Abstract
Cotton
Keywords
Introduction
Cotton (
Textiles can be rendered flame-retardant by three primary techniques: blending, copolymerization, and finishing. Among these, the finishing method is particularly favored owing to its straightforward implementation, effectiveness, and ability to enhance the fabric properties. 15 Flame retardants can be categorized into two types: inorganic and organic. Inorganic flame retardants typically include compounds containing phosphorus, boron, zinc, iron, and carbon, whereas organic options primarily consist of halogens, phosphorus, nitrogen, and silicone compounds.16,17 Historically, halogen-containing compounds have been found to be the most effective flame retardants for cotton fabrics.18,19 However, these substances release cancer-causing dioxins and harmful fumes during combustion, thereby endangering human health and polluting the environment. Consequently, several nations have banned the use of halogen-based flame-retardants. Therefore, the pursuit of eco-friendly finishing solutions for cotton textiles is aimed at enhancing the flame retardancy while minimizing environmental harm. Various natural materials such as biomass-based, protein-based, and inorganic materials have been explored as potential eco-friendly flame retardants.20–25 However, balancing the preservation of textile properties with the effectiveness of flame retardancy remains a significant challenge. 26 Recent innovations in synergistic formulations have focused on combining various flame retardants to enhance the efficiency of cotton textiles and provide additional thermal stability and mechanical properties, which our review is critically analyze in this aspect.26,27
In addition to flammability, cotton textiles are susceptible to microbial growth, which can result in odor, staining, and fabric deterioration. This vulnerability poses potential health risks, particularly in light of increased public awareness around hygiene following the COVID-19 pandemic.28–31 Although cotton textiles offer protection, their moisture-absorbing properties can create favorable conditions for microorganisms. 32 This is particularly heightened in healthcare-associated infections. Healthcare-associated infections (HAIs), also known as nosocomial infections (NIs), are infections that patients acquire during their stay in healthcare facilities and are absent at the time of admission.33,34 These infections can lead to increased patient morbidity and mortality, prolonged hospital stays, and higher healthcare costs.33–38 Studies have shown that patients with HAIs have significantly longer hospital stays and higher medical costs than those without such infections. In surgical patients, nosocomial infections result in an average of 13 additional days of hospitalization. 37 The average hospital cost for patients with nosocomial infections was also significantly higher than that for uninfected controls (US 2280 vs US 698). 39 As a result, antibacterial treatment of cotton has garnered considerable scholarly interest. 40 Conventional treatments commonly utilize synthetic biocides, such as quaternary ammonium compounds (QACs) and triclosan, which have proven effective against both Gram-positive and Gram-negative bacteria.41,42 However, these conventional methods raise environmental and health concerns, particularly regarding the persistence of these chemicals in ecosystems and their potential accumulation in the food chain.43–49 In response to these concerns, there is growing focus on eco-friendly antibacterial solutions for cotton textiles. These alternatives can be categorized into natural antibacterial agents, biotechnology-based approaches, and nanomaterials.50,51 Integrating these alternatives into textiles offers a promising pathway for the development of durable, hygienic, and environment-friendly cotton fabrics.
Moreover, the rising demand for multifunctional textiles aligns with advancements in living standards and technological progress, and there is a significant need for fabrics that are both flame-resistant and antimicrobial, particularly in the home goods and medical protective equipment sectors. This demand has led to increased interest in specialized functional agents and treatment methods for developing innovative fabrics with these dual properties.21,52–55 The fabrication of textiles with both flame-retardant and antibacterial properties typically involves either a two-step or single-step approach. 22 Although the two-step method allows for broad applicability, it may encounter challenges owing to the potential interactions between flame retardants and antibacterial substances that could hinder functionality. Conversely, single-step methods often use synthetic agents that combine both properties, albeit with complex designs and eco-friendly challenges. 40 Additionally, identifying optimal processes and functional materials remains a significant challenge in the field of multifunctional textile research. Numerous review articles have been published on flame retardant and antibacterial fabrics. These reviews have covered various aspects, including textile materials,15,56,57 biomacromolecules, 58 durable flame-retardant finishing, 15 plant extract-based antimicrobial agents,59,60 finishing technologies, flame-retardant/antibacterial agents, 40 phytic acid-based flame retardants, 61 and chitosan-based antimicrobial coatings. 62
However, a comprehensive review combining fabrics with flame retardancy, antimicrobial properties, and dual functionality has not been systematically compiled. Additionally, on January 7, 2025, California’s Los Angeles area experienced devastating wildfires, resulting in the tragic loss of 27 lives and leaving more than a dozen individuals reported missing. The fires destroyed over 13,500 structures, including homes, businesses, and fashion halls, severely affecting the community and prompting widespread evacuation. This catastrophic event underscores the urgent need for innovative solutions for fire safety, particularly within the textile industry.63–65 This review contributes to the field by systematically integrating the dual functionality of flame retardancy and antibacterial properties in cotton textiles, an area that has not been comprehensively addressed in the existing literature. While numerous reviews have focused on either flame-retardant or antimicrobial treatments, our manuscript consolidates these aspects to highlight the importance of developing multifunctional textiles that meet modern safety and health standards. Furthermore, we emphasize eco-friendly solutions that reflect the growing demand for sustainable materials in textile production. By critically examining innovative synergistic formulations and the effectiveness of various natural and sustainable agents, this review not only identifies key advancements in the development of eco-friendly treatments, but also addresses current research gaps. Additionally, we provide valuable insights and guidance for future research, manufacturing practices, and policymaking, ensuring that stakeholders are equipped to meet the evolving demands of consumers and industries. In doing so, this study serves as a vital resource that underscores the significance of safety and sustainability in the textile sector, differentiating it from previous reviews.
Review methodology
In conducting this comprehensive review of eco-friendly flame-retardant and antimicrobial finishing solutions for cotton textiles, a meticulous literature search was performed across various scientific databases, including PubMed, Scopus, Google Scholar, Science Direct, Wiley Online Library, and Web of Science, as well as reports from reputable industry websites. The search used specific keywords and phrases such as “eco-friendly flame retardants,” “antibacterial treatments,” “sustainable textile finishing,” “cotton textiles,” “biodegradable agents,” “green chemistry,” and “environmentally friendly processes” to ensure a broad and relevant dataset. Initially, over thousand references were identified through this systematic search. The review methodology was guided by the stringent inclusion and exclusion criteria. The inclusion criteria focused on peer-reviewed articles, reviews, book chapters, conference proceedings, and industry reports, that specifically addressed eco-friendly solutions for cotton textiles. Non-peer-reviewed articles, editorials, opinion pieces, and publications unrelated to core topics were excluded to maintain the quality and relevance of the literature. Following the initial search, titles and abstracts of the identified references were screened to assess their relevance. This process led to a thorough full-text assessment of those who met the established inclusion criteria. Ultimately, 304 references were selected for in-depth analysis based on their relevance to the synthesis, properties, and applications of flame-retardant and antimicrobial treatments for cotton textiles.
Data extraction involved gathering relevant information from the selected studies, including the types of eco-friendly agents employed (e.g., natural compounds and biodegradable materials), methodologies used, effectiveness of treatments, and any environmental impact assessments conducted. These data were organized in a standardized format to facilitate easier comparisons. The findings were categorized based on the types of finishing solutions (e.g., chemical and natural treatments), and performance metrics (e.g., flame resistance, antibacterial efficacy, and wash durability). This categorization enabled a structured analysis of trends, innovations, and gaps in existing research. Each study underwent a quality assessment that evaluated methodological rigor, including sample size, experimental design, and replicability of results. This assessment helped to determine the reliability of the findings and provide directions for future research. A comprehensive synthesis of the collected data was conducted to identify the common themes, innovations, and challenges within the field. The synthesis also highlighted the environmental impacts of various finishing solutions, in contrast to the traditional methods with eco-friendly alternatives. The review culminated in a discussion that contextualized the findings within the broader textile industry, addressing implications for sustainability and potential future research.
Flame retardancy in cotton textiles
Mechanisms of flammability and fire hazards in cotton textiles
The inherent flammability of cotton textiles poses significant global safety concerns. The cellulosic composition of cotton renders it highly combustible, making untreated fabrics a considerable fire hazard that requires the development and application of effective flame-retardant treatments.66,67 The high air permeability and hygroscopic nature of cotton also contribute to its propensity for rapid ignition and intense burning. 67 Although advancements have been made in flame-retardant technologies, the efficacy of current test methods for evaluating the fire behavior of cotton textiles remains a subject of debate. This underscores the need for continued research and innovation in the critical areas of textile safety. From a mechanistic standpoint, the flammability of cotton involves complex interplay between combustion and pyrolysis processes.68,69
Combustion refers to the exothermic oxidation of a material that releases heat and light. Pyrolysis, on the other hand, describes the thermal decomposition of the cellulosic polymer in the absence of oxygen, leading to the formation of flammable gases, char, and tar. As shown in Figure 1, the thermal degradation of cotton textiles can be characterized by distinct stages. The initial dehydration stage, occurring at temperatures below 150°C, involves loss of adsorbed water from the cellulose structure.
7
As the temperature rises (300°C–400°C), pyrolysis commences, triggering the irreversible breakdown of the cellulosic polymer and the generation of volatile combustible byproducts and solid char residues. As the temperature further increases (400°C–600°C), flammable gases are released, potentially igniting and leading to a self-sustaining combustion cycle. The final stage (600°C–800°C) involves the oxidation of char, producing carbon dioxide, carbon monoxide, and water vapor.70,71 Studies have shown that the thermal decomposition of cotton fabrics produces a significant amount of flammable tar and gases, along with non-flammable water vapor and char. Interestingly, the chemical aspects of pyrolysis revealed that oxygen interacts with gaseous byproducts, leading to chain branching within the cellulose fibers. This chain-branching mechanism is a critical factor that must be interrupted by effective flame-retardant treatments to enhance the fire safety of cotton textiles.
72
Schematic of thermal degradation of cellulose. Adapted from.
7
© 2023 by the authors. Licensed under CC BY. 
Working mechanisms of flame-retardants
Flame retardants play a crucial role in enhancing fire safety by becoming active when exposed to flames, heat sources, or fires. Their effectiveness arises from both their inherent chemical properties and their interactions with polymers and fibers. By employing a combination of physical and chemical actions, these substances can disrupt the combustion process at various stages in the solid, liquid, and gas phases. This interplay of mechanisms is complex and often involves multiple simultaneous processes; however, typically one stage dominates at any given moment (see Figure 2(a)).
73
To fully understand the function of flame retardants, it is essential to consider several physical phenomena that impede combustion. One primary physical method involves the formation of a protective layer, which significantly reduces the heat transfer and restricts the oxygen flow. This barrier not only lowers the degradation rates of the polymer, but also minimizes the production of pyrolysis gases, underscoring the fundamental principles of flame retardancy.74,75 Moreover, additional physical mechanisms include the cooling effects provided by various compounds such as phosphorus- and silicon-containing substances, boric acid-based additives, inorganic borates, and low-melting glasses. These materials effectively cool the substrate to temperatures below the ignition thresholds, thereby triggering endothermic processes, such as the decomposition of the additives. In conjunction with cooling, dilution plays a vital role in reducing the concentration of flammable gases in both condensed and gas phases. This is achieved by the incorporation of inert substances or additives, including fillers, hydrates, char-promoting agents, and halogen-containing compounds. During decomposition, these substances release nonflammable gases, which dilute the fuel concentration in both phases, ultimately lowering the flammable gas levels below the ignition limits.
76

In addition to these physical phenomena, significant chemical reactions also contribute to the effectiveness of flame retardants in disrupting the combustion processes. For instance, free-radical reactions in both the gas/vapor and solid/condensed phases can be interrupted by the action of flame retardants (Figure 2(b)). In the gas/vapor phase, these agents reduce the concentration of free radicals, inhibit the flaming and exothermic reactions, and provide thermal feedback to the condensed phase. Halogenated and phosphorus-based compounds are particularly effective at interfering with chain reactions, preventing interactions between hydroxide and hydrogen free radicals and oxygen and carbon monoxide. 77 In the solid/condensed phase, flame retardants can diminish the production of volatile species, leading to the formation of carbonaceous char, carbon dioxide, and water. This phenomenon is especially prevalent in nitrogen- and phosphorus-based compounds as well as heavy metal complexes, which are commonly found in both cellulosic and synthetic fibers. In synthetic fibers, flame retardants facilitate polymer breakdown, allowing the material to flow away from the influence of the flame. Additionally, these agents can create a protective layer of carbonaceous char, ceramic-like structures, or glass on the polymer surface, thereby serving as a barrier to heat and mass transfer. In the context of cellulosic fibers, significant reactions such as dehydration and crosslinking also occur.73,77 However, it is vital to note that the flammability of materials such as cotton is influenced by various factors, including the presence of oxygen, structure and composition of the fabric, and application of flame retardants.78–80 Understanding these processes not only elucidates the science behind fire resistance but also highlights the critical importance of selecting appropriate flame retardants for different materials and applications.
