Abstract
The reinforcement of polymer matrices with natural fibers has emerged as a sustainable pathway to enhance mechanical and environmental performance. In this study, sisal fibers were first mercerized and subsequently modified using potassium permanganate (0.09%) and sodium chlorite (1.5%), with the aim of improving fiber–matrix adhesion and assessing the influence of temperature on composite properties. Twill 2/2 woven laminates were fabricated via vacuum bagging and evaluated in terms of tensile, flexural, and impact performance, as well as water uptake in saline environments. Results showed that alkali treatment significantly increased tensile strength from 129.31 ± 4.32 MPa to 266.12 ± 3.56 MPa, while the addition of KMnO₄ further boosted it to 359.11 ± 2.67 MPa. Water absorption decreased after treatments but rose with temperature from 18°C to 40°C. SEM analysis confirmed improved interfacial bonding, and TGA revealed enhanced thermal stability, particularly in alkali- and KMnO₄-treated composites. The combined alkali and KMnO₄ treatment offered the most balanced improvement, highlighting its potential for marine and structural applications where durability and resistance to aggressive conditions are required.
Keywords
Introduction
In the current context, the challenges of continuous improvement in industrial processes lie in meeting the needs of the global market for sustainable textiles and materials by offering eco-friendly products, such as natural fibers from hemp, sisal, jute, flax, and kenaf. 1 These natural fibers exhibit particularly interesting mechanical and physical properties, making them ideal for use as reinforcements in thermoplastic and thermosetting resins.2,3
Natural fibers have attracted the attention of several industries for various applications such as textiles, notably in the automotive sector, due to their availability, low density, biodegradability, low cost, rigidity, flexibility.4–6 These fibers are shock-resistant, and thanks to these potentials, natural fibers have become a competitive alternative to synthetic fibers, which rely on polluting production processes that negatively impact both the environment and human health.7,8
However, the hydrophilic nature of natural fibers creates a compatibility issue with the hydrophobic nature of the polymer matrix, which can negatively influence the mechanical properties of the composite, such as tensile strength, flexural strength, and impact resistance. 9 This issue requires further research to optimize the performance of natural fiber-reinforced composites. To ensure excellent load transfer at the interface, strong adhesion between the fiber and the matrix is necessary, which can be achieved through chemical bonding between the fiber surface and the polymer matrix. 10
Furthermore, natural fibers absorb a significant amount of water, which adversely affects their mechanical properties. 11 To address this issue, surface modification of the fibers is essential to improve adhesion between the fibers and the matrix. While environmentally friendly treatments are preferred, not all chemical modification methods fully meet this criterion. Common methods include alkali treatment, benzoyl chloride, potassium permanganate, silanes, and acrylic acid treatments.12–15
The most commonly used chemical treatment for natural fibers is NaOH, which serves to reduce hydrogen bonds, remove surface impurities such as waxes, lignin, and hemicellulose, and roughen the fiber surface to promote better contact and adhesion with the polymer matrix. 17 Alkali treatment is generally applied in concentrations ranging from 2% to 10%, with treatment times and temperatures optimized depending on the fiber type and desired properties.
The effect of alkali treatment on natural fiber-reinforced composites has been extensively studied, and several recent works report significant improvements in mechanical properties. For instance, Widodo et al. 18 demonstrated that treating hemp fibers with 6% NaOH for 3 h increased tensile strength by 42% and flexural strength by 38% compared to untreated composites. Similarly, Bachtiar et al. 17 reported that 5% NaOH treatment of jute fibers enhanced tensile modulus by 35% and impact resistance by 28%, highlighting the positive effect of alkali treatment on fiber-matrix adhesion and overall composite performance. These results confirm that alkali treatment effectively removes hemicellulose and lignin, exposing reactive sites on the fiber surface, and improving interfacial bonding with polymer matrices.
Similarly, Mahesh et al. 19 found that a 5% NaOH treatment enhanced both the tensile and flexural strength of jute/epoxy composites, reinforcing the idea that alkali treatment strengthens the fiber-matrix interface. This aligns with the findings of Nimanpure et al., 20 who observed that NaOH-treated sisal fibers improved the tensile, flexural, and impact properties of epoxy composites compared to untreated fibers, further emphasizing the role of alkali treatment in enhancing fiber-matrix adhesion.
Beyond alkali treatments, other chemical modifications have been explored to improve fiber-matrix compatibility. Recent studies have investigated the effects of oxidative chemical treatments, such as KMnO₄ and NaClO₂, on natural fibers to improve interfacial adhesion and reduce moisture absorption. For example, Zhang et al. 21 studied the effect of KMnO₄ treatment on jute fibers in polypropylene composites and reported that 0.08% KMnO₄ treatment for 2 h reduced water absorption by 22% and increased tensile strength by 18% compared to untreated fibers. This confirms that KMnO₄ treatment enhances fiber-matrix bonding and improves the mechanical performance of natural fiber-reinforced composites.
Similarly, Li et al. investigated NaOH-NaClO₂ treatment on hemp/polyamide 1010 composites. Their results showed that treating fibers with 5% NaOH followed by 0.05% NaClO₂ for 90 min increased flexural strength by 25%, tensile strength by 20%, and improved fiber dispersion within the matrix, highlighting the synergistic effect of dual chemical treatments on both morphology and mechanical properties. 22
These findings collectively demonstrate that oxidative treatments, either alone (KMnO₄) or in combination with alkali (NaOH-NaClO₂), are effective strategies to reduce moisture uptake and enhance interfacial bonding, leading to superior mechanical performance in polymer composites reinforced with natural fibers.
More recently, similar conclusions were drawn by other researchers who confirmed the relevance of NaOH, KMnO₄, and NaClO₂ treatments in improving the mechanical and durability properties of natural fiber composites.23–26 However, most of these works were limited to short fibers or non-woven reinforcements, leaving woven sisal fabrics underexplored.
In addition to fiber treatment, the choice of reinforcement structure also plays a critical role in determining composite performance. Numerous studies have shown that woven fiber reinforcements offer superior tensile and flexural properties compared to non-woven structures.27–29
Recent research has shown that the architecture of fiber reinforcements plays a crucial role in the mechanical performance of natural fiber composites. In particular, Islam et al. 30 reported that three-dimensional (3D) woven flax/PLA composites with braided yarns exhibited improved tensile and flexural strength, indicating that yarn arrangement significantly affects load distribution within the material.
Similarly, Weatherburn et al. 31 found that polypropylene composites reinforced with woven fabrics provide superior mechanical performance compared to composites with randomly oriented or unidirectional fibers, highlighting the benefits of woven architectures in enhancing structural stability.
In addition, Baghaei et al. 32 evaluated woven hemp fabric composites and observed mechanical properties comparable to, or even exceeding, those of conventional engineered wood products, emphasizing their potential as sustainable alternatives in construction and other structural applications.
These studies collectively demonstrate that both the chemical treatment of fibers and the design of the reinforcement structure are key factors for optimizing the mechanical behavior of natural fiber composites, suggesting that careful selection of these parameters is essential for achieving high-performance materials.
It goes without saying that composites woven from natural fibers play a very important role in various sectors such as maritime, automotive, construction, etc. But, in the current state of our knowledge, there is no published research work on the mechanical properties of composites reinforced by woven sisal structures treated with a combination of two treatments: NaOH-KMnO4 and NaOH-NaCIO2.
From this literature, it is clear that while NaOH, KMnO₄, and NaClO₂ treatments have been extensively studied, their combined application on woven sisal fabrics has not yet been addressed. Moreover, the performance of such composites in saline environments relevant to maritime applications remains scarcely explored. This represents a critical knowledge gap, as real-world conditions such as seawater immersion and elevated temperatures strongly influence the long-term durability of natural fiber-reinforced composites.
The present study therefore aims to address this knowledge gap by manufacturing polyester composites reinforced with twill-woven sisal fibers treated with three surface modification approaches: NaOH, NaOH-KMnO₄, and NaOH-NaClO₂. The composites were fabricated using the vacuum bagging technique, which ensures good fiber wetting and consolidation. The study evaluates water absorption, mechanical, and thermal properties, with emphasis on the effects of saline immersion and temperature, providing insights into fiber-matrix interactions and long-term durability relevant for maritime applications.
By focusing on woven sisal fabrics, comparing dual chemical treatments, and simulating realistic saline exposure, this work presents a clear advancement over existing literature, offering both scientific and practical contributions.
Experimental
The Agave sisalana plant shown in Figure 1 was collected in the southern region of Marrakech, Morocco. Sodium hydroxide (NaOH, 99%) and acetic acid (CH₃COOH, 99.88%) come from SOLVACHIMIE. Sodium chlorite (NaClO2, 80%) came from SIGMA-ALDRICH and potassium permanganate (KMnO4, 98%) was obtained from LOBA CHIMIE.

