Abstract
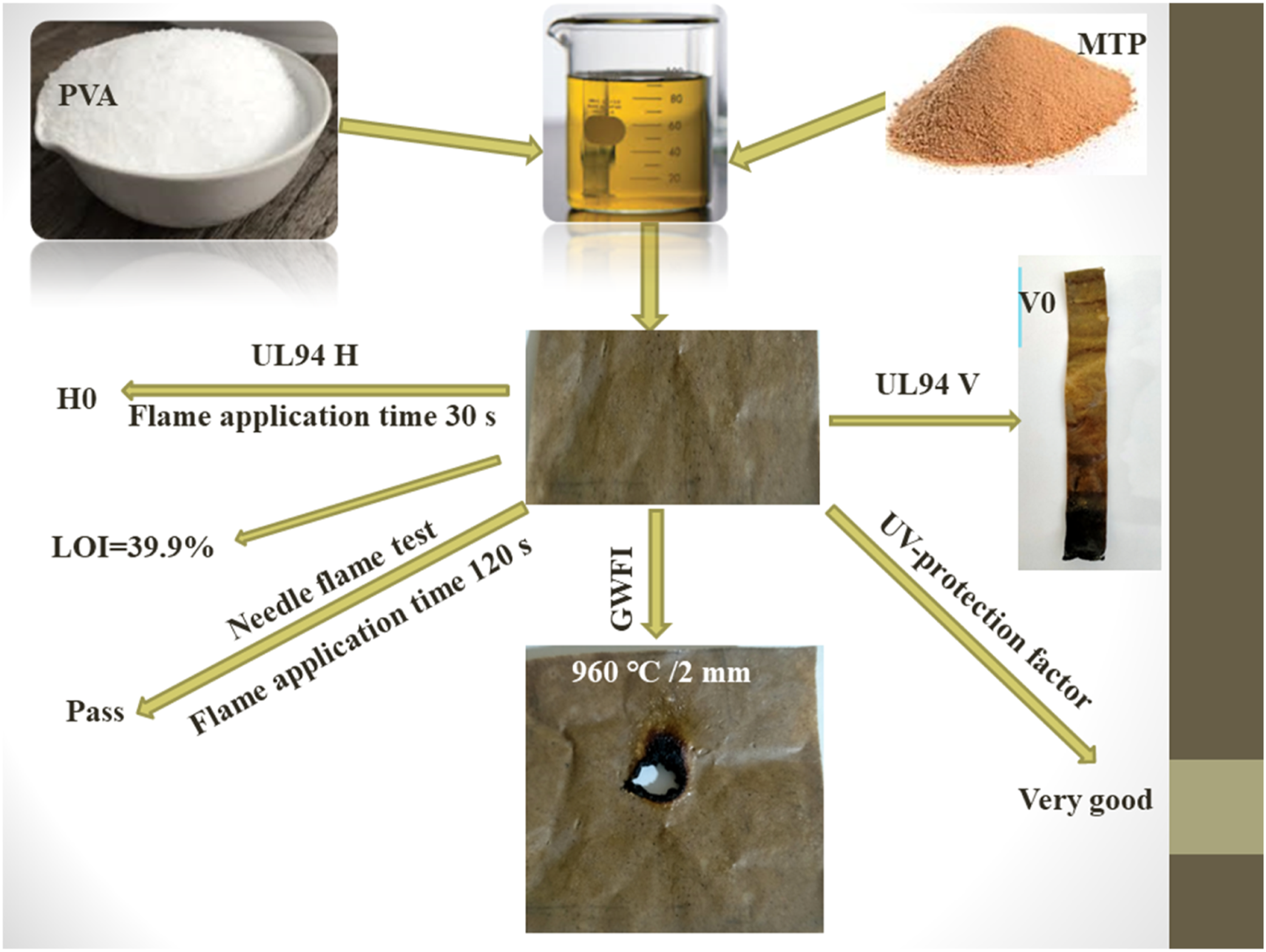
Polyvinyl alcohol (PVA) is a highly flammable material, and enhancing its flame retardancy to pass the glow wire test is a great challenge. In this work, the melamine salt of tannic phosphate (MTP) was synthesized, characterized, and combined with PVA at 8, 10, 13, and 15 wt%. The thermogravimetric analysis (TGA) results indicated that MTP enhanced the char formation of PVA at 750°C. Flammability tests showed that low concentrations of MTP can enhance the flame retardancy of PVA. The limiting oxygen index (LOI) of PVA increased from 18.8% to 39.9% in the PVA/15%MTP composite. The vertical flame spread test (UL94V) showed that PVA/13%MTP, and PVA/15%MTP composites achieved V0 rating. Glow wire test data indicated that PVA/10%MTP, PVA/13%MTP, and PVA/15%MTP composites had a glow wire flammability index (GWFI) of 960°C/2 mm. Moreover, all the composites passed the needle flame test when the flame application time was 120 s. PVA/15%MTP composite showed very good UV protection and an increase in sound absorption coefficient magnitude compared with pure PVA. The new results can increase the application of PVA in different industries.
Highlights
• PVA/melamine tannic phosphate (MTP) composites were prepared by solution blending. • PVA/15%MTP composite achieved V0 and HB classes and its LOI value was 39.9%. • Glow wire flammability index was 960°C/2 mm in PVA/13MTP and PVA/15%MTP samples. • PVA/15%MTP composite showed very good UV protection. • PVA/15%MTP sample displayed sound absorption coefficient value 0.65 at 1250 Hz.
