Abstract
Occupational and environmental exposure to extreme temperatures and harmful UV radiation poses significant health risks for workers in various industries. This study aimed to develop innovative workwear fabrics with enhanced thermal and UV protection properties. A blend of 70% cotton and 30% polyester fabric was coated with SiO2 nanoparticles and silica aerogel. The UV protection factor (UPF) was determined using standard methods (BS EN 13758-1:2002 and AS/NZ 4399:1996). Thermal properties were evaluated using thermogravimetric analysis (TGA) and thermal imaging. The coated fabrics exhibited significantly improved thermal insulation compared to the uncoated control, reducing surface temperatures by up to 7.4°C. Both SiO2 nanoparticle- and silica aerogel-coated fabrics achieved excellent UV protection with UPFs of 39.28 and 38.11, respectively. TGA analysis revealed the thermal stability of the coating materials. The developed workwear fabrics offer promising solutions for mitigating occupational heat stress and UV exposure. These innovative materials have the potential to enhance worker safety and comfort in challenging environments.
Keywords
Introduction
Humans are constantly exposed to radiation from their environment. Numerous studies have investigated the potential pathogenic effects of radiation. While the precise mechanisms by which radiation damages cells, early molecules, or organs are still being researched, it is well-established that radiation poses a significant risk to living organisms. Compared to other environmental factors, the damaging effects of radiation are more extensively understood and documented. 1
Solar thermal energy, primarily within the near-infrared spectrum (700–1100 nm), constitutes 52% of the solar energy reaching Earth. This heat energy is absorbed by the human body, leading to a rise in deep body temperature. This can result in various health complications, including heat-related diseases, thermal stress, and skin diseases. 2 Ultraviolet (UV) radiation, infrared radiation, and thermal radiation, particularly for workers in indoor and outdoor environments, pose significant health risks. These include alterations in skin melanin precursors, disruption of skin color, premature aging, loss of skin elasticity, and most importantly, skin cancer. The incidence of skin cancer has risen dramatically in recent decades. While various factors contribute to the development of melanoma and non-melanoma skin cancers, excessive exposure to UV radiation is a well-established primary cause. 3 and highlights the importance of skin protection against prolonged exposure to ultraviolet radiation.4–6 The atmospheric ozone layer serves as a highly effective absorber of ultraviolet radiation, but due to the increasing consumption of fossil fuels in recent decades, its thickness has been steadily decreasing. Decreasing the thickness of the ozone layer can cause the excessive passage of UV rays from the sun, especially UVB rays.
Protecting the skin from the sun’s harmful thermal and ultraviolet radiation is paramount. While various strategies exist, wearing personal protective clothing (PPE) is a highly effective and cost-efficient approach. PPE prevents skin contact with chemicals and reduces the risk of scratches and cuts. Moreover, it acts as a barrier against peripheral heat and harmful radiation, safeguarding workers’ health. Workwear fabrics are often composed of cotton or cotton-polyester blends, renowned for their excellent thermal conductivity and air permeability, ensuring comfort and breathability in diverse working conditions. 7
Textiles and individual protective clothing play a crucial role in safeguarding worker health and safety. Due to evolving workplace demands and emerging health concerns, there is a persistent need for innovation and improvement in textile materials and their performance. This ongoing pursuit aims to enhance worker comfort, protection, and overall well-being.8,9 Since textiles are easy to ignite, their thermal stability and flame retardant behavior are complex and challenging. 9 Unfortunately, the inherent shortcomings of natural textile fibers often limit their performance in preventing the transmission of ambient thermal radiation.10,11 However, with technological advances, there are efforts to develop textiles and reduce their weaknesses through various means such as covering fabrics with different materials that are safe. 7 Halogen derivatives and phosphorus compounds are known to have detrimental environmental effects, releasing toxic corrosive gases, smoke, and formaldehyde. In contrast, boron and silicon are non-toxic and formaldehyde-free, making them highly desirable for textile modification due to their eco-friendly properties. These elements have garnered significant attention in the field of textile research and development. 12