Agave sisalana plant.
Polyester resin, methyl ethyl ketone peroxide (used as a catalyst), and cobalt (used as an accelerator) were sourced from DETAIL CHEMISTRY. The polyester resin has a viscosity of 640 mPa-s and a density of 1200 kg/m³. In our research, we cured polyester resin with 1.9% methyl ethyl ketone peroxide and 0.25% cobalt.
Fiber treatment
The sisal fiber bundles were mechanically extracted from the Agave sisalana plant through a decortication process, which involves separating the fibers from the plant leaves using mechanical scraping, followed by drying. To enhance the surface roughness of the fiber bundles, remove wax and lignin, and improve adhesion with the polymer matrix, the fibers were subjected to an alkaline treatment using a 0.5% NaOH solution at 65°C for 40 min. These optimal conditions were determined through a design of experiments presented in our previous study. 33
In the same study, the treatment that exhibited the most significant improvement in composite performance in terms of tensile strength and water absorption involved a combination of NaOH and KMnO₄-acetone solution at a concentration of 0.09%, applied for 15 min at 40°C. The second most effective treatment used a NaClO₂ solution at 1.5% concentration, applied for 15 min at 40°C. Following these treatments, the fibers were thoroughly rinsed, neutralized with acetic acid to remove residual chemicals, and carefully washed. Finally, the fibers were dried at 105°C for 6 h in a Thermo Fisher Scientific oven to ensure complete moisture removal.
Table 1 provides detailed images of the fibers before and after the treatments, highlighting changes in surface morphology and texture. The fiber diameter was measured using a GT-B17B 2018 fiber fineness tester microscope equipped with a 506226 Hi-Plan 4x/0.1 objective. This visual analysis supports the improvements achieved through the optimized processes. 33
Microscopic images of sisal fibers before and after various surface treatments.

