Abstract
Nanomaterials have created massive opening on textile functionalization such as antimicrobial and sun protection activities of fabrics. The focal point of this paper is to impart antimicrobial and UV protection activities of textile substrates using a nanocomposite hybrid material chitosan with α-iron oxide. α-Fe2O3 nanoparticles were synthesized by self-assembly method and characterized. The average particle size was found to be 27–30 nm by X-ray diffractogram and atomic force microscopic analyses. The α-Fe2O3 nanoparticles were dispersed into the prepared chitosan solution to form the hybrid material. The chitosan-α-Fe2O3 nanocomposite thus formed was subjected to characterizations such as X-ray diffractogram, FTIR and SEM with EDX. The chitosan-α-Fe2O3 nanocomposite was dip-coated on cotton and silk fabrics and antibacterial activity was checked by zone of inhibition method (AATCC 147) against Staphylococcus aureus and Escherichia coli bacteria and the UV protection activity was analyzed using UV-DRS spectroscopy. Results revealed that the hybrid CH-α-Fe2O3 nanocomposite possess improved antibacterial as well as UV absorbing efficiency.
Introduction
Cotton has excellent moisture absorption ability. However, the moist cotton can be easily attacked by bacteria. Decomposed products of body secretions have a characteristic odor [1]. Silk has been used in textile industries for many centuries due to its superior properties. The properties of silk and its uses have attracted great attention worldwide [2]. The application of nanoparticles to textile materials has attracted considerable interest due to their novel physicochemical properties and applications. TiO2, ZnO and Fe2O3 nanoparticles were applied on textile substrates to achieve self-cleaning, abrasion resistance, water repellency, UV resistance, electromagnetic protection and infrared protection properties [3–7]. The iron oxide nanoparticles have attracted researchers from various fields such as physics, biology and material science due to their multifunctional properties such as superparamagnetism and low toxicity [8–10]. Iron oxide has been used in various medical fields such as protein and enzyme immobilization, bioseparation, immunoassay, drug delivery and magnetic resonance imaging [11,12]. The iron oxide nanoparticles coated with polymers are usually composed of the magnetic cores to ensure a strong magnetic response and a polymeric shell to provide favorable functional groups and features [13]. Synthetic polymeric coating materials include poly(ethylene-co-vinyl acetate), poly(vinylpyrrolidone)(PVP), poly(lactic-co-glycolic acid)(PLGA), poly(ethylene glycol)(PEG), polyvinyl alcohol(PVA) etc. Natural polymer systems include gelatin, dextran, chitosan (CH), etc. The modification of natural polymer offers significant advantages in biomedicine application due to their good biocompatibility and degradability [14,15]. Most of the metal oxide nanoparticles are toxic or poorly effective owing to their fast kinetics, which makes them not directly suitable for applications in medicine, filters and textiles. It has become necessary to get biocompatibility of metal nanoparticles using biopolymers [16].
Chitosan (CH) is a linear polymer of α(1→4)-linked 2-amino-2-deoxy-β-D-glucopyranose and is easily derived by N-deacetylation, to a varying extent that is characterized by the degree of deacetylation, and is consequently a copolymer of N-acetylglucosamine and glucosamine.
The amino group in CH has a pKa value of ∼6.5, which leads to a protonation in acidic to neutral solution with a charge density dependent on pH and the %DA-value. This makes CH water soluble and a bioadhesive which readily binds to negatively charged surfaces such as mucosal membranes. CH enhances the transport of polar drugs across epithelial surfaces and is biocompatible and biodegradable. Purified quantities of CHs are available for biomedical applications.
Due to its physical and chemical properties, CH is being used in widely different products and applications, ranging from pharmaceutical and cosmetic products to water treatment and plant protection. In different applications, different properties of CH are required. Industrial applications include cosmetics, water engineering, paper industry, textile industry, food processing, agriculture, photography, chromatographic separations and solid-state batteries. Biomedical applications include tissue engineering, wound healing, burn treatment, artificial skin, ophthalmology and drug delivery systems.