Today nanotechnology are used in the modification of textiles and individual protective clothing. 13 Nanoparticles are used to improve the properties of the fibers and fabrics and textile fabrics through absorption or surface treatment during the finishing process. 14 SiO2 Nanoparticle can be used as a protective agent for coating textiles. Some of the features of SiO2 nanoparticles for textiles include UV protection, anti-oxidant properties, chemical/biological protection.15–17 Nanoparticles, especially silica nanoparticles (SiO2), have received a lot of attention because of their improved optical, electrical and thermal conductivity properties. 18
Aerogels, a fascinating class of nanomaterials, have garnered significant attention in the 21st century due to their exceptional properties. These materials can be synthesized from various components, including carbon, cellulose, and silica. 19 The first of these materials was silica, which has become much more popular among aerogels, as these materials have a wide range of exceptional properties including high surface area, high porosity, very low density, very low thermal conductivity, low refractive index, and very low dielectric constant. 20 According to studies conducted in recent years, coating fabric with silica aerogel reduces dust emission, increases mechanical properties,21,22 and provides protection against ultraviolet and infrared rays, as well as thermal and sound insulation. 20 However, aerogels have certain limitations related to fragility and relatively high production costs (an energy-consuming and time-consuming production process). 23
Understanding thermal insulation and thermal comfort in textiles is a critical factor in the design and development of smart workwear across various industries. Moreover, assessing air permeability is a crucial metric for evaluating the quality and performance of workwear fabrics. It directly contributes to user protection, thermal comfort, and overall health. 24 The ability of clothing to transport air and water vapor is a critical factor in determining physiological comfort. Therefore, workwear must possess thermal comfort properties, including air permeability, water vapor transmission, water absorption, and thermal conductivity. 7 Furthermore, the fabric should demonstrate both durability and softness, incorporating tensile and bending resistance. This ensures that the fabric effectively minimizes heat transfer mechanisms, such as thermal displacement and sweat evaporation, thereby reducing thermal stress on the body. The trends in tensile strength, tear strength, and abrasion resistance are critical for evaluating fabric performance. Elevated tensile strength indicates that the fabric can withstand greater forces without breaking, essential for maintaining shape and integrity during use. Tear strength is equally important, as it measures the fabric’s resistance to tearing under stress, ensuring longevity. Lastly, abrasion resistance is crucial for fabrics subjected to friction, helping to preserve surface integrity and appearance over time. Collectively, these properties contribute to the overall durability and functionality of the fabric across various applications.25–27
Most studies that have ever been done on insulated workwear are on the work clothes of workers in high-risk groups and those with high metabolism, such as firemen’s clothes. 28 Because studies on the modification of work clothes for people who work in hot environments with moderate and low risk levels are very limited. The aim of this study is to modify the fabric of work wears for people working in warm environments with moderate and low risk.
Recent research has focused on advanced textiles with novel approaches to material synthesis and coating. These innovations aim to provide thermal comfort while safeguarding individuals from heat stress. Researchers have explored various coating materials as thermal insulators for textile fibers. Consequently, the selection and modification of coating materials for thermal and ultraviolet insulation is crucial for enhancing fabric performance.
This study pioneered the development, coating, and comparison of workwear fabrics using SiO2 nanoparticles and silica aerogel. Our research innovatively investigated the characteristics, thermal properties, and UV protection of these novel fabrics.
The treatments of the fabric with SiO2 nanoparticles and silica aerogel were crucial to this study for their exceptional thermal insulation and UV protection properties. These materials have demonstrated significant potential in enhancing the performance of workwear fabrics. By coating the fabric with these materials, we aimed to significantly improve its ability to retain heat and reduce the transfer of heat to the wearer’s body, while also providing enhanced protection against harmful UV radiation. This innovative approach has the potential to revolutionize the design and development of workwear, offering improved comfort, safety, and overall well-being for workers in various industries.
Materials & methods
Materials
Absolute Ethanol (C₂H₆O) (CAS 64-17-5), Ammonia (NH₃) (CAS 7664-41-7), Tetraethyl orthosilicate (TEOS) (SiC₈H₂₀O₄) (CAS 78-10-4), and Sodium hydroxide (NaOH) (CAS 1310-73-2) were all obtained with analytical grade purity from Merck company.