Table 2 presents the influence of the treatments on water absorption and tensile strength of the fibers. Water absorption was determined according to ASTM D570. Tensile strength was measured using a Hounsfield universal testing machine equipped with a 500 N load cell, a crosshead speed of 0.1 mm/min, and a gage length of 50 minmm, following ASTM C1557. 33
Mechanical properties of untreated and treated sisal fiber. 33

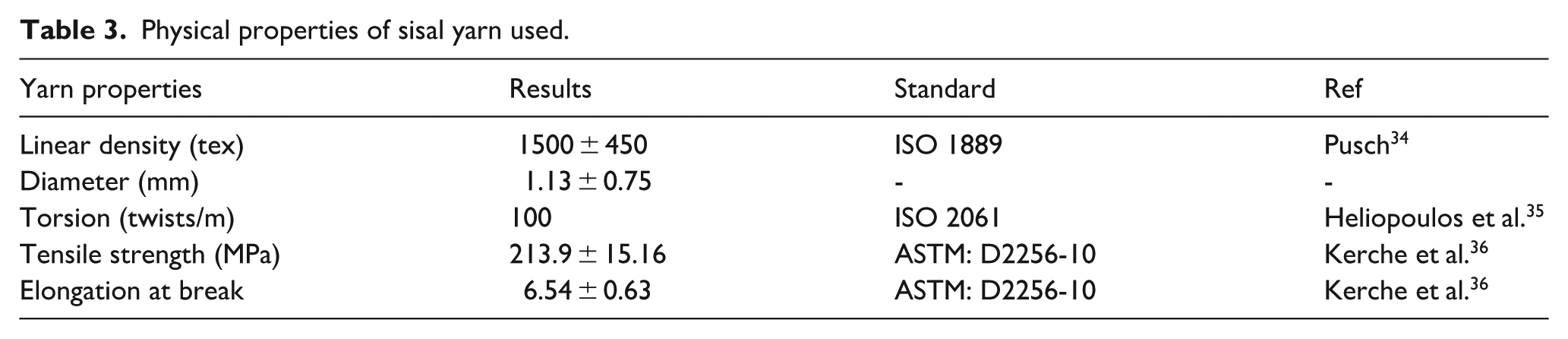
Yarn properties
The sisal yarn was produced by twisting fibers, following the method described in our previous study. 33 Using a TWISTER 3373 MESDAN-type twisting machine (Figure 2), several fibers of 50 cm ± 25 cm were twisted at 100 rpm in the vertical (Z) direction. The physical properties of the sisal yarn used are consistent to standards (see Table 3).

Twisted sisal yarn.
Physical properties of sisal yarn used.
2D woven structures
After chemical y of the yarns, the structure studied is the 2/2 twill weave, which is a derivative of the plain weave. In this weaving process, each weft yarn passes alternately under and over two warp yarns, creating a characteristic diagonal pattern. It is a variant in which the weft yarns are twisted before being interwoven with the warp yarns, producing a wavy effect in the finished fabric. The 2/2 twill fabric, although rarely used, exhibits promising mechanical properties for applications requiring durability and resistance.
Table 4 shows the mechanical and physical properties of the 2/2 twill structure. After weaving, the fabrics were cold calendared at a pressure of 10 kN to flatten and smooth the fabric and to produce composites with low density.
Properties of 2/2 twill structures.

Preparation of composites
Composites were manufactured using the vacuum bag technique as shown in Figure 3 under a pressure of 500 bar. The mold with a size of 20 cm × 20 cm and was coated with wax beforehand to prevent the composite from sticking to it. Subsequently, the fabrics were placed in one layer inside the mold, and a plastic film was placed on top to create an airtight envelope. A vacuum pump was used to evacuate the air, thus creating the required vacuum. Following this, resin was injected into the vacuum system. The fibers began to absorb the resin until saturation. Finally, the composites were left to cure for 8 h at room temperature.

Vacuum bag technique with samples.
Table 5 shows the abbreviations of the different samples, a specific nomenclature was adopted to facilitate the identification of the different materials. The term PES refers to the polyester resin used as a 100% pure matrix. The abbreviation CUN designates the untreated composite reinforced with sisal fabric, which serves as a baseline. When the fabric was subjected to a 0.5% NaOH alkaline treatment, the resulting composite was denoted as CTAL. A further modification combining the same alkaline treatment with an additional 0.09% KMnO₄ oxidation step was coded as CTALK. Finally, the composite reinforced with fabric treated with 0.5% NaOH followed by a 1.5% NaClO₂ bleaching process was referred to as CTALC. This coding system was systematically applied throughout the manuscript to ensure clarity in the presentation and discussion of results.
The abbreviations of the different samples.

In this work, pure polyester (PES) was included as a reference material representing the polymer matrix alone. Since it contains no reinforcing fibers, its mechanical behavior is not directly comparable to the treated or untreated composites. PES serves primarily to provide a baseline, allowing the effect of sisal fabric reinforcement and chemical surface treatments to be clearly assessed.
Characterization of composites materials
Water absorption
Water absorption tests on sisal-PES composites were conducted in accordance with ASTM D570-98 standard, with each sample being repeated five times. The dimensions of the samples were 25.4 mm × 76.2 mm × 10 mm. 41 All samples were pre-dried at 105°C for 1 h until weight stabilization, using a 4% NaCl solution at 18°C and at 40°C. After 24 h of immersion, the samples were carefully removed, dried with a dry cloth, and then reweighed. The samples were regularly weighed over a period of 24, 48, and 72 h, and so on, until 888 h had elapsed, at which point the water content reached saturation. The samples immersed in a 4% NaCl solution were periodically recorded on a balance with a precision of 0.001 g. Water absorption was determined according to the following equation (1) 42 :

Mt is the water absorption rate, Wi is the initial weight of the sample, and We represents the weight of sample after immersion in water.
The diffusion coefficient D was calculated using Fick’s law, based on the linear portion corresponding to the relationship between the water absorption rate Mt and the square root of time t1/2 using the following equation:

The Mt, which represents the water absorption rate of composites, can be determined by two parameters: the maximum water content (Mm) and the diffusion coefficient (D). The following equation illustrates the relationship between these parameters.