Introduction
Polyvinyl alcohol, a water-soluble polymer, is characterized by biodegradability, biocompatibility, good film formation, good mechanical properties, and high transparency. These features make PVA exceedingly used in membrane manufacturing, the textile industry, biomedical applications, adhesives, and packing materials. In addition, PVA has manifested significant implementation value in solar cells and wearable intelligent instruments.1–5 However, PVA is considered a highly flammable material because its LOI value is 19%, which restricts its industrial application. To overcome this problem, flame retardant materials are added to PVA by physical mixing or chemical reactions. The presence of many hydroxyl groups in PVA chemical structure increases the chance of using reactive flame retardant materials. 1 Zhou et al. 6 coated PVA fibre by grafting the fibre surface with acrylonitrile (AN), then reacting with hydrazine hydrate (NH2NH2.H2O) and copper sulfate pentahydrate (CuSO4.5H2O) solutions to form a flame retardant layer on the PVA surface. The results showed that the new coating was able to improve the flame retardancy of PVA. Microscale calorimeter combustion data manifested that Cu-PVA-g-AN samples had lower values of total heat release (THR) and peak heat release rate (pHRR) compared with PVA alone. Liu et al. 7 enhanced the flame retardancy of PVA by grafting its surface with 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide-g-(2,3-epoxypropoxy) propyl trimethoxysilane (DPP). Cone calorimeter data showed that grafting PVA with DPP decreased its THR and pHRR values, and the grafted samples achieved V0 rating in the vertical flame spread test (UL-94 VTM). Although reactive flame retardants can enhance the flame retardancy of PVA, they have certain drawbacks, such as the complexity of the preparation processes, and the high cost. 1 Physical blending of PVA with flame retardant materials, which can be organic or inorganic compounds, is a widely used method to prepare flame-retarded PVA. Makhlouf et al. 8 added ammonium iron (II) sulfate (AFS) to PVA to improve its flame retardancy. The authors reported that the addition of 25 wt% (AFS) to PVA enhanced its LOI to 26.1%, and the samples achieved V0 in the vertical flame spread test. Luo et al. 9 studied the synergistic effect between ammonium polyphosphate (APP) and α-zirconium phosphate (α-ZrP) to improve the fire resistance of PVA aerogels. The results indicated that APP (15 wt%) alone can enhance the flame retardancy of PVA; the samples attained V0 rating in the UL94V test, and the LOI of PVA increased to 37.5%. The addition of 2% α-ZrP to 13% APP enhanced the LOI value of PVA to reach 43.1%, and the samples realized V0 rating. Moreover, the pHRR and THR of PVA decreased by 84% and 76%, respectively. Increasing the concentration of α-ZrP to 4 wt% (and APP 11 wt%) led to a negative effect on the flammability properties of PVA, where the LOI decreased to 41.3% and pHRR and THR values increased in comparison with the PVA/2% α-ZrP/13%APP composite. It was reported that melamine polyphosphate (MPP) and graphene, dispersible graphene nanosheets (DGNS), carrageenans, and hexagonal boron nitride (h-BN)@ ZnFe2O4 can also be added to PVA to improve its flame retardancy.1,10–12
On the other hand, it is necessary during the designation of electrotechnical products to consider the fire risk and the potential hazards associated with fire. The process of choosing materials and product design should aim to reduce the potential for fire risk to acceptable levels during normal operation, malfunction, and/or failure.13–15 In electrotechnical products, the overheated metal portions can behave as ignition sources. The glow wire test apparatus contains a red-hot glowing wire to simulate these ignition sources. The standard test method IEC 60,695-2-12 explains in detail the glow wire test procedure that is executed to test specimens of solid materials for flammability to measure the glow-wire flammability index (GWFI). According to the IEC 60,695-2-12 standard test method, GWFI is defined as the highest temperature at which the material under test fulfils the following conditions: (1) the material doesn’t ignite during the test; or (2) if ignition takes place, it extinguishes within 30 s after elimination of the glow-wire, the material is not totally combusted, and the molten drips, if they are found, do not cause ignition for the wrapping tissue. 14 It was reported in the literature that various types of flame retardant materials were applied to different kinds of polymers, such as poly (ethylene-co-butyl acrylate), glass fibre reinforced polyaminde 6,6, poly (lactic acid), polystyrene, recycled glass fiber reinforced flame retardant nylon 46 (RGFFRPA46), ethylene-propylene-diene (EPDM) rubber, polypropylene (PP), glass fibre–reinforced polyester (GFRP), recycled glass fiber-reinforced flame retardant poly (butylene terephthalate) (RGFFRPBT), polycarbonate (PC), poly (ethylene terephthalate) (PET), and poly (butylene terephthalate) (PBT) and polyaminde 6,6, to improve their GWFI and glow wire ignition temperature (GWIT) values.16–31 The flammability properties, including GWFI and GWIT, of these polymer composites are summarized in Table S1 in the supplementary data.
Noise contamination has become a critical problem all over the world because it causes harm to occupational surroundings and humans in the environment. The application of sound-absorbing materials to mitigate the noise was studied in the literature.32–36 The materials used for sound absorption were effective at low and middle frequencies. Following the sound absorption fundamentals, materials that can absorb sound are generally divided into (1) resonant sound-absorbing structures and (2) porous sound-absorbing structures. When sound waves get into the resonance-absorbing structures, they are resonating within the structure and hence scattering large amounts of acoustic energy.32–36
According to our literature survey, improving the GWFI value of PVA and, hence, increasing its application in electrical industries was not studied before in the literature. Therefore, this work was directed to enhance the GWFI of PVA by adding the melamine salt of tannic phosphate (MTP). Moreover, the addition of MTP to PVA is expected to increase its application in different industrial sectors such as food backing, paper, coating, and textiles. The thermal stability and combustion properties of PVA composites were measured. The mechanism of flame retardation was suggested. In addition, the acoustic and UV protection characteristics of the prepared composites were measured and evaluated.
Experimental work
Materials
Poly (vinyl alcohol), with a degree of hydrolysis of 86%–89% and a molecular weight (M.W.) of 85,000–124,000 g/mol, and tannic acid, with a M.W. of 1701.22 g/mol, were obtained from S D Fine-Chem Limited (SDFCL), India. Melamine was supplied by Alfa Aesar Company, Germany. Phosphoric acid (85%) and methanol (99%) were purchased from Sigma-Aldrich Company, Germany. Deionized water was produced in our laboratory using a water purification unit that was supplied by Purite Limited Company, UK.