Traditional flame retardant treatments
Traditional flame retardants are essential components of fire safety measures for textile fabrics, significantly mitigating fire hazards across various sectors, including clothing, upholstery, and industrial applications. These chemical compounds work by impeding or delaying flame ignition, combustion, or spreading, thereby enhancing overall fire safety. 81 They can be categorized into two main groups, halogenated compounds and non-halogenated compounds, each with distinct mechanisms of action and application. Halogenated flame retardants, particularly those containing bromine or chlorine, have been widely adopted owing to their high effectiveness in interrupting combustion. These compounds primarily operate in the gas phase, where they interfere with radical chain reactions that sustain flames. However, its use raises significant environmental and health concerns. For instance, the combustion of halogenated compounds can produce toxic byproducts such as halogenated dioxins and furans, which leads to serious health risks. 82 Furthermore, brominated flame retardants (BFRs) have been detected in wildlife far from their original sources, highlighting their persistence and long-range transport capacity. This bioavailability in both terrestrial and aquatic ecosystems raises concerns about their potential to bioaccumulate, magnifying risks at various trophic levels and posing significant threats to wildlife and human health. 83
Phosphorus-based flame retardants function by promoting charring and forming a protective barrier that inhibits further combustion. They can be implemented as reactive systems or additives in various applications, including electronics, automobiles, and construction materials. Although these compounds often enhance fire resistance, they have limitations. Their effectiveness can vary depending on the material and specific application, and some phosphorus-based retardants may negatively affect the mechanical properties. 84 Consequently, current research is focused on developing phosphorus compounds that minimize health and environmental hazards while maintaining effective flame retardancy.85–87 In addition, inorganic flame retardants, such as metal hydroxides, release water vapor upon heating. This process cools the material and dilutes the flammable gases, thereby reducing the likelihood of ignition. Typically, halogen-free inorganic compounds are less toxic than their halogenated counterparts. 88 For example, metallic oxides serve as both flame retardants and smoke suppressants, thereby enhancing the fire safety. 89
Although metal oxide flame retardants offer some benefits, they present notable drawbacks. These compounds often require high loading levels to achieve adequate flame retardancy, which can negatively affect the properties of cotton fabrics. This high loading requirement may lead to decreased comfort, reduced breathability, and a stiffer hand feel for the treated fabrics.
90
Additionally, the use of metal oxides may result in decreased mechanical strength of the cotton fabric owing to the formation of crosslinks under acidic conditions and at high curing temperatures.
90
This trade-off poses challenges for manufacturers striving to maintain product integrity and performance, while ensuring fire safety.
91
In light of the limitations associated with traditional flame retardants, there is growing interest in alternative materials. Researchers are exploring compounds such as ammonium polyphosphate, pentaerythritol, melamine, metal salts, and organic-inorganic hybrid materials for their potential to enhance fire resistance while minimizing negative impacts. For instance, Xu et al.
92
investigated alternative flame-retardant approaches, such as single-walled carbon nanohorns in combination with ammonium polyphosphate (APP). This combination can achieve high flame retardancy at low addition levels. These emerging alternatives often combine the benefits of both organic and inorganic flame retardants, providing improved performance characteristics and a reduced environmental footprint. It summarized that, while halogenated flame retardants have historically been effective, their environmental and health implications have spurred the exploration of safer alternatives such as phosphorus-based and inorganic compounds. By 2022, non-halogenated products, including phosphorus-based and inorganic options, accounted for over 59% of the market share (see Figure 3(a)). The global non-halogenated flame retardants market was valued at USD 5.62 billion in 2023 and is projected to grow at a CAGR of 8.25% from 2024 to 2030 (Figure 3(b)). Phosphorus-based retardants strike a balance between performance and safety, while inorganic options provide a non-toxic alternative, albeit with some limitations in material properties. The ongoing development of new flame retardants is driven by the pressing need for safer and more environmentally friendly solutions.82,85,88,89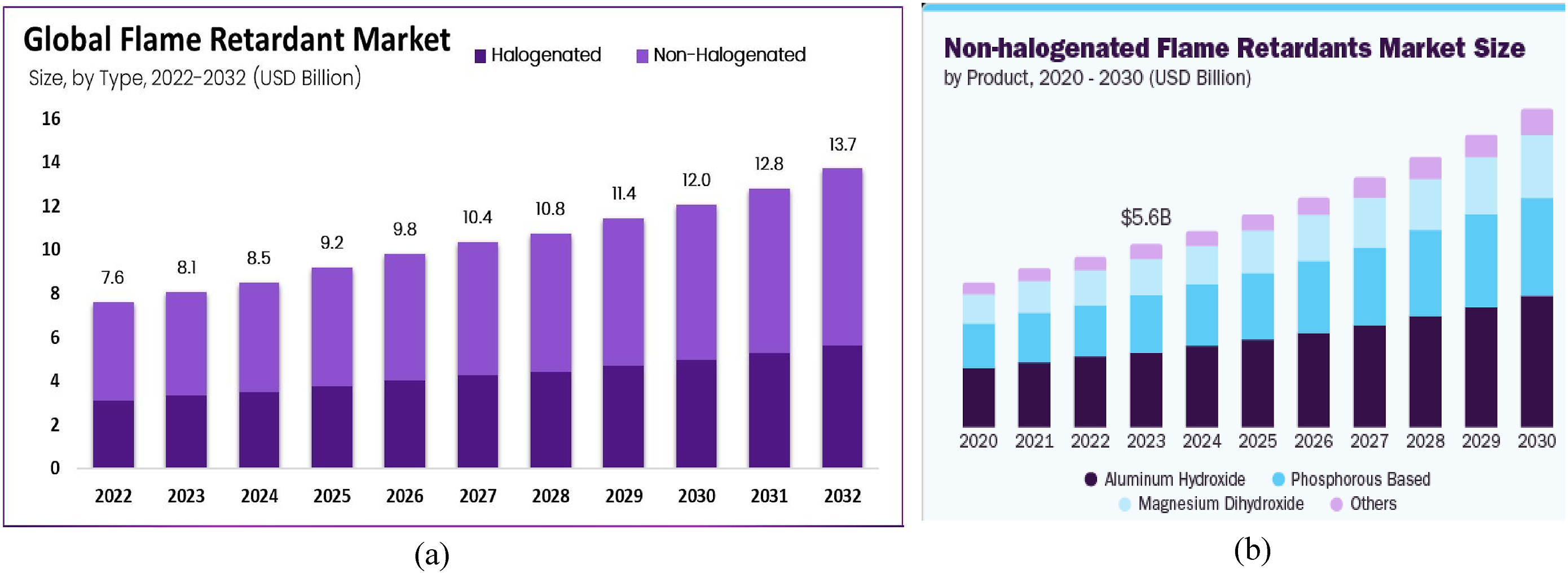
Eco-friendly flame retardant approaches
Eco-friendly flame-retardant approaches (non-halogenated, naturally based, and inorganic) for cotton textiles are becoming increasingly important owing to the aforementioned environmental and health concerns associated with halogenated flame retardants. Natural/biobased flame retardants can be divided into two categories according to their origin: animals (chitosan, DNA, and others) and biomass (cellulose, starch, tannins, phytic acid, lignin, plant extracts, and oils), as shown in Figure 4.
12
Notably, synergistic effects are particularly important for the development of effective flame retardant systems. This section briefly discusses the ecofriendly flame retardants and their synergistic effects. Schematic representation of eco-friendly flame retardants. Reprinted with permission from.
12
© 2020 Elsevier B.V.
Natural-based flame retardants
Natural-based flame retardants are gaining attention as eco-friendly alternatives to the traditional halogenated flame retardants. These materials can be categorized into two main groups: biomass-based compounds, such as phytic acid, tea polyphenols, lignin, tannic acid, and laccase, and animal-based materials, including chitosan and DNA. Researchers are increasingly exploring these natural substances for their potential to create effective flame-retardant coating systems for cellulose fabrics, particularly cotton, owing to environmental and health concerns associated with conventional flame retardants.23,92
Biomass-based compounds
Phytic acid (PA)
Phytic acid (PA), also known as inositol hexakisphosphate, is a naturally occurring compound that contains six phosphate groups and 12 hydroxyl groups, allowing it to interact with a wide range of metal ions and positively charged compounds.
61
It is readily available and can be extracted from various plant sources including oil seeds, beans, and cereal grains.
94
The structure and key features of the PA are illustrated in Figure 5. Importantly, the increasing demand for PA as a flame retardant can be attributed to its unique structure, high phosphorus content (28 wt % relative to its molecular weight),17,95 and its ability to adhere effectively to cellulose, proteins, and a variety of fabrics, including cotton, wool, nylon, silk, and poly (lactic acid) nonwoven materials.
95
Furthermore, PA and its salts are recognized as effective partners for positively charged counterparts in layer-by-layer (LbL) assemblies, a technique widely used to create flame-retardant fabrics.
61
In experimental studies, cotton treated with PA exhibited self-extinguishing behavior in vertical flame tests, whereas untreated samples burned completely.
95
It also facilitates dehydration reactions and supports carbonization during burning.
96
The other benefit of using PA in flame-retardant fabrics is the formation of a protective layer prior to decomposition of the untreated fabric.
61
Consequently, extensive research has focused on the effectiveness of PA as a flame-retardant agent for cotton textiles, as summarized in Table 1. Structure of phytic acid. Adapted from.
61
© 2021, © 2021 Elsevier. Licensed under CC BY 4.0. Summary of various studies on the effectiveness of phosphorus-based agents as flame-retardant treatments for cotton fabrics. Adapted and modified from.
61
© 2021 Elsevier. Licensed under CC BY 4.0.

Flame retardant properties of phytic acid for cotton textiles, employing various methods of application and evaluating performance through LOI, vertical flammability, and other tests.

Tannins and starch-based flame retardants
In addition to phytic acid, other biomass-based flame retardants such as tannins and starch are gaining traction as eco-friendly alternatives. Tannins are naturally occurring polyphenolic compounds extracted from various biomass sources that are safe, cost-effective, and widely available. There are three categories of tannin such as hydrolyzable, complex, and condensed tannins that account for 90% of global production. 105 Among others, condensed tannins have emerged as promising flame-retardant agents for cotton textiles, providing a sustainable alternative that addresses environmental concerns while meeting the fire safety requirements. 106 The flame-retardant properties of tannins are primarily attributed to their phenolic hydroxyl groups, which play crucial roles in their effectiveness. The application method typically involves a facile adsorption technique under weakly acidic conditions, which ensures good and durable flame retardancy. 106 The mechanism through which tannins operate on cotton textiles is primarily based on a condensed-phase mechanism. During combustion, tannins facilitate the formation of a char layer on the fabric surface, acting as a barrier to heat and mass transfer and inhibiting further burning.9,106 This char formation resulted from the dehydration and crosslinking reactions of tannin molecules under heat, creating a protective carbonaceous layer.
The key findings of studies on tannin-based flame retardants highlights several important aspects. The treated fabrics demonstrated improved LOI values, exceeding 27%, indicating enhanced flame resistance. In addition, the char length of the treated fabrics remained below 12 cm after the vertical burning tests, even after multiple washing cycles, demonstrating their durability. Furthermore, tannin-treated fabrics exhibit multifunctional properties, including antibacterial and antioxidant activities, thereby broadening their potential applications. 106 Additionally, thermogravimetric analysis (TGA) revealed the improved thermal stability of the tannin treated fabrics, further supporting the effectiveness of the condensed-phase flame retardant mechanism.9,106 Tannic acid has also shown potential as a flame retardant in cotton. When used in combination with sodium ions, tannic acid produced an intumescent flame-retardant effect, reducing the heat release capacity by 82% and increasing the LOI by 30.2% compared with untreated cotton. 107 This approach highlighted the effectiveness of tannins in enhancing the fire resistance of cotton textiles.