T represents the time, Mm represents the maximum water content of the sample, Dt represents the diffusivity, and h represents the thickness of the composite.
Although polymer–fiber composites exhibit heterogeneous microstructures (fiber lumens, interfacial gaps, and microcracks), the initial water uptake stage of the tested composites followed a nearly linear relationship with t1/2, which is characteristic of Fickian diffusion. Similar assumptions have been reported for natural fiber composites in recent studies.43,44 Therefore, Fick’s law was considered a valid approximation to extract effective diffusion coefficients and maximum water contents. It is acknowledged, however, that deviations from ideal Fickian behavior may occur at later stages due to fiber swelling and interface debonding.
The samples were immersed in a 4% NaCl solution for 37 days at 18°C and 40°C to investigate the effects of water immersion and temperature on the tensile, flexural, and impact properties of the composites. After immersion, the samples were dried in an oven at 105°C for 1 h until their weight stabilized to evaluate the post-immersion properties.
Mechanical properties
The tensile properties were assessed using a Universal Testing Machine GT-C01-3, with a 50 kN capacity cell and a speed of 2 mm/min, in accordance with the UNE-EN ISO 527-4:1997 standard. This test is crucial for determining the tensile strength and elasticity of the composites, providing information on their behavior under stress. For each treatment, a total of 10 samples, with dimensions of (250 mm × 25 mm × 10 mm), were cut and tested. 45
The flexural tests were carried out in accordance with the ISO 14125 standard to determine the flexural strength of Sisal/PES composites using the three-point bending method, with a crosshead speed of 1 mm/min. This test is essential for evaluating the materials’ ability to resist bending forces without deformation or failure. The results were obtained from 10 samples with dimensions of (80 mm × 15 mm × 10 mm), using a Universal Testing Machine GT-C01-3 with a 50 kN capacity. 46
The Charpy impact test was conducted in accordance with the NF ISO 179 standard using a motorized pendulum impact tester, INSTRON CEAST 9050. The initial impact energy of the pendulum was 25 J. This test measures the resistance of the composites to impact and sudden loads, which is a key parameter for assessing their durability and performance under extreme conditions. Five samples with dimensions of (60 mm × 15 mm) were tested. 47 The test specimens were placed horizontally on two supports, spaced 62 mm apart. The absorbed surface energy of the specimen was precisely determined based on the movement of the pendulum arm using the following equation:

With:
E = energy absorbed by the specimen (kJ/m²)
m = mass of the specimen (g)
L = length of the pendulum hammer (m)
g = gravitational acceleration (9.8 m/s²)
α = angle reached by the pendulum arm after impact.
Statistical analysis
To evaluate the influence of chemical treatments and immersion conditions on the mechanical performance of polyester composites reinforced with sisal fibers, statistical analyses were performed. Data are expressed as mean ± standard deviation (n = 5). Tensile, flexural, and impact properties were assessed using a two-way ANOVA with Treatment (UNTREATED, NaOH, NaOH +KMnO₄ 0.09%, NaOH +NaClO₂ 1.5%) and Condition (Before immersion, Immersed at 18°C, Immersed at 40°C) as fixed factors, including their interaction. Statistically significant differences are indicated by different superscript letters. This approach has been successfully applied in recent studies on natural fiber-reinforced polymer composites to determine the effects of chemical treatment and environmental exposure on mechanical performance. 48
SEM
The surface of untreated and treated composites was cut and analyzed using a scanning electron microscope (SEM, JEOL JCM 6000 Therm Fisher Scientific, Waltham, MA, USA, at a voltage of 15 kV with a spot size of 56).
TGA
The thermal decomposition of the composites sisal-PES was investigated using a thermogravimetric analysis (TGA) apparatus, specifically the Cahn Versa Therm Instruments. The analysis was performed in a nitrogen atmosphere with a purge flow rate of 10 ml/min and a heating rate of 10°C/min. The temperature range for the analysis extended from 32°C to 700°C.
Results
Water absorption behavior
Figure 4(a) and (b) illustrate the water absorption (Mt) of the composites over time in a 4% NaCl solution, at temperatures of 18°C and 40°C respectively.
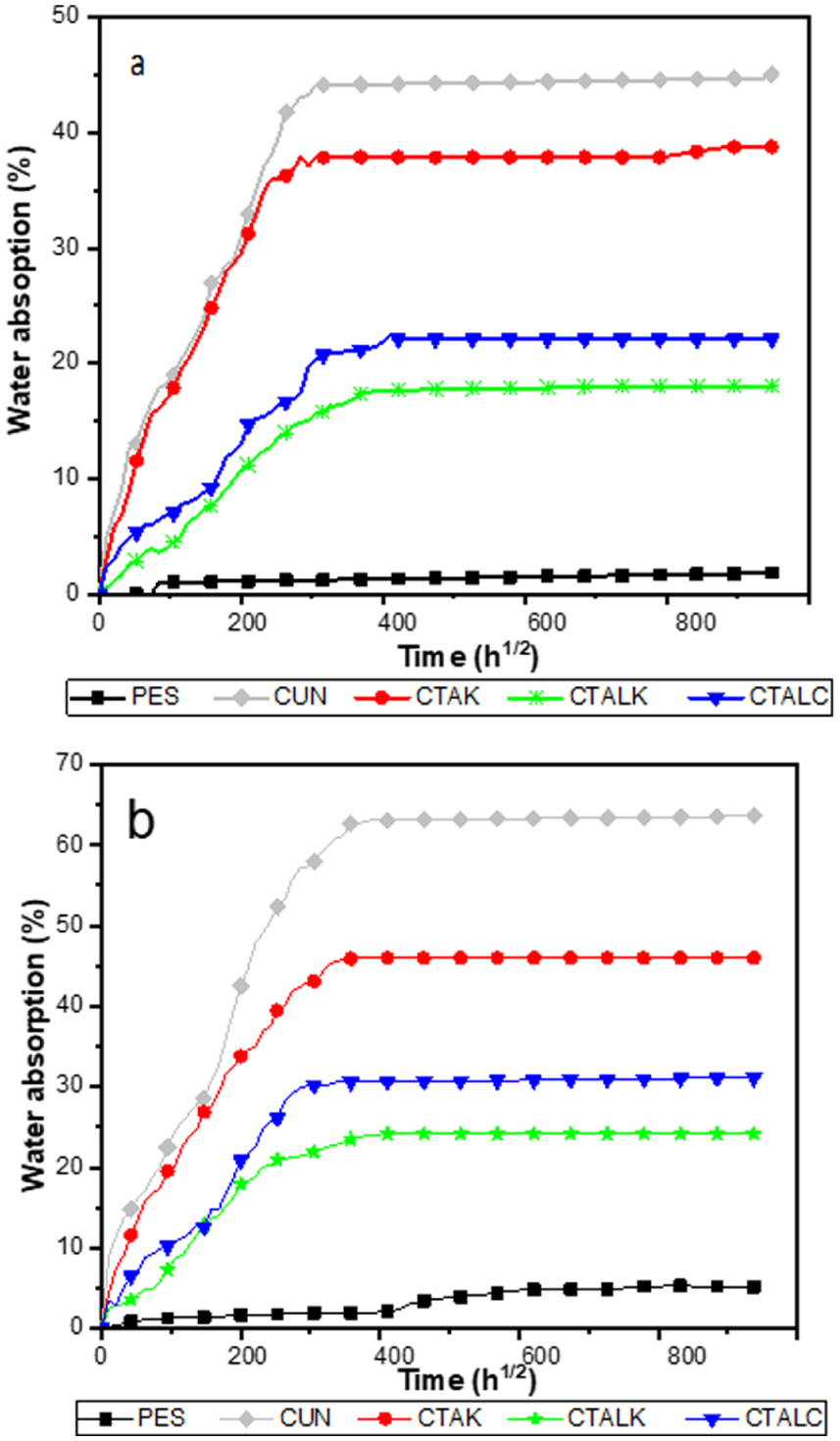
Water absorption curves of treated and untreated sisal fabric composite: (a) at 18°C and (b) at 40°C.
Table 6 displays the maximum water content (Mm) and diffusivity (D). It is clear that the values of Mm and D of the composites decrease after the various treatments carried out.
Maximum water content (Mm) and diffusivity of composites at 18°C and 40°C.