Preparation of MTP
The melamine salt of tannic phosphate (MTP) was synthesized following the procedure in our previous work. 37 In a 500 mL glass beaker placed on a ceramic hot plate with a magnetic stirrer, 0.5 mol of phosphoric acid was added to 0.01 mol of tannic acid to form the intermediate compound tannic phosphate (TP). The reaction carried on for 1 h at 100°C until a solution with a black colour (with a pH = 0.01 pH unit) was obtained. The temperature of the hot plate was adjusted to 120°C, and then the TP solution was appended dropwise to melamine (0.24 mol of melamine that were scattered in 500 mL of methanol in a 1 L round bottom flask with a condenser) within 30 min. The reaction carried on for 6 h, and the final product (MTP) was clarified and rinsed with methanol till the pH value reached 6.79 pH units. MTP was dried at ambient temperature for 80 h, and it was floured to a smooth powder. The FTIR spectra of TP and MTP are presented in Figure S1 in the supplementary data. The FTIR spectrum of TP in Figure S1 shows absorption peaks at 1612 and 1441 cm−1 which are referred to aromatic (C=C). The peak at 761 cm−1 is assigned to C-H of aromatic ring. The peaks at 973 and 870 cm−1 are for P-O-C. The peaks at 1137 and 465 cm−1 is for P=O and O-P-O groups.38,39 The FTIR spectrum of MTP in Figure S1 displays peaks at 3416 and 3470 cm−1 which are referred to NH2 groups. The peaks at 3146 and 1405 cm−1 are assigned to +NH3. The peak at 3370 cm−1 is due to H-bonded OH groups. The peak at 2685 cm−1 is attributed to the OH of O = P-O–H. The absorption peaks at 1673, 1562 and 815 cm−1 are for the triazine ring of melamine. The peaks at 1252, 1173 and 1111 cm−1 are referred to P=O, P–O and P–O–C. The peaks at 501 and 620 cm−1 are for O–P–O stretching, and the peaks at 1615, 1423, and 782 cm−1 are attributed to C–C aromatic and C-H aromatic of tannic acid. The absorption peaks at 2362, and 1713 cm−1 are for O–C =O and C=O of tannic acid, respectively. The FTIR data of MTP are consistent with the data reported in the literature. 37 The 1H NMR spectrum of MTP agrees with literature data, where it shows the absence of the peak of the tannic acid proton at 5.3 ppm. 37 In addition, the strength of protons peaks between 6 and 8 ppm is boosted which may be attributed to the interaction between the protons of the tannic acid aromatic ring and the protons of NH2 and OH of the melamine and phosphate groups, respectively.
Preparation of PVA/MTP composites
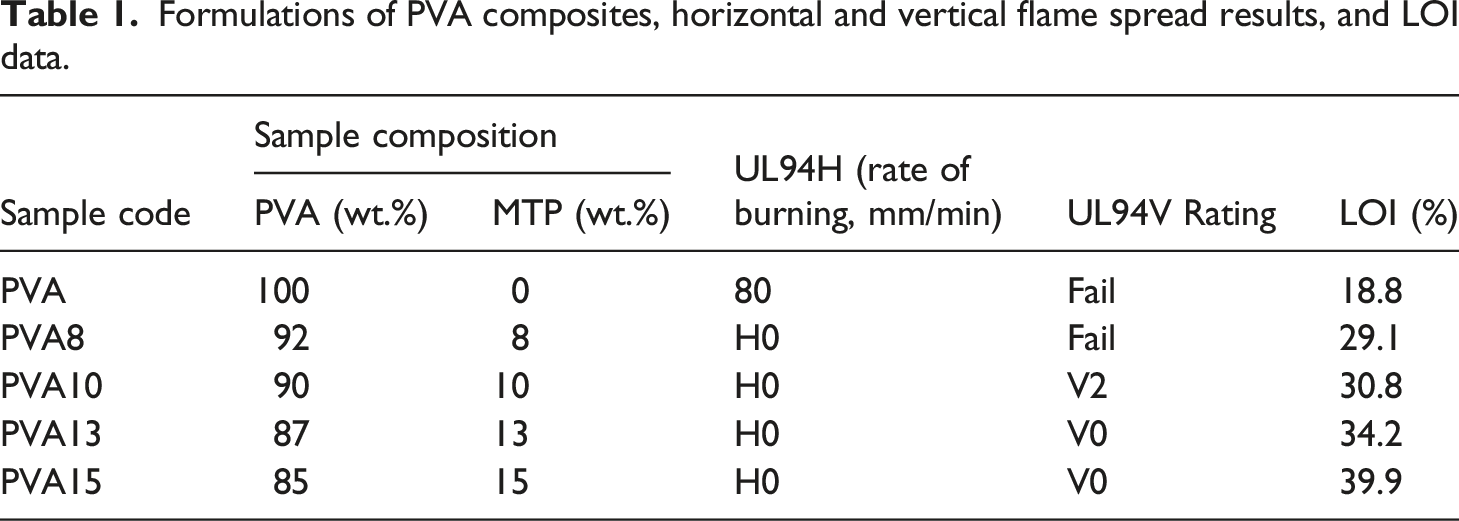
Formulations of PVA composites, horizontal and vertical flame spread results, and LOI data.
Characterization

The sound absorption attitude was tested using a two-microphone impedance tube (type: Brüel and Kjaer 4206, Denmark) according to international standards ASTM E1050 and ISO10534–2.47,48 Two cylindrical metal tubes of various diameters were used to measure acoustical features within two ranges of frequency. The large tube, with an inner diameter of 100 mm, was used to measure sound absorption in the frequency range between 63 and 1600 Hz. The small tube, with an inner diameter of 30 mm, was applied to measure sound absorption in the frequency range between 2000 and 6300 Hz. The powerful sound waves were tested within the range of 200 to 6300 Hz at 25°C. Each sample was tested four times to obtain consistent and representative results. The thickness of the samples was 2 mm.