Starch-based flame retardants are promising eco-friendly alternatives to cotton textiles. Typically, these flame-retardants incorporate phosphorus-containing groups to enhance their effectiveness. One notable example is ammonium starch phosphate (ASTP), a novel phosphorus-containing flame retardant synthesized from biomass starch. 108 The structure of ASTP was characterized using 1D and 2D NMR spectroscopy, which confirmed its composition. The application method of ASTP involves treating cotton fabrics with 30% ASTP solution. This treatment significantly increased the LOI of the fabric to 45.2%; impressively, the LOI remained at 32.1% even after 50 laundry cycles, demonstrating both high flame retardancy and semi-permanent wash durability. 108 The durability of this treatment is attributed to the formation of P-O-C covalent bonds between ASTP and the cotton fibers, as evidenced by FTIR and SEM analyses. The flame-retardant mechanism of starch-based treatments, such as ASTP, primarily operates in the condensed phase. This is supported by the results of vertical flammability tests, TGA, and cone calorimetry, all of which showed excellent flame retardancy for the treated cotton fabrics. 108 The phosphorus-containing groups in ASTP likely promote char formation and reduce the heat release during combustion, similar to other phosphorus-based flame retardants.9,109 While starch-based flame retardants, such as ASTP, show promising results, they are not without drawbacks. This treatment can lead to a reduction in the physical properties of cotton fabrics. 108 However, this trade-off may be acceptable, given the significant environmental benefits of using biomass-derived flame retardants. Moreover, starch-based flame retardants provide a formaldehyde-free alternative to traditional phosphorus-based flame retardants, thereby addressing the growing environmental concerns.9,108–110 Further research is needed to optimize their performance and minimize any negative impacts on fabric properties.
Cellulose, alginate and lignin
Other biomass-based flame-retardant agents such as cellulose, alginate, and lignin have been extensively studied to enhance the fire safety of cotton textiles. While cellulose itself is not a flame retardant, it can be effectively used in conjunction with various flame retardants to improve the fire resistance of cotton fabrics. These materials are particularly attractive owing to their renewable and biodegradable characteristics. Various flame-retardant groups, mechanisms, and application methods have been investigated to improve the fire resistance of cotton fabrics. Common flame-retardants include nitrogen (N), sulfur (S), phosphorus (P), and silicon (Si) compounds. 9 They can be categorized as polymeric, nonpolymeric, or hybrid materials. Recent developments include a novel flame-retardant coating made from vinyl phosphonic acid and 1,3,5,7-tetravinyl-1,3,5,7-tetramethylcyclotetrasiloxane via an oxygen-plasma-induced polymerization process. 109 Additionally, researchers have synthesized fluorinated phosphoric acid ester-modified silicone resins by combining the flame-retardant properties of phosphoric esters with the hydrophobic qualities of fluorine. 111
The mechanisms underlying flame retardancy typically involve the formation of a stable char layer that serves as a barrier to heat and oxygen. For example, the synergistic effect between (vinyl phosphonic acid) and polysiloxane segments in flame-retardant cotton fibers resulted in the earlier decomposition of cellulose and enhanced stable char formation under thermal conditions. 109 Another study highlighted that ammonium acryloyl phosphite (AAP) exhibits a condensed-phase flame retardant mechanism. 112 Methods for incorporating flame retardants into cotton textiles include chemical bonding, coating, and dip-coating techniques. The dip-coating method was effectively used to apply fluorinated phosphoric acid ester-modified silicone resins to cotton fabrics in a simple one-step process. 111 In another approach, cotton cellulose was treated with the flame retardant Pyrovatex CP New and a cross-linking agent, Knittex CHN, in the presence of catalysts. 113 Several key findings have emerged from these studies. First, the combination of phosphorus and silicon-based compounds demonstrated excellent flame retardancy and thermal stability. 109 Second, multifunctional fabrics displaying both flame-retardant and hydrophobic properties were achieved using fluorinated phosphoric acid ester-modified silicone resins. 111 Additionally, the use of TiO2 as a co-catalyst improved flame retardant properties while reducing the loss of tearing strength in cotton fabrics. 113 AAP-treated cotton exhibits exceptional flame retardancy and durability, maintaining high LOI values even after 50 laundering cycles. 112 Finally, an “H”-shaped flame retardant (TBSA) synthesized from hydroxyethyl methylene phosphate, pentaerythritol diborate, and cyanuric chloride showed high efficiency, durability, and enhanced fabric strength. 112 These advancements in cellulose-based flame retardants for cotton textiles demonstrate their potential for developing environmentally friendly, durable, and effective fire-resistant materials for various applications.
Alginate-based flame retardants have also gained attention owing to their environmental friendliness and effectiveness. These flame retardants typically incorporate phosphorus-, nitrogen-, and silicon-based compounds to enhance fire resistance. The mechanisms of alginate treatment of cotton textiles involve both condensed and gas-phase actions. In the condensed phase, alginate forms a char layer on the cotton surface, which acts as a barrier to heat and oxygen transfer. 114 Char formation is often enhanced by phosphorus-containing compounds that promote the dehydration and crosslinking of cellulose. In the gas phase, nitrogen-containing compounds may release nonflammable gases that dilute combustible gases and reduce the heat release rate. Application methods for alginate-based flame retardants typically involve dip-coating or pad-dry-cure techniques, 115 which allow for the formation of a stable layer on textile fibers, which is crucial for durability and effectiveness. Some researchers have explored microencapsulation techniques to incorporate phase change materials (PCMs) along with flame retardants, providing both thermal comfort and fire protection. 115
Key findings from various studies have highlighted the potential of alginate-based flame retardants in cotton textiles. For instance, the incorporation of clay nanoparticles into alginate-based microcapsules significantly improves the thermal stability and flame retardancy of treated cotton fabrics. 115 Additionally, the synergistic effects between alginate and other flame-retardant components, such as phosphorus-containing compounds, have been observed to enhance overall fire protection performance. 114 Despite these advancements, the development of effective and environmentally friendly flame-retardant coatings for textiles remains an ongoing challenge in the field,9,110 and lignin-based flame retardants have shown promise in enhancing the fire resistance of cotton fabrics. For instance, sodium lignin sulfonate (SLS) treatment on cotton fabric resulted in an LOI value of 28.5 and self-extinguishment with a minimum char length of 4 cm. Additionally, SLS treatment imparted a naturally attractive yellow color and UV protective properties to the fabric without compromising its physical strength. 116 This multifunctional approach underscores the versatility of lignin-based flame retardants for cotton textiles. It is summarized that, cellulose, alginate, and lignin represent promising avenues for the development of natural flame retardants for cotton textiles. These materials not only enhance fire safety, but also align with sustainability goals by being renewable and biodegradable. Continued research in this area is essential to optimize their effectiveness and address any potential drawbacks, ensuring a safer and more eco-friendly future for textile applications.
Plant based extracts
Plant extracts, including bio-macromolecules, have been utilized in natural dyeing, antimicrobial finishing, textiles, and aroma finishing. Some plant extracts and waste products contain phosphorus and positive metal ions, which can serve as flame-retardant agents for cellulosic, lignocellulosic, and protein fiber-based textiles.
117
The exploration of plant-based liquids, such as false banana (
Similarly, Banana pseudostem sap (BPS) is extracted from the pseudostem of banana trees and contains phosphorus, nitrogen, and other metallic constituents. It was used to enhance the flame-retardant properties of cotton by mordanting it with 5% tannic acid and 10% alum, followed by impregnation with a 1:10 ratio BPS solution. The treated fabrics were dried at 110°C for 5 min. This treatment significantly altered the flame-retardant behavior of the cotton substrate, increasing the total burning time and decreasing the burning rate, resulting in an increase in the LOI values from 18 to 30.
119
Figure 6 shows the vertical burning behavior of the control and BPS-treated samples. The results showed that the BPS-treated sample increased the total burning time by 8.5 times compared to the control sample. Additionally, the alkaline extraction method was employed to obtain BPS extract, which was applied to bleached and mercerized Egyptian cotton fabric, resulting in improved flame retardancy and a higher LOI at 10% BPS concentration compared to untreated fabrics. In the vertical flammability tests, the treated fabric showed flames for only a few seconds before self-extinguishing, while the horizontal flammability tests indicated a significantly lower propagation rate of 7.5 mm/min compared to the control fabric.
120
Comparison in burning behavior of control and BPS treated cotton fabrics at different time interval. Reproduced with permission from.
119
© 2015, Emerald Publishing Limited.
The mechanism of fire retardancy imparted by both EPS and BPS in textiles can be attributed to their metallic elements such as potassium, sodium, phosphorus, aluminum, chlorine, and calcium, which can form inorganic salts or metal oxides. These elements were present in lower quantities on the treated fabric surface. The char morphologies of treated cellulose and protein fibers exhibit thickening, swelling, and axial failure, indicating condensed-phase intumescent fire-retardant behavior.118,121 Pomegranate rind extract (PRE), a waste agricultural product, contains nitrogen and various components, including aromatic phenolic groups and inorganic metallic salts, which can also impart flame-retardant properties to cellulosic textiles.
122
Spinach juice (SJ), derived from the fleshy leaves of spinach plants (
Another promising plant-based flame retardant is the waste tea extract. When applied to cotton fabrics, it displayed remarkable flame-retardant properties, achieving an LOI value of 29 compared to 18 for the control fabric, and exhibited self-extinguishing behavior with a char length of 30 mm in vertical flammability tests. Gas chromatography and mass spectroscopy (GC-MS) revealed that the tea extract contained various large molecular weight polyphenolic compounds and tannins responsible for its flame retardancy action. 123 The mechanism of flame retardancy of plant extracts typically involves the formation of a stable char layer on the textile fiber surface. 123 Similarly, tannin-based plant bio-macromolecules have shown potential as flame retardants in various textiles, including cotton, wool, silk, and synthetic polymers. 124 This indicates that plant extract-based flame retardants are a sustainable and effective alternative to traditional synthetic flame retardants for cotton textiles. Although plant-based flame retardants show promising results, further research is needed to improve their durability and washing fastness to meet industry standards for large-scale applications.
Recent approaches to biomass-based flame retardants
Biomass-based flame retardants such as cellulose, starch, lignin, phytic acid, plant extracts, and tannins have shown promise as eco-friendly alternatives for treating cotton fabrics. However, when used individually, these materials often face limitations in terms of durability, effectiveness, and impact on the fabric properties. For example, starch-based flame retardants such as ammonium starch phosphate (ASTP) demonstrate high flame retardancy and semi-permanent wash durability for cotton but can reduce the physical properties of the treated cotton fabric. 108 Similarly, although lignin-based flame retardants exhibit good flame-retarding properties, they may cause thermal degradation of the polymer matrix when used alone. 125 To overcome these limitations, researchers have explored the combination of different biomass-based flame retardants or their integration with other materials. For instance, the combination of lignin and phytic acid in PLA composites has shown synergistic effects, where phytic acid improves the dispersion of lignin particles, whereas lignin reduces the hygroscopicity induced by phytic acid. This combination resulted in significant improvements in the flame retardancy and mechanical properties. 125 A novel flame-retardant system combining phytic acid, hydrolyzed silk protein, and potato starch was also developed for polyacrylonitrile (PAN) fibers, which could potentially be adapted for cotton. 126 This fully biomass-based system significantly reduced the peak heat release rate and peak smoke production rate.
Summarized applications of biomass-based flame retardants and its synergetic effect for cotton fabrics: effectiveness measures and key findings.

Animal based-compounds
Chitosan biopolymers
Chitosan (CS), a deacetylated derivative of chitin, is a bio-based material that has been effectively used in flame-retardant applications owing to its inherent nitrogen content and ability to form protective char layers upon combustion.56,145,146 However, chitosan coatings often exhibit limited flame-retardant effects, necessitating the use of composite materials such as layer-by-layer (LBL) systems or chemical modifications to create intumescent flame retardants (IFRs) that achieve the desired fire resistance. The cellulose units in cotton fabrics serve as carbon sources, allowing IFRs to generate reactive phosphorus-containing acids in-situ at high temperatures. These acids promote dehydration and produce water vapor, enhancing the formation of a thermally stable char structure that improves the fire resistance of cotton fabrics. In addition to its flame retardancy, chitosan can also provide antibacterial activity. This effect occurs because CS disrupts the cell wall of bacteria, infiltrating and disturbing their normal physiological functions owing to the positive charge of -NH3+ in chitosan.147,148 Research by Patankar et al. 149 supported the flame-retardant potential of chitosan chemically modified with melamine and sodium pyrophosphate, which serve as nitrogen and phosphorus sources, respectively. The modified chitosan-treated cotton fabrics exhibited not only fire-retardant properties, but also UV protection and antibacterial effects, although the mechanical properties of the fabrics were slightly compromised, remaining within acceptable textile standards. The review presented by Islam and Ven 9 highlighted the broader trend towards sustainable and eco-friendly fire safety solutions, emphasizing chitosan and other bio-based flame retardants.
Furthermore, the incorporation of carbon-based nanomaterials with chitosan and phytic acid has led to the development of smart textiles that possess both flame-retardant and fire-warning functions.