At 40°C, the composites reach saturation more quickly than at 18°C, demonstrating the strong impact of immersion temperature on diffusion kinetics. Weight gain during immersion at 40°C is attributed to accelerated water diffusion, leading to microcrack development and hydrolytic degradation of PES chains. Specifically, hydrolysis involves the cleavage of ester bonds in the polyester backbone, forming carboxylic acid and hydroxyl end groups, which reduce chain length and compromise mechanical integrity, weakening the fiber-matrix interface.49–51
These findings align with recent studies showing that elevated temperatures accelerate water uptake and interfacial degradation in natural fiber-reinforced polyester composites.52,53 In a 2024 study, Kumari et al. 54 observed that increasing immersion temperature led to faster water diffusion and greater mechanical degradation in natural fiber-reinforced composites, confirming the thermal impact on composite durability. Additionally, Sari and Sutjea 55 demonstrated that water absorption at 40°C accelerates hydrolytic degradation of polyester, reducing stiffness and mechanical strength of composites, highlighting the importance of temperature in long-term composite performance.
These findings further emphasize that dual chemical treatments (NaOH + KMnO₄) not only improve interfacial adhesion but also limit hydrolytic cleavage of polyester chains by reducing water pathways and enhancing fiber surface hydrophobicity, thereby mitigating molecular-level degradation.
These findings align with recent research indicating that elevated temperatures accelerate water uptake and interfacial degradation in natural fiber-reinforced polyester composites. For instance, Pavlovic et al. 10 reported that moisture absorption can cause reductions in tensile strength of up to 40% and in elastic modulus by 20%–30%, depending on fiber type, mass fraction, and surface treatments. Similarly, Mahdi et al. 56 observed that water absorption led to a reduction in tensile strength and modulus of DPLFRP composites by 39%–47% and 21%–32%, respectively.
Overall, the CTALK composites demonstrate the highest resistance to water absorption, confirming that the dual chemical treatment effectively enhances interfacial adhesion and hydrophobicity, thereby improving the long-term durability of sisal-reinforced polyester composites.
Effect of water absorption in mechanical properties
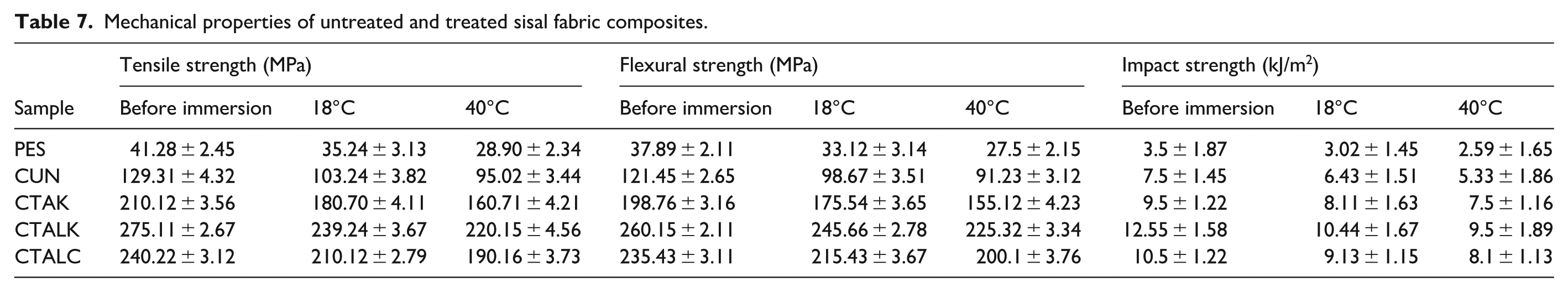
Table 7 evaluates the influence of temperature on the mechanical properties, including tensile, flexural, and impact resistance. Figures 5 –7 display the tensile strength, the flexural strength and the impact strength, respectively, of composites before and after immersion at 18°C and 40°C.
Mechanical properties of untreated and treated sisal fabric composites.

Tensile strength of composites before and after immersion at 18°C and 40°C.

Flexural strength of composites before and after immersion at 18°C and 40°C.

Impact strength of composite before and after immersion at 18°C and 40°C.
The tensile strength of composites reinforced with fabrics treated using alkaline and KMnO₄ methods increased by from 129.31 to 275.11 MPa. This represents the highest tensile strength observed across all treatments. The enhancement can be attributed to the formation of permanganate ions (Mn³⁺), which react with the -OH groups of cellulose, forming cellulose-manganate. This process significantly enhances adhesion between the fiber and the matrix. These findings are consistent with earlier results, confirming that fibers alkalized and treated with 0.09% KMnO₄ exhibit superior tensile strength. 33
However, the tensile strength of all composites decreases after immersion at 18°C and 40°C as shown in Figure 5. This reduction is primarily due to the development of micro-cracks between the fiber and the matrix, which results from fiber swelling after water absorption. The detachment of fibers from the matrix, combined with the hydrolysis of the polyester (PES) matrix caused by water absorption, further reduces tensile strength. Hydrolysis leads to the breaking of macromolecular chains in the polyester resin. These findings align with those of various researchers.57–59
Table 7 also highlights that PES composites immersed at 40°C exhibit the lowest tensile strength. This reduction is likely due to the higher rate of water absorption, which exacerbates damage at the fiber-matrix interface.