Results and discussion
FTIR analysis of PVA and PVA/MTP composites
FTIR analysis in Figure 1 shows that PVA has a broad absorption band in the region of 3000 to 3600 cm−1, which is attributed to stretching of H bonded to OH groups. The bands at 2923 and 2857 cm−1 are referred to CH and CH2 stretching. The weak band at 1742 cm−1 represents the C=O of the vinyl acetate group in PVA. The peaks at 1630 and 1087 cm−1 are referred to C=C and C-O of PVA.49,50 The FTIR spectrum of PVA15 composite shows nearly the same absorption bands of PVA but with relatively higher intensity. This is very clear in the absorption band that locates in the region 3000–3600 cm−1 and the band at 1630 cm−1. This is attributed to the interaction between MTP, which has NH2, phenyl, and P-OH groups in its chemical structure, and PVA. In addition, the PVA15 spectrum shows absorption peaks at 713 and 625 cm−1 that can be attributed to the aromatic ring of tannic acid. FTIR analysis of PVA and PVA15 composite.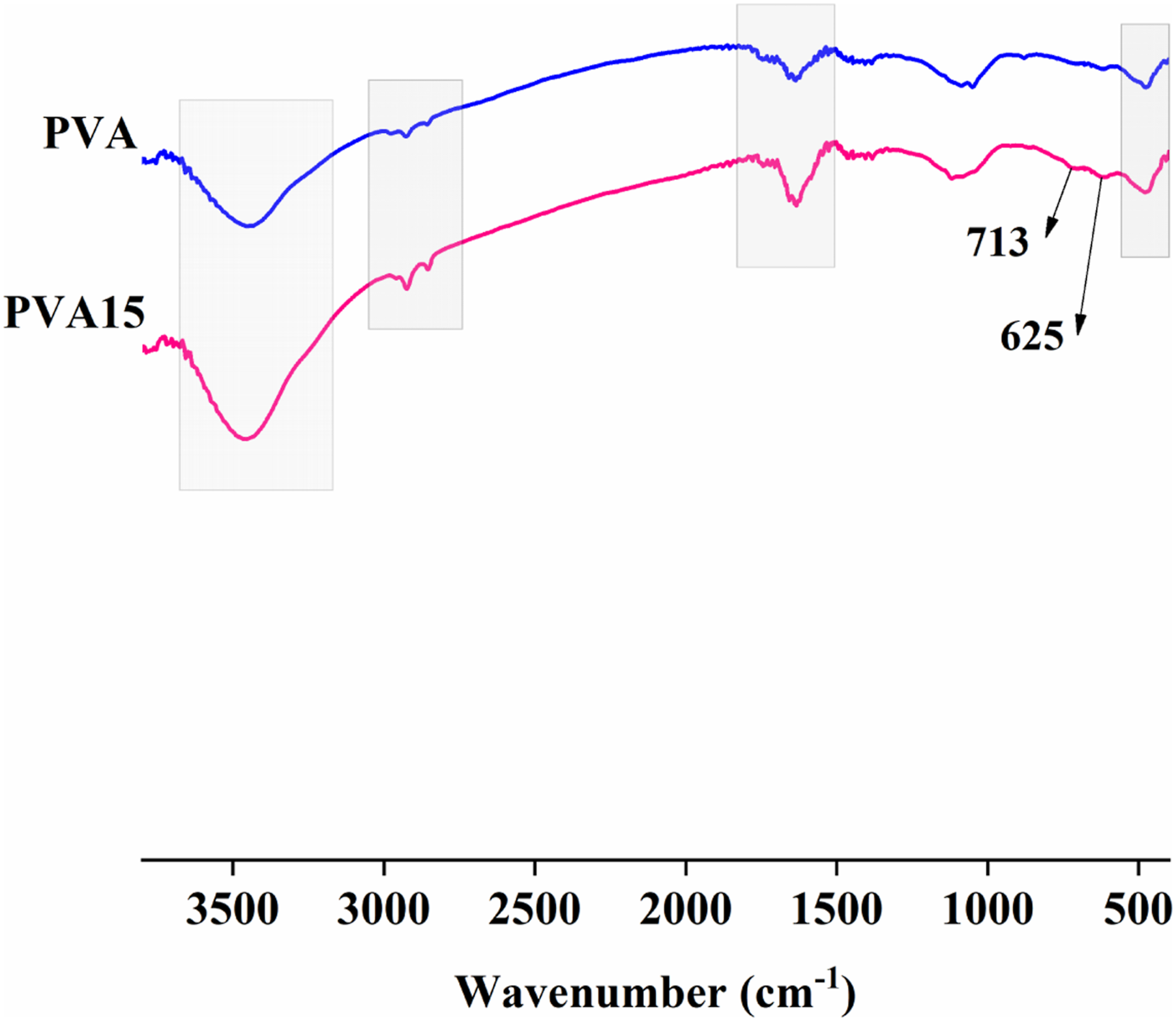
Flammability properties of PVA and its composites
Horizontal and vertical flame spread data
The data for horizontal (UL94H) and vertical (UL94V) flame spread tests of PVA and its composites are presented in Table 1. PVA is an easily flammable material in the air. The linear burning rate of PVA is 80 mm/min, and the falling of flammable driblets is observed during the test. The addition of MTP to PVA enhances its flame retardancy, and the flame fails to propagate in the samples. Therefore, all PVA composites achieve HB class in the UL94H test. The data in Table 1 shows that PVA and PVA8 samples fail to realize class in the UL94V test. Figure 2(a1-a4) displays that PVA burns completely in vertical flammability test. In contrast, PVA10 composite attains V2 class. Moreover, PVA13 and PVA15 composites succeed in realizing V0 class. Figure 3(a) shows a digital photograph of the PVA15 composite after the UL94V test. The great enhancement in the flame retardancy of PVA composites is attributed to the formation of an intact and coherent char layer on the polymer surface that prevented the spread of flame in composites matrices.