145
The proposed flame-retardant mechanism of phosphorylated chitosan (PCS) for cotton fabrics involves the degradation of PCS, which generates phosphoric and polyphosphoric acids (see Figure 7). These acids enhance dehydration and carbonization by utilizing excellent charring properties of CS to form a stable char barrier. This barrier impedes the transmission of heat, oxygen, and flammable volatiles, thereby effectively safeguarding underlying cotton. The process also generates non-flammable gases, such as H2O, CO2, and NH3, which dilute flammable volatiles and reduce the temperature of the system. Consequently, the treated cotton fabrics exhibited highly effective flame retardancy.
147
Proposed flame-retardant mechanism of phosphorylated chitosan (PCS) coated cotton fabrics. Reproduced with permission from.
147
© 2022, Springer Nature B.V.
Importantly, the flame-retardant properties and durability of chitosan and chitin derivatives have been enhanced using various methods. LbL assembly has been employed to deposit bio-based chitin derivatives onto cotton fabrics, resulting in improved thermal stability and reduced peak heat release rates (Figure 8(a)).
150
Li et al.
151
used the LBL technique to create a composite coating of chitosan and lignosulfonate on cotton fabric. This coating exhibits exceptional synergistic flame-retardant properties. The treated cotton fabric with 25.2% loading level achieved LOI of 26.0%. Li et al.
147
developed a modified chitosan derivative containing N-methylene phosphonic groups using the Mannich reaction (Figures 8(b) and (c)). This compound was then utilized to improve the fire resistance of cotton fabrics. When applied at 14.2% concentration, the treated fabric achieved a LOI of 28.0% and successfully satisfied the requirements of the vertical flame test (VFT). However, despite its impressive performance, this application method has several drawbacks. This requires multiple rounds of immersion, drying, and reimmersion, leading to an inefficient overall process. In addition, the production and refinement of phosphorus-containing chitosan derivatives require significant quantities of chemical reagents. (a) Scheme of LBL assembly of chitosan (CH) and phosphorylated chitin (PT). Adapted and reproduced with permission from.
150
© 2014 Elsevier Ltd.; (b) Preparation of phosphorylated chitosan (PCS); (c) Schematic preparation process of treated cotton fabrics. Reproduced with permission from.
147
© 2022, Springer Nature B.V.
Therefore, in recent years, researchers have frequently utilized high concentrations of phosphorus-based inorganic and organic acids to solubilize chitosan, resulting in chitosan phosphonate flame retardants and coatings that exhibit promising fire resistance when applied to cotton fabrics.78,152 However, many studies have employed excess phosphorus-containing acids, which exceed the amount required for amino protonation. This overabundance can create an overly acidic environment, which deteriorates both chitosan and cellulose and negatively impacts fabric quality. Although neutralizing surplus phosphoric acid can mitigate these issues, it also generates chemical waste. Furthermore, small-molecule phosphate components produced during neutralization remain on the fabric surface, raising safety concerns.
142
Conversely, Huang et al.
142
developed a single-component macromolecular chitosan-based coating using amino trimethylene phosphonic acid (ATMP) (Figure 9). This coating was applied through a padding-drying-curing process to evaluate various properties, such as thermal stability, flame-retardant behavior, and antibacterial activity. The fabric achieved a LOI of 29.7% and passed the VFT when the load capacity reached 11.5%. This study presents a simple, environment-friendly, and efficient fabric coating approach, potentially expanding the use of chitosan-based materials while addressing previous challenges. Generally, chitosan biopolymers offer a versatile approach to enhance the flame-retardant properties of cotton textiles while providing antibacterial benefits. Ongoing research is focused on optimizing the application methods and addressing the challenges associated with using phosphorus-based compounds in chitosan to ensure the safety and efficacy of these treatments in practical applications. (a) Equation of CS and amino trimethylene phosphonic acid (ATMP) reaction. (b) Schematic of preparation of flame-retardant fabric (ATMP-CS@Cx). The green arrows represent mechanical mixing. The other arrows represent the processing steps.
142
© 2024 by the authors. Licensed under CC BY.
Deoxyribonucleic acid (DNA) as flame retardant for cotton
Deoxyribonucleic acid (DNA) is a biomacromolecule with remarkable flame retardant properties. It comprises deoxyribose units, nitrogen-containing bases that release ammonia, and phosphate groups that degrade into phosphoric acid upon exposure to flames or irradiation. This dehydration process promotes the formation of a stable protective char, limiting the heat and mass transfer between the flame and burning fabric. The resulting multicellular swollen carbonaceous structure acts as an effective physical barrier capable of stopping combustion reactions and extinguishing flames.153,154 A pioneering study in 2013 utilized commercially available herring sperm DNA to treat cotton fabrics.
155
Similar to the biopolymer discussed above (chitosan), DNA powder was suspended in water at a 2.5 wt % concentration to impregnate the cellulosic material, achieving a final dry add-on of 19 wt%. The treated fabric demonstrated self-extinguishing properties in horizontal flame-spread tests, exhibiting slow burning and extinguishing within 2 s of ignition. Notably, the sample resisted reignition, even after repeated flame exposure. LOI tests confirmed these findings, with the treated fabric achieving an LOI of 28% compared with 18% for untreated cotton. In the forced combustion analyses, the treated fabrics were not ignited under a standard 35 kW/m2 irradiative heat flux. The observed fire resistance was attributed to the ability of DNA to form char and the gas-phase dilution effect resulting from the breakdown of the purine and pyrimidine bases. These bases generate azo compounds, which promote char formation and produce noncombustible gases such as carbon dioxide and nitrogen. Encouraged by these results, further research has focused on optimizing the final dry add-on for treated cotton, leading to the preparation and analysis of fabrics with 5%, 10%, and 19% add-ons.
156
Representative SEM images are shown in Figure 10. SEM micrographs of COT (a), COT_DNA 5% (b), COT_DNA 10% (c), and COT_DNA 19% (d) at 5000x magnification, and elemental analyses.
156
© 2024 by the authors. Licensed under CC BY.
Despite their promising flame-retardant properties, the use of DNA faces challenges in terms of scalability and practical implementation. A significant concern is the cost-effectiveness of large-scale DNA use for textile production. Although DNA exhibits excellent flame retardancy, it may be more expensive than traditional synthetic flame retardants, potentially limiting its adoption in the textile industry. 58 Additionally, the extraction and purification of DNA from biological sources for industrial use poses logistical and economic challenges. Although DNA presents a green flame-retardant option for textiles, further research is necessary to address scalability issues and develop cost-effective methods for large-scale applications. The shift of the textile industry towards sustainable and environmentally friendly flame retardants 124 may drive innovation in this field, potentially leading to more efficient production methods and wider adoption of DNA-based flame retardants in the future. Balancing the excellent flame retardancy of DNA with the practical considerations of cost and scalability is crucial for its viability in commercial textile production.
Other animal-based flame retardants
Other animal-based or protein-based flame retardants, such as casein, whey protein, hydrophobins, and chicken feathers, have emerged as eco-friendly alternatives to traditional toxic flame retardants for cotton fabrics. These alternatives have shown promising results for enhancing the fire resistance of cotton fabrics through various mechanisms. Casein and whey proteins derived from milk have demonstrated effective flame-retardant properties when applied to cotton fabrics. These proteins form a protective char layer on the fabric surface during combustion, acting as a barrier to heat and oxygen. 157 Research has shown that alpha casein, when fused with a cellulose-binding domain (CBD), increases the flame retardancy of cotton fabrics by 43.3% in vertical burning tests. 158 The fusion protein displayed good durability and remained effective even after washing. The char-forming characteristic of casein-based coatings has been found to be more effective than that of whey protein in protecting the underlying fabric from heat flux. This intumescent char layer helps slow the spread of flames and reduces the production of flammable volatiles. Chicken feather protein-based flame retardants have shown promise when combined with other compounds. A synthesized flame retardant from chicken feather protein, melamine, sodium pyrophosphate, and glyoxal demonstrated improved flame retardancy when applied to cotton fabrics. 159
Summary of bio-sourced protein based flame retardants applied to cotton textile and their outcomes. Adapted and modified from. 117 © 2016 Elsevier Ltd.

Note: [HZ]-Horizontal flammability, [V]-Vertical flammability, DS-Herring sperm DNA, DT- Herring testis DNA, Y-Borax + Boric acid, B.R-burning rate, B.T-burning time, C’L- char length. The asterisk (*) indicates that the LOI value was not measured or significantly influenced by the bio-molecule treatment, affecting direct comparison.
Summary of natural-based flame retardants
Natural-based fire retardants are a promising solution in the fields of fire safety and materials science. Extracted from renewable resources, these compounds leverage the intrinsic characteristics of polysaccharides to enhance their fire resistance of various materials. Natural-based fire retardants function primarily by forming char when exposed to heat, thereby creating protective barriers and emitting noncombustible gases. This approach provides a sustainable, eco-friendly, and efficient method for improving the fire performance across a wide range of substrates, including textiles and polymers. Figure 11 shows a summary of the mechanism of action of the natural fire retardants. Table 5 outlines the primary flame-retardant mechanisms facilitated by the various categories of bio-derived products applied to textile materials. Notably, the fire-resistant action of these natural-based retardants occurs in the condensed phase, resulting in the formation of durable, aromatic, and protective char. This process typically involves dehydration reactions that occur during the breakdown or activation of bio-based flame-retardant additives.
162
In addition to their condensed-phase activities, some bio-based flame retardants can also function in the gas phase via two mechanisms. First, they produce a dilution effect by releasing inert gases during decomposition or activation, which dilutes combustible gaseous compounds that sustain the flame. Secondly, they can perform a “scavenging action,” effectively trapping the reactive free radicals responsible for flame propagation.
163
Mechanism of natural based fire-retardants.
81
© 2023 MAP. Licensed under CC BY 4.0. Main fire-retardant mechanisms of the bio-sourced FRs applied to textiles.
162
© 2024 by the authors. Licensed under CC BY 4.0.

Inorganic flame retardants
Natural inorganic flame retardants for cotton textiles include a range of substances including nanoparticles, silica- and silicate-based materials, and intumescent compounds. Metal hydroxides, such as aluminum hydroxide, enhance flame retardancy by endothermically decomposing and releasing water, which cools the material and dilutes flammable gases (Figure 12).
164
Silica-based flame retardants, often used in hybrid organic-inorganic intumescent forms, can be applied to cotton fabrics to improve flame resistance and hydrophobicity. For instance, polyethyleneimine-wrapped nano-silica combined with polyphosphoric acid was used in a layer-by-layer (LBL) assembly process.
165
Moreover, intumescent flame retardants are particularly noteworthy for their ability to form a stable foam layer that insulates and protects the underlying material from heat and flames. This category includes compounds that serve as acid, gas, and carbon sources, all essential components of intumescence Flame retardancy technology for cotton fabrics using functional inorganic compounds.
7
© 2023 by the authors. Licensed under CC BY.
Inorganic nanoparticles
Extensive research has been conducted on nanotechnology owing to its potential as a method for creating non-toxic and high-performance flame-retardant systems, which are of significant scientific and commercial interest. 7 These nanoparticles, measuring 1-100 nm in at least one dimension, can be incorporated into fibers or applied to fabric surfaces to enhance the flame-retardant properties and improve the mechanical strength and thermal stability of treated textiles. When evenly distributed, a small quantity of inorganic filler (typically less than 5 wt %) can decelerate the diffusion of gas molecules through the polymer matrix and reduce the burning rate of materials. The research indicated in refs.166–168 that nanoparticles can be applied to textiles using LbL techniques or nanoparticle adsorption. During combustion, these coatings generate intumescent barriers, which primarily function to create expanded char structures on the surface of the burning fabrics. This process reduces both the heat transferred by the flame and the release of the flammable volatiles. This mechanism mimics the flame retardancy of conventional intumescent coatings, which are larger in scale and reach thicknesses of hundreds of micrometers.169,170
Nanoclay, carbon nanotube and graphene
Inorganic nanoparticles, such as nanoclays, carbon nanotubes, and graphene, have shown promising results as flame-retardant additives for cotton fabrics. These nanoparticles can enhance the thermal stability and flame retardancy of cotton fabrics via various mechanisms. Nanoclays have been demonstrated to outperform carbon nanotubes in improving the fire properties of intumescent formulations. When incorporated into polymers, nanoclays enhance the formation of intumescent chars, which act as protective barriers during combustion. 171 This char formation helps reduce the heat release rate and improve the overall flame retardancy of the fabric. Carbon nanotubes (CNTs) have also been explored as flame-retardant additives in cotton fabrics. Although they may not be as effective as nanoclays in some aspects, CNTs can still contribute to the improved thermal stability and mechanical properties of treated fabrics.171,172 However, it is worth noting that CNTs may form strong networks on the flaming surfaces during combustion, which can potentially restrict intumescence. 171 Graphene and its derivatives such as graphene oxide (GO) and functionalized graphene oxide (FGO) have shown excellent potential as flame-retardant coatings for cotton fabrics. For example, FGO nanocomposite coatings have been reported to significantly improve the flame retardancy of cotton fabrics. In one study, FGO-coated cotton fabric was able to withstand fire for more than 325 s compared to untreated cotton fabric, which burned completely in 5 s. 173 Graphene polymers functionalized with trimethyl phosphate (GPTMP) demonstrated excellent flame retardancy, with a coated fabric resisting flame for up to 540 s and achieving an LOI of 35%. 174
Interestingly, combining these nanoparticles with other flame-retardant additives can lead to synergistic effects. For example, graphene sheets decorated with nanoparticles have shown enhanced flame retardancy when used in polymer nanocomposites. 175 Additionally, the incorporation of silver nanoparticle-loaded halloysite nanotubes (Ag@HNTs) in flame-retardant coatings has been reported to improve both fire safety and smoke suppression in polyester-cotton fabrics. 176 Regarding eco-friendliness, these inorganic nanoparticles offer potential alternatives to halogenated flame retardants, which pose threats to human health and the environment. 92 The development of environmentally friendly flame retardants that achieve high efficiency at low additive levels is crucial for both human safety and environmental protection. However, there are limitations to using inorganic nanoparticles as flame retardants in cotton fabrics. One major challenge is the durability of antimicrobial and flame-retardant properties after washing. 177 Although each type of nanoparticle has its own strengths and limitations, its effectiveness can be further improved through functionalization, combination with other additives, or incorporation into composite coatings. Future research should focus on optimizing these nanoparticle-based flame-retardant systems for improved performance, durability, and environmental friendliness.