An increasing trend in flexural strength was observed for the CTALK and CTALC samples as shown in Figure 6. Flexural strength follows the order: PES < CUN < CTAL < CTALN < CTALK. Untreated fibers demonstrated the lowest flexural strength, measured at 121.45 ± 2.65 MPa. This outcome is attributed to the weak bonding at the fiber-matrix interface and the limited stress transfer capability in untreated composites.
Composites reinforced with fibers treated with a combination of 0.5% NaOH and KMnO₄ exhibited the highest average flexural strength, reaching 260.15 ± 2.11 MPa. This was followed by composites reinforced with fibers treated with 0.5% NaOH and 1.5% NaClO₂, which achieved 235.43 ± 3.11 MPa. These results can be explained by the enhanced stress transfer mechanism between the PES matrix and the treated sisal fibers. 60
After immersion in water, all composites experienced a decline in flexural strength. The composites immersed at 40°C showed lower strength compared to those immersed at 18°C, indicating that higher temperatures intensify water absorption. This behavior is attributed to the formation of hydrogen bonds between water molecules and the hydroxyl groups of cellulose, leading to reduced water resistance. Thus, it can be concluded that water absorption adversely affects the flexural properties of composites. These results are consistent with the tensile strength trends depicted in Figure 5.
These findings align with those of Wang et al., 61 who reported that moisture absorption leads to a significant reduction in the mechanical properties of plant fiber-reinforced composites, including tensile and flexural strength, due to fiber swelling and degradation of the fiber-matrix interface.
Figure 7 clearly illustrates a notable increase in the impact resistance of the composites following all treatments. These findings align closely with the previously discussed tensile and flexural strength results, reinforcing the conclusion that the combination of NaOH-KMnO₄ and NaOH-NaClO₂ treatments significantly enhances the mechanical properties of PES/sisal fabric composites. Although a slight improvement in impact resistance can be attributed to better interfacial adhesion for the treated composites with NaOH, however, it is not enough to improve the adhesion between sisal fiber and polyester matrix. The impact resistance follows this increasing order: PES < CUN < CTAL < CTALN < CTALK with the alkaline-KMnO₄ treatment giving the best interfacial adhesion.
The improvement in impact resistance can be attributed to a slightly better interfacial adhesion between the treated fibers and the polyester matrix.
The untreated composite (CUN) exhibited an impact resistance of 7.5 kJ/m², which rose markedly to 12.55 kJ/m² after the alkaline-KMnO₄ treatment. This improvement underscores the effectiveness of the chemical treatments in enhancing the composite’s durability.
However, the figure also indicates a decline in impact resistance after aging, highlighting that water absorption negatively affects the mechanical performance of the composites. This reduction can be attributed to the degradation caused by water infiltration, which weakens the fiber-matrix interface and compromises the structural integrity of the composites. 62 Untreated composites experienced more pronounced degradation compared to their treated counterparts, likely due to the greater vulnerability of the PES matrix and untreated sisal fibers to water-induced damage. 63
The reduction in impact resistance was particularly evident in composites immersed at 40°C, which absorbed significantly more water than those immersed at 18°C. Conversely, composites treated with NaOH-KMnO₄ and NaOH-NaClO₂ and immersed at 18°C for 37 days exhibited minimal changes in impact resistance compared to untreated composites, indicating the effectiveness of these treatments in mitigating water-related degradation.
These results are further supported by previous studies. For example, Acharya et al. 64 demonstrated that chemical treatments significantly enhance fiber–matrix adhesion, thereby reducing moisture absorption and improving tensile, flexural, and impact properties in sisal composites. Similarly, Prome et al. 65 reported that natural fiber reinforced polyester composites exhibit markedly improved mechanical performance as a result of enhanced interfacial bonding, particularly at higher fiber loadings.
Statistical analysis
A two-way Analysis of Variance (ANOVA) was conducted to assess the influence of chemical treatments and exposure conditions on the mechanical properties of sisal fiber-reinforced composites. The results are summarized in Table 8 below:
Two-way ANOVA of mechanical performance of sisal fiber composites under different chemical treatments and immersion conditions.