51
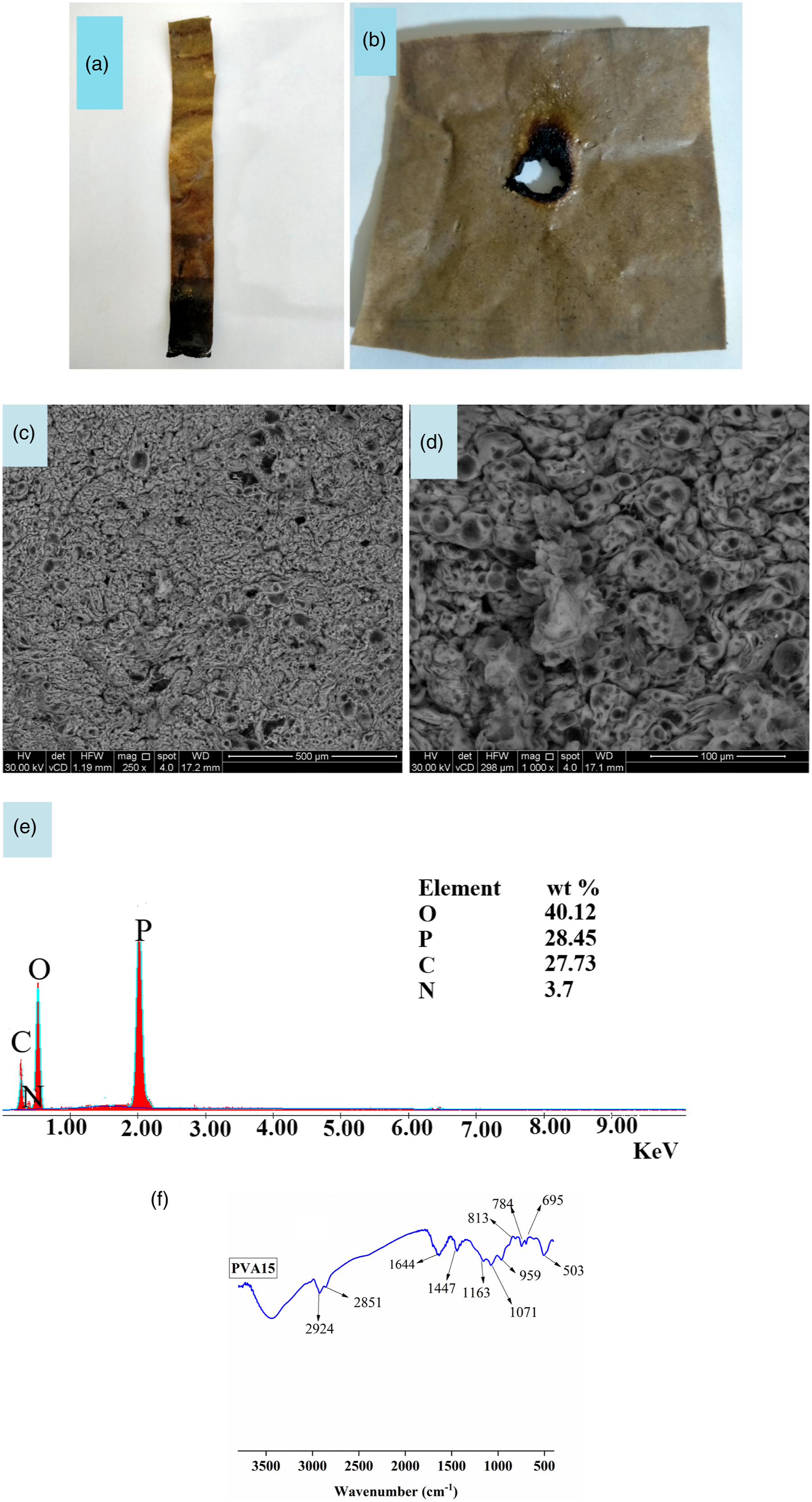
(a1-a4) flame propagation in PVA sample during UL94 vertical test. (a),(b) Digital photographs of PVA15 composite after UL94V test and glow wire test at 960°C. (c),(d) SEM images, (e) EDXS analysis, and (f) FTIR analysis of char residue of PVA15 composite after UL94V test.
LOI data
The LOI values of PVA and its composites are displayed in Table 1. It can be seen in Table 1 that the minimum concentration of oxygen (in the oxygen/nitrogen mixture) necessary to support flame in the PVA sample is 18.8%. This value is increasing with the addition and growth of MTP concentrations in the polymer matrix. The addition of 8, 10, 13, and 15 wt % from MTP to PVA enhances its LOI value to reach 29.1%, 30.8%, 34.2%, and 39.9% in PVA8, PVA10, PVA13, and PVA15 composites, respectively. The LOI data combined with the UL94V and UL94H results reveal that MTP is a highly efficient flame retardant for PVA.
Glow wire test data
Glow wire test results of PVA and its composites.
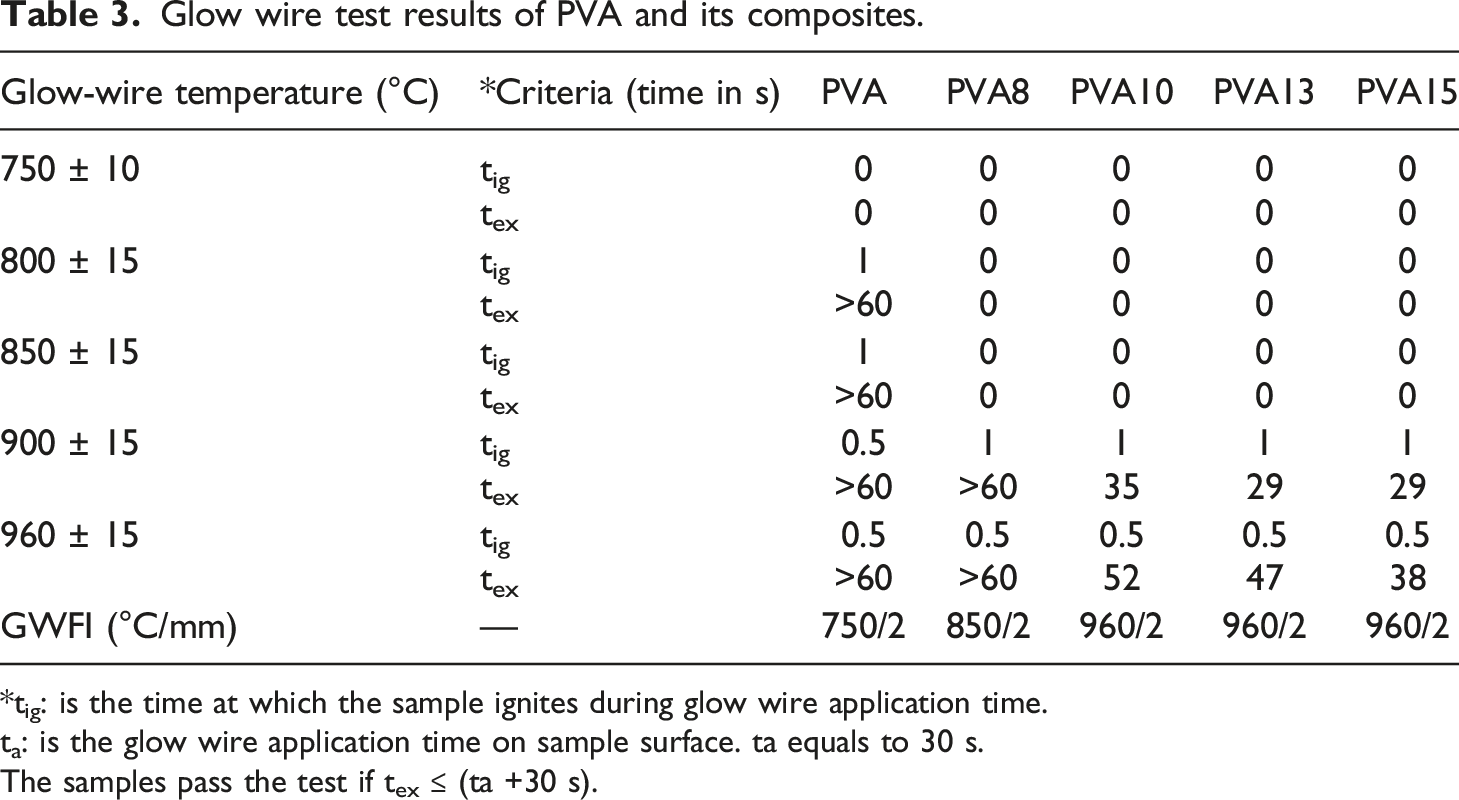
*tig: is the time at which the sample ignites during glow wire application time.