Metal-based nanoparticles
Results from forced-combustion, flammability, and thermogravimetric analyses for cotton treated with metal-based nanoparticle additives. 7 © 2023 by the authors. Licensed under CC BY.

aNanosol-treated sample before washing.
bNanosol-treated sample after washing.
Additionally, agents such as magnesium hydroxide (Mg(OH)2) and aluminum tri-hydroxide (Al(OH)3) are preferred because of their non-toxic combustion products and lack of harmful emissions.
179
These inorganic compounds exhibit high specific heat capacities, enabling them to absorb significant amounts of heat before decomposition. They also absorb heat during evaporation, which helps dilute the combustible gas concentrations. The thermal decomposition of these hydroxides leads to the formation of metal oxides with high melting points, creating a physical barrier on the surface of the cotton fibers. These oxides promote dehydration into char and prevent flame spread. Additionally, they act as catalysts in redox and cross-linking reactions, converting carbon monoxide to carbon dioxide and thus reducing hazardous CO generation.
180
Ji et al.
180
explored a novel technique for producing flame-retardant cotton fabrics using magnesium hydroxide (Mg(OH)2) nanoparticles. These nanoparticles were applied using the pad dry cure method. Thermogravimetric analysis in a nitrogen atmosphere indicated that the treated fabrics exhibited a final residue of 28% at 800°C compared to 13.3% for the untreated samples. In air, the final residue increased from 7.2% for untreated cotton to 20.3% at 800°C. These findings demonstrate that Mg(OH)2 effectively inhibits cracking and significantly reduces fabric weight loss. The enhanced thermal stability of the Mg(OH)2-coated cotton fabrics is attributed to the breakdown of magnesium hydroxide crystals into magnesium oxide (MgO) and water vapor, with MgO forming an insulating barrier against heat transfer. Vertical flame-spread tests revealed that untreated cotton fabric ignited instantly and burned completely, while the Mg(OH)2-treated fabric exhibited superior flame-retardant properties, igniting after 48 s and continuing to glow for 52 s (Figure 13). However, it is worth noting that, inorganic hydroxides often have limited flame-retardant effectiveness and require considerable quantities to achieve the desired results, impacting the physical characteristics of materials such as cotton fabrics. Therefore, these substances are frequently used in combination with other flame retardants.
57
TGA curves of pristine and coated cotton fabrics under (a) N2 and (b) air atmospheres; (c) digital images of pristine and magnesium hydroxide (MH)/polydimethylsiloxane (PDMS)-coated cotton fabrics after vertical flame testing. Reproduced with permission from.
180
© 2019, Springer Nature B.V.
Silica and silicate-based compounds
Silica- and silicate-based compounds have gained significant attention as promising flame-retardant materials for cotton fabrics because of their excellent thermal stability and eco-friendly nature. 16 These silicone-based substances can produce siliceous char that acts as a barrier against heat, fuel, and oxygen, thereby protecting the underlying cotton from fire propagation. 181 The incorporation of silicon-containing elements can further enhance the synergistic effects of flame retardants. 182 For example, silica is noted for its thermal stability and ability to form a protective barrier on material surfaces, helping slow the combustion process. 183 Moreover, silicate compounds such as sodium silicate have been used alongside other agents such as urea and melamine to improve the flame retardancy of coatings, promote the formation of a silicon-containing char layer, and enhance the uniform dispersion of flame retardants in waterborne acrylic coatings. 184 However, these compounds can face challenges regarding their dispersion within polymeric matrices, which may decrease their flame-retardant efficiency if not adequately addressed. When combined with other materials or modifiers, silica- and silicate-based compounds often exhibit synergistic effects, which significantly improve their performance as flame retardants. Li et al. 182 developed a new intumescent flame retardant using nanosilica (SiO2), polyethyleneimine (PEI), and phytic acid (PA) coated on cotton fabrics. The LBL self-assembly process resulted in an enhanced flame retardancy, achieving a LOI value of 33.7% and a char length of 7.5 cm. The coated cotton exhibited 40.7% char residue at 600°C compared to only 8% for the uncoated fabric. Cone calorimetry tests confirmed a 75% reduction in the peak heat release rate (PHRR) and a 52% decrease in the total heat release (THR), with minimal impact on the fabric tensile strength.
Intumescent systems
Flame retardant properties of inorganic compounds for cotton textiles.

Antibacterial properties of cotton textiles
The demand for antibacterial textiles, particularly those made from cotton, has become increasingly important for combating microbial proliferation, which can lead to hygiene issues and fabric deterioration. Cotton, known for its hydrophilic nature and large surface area, provides an ideal environment for bacteria and other microorganisms to thrive, particularly under humid conditions.
191
This is critically relevant in medical textiles, where the prevention of nosocomial infections is crucial.
192
Thus, the development of functional textiles with antibacterial properties is essential for protection against airborne bio-particles and microorganisms.
193
The market for antimicrobial textiles was estimated at $10.7 billion in 2021 and is projected to reach $14.7 billion by 2026, with a compound annual growth rate (CAGR) of 6.5% during this period.
194
As shown in Figure 14, the antimicrobial textile industry is anticipated to experience substantial growth in the coming years, driven by the increasing demand for medical textile and apparel applications. Global antimicrobial textile market trends.
194

Synthetic antibacterial solutions
Overview of the chemical structures and mechanisms of action of conventional antibacterial agents, along with the primary fibers in which they are applied.
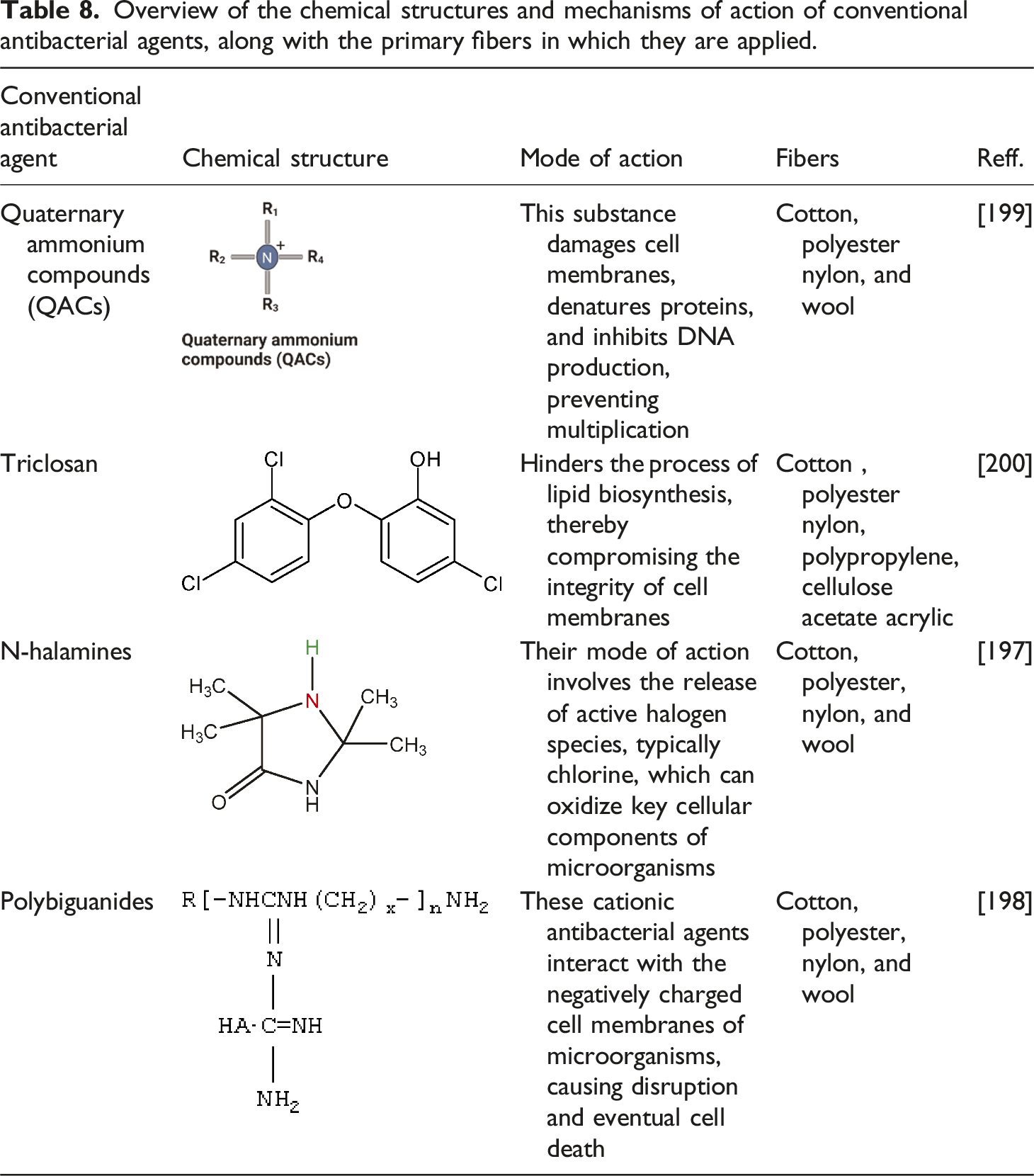
Synthetic antimicrobial treatments for cotton, including triclosan, quaternary ammonium compounds (QACs), N-halamines, and polybiguanides, employ various mechanisms to combat bacterial growth and proliferation on textile surfaces. Triclosan inhibits bacterial fatty acid synthesis and disrupts cell membrane functions. It is effective against a wide range of microorganisms but has been scrutinized due to environmental concerns and potential endocrine disruption in humans. 200 On the other hand, QACs are cationic compounds that disrupt bacterial cell membranes through electrostatic interactions and hydrophobic alkyl chain penetration. They are stable, easily manufactured, and exhibit a broad spectrum of antimicrobial activities. 199 N-halamines operate through a different mechanism, releasing active chlorine upon contact with moisture, which oxidizes the microbial cell components. This allowed for the regeneration of the antimicrobial activity after washing. 197 Polybiguanides, such as polyhexamethylene biguanide (PHMB), disrupt cell membranes and cause cytoplasmic coagulation. Interestingly, while these synthetic antimicrobials are effective, there is growing concern about bacterial resistance, particularly to QACs and triclosan. 198 This has led to increased interest in bound or non-leaching antimicrobials, including QACs, PHMB, and N-halamines. These agents offer durable antimicrobial protection and potentially reduce the risk of developing resistant strains. 199
It is essential that the antibacterial treatment of textiles meet various requirements, including effectiveness against microorganisms and non-toxicity to consumers. This includes consideration of cytotoxicity, potential allergies, irritation, and sensitization. 201 However, conventional antibacterial treatments of cotton textiles have raised significant environmental and health concerns. One major drawback is the potential for bioaccumulation of toxic substances in living organisms, which can lead to adverse health effects owing to their persistence and accumulation. 43 Additionally, halogenated compounds used in some antibacterial treatments have been found to be toxic and capable of bioaccumulating in the food chain, raising serious environmental concerns. 202 Environmental impacts include water pollution, ecosystem disruption, and health risks to both wildlife and humans. The persistence of these chemicals in the environment and their accumulation in the food chain highlight the urgent need for more sustainable and less harmful alternatives to conventional antibacterial treatments.43–45,202,203
Eco-friendly antibacterial solutions
Eco-friendly antibacterial solutions for cotton textiles are increasingly being sought to address both microbial resistance and environmental concerns. These solutions can be broadly categorized into natural antibacterial agents, biotechnology-based approaches (although not widely popular), and nanomaterials. Natural antibacterial agents are derived from plants and other organic sources and provide effective antibacterial properties while minimizing the environmental impact and potential health risks associated with synthetic antimicrobials. 51 Integrating these solutions into textiles is a promising avenue for developing durable, hygienic, and environment-friendly cotton fabrics. Nanomaterials, on the other hand, offer unique antibacterial properties owing to their high surface area and reactivity. These materials, such as silver nanoparticles and zinc oxide, can be incorporated into cotton fabrics to enhance their antibacterial efficacy without compromising their integrity.204,205 This section explores both natural and nanomaterial-based antibacterial solutions for cotton, discussing their effectiveness, applications, and potential challenges.
