toxicity and biodegradable nature, they are also perfect for environmentally friendly uses in technical materials, household textiles, and clothing. Making use of plant-derived solutions is a big step toward more environmentally friendly, high-performing textile finishing techniques that support sustainability objectives.250,251 For example, multifunctional wool fabrics were produced by treating them with silver (Ag) nanoparticles in a single-stage treatment using trisodium citrate (TSC) as a reducing and capping agent. The treated wool fabric showed excellent antibacterial activity, UV radiation absorption capacity, and also very good antistatic properties, along with an excellent colorfastness to washing. 252 Another study was done on the functionalization of silk with antimicrobial and ultraviolet protective properties using a dye extract from the roots of Rubia cordifolia L. in conjunction with chitosan as a bio-mordant. Alum mordant was also used in the dyeing process alongside chitosan for comparison purposes. 253 Figure 21 illustrates a study on turmeric (Curcuma longa L.) dye extract is combined with chitosan to markedly enhance both the antibacterial activity and UV-shielding properties of silk.

Schematic representation of turmeric dye extract is combined with chitosan as a multifunctionality. 253
Furthermore, finishing solutions derived from animals provide a multipurpose and sustainable way to improve textiles, especially those that are high in protein. These finishes, which are made from lipids like lanolin and beeswax, proteins like casein, keratin, and gelatin, or polysaccharides like chitosan, offer a number of advantages. 254 They can provide enhanced softness and handling, UV protection, water and oil repellency, antioxidant activity, and antibacterial and antifungal qualities. 255
Synergetic effect of green solution with multi-functionality
A complex strategy that incorporates a variety of environmentally friendly finishing techniques is necessary to achieve multi-functionality in textiles, as was covered in the preceding section. Instead of relying solely on one environmentally friendly solution, such as plant extracts, keratin, chitosan, or nanomaterials, textile research has shown a great deal of interest in the synergistic effect of integrating several strategies. The synergistic effects of green solutions offer a multifunctional approach for enhancing the functionality of protein-based fibers. Metal nanoparticles (NPs) in combination with plant-derived extracts can provide multi-functionality such as antibacterial, antioxidant, and UV-protective qualities while preserving the integrity of the fiber. Likewise, animal-based functionalizing materials combined with plant extract as well as metal nanoparticles can improve bioactivity, durability, and mechanical strength.256,257
Protein fibers, such as silk and wool, can achieve sophisticated functions without the use of hazardous chemicals by incorporating bio-inspired techniques. This method demonstrates how eco-friendly, sustainable nanotechnology can be used in textile engineering to create fibers with enhanced performance, versatility, and biocompatibility. 258 For example, multipurpose wool fabric was developed by Bulfa et al. 259 The study employed nitrogen-doped ZnO/TiO2 photocatalysts enhanced with silver. Even while this nanocomposite only had a 4% reflectance in the 200–380 nm region, it greatly enhanced UV protection. Strong antibacterial and antifungal properties without cytotoxicity were also supplied, along with an increase in tensile strength, allowing for long-lasting, multipurpose fabric performance. Multifunctional wool fabric finished with silver- modified N-doped ZnO/TiO2 is shown in Figure 22. Another study on silk multifunctionality was done to improve its poor resistance to ultraviolet (UV) light and to wrinkles. Nano-silica (nano-SiO2) is applied to the surface of silk fabric as a finishing agent. The result demonstrates that after completing in a solution containing 10 g/L nano-SiO2 and 20 g/L silane coupling agent (KH570) at 80°C, the UV protection factor (UPF) value recorded reaches a maximum of 84.52. The treated silk materials also shown enhanced hydrophobicity and resilience to wrinkles.