CH along with iron oxide nanoparticles has been utilized as a stabilizing agent due to its excellent film-forming ability, mechanical strength, biocompatibility, non-toxicity, high permeability towards water, susceptibility to chemical modifications and antibacterial activity. Moreover, amino groups of CH provide a hydrophilic environment compatible with the biomolecules. Many attempts have been made to improve the biocompatibility and activity of the CH by structural modification by fabrication of nanocomposites with metal oxide nanoparticles, etc. [17–29]. In the present work, CH-iron oxide composite was synthesised and coated on cotton and silk fabrics for imparting antibacterial and UV resistance properties.
Experimental
Chemicals such as ferric chloride (FeCl3), urea (CH4N2O), tetra-n-butylammonium bromide (C16H36NBr), ethylene glycol (C2H6O2), potassium hydroxide (KOH), sodium hydroxide (NaOH), zinc chloride (ZnCl2) and ethanol (C2H5OH) were purchased from Merck and used as such without further purification.
Synthesis of iron oxide nanoparticles by self-assembly method
FeCl3 (125 mM), urea (125 mM) and tetra-n-butylammonium bromide (62 mM) were added to 100 mL ethylene glycol in a conical flask. The red solution thus obtained was stirred with a magnetic stirrer and heated at 200℃ for 1 h. After cooling, the as-synthesized iron oxide precursor was collected as a green precipitate (α-Fe2O3). The α-Fe2O3 precipitate thus formed was washed with ethanol four times and dried using hot oven at 300℃ for 4 h [30].
Preparation of CH
For the preparation of chitin, the crab shells were obtained from the coastal water of Nagapattinam town. The shells were washed well with sea water followed by wash in fresh water, dried and then powdered using mortar. About 50 g of powdered shell was taken in a 500-mL beaker. A total of 250 mL of 5% HCl was added to remove the calcium carbonate present in the shell. The mixture was allowed to stand for about 2 h. It was then filtered in a muslin cloth and the residue was transferred into a 500-mL beaker; 250 mL of 5% NaOH was added to it slowly to remove the protein present. The mixture was then allowed to stand for about 3 h and filtered through muslin cloth to get chitin. In all, 2 g of this chitin was added to 60% anhydrous ZnCl2 and heated in boiling water bath for 30 min. The mixture was dissolved in dilute acetic acid and filtered to remove the unreacted chitin and other impurities. CH was precipitated from the filtrate using 20% NaOH solution, filtered and then air-dried.
Coating of fabric
The iron oxide, CH and CH-iron oxide composites were coated separately on both cotton and silk fabrics by dip-coat method. For this, 50 mg/L of iron oxide nanoparticle was taken by diluting it with distilled water. The test fabric was immersed in to the solution and kept for 10 min. Then fabric was taken out, washed with water and then air-dried. This procedure was repeated with CH and chitosan-iron oxide composite (3 : 1 ratio) solutions.
Characterization
The synthesized materials were characterized by X-ray diffractogram (XRD), FTIR, SEM, atomic force microscopic (AFM), techniques. X'Pert PRO diffractrometer instrument was used for studying crystallography of the resultant. Shimadzu 8400 FTIR instrument was used for functional group analysis. Hitachi S3000H Scanning Electron Microscope operating at an accelerating voltage of 20 kV was used for SEM imaging. Veeco II AFM instrument was used to observe the 3D morphology of the iron oxide nanoparticles. The CH-iron oxide composite-coated fabrics were characterized by XRD and SEM. The coated fabrics were recorded using UV-Vis diffuse reflection spectra (DRS) with Shimadzu UV-3101PC spectrophotometer equipped with an integrating sphere and barium sulphate as the reference.
AATCC 147(qualitative test)
The finished materials were tested for antibacterial activity by AATCC 147 standard method. Sterile AATCC bacteriostasis agar medium was dispensed into the sterile Petri dishes. Overnight culture was used as an inoculum by using sterile swab. The test organism was inoculated over the surface of the agar plate and gently pressed in the centre of the Mat culture. The plates were then incubated overnight at 37℃ for antibacterial activity against E. coli and S. aureus.