Natural antibacterial agents
Plant-based extracts and essential oils
Numerous plant species around the world remain to be explored for their potential medicinal uses and other applications. The value of these plants lies in their diverse range of phytochemicals. Phytochemicals are naturally occurring compounds within plants that can exhibit antibacterial properties.
206
As shown in Figure 15(a), parts of a plant are rich in valuable phytochemicals or secondary metabolites that can be extracted for various purposes.
208
Certain phytochemicals, including phenolic compounds, quinones, flavonoids, tannins, coumarins, terpenoids, alkaloids, and saponins, have demonstrated antibacterial activities.
209
These substances can combat bacteria by disrupting their essential components (Figure 15(b)).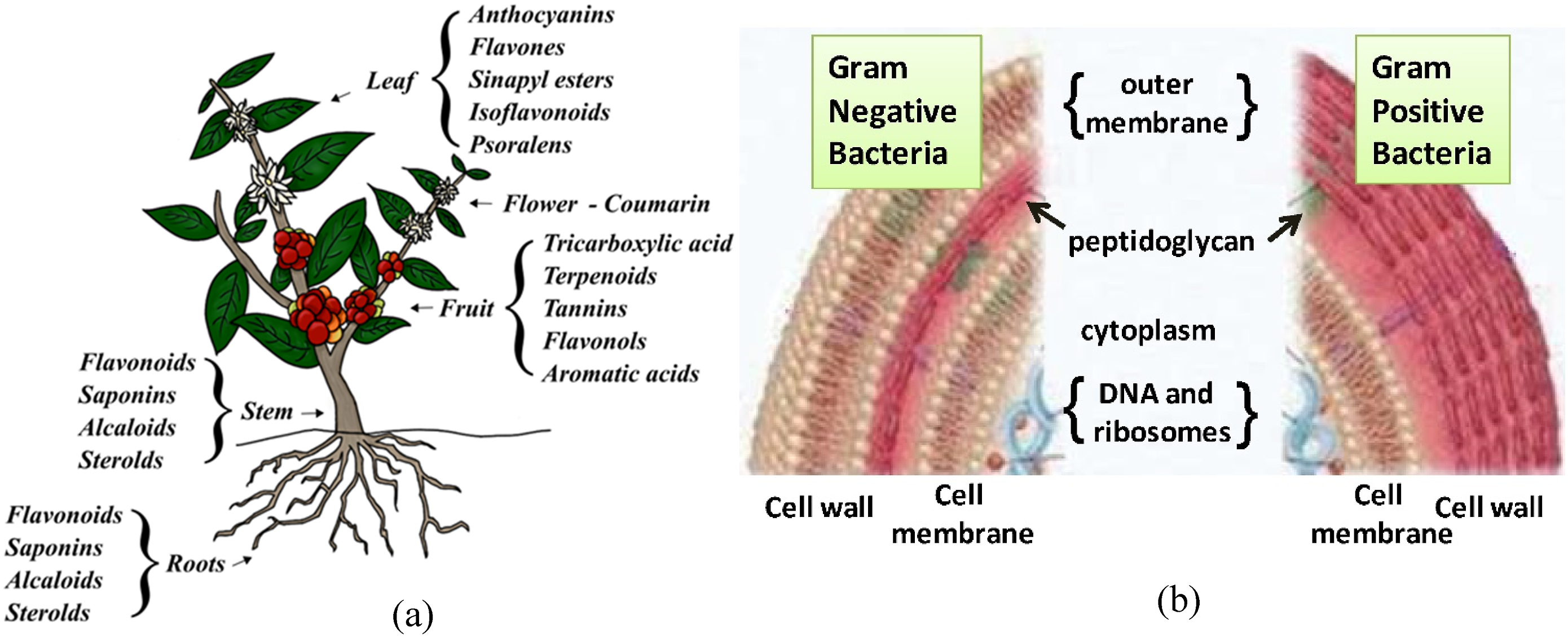
Phytochemicals impede bacterial growth by interfering with the ribosomes, cell walls, DNA, and cell membranes. They inhibit protein synthesis by targeting the 30S (small) and 50S (large) ribosomal subunits, creating pores that allow for increased penetration and disruption of bacterial components.
210
They also attack the cell wall by targeting enzymes crucial for peptidoglycan synthesis, which provide structural integrity to the cell wall.
211
In addition, phytochemicals interfere with DNA gyrase, blocking DNA replication and bacterial growth.
209
Notably, the composition and concentration of phytochemicals differs across plant parts.
208
Research has indicated that the highest antibacterial properties are found in leaves, which are often preferred for medicinal applications.
212
Aloe vera, extensively studied for its diverse applications, shows significant potential in areas such as wound healing and medical textiles owing to its antibacterial and antifungal properties (Figure 16).
213
For instance, chitosan coatings loaded with Aloe vera extract and Application of aloe vera-finished textiles in wound healing and UV-resistant textiles. Reproduced with permission from the ref.
213
© 2020, Springer Nature. The antibacterial efficacy of plant extract and essential oil in cotton fabrics.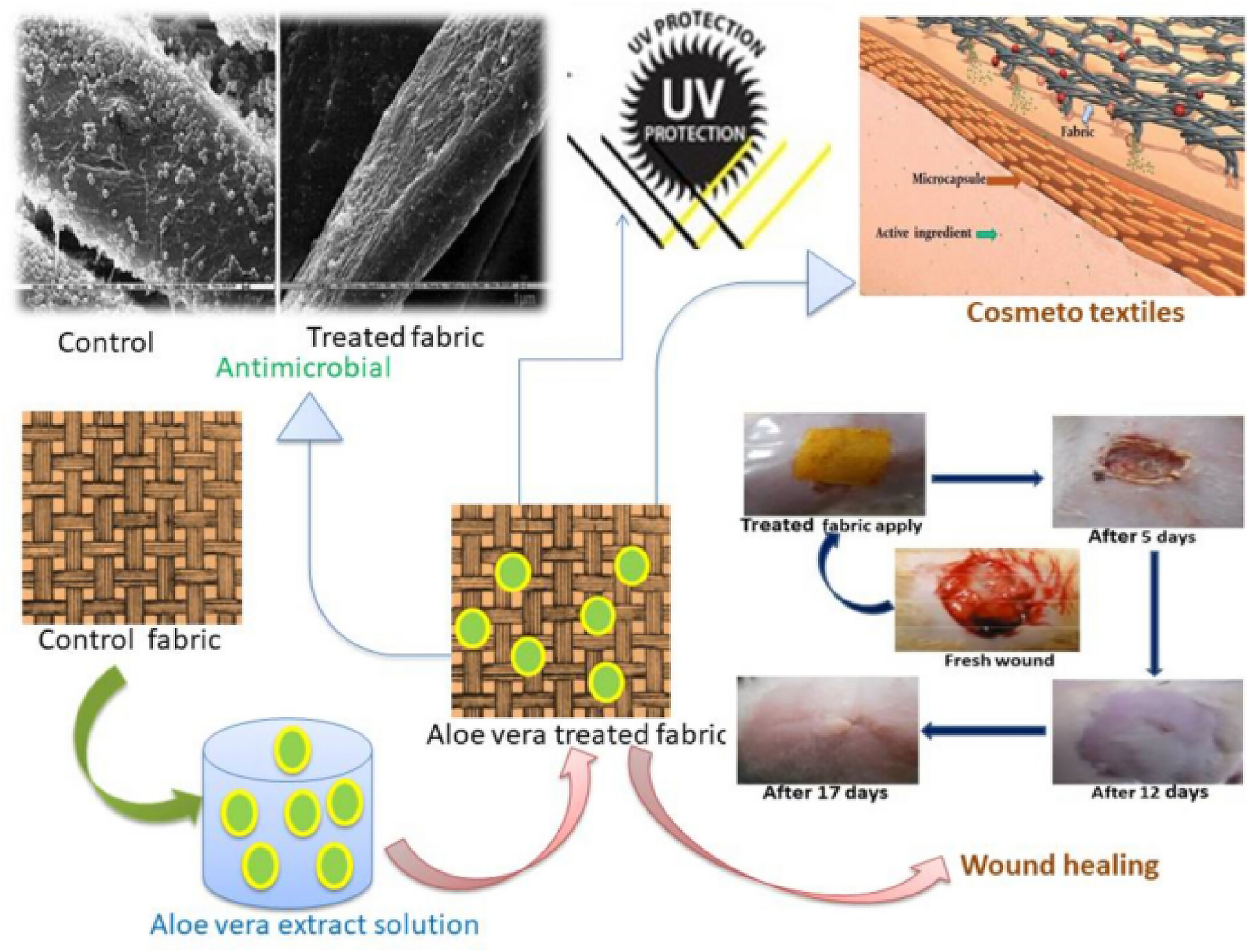
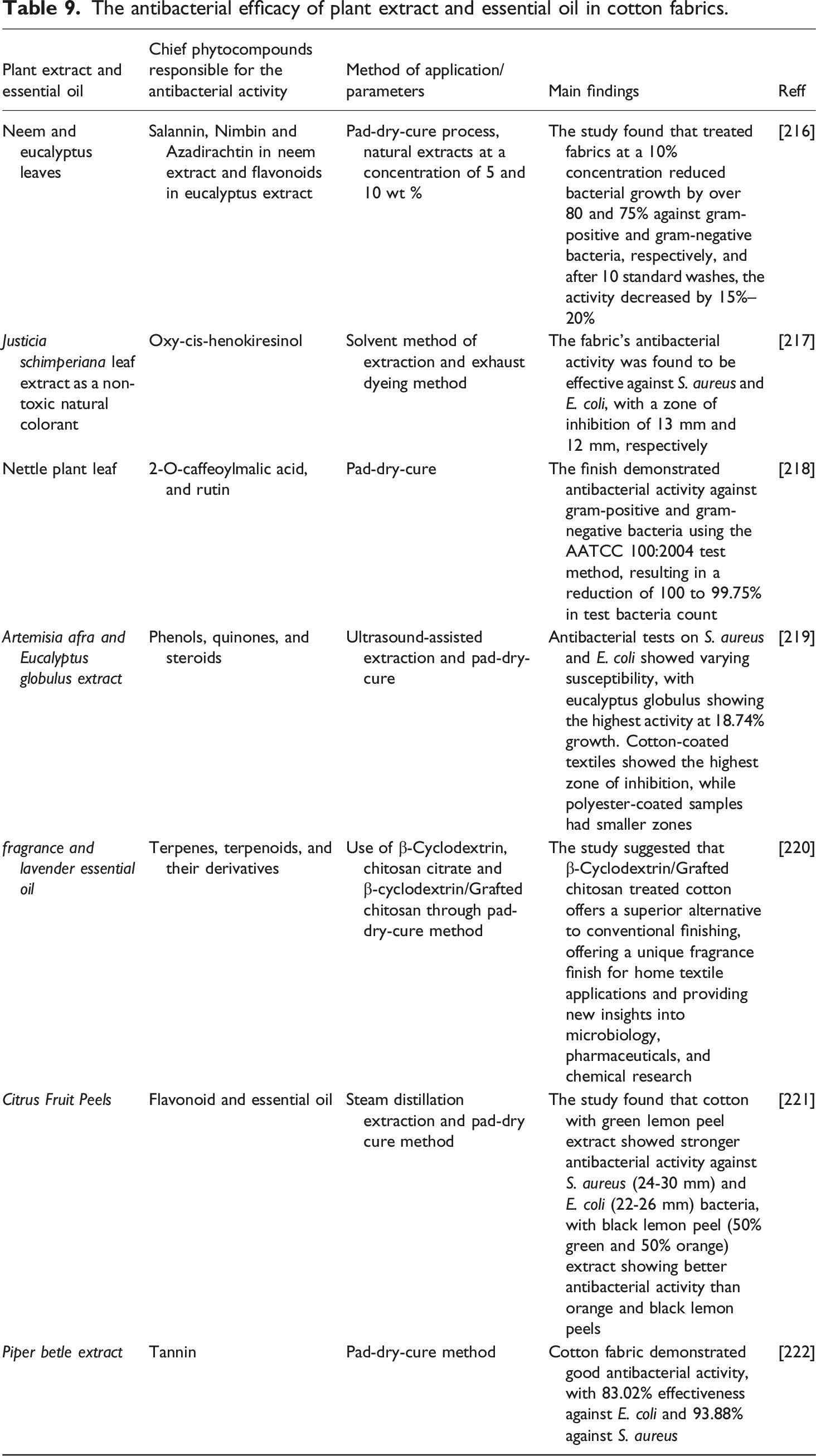
Apart from plant extracts, Essential oils (EOs) are characterized by their complex chemical compositions, including terpenes, phenylpropanoids, aldehydes, ketones, alcohols, esters, phenols, and ethers. These oils represent novel alternatives to conventional medications because of their biological efficacy against various pathogenic microorganisms.