Multifunctional wool fabric finished with silver-modified N-doped ZnO/TiO2 photo catalysts. 260
Furthermore, modern textile finishing techniques have made great strides in creating multi-functional textiles with properties like UV protection, wrinkle resistance, and flame retardancy. 147 Layer-by-layer (LbL) self-assembly has become one of the most successful methods among them because it allows for the integration of several functionalities in a single treatment and provides perfect control over the deposition of functional materials. 261 Other promising methods besides LbL include chemical grafting, which permits covalent attachment of functional moieties for long-lasting effects; plasma treatment, which improves surface reactivity and adhesion of functional layers; and sol-gel coatings, which provide excellent durability and uniform coverage of functional nanoparticles. 262 By the combination of various techniques or their selection based on the performance criteria, researchers can create customized multifunctional finishes that satisfy the increasing needs of high-performance functional textiles.
Durability challenges and solutions
The major problem in improving the functionality of protein fibers, like wool and silk, is the durability challenge. Because of their softness, luster, and comfort, these natural fibers are widely prized; yet, their resilience to microbial attack, shrinkage, UV deterioration, washing, and mechanical abrasion compromises their durability.263,264 Though they frequently have poor long-term stability, conventional surface modifications, such as layer-by-layer (LbL) self-assembled coatings, have shown promise in imparting multifunctionality. However, over time, the coating’s effectiveness diminished due to peeling or leaching caused by mechanical stress, repeated washing, and exposure to environmental factors. To overcome these limitations, covalent grafting techniques have emerged as a more robust alternative, enabling permanent chemical bonding between the functional material and the fiber substrate. For example, wool fiber was graft copolymerized with poly (styrene sulfonate) to enhance its fire-retardancy, antibacterial activity, antistatic properties, stain-resistance, and tensile strength as shown in Figure 23.

A facile surface phosphorylation grafting strategy to fabricate durable flame-retardant wool fabric. 265
An alternative to covalent grafting is provided by sol-gel processes, which produce inorganic–organic hybrid networks that are firmly bonded to the fiber surface, improving resistance to washing and mechanical wear. A common surface activation technique is plasma treatment, which produces reactive functional groups on wool or silk that improve the binding of antibacterial or flame-retardant substances. 266 Additionally, hydrogel-based coatings, which combine antibacterial, water-retention, and flame-retardant qualities, are a potential breakthrough. Although protein fibers must be optimized to avoid stiffness or loss of breathability, their capacity to create robust crosslinked networks on textiles allows them to function as moisture reservoirs and heat barriers. 267 By providing mechanical strength, antibacterial activity, shrink-proof properties, and UV protection, nanomaterial incorporation, such as graphene oxide, silica nanoparticles, or silver nanoparticles, offers yet another long-lasting alternative. In addition to bio-inspired techniques, materials like in-situ polymerization or nanoparticle grafting that are implanted into protein fibers do not leak even after multiple washings and physical stress.268,269 These multipurpose tactics not only improve durability but also support sustainable and eco-friendly practices, which are becoming more and more crucial in the textile sector.
In conclusion, improving the durability of protein fibers requires a combination of methods such as covalent grafting, sol-gel hybridization, plasma activation, hydrogel incorporation, nanomaterial embedding, and bio-inspired coatings. By integrating these methods, it is possible to achieve long-lasting multifunctional protein-based textiles that retain their esthetic and mechanical properties while providing enhanced fire safety, antibacterial performance, UV protection, and wear resistance, thereby meeting the growing demand for sustainable, high-performance fabrics.270–272
Cost-effectiveness and feasibility of multi-functionality for mass production
Large-scale manufacturing of multifunctional textiles needs economical and ecologically friendly finishing techniques. Although they offer good performance, conventional chemical finishes like heavy-metal-based antimicrobials and halogenated flame-retardants are becoming less and less available because of health and environmental issues, as well as the high cost of waste disposal. 273 To address this, eco-friendly chemical strategies have emerged as promising alternatives. Covalent grafting with bio-based agents such as tannic acid, chitosan, and phosphorylated lignin provides durability and multifunctionality at a comparatively low cost because these raw materials are renewable and plentiful. This process creates strong coatings that provide flame retardancy, UV protection, and antibacterial properties without requiring a lot of energy or chemicals.257,274 Similar to this, chemical grafting with plasma assistance improves fiber reactivity and makes it possible to fix functional molecules effectively, which minimizes the need for repeated treatments and lowers overall production costs. 258
Nanomaterial incorporation, when derived from sustainable sources such as bio-silica or plant-based nanocellulose, green-synthesized nanoparticles provide multifunctionality while avoiding expensive synthetic nanoparticles.259,275 Crucially, environmentally friendly materials show promise in providing flame retardancy, UV protection, and other properties while preserving breathability using scalable padding or dip-coating techniques. They are easy to integrate with current textile finishing lines, minimize waste, and lessen reliance on expensive raw materials.147,261 From an industrial point of view, water-based chemical processes involve fewer process steps and rely on renewable ingredients, making them the most cost-effective. Table 8 shows the comparison of some selected metal nanoparticles with respect to cost, toxicity, and UV-protection efficiency. The majority of chemical agents are dissolved in water to make a finishing bath. Following this, fabrics are treated using padding, dipping, or exhaustion techniques, in which the chemicals are carried by water. 262 Therefore, using sustainable chemical and bio-based finishes that offer long-lasting performance while meeting environmental and regulatory requirements is essential to the viability of mass-producing multifunctional textiles.
Comparison of some selected metal nanoparticles with respect to cost, toxicity and UV-protection-efficiency. 276