Results and discussion
Characterization
The representative XRD of α-iron oxide displaying the structural information and crystallinity are shown in Figure 2(a). Diffraction peaks of 2θ at 24.8°, 33.3°, 35.6°, 39.3°, 43.1°, 54.1°, 56.4°, 62.4°, 64.7° and 72.6° are seen. The d-space values of these main peaks were 3.68, 2.69, 2.51, 2.29, 2.07, 1.69, 1.63, 1.48, 1.45A°, which correspond to h k l planes of 012, 104, 110, 006, 202, 116, 211, 214, 300 and 119, respectively. This data attributes the formation of α–Fe2O3 nanoparticle [JCPDS 80-2377]. The grain size was calculated using Scherrer formula and found to be 27 nm. In Figure 2(b), the main diffraction 2θ peak at 20° was observed for CH [18]. Figure 2(c) and (d) shows peak at around 20° that corresponds to fabrics such as cotton and silk. The dispersed α-Fe2O3-CH composites are perfectly embedded on textile matrix. The XRD patterns displayed are consistent with earlier reports on this composite. This is the advantage of this study for better bio-compatibility of synthesized nanocomposite [16].
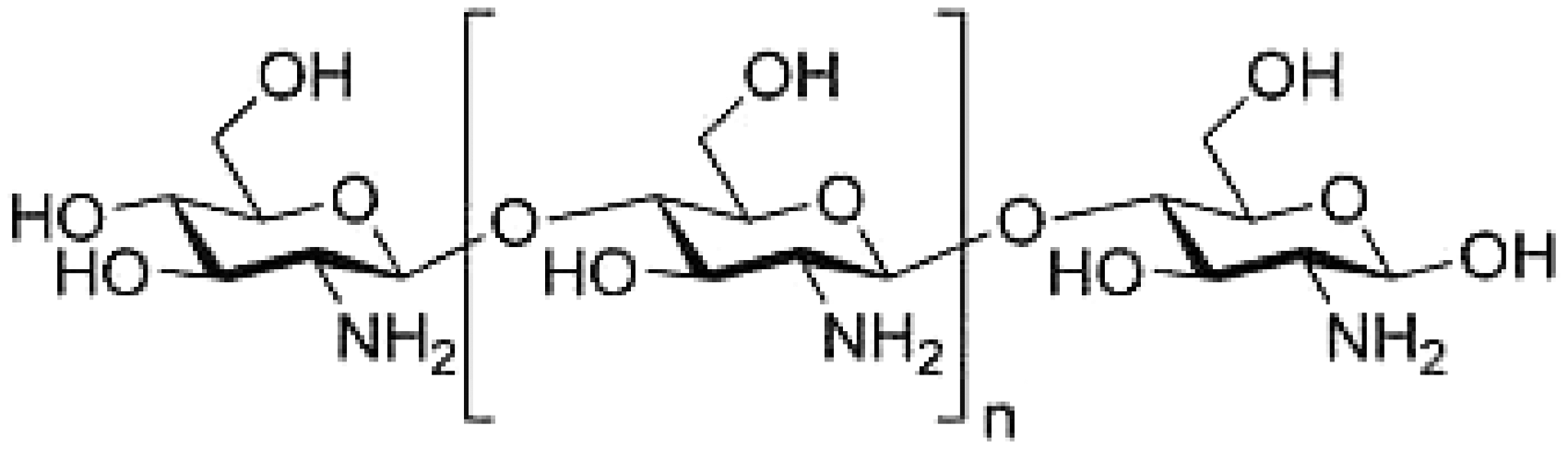
Structure of chitosan.

X-ray diffractogram (XRD) patterns of (a) α-Fe2O3 nanoparticles, (b) chitosan, (c) CH-α-Fe2O3-coated cotton, (d) CH-α-Fe2O3-coated silk.
FTIR spectra of CH, CH-α-Fe2O3 nanocomposite and α-Fe2O3 nanoparticles (Figure 3) show the peak around 3400 cm−1, which is responsible for the presence of −OH group. FTIR spectrum of CH (image a) shows the band of 1596 cm−1 for N–H bending vibration and the peak of 1384 cm−1 for C–O stretching of primary alcoholic group. FTIR spectrum of Fe2O3–CH composite (image c) exhibits characteristic bands of the functional groups corresponding to CH and Fe2O3 nanoparticles [31,32]. Iron oxide particles exhibit strong bands in the low frequency region at 676 cm−1 [33] and could be seen from figure that all the characteristic peaks of CH were present in the spectrum of Fe2O3–CH nanocomposite. The FTIR spectra showed that CH was covered entirely on Fe2O3 nanoparticles [34].