223
For example, thyme essential oil has been applied to linen-cotton blends, resulting in significant antibacterial and antifungal activity without compromising fabric strength.
214
Despite their potential, commercial applications of essential oils are limited owing to issues such as evaporation loss and difficulties in controlled release. To address these challenges, essential oils can be formulated into emulsions, microcapsules, and gels. Integrating essential oils into biopolymers allows for their protection and gradual release over time.224,225 Rosu et al.
223
developed materials with aromatherapeutic and antibacterial properties by applying emulsions based on peppermint essential oil (PEO) onto cotton fabrics. The emulsions showed good inhibitory effects against Gallic acid functionalized polylysine for endowing cotton fiber with antibacterial, antioxidant, and drug delivery properties. Reproduced with permission from.
228
© 2022 Elsevier B.V.
Natural dyes as antibacterial agents
The introduction of natural dyes as antibacterial agents in textiles is essential to enhance the health-protective features of fabrics. This innovation has wide-ranging applications, including medical textiles, hygiene-focused fabrics, and personal protective equipment (PPE) (Figure 18).
51
Researchers have extensively investigated natural dyes derived from various biological sources, such as plants, microbes, and fungi, along with different mordants, to impart antibacterial properties to textiles.
51
These natural dyes, when applied using conventional dyeing techniques or mordants, have shown promising results in inhibiting the growth of pathogenic microbes. Baseri
229
developed a method for producing antibacterial and biodegradable cotton fabrics using natural dyeing techniques. The dye was extracted from boiled pomegranate rinds and processed into fine powder. Whey protein isolate was used as an eco-friendly mordant to enhance the affinity of the fabric for pomegranate rind extract. This eco-friendly method maintained desirable antibacterial properties against pathogenic and spoilage bacteria even after 10 washing cycles, while providing excellent protection against ultraviolet radiation. This procedure presents a promising option for biomedical applications without adverse environmental effects, with the natural mordant serving as a good alternative to the commonly used metallic mordants. Personal protective clothing with newly developed, cost-effective technology can reduce the presence of enveloped viruses such as SARS-CoV-2 and phi-6 as well as methicillin-resistant bacteria such as 
Additionally, some studies have explored plasma pretreatment for fiber surface modification, demonstrating its effectiveness in replacing ultrasonic bath coatings for eco-friendly cotton bio finishing. Li et al.
230
utilized plasma fiber surface modification combined with soy protein coating to enhance natural dyeing ability of cotton and achieve functional finishes using the herbal plant
Protein-based antibacterial agents
Protein-based antibacterial agents represent another class of natural antibacterial solutions for cotton textiles, gaining significant attention owing to their natural origin and potential biomedical applications. Key examples include chitosan, lysozyme, lactoferrin, and silk fibroin, all of which have shown promise as effective antibacterial agents. Chitosan, which is derived from chitin, is a biopolymer known for its inherent antibacterial properties, making it particularly suitable for cotton textiles. It is noted for its ability to inhibit fungal growth, compatibility with cotton, and nontoxicity.231,232 The antibacterial action of chitosan is believed to involve electrostatic interactions between the positively charged amine groups (NH3+) at the C2 position of glucosamine monomers and the negatively charged components on the bacterial and fungal cell surfaces. These interactions compromise cell integrity and enhance permeability, leading to leakage of crucial cellular elements and eventual microbial death.
233
Silva et al.
234
explored chitosan nanocapsules (CNs) as potential self-disinfecting agents for cotton fabric. This study demonstrated that attaching CNs to cotton surfaces using polyacrylate resulted in significant changes in the surface characteristics of the fabric (Figure 19). The CN/polyacrylate coating exhibited antibacterial efficacy against (a) Ester (–OCO–) and amide (–NHCO–) bonds form between chitosan nanocapsules (CN) and polyacrylate in the binder through aminolysis (blue bar), condensation (green bar), transesterification (orange bar), and esterification (yellow bar). The binder adheres to the cotton fibers’ surface due to reactions between hydroxyl (OH) groups on cellulose chains and ester and carboxylic acids in polyacrylate. These interactions also contribute to the formation of (b) CN/binder aggregates and (c) a binder film on the cotton fiber’s surface, with nanocapsules trapped inside (CN/polyacrylate coating).
234
Copyright © 2023, American Coatings Association.
It is worth noting that, the effectiveness of chitosan can be enhanced by combining it with other substances, such as metal nanoparticles or high-polymer antibacterial agents. For example, cotton fabrics modified with both chitosan and nanoparticles exhibit greater antibacterial activity than fabrics treated with either agent alone.
232
In addition, researchers are developing highly active antibacterial dressings using N-halamine biocides and chitosan. Nawaz et al.
235
modified a nonwoven cotton fabric using eco-friendly precursors, achieving strong antibacterial properties without compromising fabric strength. Similarly, Gadkar et al.
236
studied a self-assembled coating on a cotton fabric using the LBL technique to impart antimicrobial properties. They employed up to 15 bilayers of poly (styrenesulfonate) (PSS) and silver-loaded chitosan (CS-Ag) nanoparticles as anionic and cationic agents, respectively. Both qualitative and quantitative analyses demonstrated enhanced antibacterial activity of the CS-Ag-coated fabric compared to chitosan-only coatings, supported by sustained Ag+ release measured by atomic absorption spectroscopy (Figure 20). Importantly, the L-B-L coating did not adversely affect the physical properties of the fabric, including air permeability, tensile strength, and flexural rigidity. Chitosan has also been employed in grafting copolymerization to create high-polymer antibacterial agents, achieving long-lasting antibacterial effects while improving the mechanical properties.
237
When used in pigment printing, chitosan demonstrated up to a 96% reduction in Antibacterial efficacy of silver loaded chitosan nanoparticles on layer-by-layer self-assembled coated cotton fabric. Reprinted with permission from.
236
© 2020 Elsevier B.V.
Lysozyme and lactoferrin have also been studied as potential antibacterial agents in cotton. Lysozyme is notable for its ability to hydrolyze the peptidoglycan layer of bacterial cell walls, although its effectiveness against gram-negative bacteria is limited.
240
Lactoferrin, often used in combination with lysozyme, has shown enhanced antibacterial activity against Sericin-TiO2 nanocomposite treated cotton fabrics for enhanced antibacterial and self-cleaning properties.
244
© 2024 Elsevier B.V. CC-BY-NC license.
Synergetic effect of chitosan and plant extract/essential oils
There is growing interest in the benefits of combining chitosan coatings with eco-friendly additives such as plant extracts and essential oils.
231
These components are known for their antibacterial, antifungal, and biodegradable properties, as well as their safety for human health, making them suitable for use in traditional and alternative medicine, personal care products, and natural preservatives. Essential oils, which are rich in phenolic compounds such as carvacrol and thymol, exhibit significant antibacterial and insecticidal properties. For instance, Ibrahim et al.
245
found that essential oils such as lavender, clove, aloe vera, and cinnamon enhance the antibacterial properties of cotton fabrics modified with MCT-βCD. Similarly, plant extracts with bioactive compounds, such as flavonoids and tannins, positively influence the biocidal properties of textile materials. Research by Javid et al.
246
indicated that increasing the concentrations of chitosan, essential oils, and surfactants, particularly eucalyptus and sandalwood oils, improved the antibacterial properties of modified cotton fabrics. Kumar et al.
247
reported that combining chitosan with frankincense oil enhanced antibacterial, antioxidant, and mosquito repellent properties, while also improving flame retardancy. Chandrasekar et al.
248
utilized chitosan compositions with medicinal plant extracts from Bioactive and biodegradable cotton fabrics are produced via the synergistic effects of plant extracts and essential oils in chitosan-coated systems.
214
© 2024 Springer Nature. CC-BY-NC license.
Nanomaterials for antibacterial applications
Cotton textile antimicrobial finishing utilizes metal and metal oxide nanoparticles as well as nanofibers and nanocomposites to enhance the antimicrobial properties of cotton fabrics.39,249 Notable metal and metal oxide nanoparticles, such as silver, zinc oxide, copper, and titanium dioxide, are recognized for their effective antimicrobial activity.250–253 However, traditional synthesis methods often involve toxic chemicals and nonpolar solvents, leading to hazardous byproducts.
254
Recent advancements have identified high-yield, non-toxic, and environmentally friendly methods for synthesizing nanoparticles using bacteria, plant extracts, fungi, and algae. These biological agents act as reducing, stabilizing, and binding agents for the in situ synthesis of nanoparticles on cotton fabrics.
255
The incorporation of these nanoparticles not only imparts antimicrobial properties but also enhances other functionalities, such as photocatalytic activity.
256
For example, Cuk et al.
254
developed an environmentally friendly textile material by synthesizing silver nanoparticles (AgNPs) directly on cotton fabrics using water extracts from food waste and invasive plants. The highest concentration of Ag NPs was found in samples treated with (a) Antibacterial and UV-protective cotton fabrics using plant food waste and alien invasive plant extracts as reducing agents for the in situ synthesis of AgNPs.
254
© 2021, CC BY license; (b) multifunctional antibacterial and ultraviolet protective cotton cellulose developed by the in situ biosynthesis of AgNPs into a polysiloxane matrix mediated by sumac leaf extract.
257
© 2021, CC BY license.
However, functional nanoparticle coatings often exhibit low antimicrobial performance, limiting their effectiveness. To enhance the binding strength to cotton surfaces, various strategies, such as the chemical modification of nanoparticles and textile substrates, have been employed. For instance, Stular et al.
257
developed an eco-friendly method to create multifunctional antibacterial and UV-protective cotton surfaces. This process involves pre-modifying cellulose fibers with a polysiloxane matrix and green in situ biosynthesis of AgNPs using sumac leaf extract, overcoming the limitations of traditional methods (see Figure 23(b)). The modified cotton surface exhibited strong antimicrobial and UV protection properties, with AgNPs and sumac leaf extract demonstrating biocidal activity against
While AgNPs have been widely used, concerns about bacterial resistance have shifted the focus towards other metals, such as titanium and copper, and their oxides. These materials have shown promise owing to their antimicrobial efficacy and potential for developing effective antimicrobial cellulose wound dressings.
258
Xue et al.
259
developed a Schiff base for chitosan modified with salicylaldehyde and titanium dioxide (SCSB-Si-TiO2). This was coated onto cotton fabrics using a layer-by-layer (LBL) self-assembly method, resulting in functional cotton fabrics with excellent antibacterial and UV protection properties, achieving inhibition rates of 95.22% against (a) Functional cotton fabrics with antibacterial and UV protection. Reprinted with permission from.
259
© 2024 ACS; (b) Sonochemical treatment for enhanced properties. Reprinted with permission from.
260
© 2022 Elsevier. (c) Diagram presentation of the mechanisms of the antimicrobial activity of textile materials functionalized with CuO NPs: (a) release of copper ions, (b) contact of CuO NPs with bacteria, and (c) reactive oxygen species.
261
© 2021, CC BY license.
Summary of the functionalization of cotton fabrics with mixtures of nanoparticles and their applications. 268 © 2022, CC BY license.

Eco-friendly finishing solutions with dual functionalities
The textile industry has made significant strides in the development of eco-friendly finishing solutions that integrate flame retardancy and antimicrobial properties. Although previous sections have discussed these functionalities separately, their convergence into a single treatment represents a pivotal advancement. This integrated approach enhances fabric safety and durability, while aligning with modern environmental standards. By addressing safety concerns in home textiles and apparel, and combating microbial growth, these dual-function treatments provide comprehensive solutions for hygiene and durability.26,100,269,270 Researchers are increasingly employing natural agents, biopolymers, and eco-friendly synergetic innovations to create sustainable cotton textiles that embody these dual functionalities, marking an essential step toward a greener textile industry.