Challenges and future directions
Eco-friendly flame-retardant UV-protection and shrink-proof finishing solutions for protein textiles face several challenges, but also present significant opportunities for future research. The primary challenge lies in developing durable and effective treatments that maintain the intrinsic properties of the fabric without compromising environmental sustainability. For example, a durable flame-retardant silk fabric was developed by the combination of Gallic acid, polyethyleneimine and metal ions. The result shows a better flame retardancy up to 50 wash cycles, as shown in Figure 24. Additionally, bromelain, a proteolytic enzyme, was used with salt in an acidic environment to prevent surface hydrolysis, making wool fabric more environmentally friendly and long-lasting. Following the Box-Behnken design, a two-step acidic bromelain treatment was developed with optimal parameters to reduce area shrinkage, weight loss, and strength loss while improving fabric softness and overall performance. 263

Schematic representation of the eco-friendly flame-retardant treatment on silk fabric. 264
Future studies should focus on exploring these synergistic combinations of biomass organic and inorganic finishing agents by considering the cost of finishing to achieve multifunctional properties. Advanced finishing techniques, such as sol-gel processing, sol-gel-derived hybrid coatings, cross-linking with eco-friendly binders, and nanoparticle incorporation, should also be explored to enhance durability and functionality while ensuring sustainability. Furthermore, eco-friendly textile alterations such as enzymatic, supercritical CO2 dyeing/finishing, and plasma treatments might greatly lessen reliance on harsh chemicals. Careful evaluation of lifecycle implications, such as resource consumption, energy use, and waste creation during large-scale production, is necessary to ensure their sustainability. Market changes and consumer acceptance, which impact adoption, are equally significant. Gaining insight into customer preferences can help direct marketing tactics and increase the adoption of eco-friendly textiles. Targeted consumer perception surveys and retailer partnerships to promote improved finishes should be the main areas of future study.
Conclusion
The development of green solutions for enhancing flame-retardant, shrink-proof and UV-protection finishing of protein textiles is one of the great steps forward in the textile industry. This review highlighted a trend toward the use of natural materials, such as plant-based extracts, bio-based polymers, and nanomaterials, as efficient flame retardants, as well as UV protection and shrink-proof finishing. It seems that the efficiency of these eco-friendly solutions depends on their origin and formulation, and some of them could show a synergistic effect when combined with different natural agents. This review examines the use of underlying natural agents, biotechnological approaches, and nanomaterials to achieve multifunctionality. The application of compounds such as essential oils, metal nanoparticles, such as ZnO and TiO nanoparticles, together with novel synthesis methods, represents new perspectives in producing textiles. The development of such dual-functional solutions has taken into account flame-retardant, shrink-proof, and UV-protection finishing, key concerns for safety and hygiene. Further, different innovative techniques such as layer-by-layer self-assembly and covalent grafting have opened up new opportunities for durable multifunctional fabrics that could meet modern consumer demands. However, despite these, scalability, durability, washability, and cost-effectiveness continue to remain bottlenecks to the widespread use of eco-friendly treatments. Most of these promising methodologies remain at the research level and need further refinement to make them viable industrially. Further research on optimization of formulations with respect to durability-performance aspects, studying practical applications to bridge the gap between laboratory innovations and real-world applications, is needed.
Footnotes
Acknowledgements
The authors express gratitude to the current and previous group of researchers in the field of textile finishing.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Author contributions
Girma Moges: Preparation of the original draft, outlining, editing, and revision; Seid Ahmed: Editing and reviewing; Mihret Pawlos: Editing the draft and improvement of intellectual content.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