ta: is the glow wire application time on sample surface. ta equals to 30 s.
The samples pass the test if tex ≤ (ta +30 s).
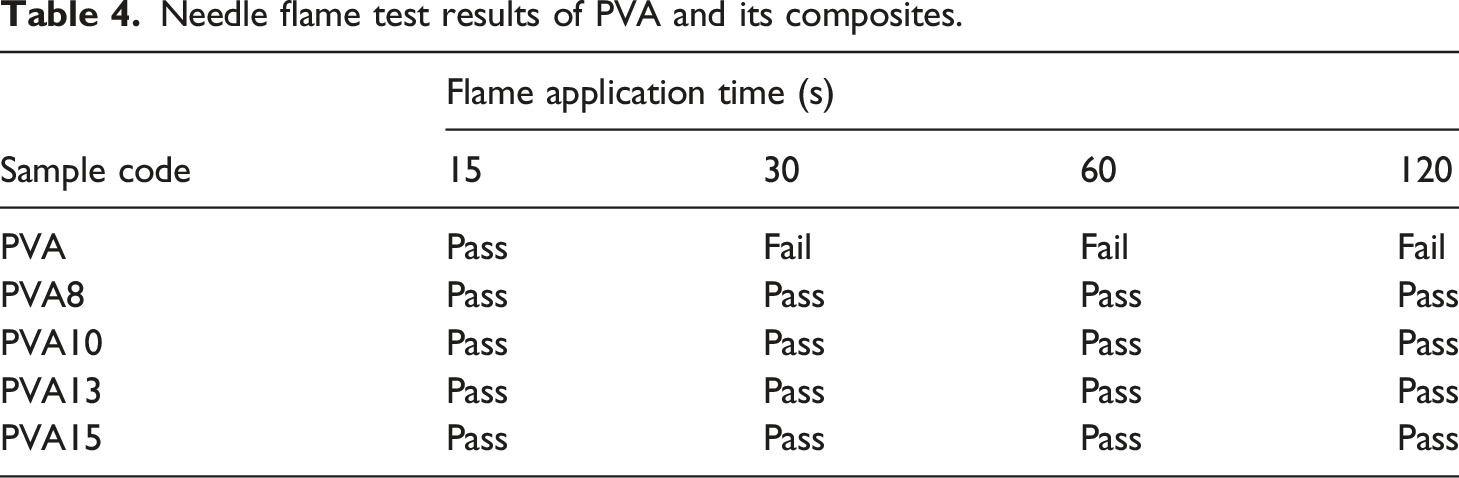
Needle flame test data
Needle flame test results of PVA and its composites.
Char analysis and flame retardation mechanism
The digital photographs in Figure 2(a1-a4) display that PVA sample burns completely without leaving any char residue in vertical flammability test. Figure 3(a),(b) shows digital photos of PVA15 composite after the UL94V and glow wire tests. Char residue after the UL94V test of PVA15 composite was analysed by SEM, EXDS, and FTIR. SEM images in Figure 3(c),(d) show the formation of an intact, continuous, and coherent char layer on the polymer surface. The elemental analysis of this char in Figure 3(e) shows that it contains C, P, N, and O, and the weight percentages of these elements are 27.73, 28.45, 3.7, and 40.12 wt %, respectively. FTIR analysis of the char residue formed after the UL94V test shows that the char layer contains polyaromatic structure, polyphosphate and/or pyrophosphate, C-N, C=C, and alkyl groups. The absorption bands for aromatic ring appear at 813, 784, and 695 cm−1. The absorption band at 1644 cm−1 is for the C=C group. The absorption bands at 1447 cm−1 and 503 cm−1 are attributed to the C-N group. The peak at 959 cm−1 is assigned to the presence of pyrophosphate and/or polyphosphate. The peaks at 1071 and 1161 cm−1 are for P-O and P-O-C. The absorption bands at 2924 and 2851 cm−1 are for alkyl CH stretching (asymmetric) and alkyl CH stretching (symmetric). The broad peak in the region 3000 to 3500 cm−1 is attributed to OH of adsorbed moisture.37–39,52
According to SEM, EDXS, and FTIR analysis of char residue after the UL94V test, it is expected that the PVA sample decomposed during combustion and produced heat, flame, flammable driblets, CO and CO2, water vapour, and flammable gases (such as CH4, C2H6 and C3H8). 37 The flame, oxygen, and heat were sufficient to form a sustained flame and burn the whole PVA sample. The flammable driblets supported the flame spread in other compartments nearby the PVA sample, such as the cotton patch sample that was placed underneath the polymer sample. In contrast, when PVA15 composite is subjected to the ignition source, it is expected to produce water vapour, CO, and CO2 in the gas phase. In the condensed phase, MTP degraded and formed polyphosphoric acid, which interacted with PVA chains to form a crosslinked char residue containing polyaromatic structure, polyphosphate, alkyl, C-N, and C=C groups. The char was very compact and coherent, and protected the sample from the effects of flame, oxygen, and heat.37,50–55 Therefore, the flame didn’t propagate in the PVA15 sample, and the intact char layer prevented the formation of flammable driblets. As a result, PVA15 composite is considered much safer than pure polymer. These findings were confirmed by the other flammability tests. For instance, in the glow wire test, MTP was able to provide the necessary protection for PVA against heat, and the sample passed the test at 960°C according to IEC 60,695–2–12. 15 In the needle flame test, PVA composites did not ignite after abstraction of the ignition source. This means that PVA/MTP composites can withstand the small flame that may be produced from a combustible material that can be found nearby when it is used in manufacturing electrical parts. The LOI data confirmed that PVA15 composite isn’t an easily flammable material, and the concentration of oxygen necessary to support flame is much higher than the oxygen required for supporting flame propagation in PVA alone. The higher LOI values of PVA composites assisted in protecting the samples when they were subjected to the ignition source. In conclusion, the addition of MTP to PVA makes it safer and more suitable for industrial applications.