FTIR of (a) chitosan (b) CH-α-Fe2O3 composite (c) α-Fe2O3 nanoparticles.
The SEM image of synthesized Fe2O3 is shown in Figure 4(a), which reveals the presence of agglomerated spherical nanoparticles. The porous film of CH contains pin holes as shown in Figure 4(b). The Fe2O3 nanoparticles are dispersed uniformly within the porous network of CH (Figure 4(c)). From the EDX analysis, we confirm the presence of α-Fe2O3 nanoparticles-CH composites (Figure 4(d)). The AFM image showed spherical shape for the α-Fe2O3 nanoparticles synthesized (Figure 5) and the average particle size was derived as 27 nm.
SEM images of (a) α-Fe2O3 nanoparticles, (b) chitosan, (c) CH-α-Fe2O3 composite, (d) EDX of CH-α-Fe2O3 composite. Atomic force microscopic (AFM) image of α-Fe2O3 nanoparticles.

The SEM images of control and finished fabric sample are shown in Figure 6. The images depict clearly the existence of α-Fe2O3, CH, CH-α-Fe2O3 composite on silk and cotton fabric. It was felt that control samples were less smooth than their finished counterparts [35,36].
SEM images of (a) control silk and coated silk fabrics with (b) α-Fe2O3 nanoparticles, (c) chitosan, (d) CH-α-Fe2O3 composite, (e) control cotton fabric and coated cotton fabrics with (f) α-Fe2O3 nanoparticles, (g) chitosan, (h) CH-α-Fe2O3 composite.
AATCC 147 (qualitative test)
The antibacterial test by zone of inhibition method was performed against E. coli and S. aureus organisms (Figure 7) for α-Fe2O3-coated fabric and CH-α-Fe2O3 composite-coated fabrics [37]. The results showed that the coated fabrics had better inhibitory effect against E. coli and S. aureus (Table 1), when compared to α-Fe2O3-coated fabrics. When the concentration of α-Fe2O3 and CH-α-Fe2O3 composite was increased in coating, the zone of bacterial inhibition also increased accordingly for both cotton and silk fabrics (Figure 8).
Antibacterial test by zone of inhibition method against S. aureus (Gram positive) organism by (a) Cotton with α-Fe2O3 nanoparticles, (b) cotton with CH-α-Fe2O3 composite, (c) silk with α-Fe2O3 nanoparticles, (d) silk with CH-α-Fe2O3 composite, test against E. coli (Gram negative) organism (e) cotton with α-Fe2O3 nanoparticles, (f) cotton with CH-α-Fe2O3 composite (g) silk with α-Fe2O3 nanoparticles, (h) silk with CH-α-Fe2O3 composite. Increasing the concentration of CH-α-Fe2O3 composite tested against E. coli (Gram negative) (a) cotton (b) silk and increasing the concentration of CH-α-Fe2O3 composite tested against S. aureus (c) cotton, (d) silk. Antibacterial assessment by zone of inhibition method.


UV-visible reflectance spectra
Figure 9 shows UV-Vis reflectance spectra of control and finished textiles that were checked between 200 and 800 nm. The UV absorbing efficiency of finished textiles gave superior results when compared to control samples. This may be due to uniform spread of composite materials used for coating as evidenced from the SEM images.
UV-Vis diffusive reflectance spectra of (a) control silk, (b) control cotton, (c) CH-α-Fe2O3 composite-coated silk, (d) CH-α-Fe2O3 composite-coated cotton.
Conclusion
Formation of α-Fe2O3 nanoparticles was confirmed from XRD and AFM results. CH-α-Fe2O3 composite was prepared and dip-coated smoothly on cotton and silk. XRD results depict that composite is incorporated into the cotton and silk. SEM images also confirm the coating of composite. Antibacterial and UV protection effect on coated cotton and silk substrates were successfully developed using the composite CH-α-Fe2O3. From these antibacterial and UV protection results, it is concluded that this novel CH-α-Fe2O3 nanocomposite-coated fabrics could be well exploited for medical and protective textile applications. Cotton and silk textiles were functionalized with low-cost material. It is a new avenue for cost-effective scheme for the design of medical textile products.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