p < 0.001: Hautement significative.
The application of chemical treatments to sisal fibers markedly improved the mechanical performance of polyester composites, including tensile, flexural, and impact strengths. This enhancement is likely due to surface modifications that strengthen fiber–matrix adhesion and reduce internal moisture and porosity.66–68
Immersion conditions, such as exposure to water at different temperatures, also affected the mechanical behavior. Environmental factors, including moisture and temperature, can influence the durability and performance of natural fiber composites. 69
A significant interaction between treatment and immersion condition was observed, indicating that the effectiveness of chemical treatments depends on environmental conditions. Composites CTAK, CTALK, and CTALC retained higher mechanical performance after immersion than untreated composites, emphasizing the importance of selecting appropriate treatments for specific applications. 66
Scanning electron microscopy studies
Figure 8 illustrates the microstructural features of sisal fiber-reinforced polyester composites under different chemical treatments and provides insight into fiber-matrix interfacial adhesion where Figure 8(a) shows a clear gap between the outer wall of the untreated fiber and the polyester matrix indicating poor bonding which can lead to stress concentration and reduced mechanical performance as reported in untreated natural fiber composites. 70

SEM micrographs of fractured sisal fabric composites showing fiber–matrix interfacial behavior at 100× magnification: (a) clear fiber pull-out evidencing weak interfacial adhesion, (b) voids and gaps created after fiber detachment, (c) partial fiber debonding with matrix residues attached to the fiber surface, and (d) longitudinal pull-out of fibers indicating interfacial failure mechanism.
Figure 8(b) reveals holes and voids resulting from fiber pull-out demonstrating that treatment with NaOH alone is insufficient to improve adhesion between the fiber and matrix with incomplete wetting and weak bonding consistent with previous findings on single-alkali-treated fibers 71 Figure 8(c) presents fibers fully embedded in the polyester matrix with resin coating the fiber surfaces and no visible pull-out suggesting that the dual NaOH-KMnO₄ treatment effectively removes surface impurities and enhances fiber roughness thereby promoting strong interfacial adhesion and correlating with the enhanced mechanical properties observed in tensile and impact tests 72 Figure 8(d) shows a reduced gap between fiber and matrix after NaOH-NaClO₂ treatment with increased cellulose exposure on the fiber surface indicating improved chemical interactions between the sisal fibers and the polyester matrix which contributes to better adhesion and overall composite performance consistent with recent studies on dual chemical treatments 73 Overall the SEM analysis confirms that dual chemical treatments markedly improve fiber-matrix interfacial bonding compared to untreated or singly treated fibers and these microstructural improvements provide a clear explanation for the observed enhancements in mechanical strength thermal stability and resistance to fiber pull-out highlighting the effectiveness of the proposed treatments for high-performance polyester composites.
Thermal stability
Figure 9 shows the thermogravimetric curves for untreated and chemically treated PES/sisal composites. Two distinct stages of degradation are observed. The first stage, occurring between 29°C and 140°C, corresponds to the evaporation of physically bound water. The second stage, between 140°C and 400°C, is associated with the decomposition of hemicellulose, cellulose, and lignin. According to previous studies, hemicellulose degrades between 200°C and 310°C, cellulose between 230°C and 350°C, and lignin over a broader range of 160°C–700°C due to its aromatic nature.74,75
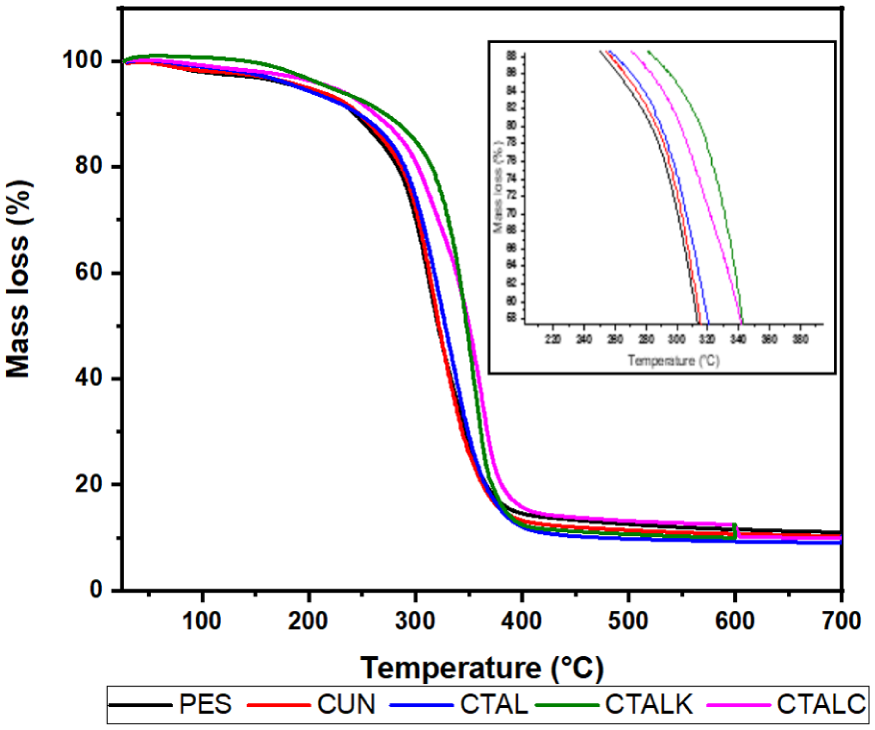
TGA analysis of untreated and treated composites.
Table 9 shows the residual mass at selected temperatures. The PES, CUN, and CTAL composites display similar decomposition behavior, indicating that mild alkali treatment does not significantly alter the thermal stability of sisal fibers. In contrast, the CTALK composite shows a delayed onset of weight loss and the highest residual mass at 600°C (12.46%), suggesting superior thermal stability.
Residual mass percentage of untreated and treated composites.

This improvement can be attributed to the formation of cellulose manganate during KMnO₄ treatment, which promotes char formation and enhances thermal resistance, as reported by Abisha et al. 62 Similarly, CTALC composites retain more mass than untreated samples, which may be due to the selective removal of hemicellulose and partial delignification induced by NaClO₂, leading to a more compact fiber structure and better fiber–matrix adhesion. 60
These observations highlight that oxidative treatments (KMnO₄ and NaClO₂) improve the thermal stability of sisal/polyester composites compared to untreated or solely alkali-treated fibers. This trend is in agreement with other natural fiber composites, such as flax and jute systems, which have shown increased char yield and delayed degradation after chemical modification or partial delignification 76
In those studies, chemically treated bast fibers exhibited not only higher decomposition onset temperatures but also greater residual char, confirming that surface oxidation and lignin modification enhance interfacial bonding and the formation of thermally stable residues. 77
Conclusion
This study investigated the effects of KMnO₄ and NaClO₂ surface treatments, activated by NaOH in fabric form, on the performance of thermosetting unsaturated polyester composites reinforced with sisal fibers. The results demonstrated that untreated composites exhibited higher moisture content (Mm) and diffusivity (D) compared to treated ones, confirming that chemical modification enhances resistance to water absorption.
Mechanical performance was also significantly improved by surface treatments. The composite treated with alkaline-0.09% KMnO₄ followed by alkaline-1.5% NaClO₂ achieved the highest tensile strength, while the NaOH–KMnO₄ combination provided superior flexural and impact resistance.
Immersion in a 4% NaCl solution at 40°C led to a marked reduction in tensile, flexural, and impact strengths compared with immersion at 18°C, underlining the sensitivity of the composites to elevated saline conditions. This finding emphasizes the need for protective strategies to mitigate performance degradation in harsh marine environments.
Overall, these results highlight the potential of chemically tailored sisal fibers to produce durable and sustainable composites. As a renewable and biodegradable resource, sisal also supports global efforts to reduce reliance on synthetic fibers and to promote eco-friendly alternatives.
Looking ahead, future studies should prioritize long-term seawater immersion trials, fatigue and cyclic loading evaluations, and eco-toxicological assessments to better predict service life and environmental impact. Furthermore, research on scaling up fiber treatment and composite fabrication while ensuring cost-effectiveness and environmental compatibility will be crucial for industrial implementation. Taken together, these directions will consolidate the role of natural fiber–reinforced composites as credible and sustainable alternatives to conventional materials in marine and other demanding applications.
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
This article does not contain any studies with human or animal participants.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