TGA data of PVA and its composites
TGA and Dr.TGA data of PVA and its composites.

a,b,c: are the temperatures at which 10%, 50% and 70% weight loss take place. d,e,f: are the temperatures at which the maximum degradation take place during the first, second and third decomposition stages, respectively. g: the value in bracket is the calculated char residue.
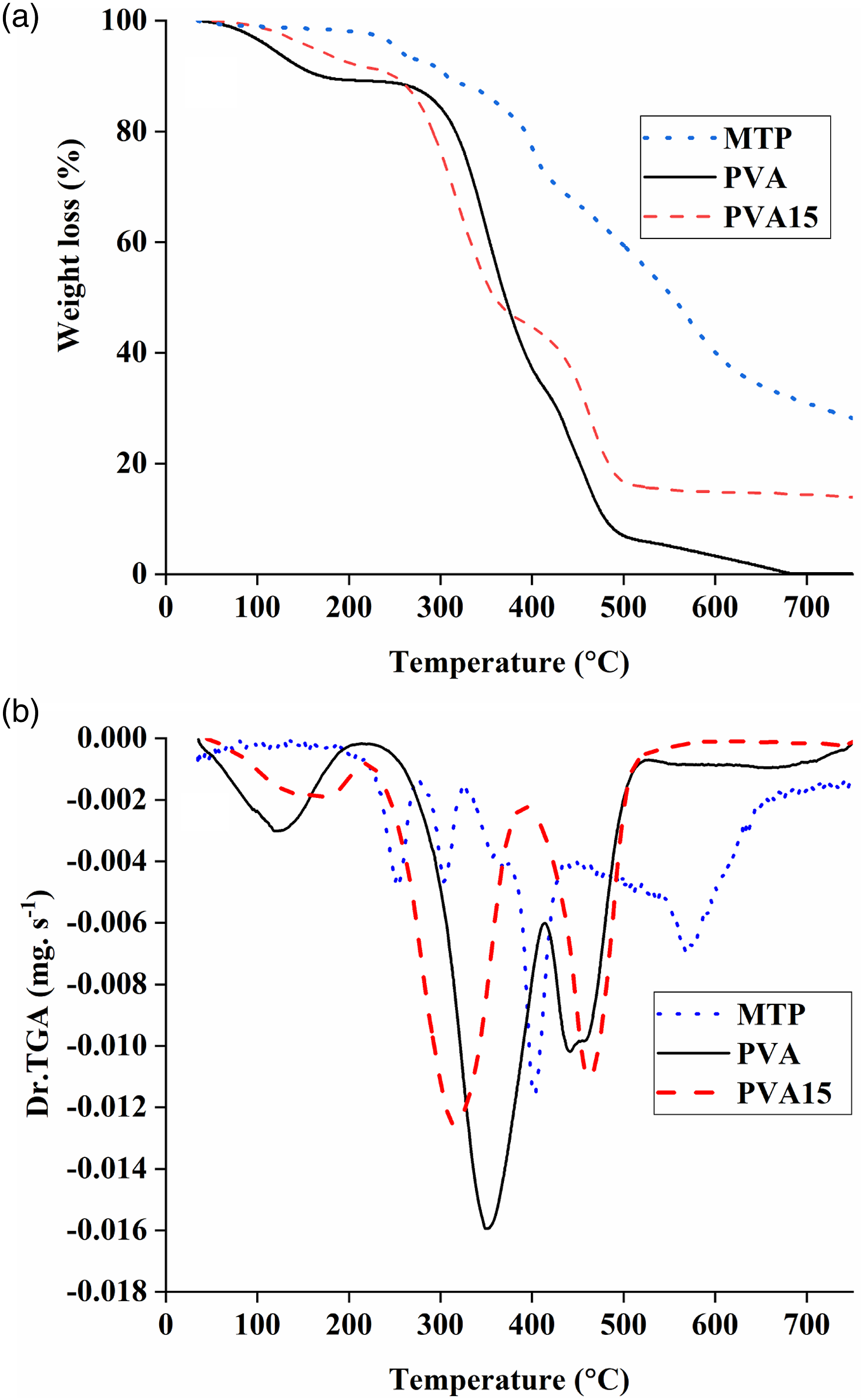
(a) TGA and (b) Dr.TGA curves of MTP, PVA and PVA15 composite.
UV protection data of PVA and its composites
UPF values of PVA and its composites.