Plant-based extracts with dual functionalities
Plant-based extracts and essential oils have emerged as promising alternatives for cotton textiles with both flame retardancy and antimicrobial properties.
269
This dual functionality is advantageous because it enhances performance while minimizing the environmental impact associated with traditional chemical treatments. For example, (a) Natural bioactive formulations of biodegradable cotton eco-fabrics with antibacterial and fire-shielding properties. Reprinted with permission from.
272
© 2023 Elsevier B.V.; (b) eco-friendly flame retardants and antibacterial coatings on cotton fabrics via LBL self-assembly. Reprinted with permission from.
52
© 2020, Springer Nature B.V.
Moreover, some flame-retardant chemicals possess inherent antibacterial properties, which further blur the lines between these functionalities. Fang et al.
53
reported the successful application of potassium alginate (PA) coupled with polyhexamethylene guanidine phosphate (PHMGP) to create a coating that conferred both flame retardancy and antibacterial effects on cotton fabrics. This coating effectively reduced the peak heat release rate (PHRR) and total heat release (THR), indicating enhanced flame resistance while also exhibiting significant antibacterial activity against pathogens such as
Innovative approaches and perspectives
Other studies have demonstrated the potential of various natural compounds along with special coating techniques to impart these dual functionalities. The use of bio-based IFR systems, microencapsulation techniques, and reactive antibacterial finishes represents an innovative approach to achieving multifunctional cotton textiles.26,100,269,270 For instance, atmospheric-pressure dielectric barrier discharge plasma treatment followed by the deposition of nano-TiO2/SiO2 improved both the flame retardancy and antibacterial activity of cotton textiles. 273 Additionally, chitosan-based coatings protonated with amino trimethylene phosphonic acid (ATMP) exhibit excellent flame retardancy and antibacterial rates against common bacteria. 142 Furthermore, impregnating polyester-cotton blended fabrics with guanidine phosphate (GP) and polyethylenimine (PEI) results in textiles with high flame retardancy and significant antibacterial properties. 21 Innovative step-by-step dip-coating and spraying technologies have produced durable superhydrophobic and flame-retardant cotton fabrics that maintain their properties even after extensive washing and abrasion. 274 The synthesis of an N-heterocyclic compound with ammonium phosphate ester groups has been shown to enhance both the flame-retardant and anti-wrinkle properties of cotton fabrics, demonstrating remarkable durability and restorability after washing. 26 Other formulations, such as combinations of antimony pentoxide sol (Sb2O3), aluminum hydroxide sol (Al(OH)3), and fluoroalkyl silanes, have yielded cotton fabrics with superhydrophobicity, self-cleaning abilities, and enhanced flame retardancy. 55
Chitosan-based compounds with dual functionalities
Chitosan-based compounds have emerged as particularly effective dual-functional finishing agents for cotton fabrics, imparting both flame-retardant and antibacterial properties.
27
Numerous studies have highlighted the effectiveness of chitosan formulations for achieving these dual functionalities. For instance, a two-stage finishing process utilizing chitosan in conjunction with citric acid and phytic acid has been shown to impart multiple performance characteristics, including flame retardancy and antibacterial properties, to linen fabrics.
275
Similarly, a LBL technique using chitosan in a citric acid solution combined with sodium lignin sulfonate and boric acid resulted in a cotton fabric with excellent flame retardancy and antibacterial activity.
276
However, a notable advancement in this area was reported by Huang et al.,
142
who developed a single-component chitosan-based multifunctional coating (ATMP-CS) that effectively conferred both flame-retardancy and antibacterial properties to cotton fabrics. This formulation achieved a LOI of 29.7% and high antibacterial rates against Fabrication of chitosan-based finishing agents for flame-retardant, UV-protective, and antibacterial cotton fabrics. Reprinted with permission from.
149
© 2022 Elsevier Ltd.
Interestingly, the source of chitosan can vary significantly, with one innovative study utilizing chitosan extracted from waste shrimp shells to create a multifunctional finishing formulation for cotton. 277 This approach not only imparts the desired properties but also contributes to sustainable practices by repurposing waste materials, aligning with the growing demand for eco-friendly solutions in textiles. The synergistic effects of chitosan with other compounds have been substantiated in several studies. For instance, the combination of TiO2 nanoparticles and chitosan phosphate results in improved flame retardancy and antibacterial properties in treated cotton fabrics. 278 Additionally, employing chitosan as a nitrogen source during the phosphorylation process has demonstrated a synergistic effect with phosphorus, further enhancing the flame retardancy of treated cotton fabrics. 27 It is summarized that, chitosan-based compounds represent a significant advancement in the dual-functional finishing of cotton fabric. These formulations not only provide effective flame retardancy and antibacterial characteristics but also align with the increasing demand for sustainable and eco-friendly textile treatments. The versatility of chitosan, its compatibility with other compounds, and its potential to be sourced from waste materials make it an attractive option for future innovations in textile finishing. As the textile industry continues to evolve, these eco-friendly solutions will play a vital role in addressing both consumer demand and environmental challenges, paving the way for a more sustainable future for textile production.
Synergetic effect of eco-friendly solution with dual functionalities
As discussed in the previous section, achieving dual functionality in textiles requires a multifaceted approach that integrates various environment-friendly finishing methods. Rather than relying solely on a single eco-friendly solution such as plant extracts, chitosan, or nanomaterials, the synergistic effect of combining multiple techniques has garnered significant interest in textile research.
54
This section explores innovative strategies employed to enhance both the flame retardancy and antibacterial properties of cotton textiles, emphasizing the importance of collaborative methodologies. For instance, Nabipour et al.
54
developed guanazole complexes with silver and zinc ions that were applied to cotton fabrics to create textiles that exhibited both flame-retardant and antibacterial characteristics. The effectiveness of these complexes was rigorously evaluated using various methods, including assessments of fabric morphology, flammability, and thermal degradation. Remarkably, the guanazole-zinc- and guanazole-silver-treated fabrics demonstrated high LOI values and self-extinguishing properties during vertical burning tests. Furthermore, TGA confirmed that these complexes promoted thermal decomposition, resulting in increased char yield. Microscale combustion calorimetry measurements indicated a significant reduction in PHRR and THR compared with untreated cotton, showing enhanced flame retardancy. In addition to these fire safety benefits, the treated fabrics exhibited substantial antibacterial activity against pathogens such as An environmentally friendly approach to fabricate flame-retardant, antibacterial, and antifungal cotton fabrics via the self-assembly of guanazole-metal complexes. Reprinted with permission from.
54
© 2020 Elsevier Ltd.
Based on these findings, Zhang et al.
21
introduced a treatment for polyester-cotton-blended fabrics utilizing guanidine phosphate (GP) and polyethylenimine (PEI). This innovative treatment achieved a LOI of 30.5%, simultaneously enhancing flame retardancy while maintaining excellent antibacterial properties. This example illustrates that integrating multiple eco-friendly agents can yield significant improvements in both flame resistance and microbial protection, without compromising the mechanical integrity of the fabric. In a related study, Jin et al.
279
synthesized a new material, DMH-BPMS, from hydantoin-bromopropylmethylsiloxane, which was then mixed with ammonium polyphosphate (APP) and sodium alginate for coating cotton fabrics. The resulting textiles exhibited a LOI of 29.8% and a char length of 9.6 cm at a specific mass ratio, indicating effective flame retardancy. Cone calorimetry tests further demonstrated a remarkable 36% decrease in THR, CO2 release, and total smoke production compared to APP-coated cotton. Additionally, the DMH-BPMS/APP-treated fabrics displayed impressive antibacterial effects against Flame-retardant and antibacterial cotton fabrics were fabricated by incorporating a linear polysiloxane-based coating. Reprinted with permission from.
279
© 2022 Elsevier B.V.
Furthermore, recent advancements have highlighted the efficacy of LbL self-assembly techniques in developing multifunctional finishes that exhibit both flame-retardancy and antibacterial properties. This method has gained traction among researchers owing to its precise control over the deposition of functional materials, which allows the integration of multiple characteristics. For example, Chen et al.
280
developed a multifunctional cotton fabric using LbL self-assembly, incorporating a flame-retardant and antibacterial linear polymer (P(AA-ADMH)) along with phytic acid and γ-aminopropyl triethoxysilane (see Figure 29(a)). The modified fabrics exhibited improved LOI values and a reduced PHRR, whereas the N-halamine structure conferred enhanced antibacterial efficacy within 30 min. The resulting contact angle of approximately 110° indicated reduced bacterial adhesion, further enhancing the hygienic properties of the fabric. (a) Schematic illustration of the preparation of modified cotton fabrics. Reprinted with permission from the ref
280
; (b) durable flame-retardant, anti-UV, photothermal, and antibacterial cotton fabrics based on lignin and ammonium polyphosphate. Reprinted with permission from.
281
© 2024 American Chemical Society.
Durability challenges and solutions
Although the demand for durable multifunctional cotton fabrics is increasing, LbL self-assembled coatings are facing several durability challenges. Mechanical stress can lead to the wear of these coatings, diminishing their efficacy over time. Frequent washing may lead to peeling or deterioration, compromising the intended multifunctional characteristics. To mitigate these concerns, covalent grafting techniques are often employed to establish stronger, longer-lasting connections between the functional materials and fabric substrates. Liu et al. 281 developed a durable multifunctional cotton fabric by covalently grafting phosphorylated lignin (LP) and ammonium polyphosphate (APP) onto the fabric surface. This innovative approach yielded a fabric that not only offers UV protection and photothermal conversion but also demonstrates fire safety and antibacterial properties. Remarkably, the fabric maintained a LOI of 48.5% and achieved self-extinguishment in the vertical burning tests, showing excellent durability even after 50 washing cycles, with an LOI value of 31.5% (see Figure 29(b)).
In addition to these advancements, the integration of hydrogel polymers into textile treatments is a novel approach for enhancing flame retardancy while maintaining water retention. Yu et al.
282
developed a novel fire-preventing triple-network (TN) hydrogel composed of poly(N-isopropylacrylamide) (PNIPAAm), sodium alginate (SA), and polyvinyl alcohol (PVA). This hydrogel was applied to a cotton fabric and subjected to ionic coordination crosslinking in CaCl2 solution to establish a stable structure. The hydrogel-fabric laminates demonstrated impressive fire resistance, remaining largely intact after exposure to fire for 12 s, unlike the untreated fabrics. This fire resistance can be attributed to energy absorption from the water within the hydrogel, which heats and evaporates, creating a protective barrier. Additionally, the hydrogel exhibited excellent antibacterial properties, demonstrating over 96% efficacy against (a) Schematic diagram of the preparation of fire-resistant hydrogel-fabric laminates. (b) Interpenetrating polymer network structure and antibacterial activity of the triple-network composite hydrogels. (c) Vertical fire-retardant testing of untreated cotton fabric and hydrogel-fabric laminates. (d) Schematic illustration of the mechanism of fire resistance of hydrogel-cotton fabric laminates. (e) Photograph of bacteria recovered from the samples against Summary of eco-friendly finishing solutions for enhanced flame retardancy and antibacterial performance of cotton textiles.

Challenges and future research directions
Overview of challenges, past actions, remaining issues, and future research directions in eco-friendly flame retardants and antibacterial finishes for cotton textiles.

Conclusion
The development of the eco-friendly flame-retardant and antibacterial finishing of cotton textiles is one of the great steps forward in textile technology. This review highlighted a trend towards the use of natural materials, such as plant-based extracts, bio-based polymers, and nanomaterials, as efficient flame retardants. It seems that the efficiency of these eco-friendly solutions depends on their origin and formulation, and some of them could show a synergistic effect when combined with different natural flame retardants. The present review further deals with eco-friendly antibacterial treatments, underlining natural agents, biotechnological approaches, and nanomaterials. The application of compounds such as essential oils and silver nanoparticles, together with novel synthesis methods, represents new perspectives in producing textiles capable of effective microbial growth inhibition. Most importantly, the synergistic interaction, especially that between chitosan and plant extracts, enhanced the overall safety and efficacy of the treated cotton textiles. The development of such dual-functional solutions has taken into consideration flame retardancy and antimicrobial properties, key concerns for safety and hygiene. Further, different innovative techniques such as layer-by-layer self-assembly and covalent grafting have opened up new opportunities for durable multifunctional fabrics that could meet modern consumer demands. However, despite these, scalability, durability, washability, and cost-effectiveness continue to remain bottlenecks to the widespread use of eco-friendly treatments. Most of these promising methodologies remain at the research level and need further refinement to make them viable industrially. Further research on optimization of formulations with respect to durability-performance aspects, studying practical applications to bridge the gap between laboratory innovations and real-world applications, is needed.
Footnotes
Acknowledgments
The authors express gratitude to the current and previous group of researchers in the field of textile finishing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