Sound absorption behavior
Figure 5 shows the sound absorption coefficient (SAC) of PVA and its composites. We can see from Figure 5 that materials of small thickness (2 mm) have, to some extent, sound absorption properties, especially at low frequencies. It is clear in Figure 5 that the PVA has a resonance peak at the frequency of 1250 Hz with a SAC magnitude of 0.37. While the composites PVA8, PVA10, PVA13, and PVA15 reveal a small shift in their resonance. The shift is observed in the direction of increasing the frequency, and the magnitude of SAC is increased for all composites. Both composites PVA8 and PVA10 show absorption at the frequency of 1600 Hz with SAC magnitudes of approximately 0.39 and 0.54, respectively. The other composites, PVA13 and PVA15, have the resonance peak recorded at 1250 Hz with SAC magnitudes of 0.56 and 0.68, respectively. Also, we can observe a small broadening in the resonance peak of the PVA composites. This means that the absorption is still better in two or three frequency bands. The side part in Figure 5 shows the sound absorption frequencies with their amplitudes, for PVA and its composites, concentrated within 1000–2000 Hz (low frequency). It is clearly seen that the PVA15 sample has the highest absorption attitude. The improvement in acoustic damping may depend on the mechanism of absorption. The mechanism of acoustic absorption here may be due to the viscoelasticity of air, heat conduction, and relaxation of the molecular vibration of air. Although PVA composites samples have a 2 mm thickness, which may limit their sound absorption ability from one side, they have the advantage of being light in weight.32–35 Sound absorption coefficient performance versus 1/3 octave band frequency for PVA and its composites.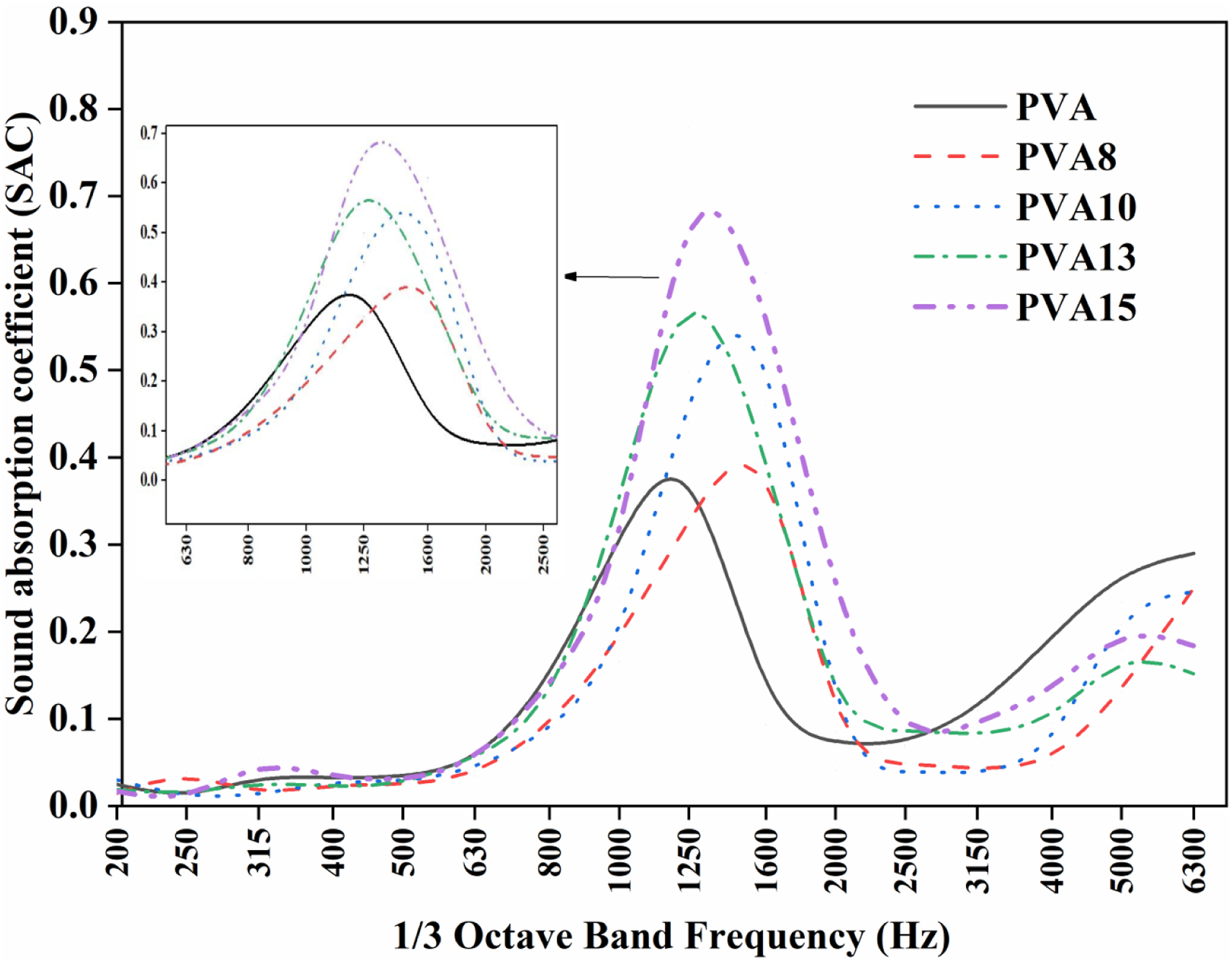
Conclusions
The single molecule intumescent flame retardant, melamine salt of tannic phosphate (MTP), was added to PVA at 8, 10, 13, and 15 wt %. TGA data indicated that MTP enhanced the thermal stability and char formation of PVA at high temperatures. The practical char residue of PVA15 composite at 750°C was more than three times the calculated char residue. LOI results showed that MTP enhanced the LOI value of PVA. Δ LOI values were 10.3%, 12%, 15.4%, and 21.1% in PVA8, PVA10, PVA13, and PVA15 samples, respectively. The horizontal flame spread test indicated that all PVA composites achieved H0 class according to ASTM D 635, and the samples PVA13 and PVA15 attained V0 class following ASTM D 3801. GWFI data manifest that PVA had a GWFI of 750°C/2 mm while PVA10, PVA13, and PVA15 had a GWFI of 960°C/2 mm. Moreover, all PVA composites passed the needle flame test when the flame application time was 30, 60, and 120 s. UPF values indicated that PVA did not have anti-UV properties, while PVA15 samples had very good UV protection. Also, PVA composites (with 2 mm thickness) showed, to some extent, sound absorption properties at low frequencies (1000–2000 Hz). The new characteristics that were provided to PVA after adding MTP can widen its industrial applications.
Supplemental Material
Supplemental Material - Reducing fire hazards and enhancing the thermal stability, acoustical, and UV protection of polyvinyl alcohol using bio-based flame retardant
Supplemental Material for Reducing fire hazards and enhancing the thermal stability, acoustical, and UV protection of polyvinyl alcohol using bio-based flame retardant by Aksam Abdelkhalik, Ghada Makhlouf, and Tarek M. El-Basheer in Journal of Thermoplastic Composite Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