This study was conducted on cotton-polyester fabric, which is usually used as common workwear fabric in environments exposed to heat stress. This type of fabric, with a simple weave of 70% cotton and 30% polyester, weighing 246 g/m2, was obtained from Yazd Weaving Company in Yazd, Iran.
Preparation of fabric
Before starting the coating process, two preparation steps for the fabric were carried out. The fabric was cut to dimensions of 5 * 5 cm. Each sample was immersed in ethanol for 20 min on a shaker. Subsequently, the samples were washed with distilled water and dried in an oven at 80°C for 30 min. 29 This step was done to wash and remove any previous contaminants. In the second step, a 10% w/v NaOH solution was used to activate the samples and improve material absorption on the surface. 30 The fabrics were immersed in the NaOH solution for 2 h in an ultrasonic bath (PARSONIC 2600s model, 220 AC voltage, 50 Hz frequency, 50 W’ power, 20°C temperature). Then, the fabrics were removed from the solution, washed with distilled water, and dried in an oven at 80°C for 30 min. Finally, after drying, the fabric was weighed as the initial weight (before coating).
Coating of fabric with silica nanoparticles (SiO2)
For the synthesis of SiO2 nanoparticles, fabric was shaken in 15 mL of ethanol and 4 mL of distilled water for 10 min. Then, to increase the rate of hydrolysis, a solution containing 1.5 mL of ammonia was added to it. After that, 1 mL of Tetraethyl orthosilicate (TEOS) was added to the solution and shaken for 30 min until a milky solution was obtained. The fabric in contact with the solution was kept for 2 h in an ultrasonic bath at a temperature of 60°C. Finally, the fabric was rinsed three times with distilled water and dried in an oven at 80°C overnight. 31 After coating, the fabric was weighed again, and the coating percentage was calculated.
Coating of fabric with silica aerogel
Merck silica gel with indicator (orange gel) used for coating in this study was obtained from Tamadkala company. In order to convert the hydrophobic silica aerogel to hydrophilic, the sample was placed in the oven at 400° for 4 h. Then 0.06 g of silica gel powder was mixed in 20 mL of distilled water for 10 min on a shaker with a cloth. Afterward, this solution containing silica aerogel and cloth was incubated for 2 h in an ultrasonic bath. 21
UV protection factor
The ultraviolet transmission spectrum (T%) of the samples will be measured using a spectrophotometer (model Cary 100, Varian, Australia) in the range of 280–400 nm with a step size of 5 nm. A solid sample spectrophotometer with an angular divergence of ±5° about the beam axis is required.
32
The properties of nanofiber protection against ultraviolet radiation will be assessed by measuring the amount of ultraviolet transmission according to the BS EN 13758-1:2002 method. Additionally, the ultraviolet protection factor (UPF) in the UV-A region (315–400) and the protection in the UV-B region (280–315) will be calculated using the AATCC 2010-183 test method.
33
Finally, three repetitions will be carried out for each sample in different areas, and their average will be taken into account. The protection factor of fabrics will be calculated using the AS/NZ 4399:1996 standard
32
:
The effectiveness of fabrics in protecting against ultraviolet rays is expressed by the term ultraviolet protection factor (UPF), which indicates the amount of radiation absorbed by the fabric. Based on different standards, fabric and product protection is classified as insufficient protection (UPF <20), good protection (20 < UPF < 30), very good protection (30 < UPF < 40) and excellent protection (UPF >40). 34
Thermal analysis
Thermogravimetric analysis (TGA) was conducted to study the thermal properties of bare fabrics and fabrics coated with SiO2 nanoparticles and silica aerogel. This analysis is widely used to study the thermal stability of polymers and compounds as a function of temperature. The role of temperature in the degradation study of samples was examined through gravimetric. 23 Additionally, gradient changes in the curve are important features of sample analysis. In these cases, the derivative curve of TGA, called DTG (Derivative Curve), is very useful. 35
Thermogravimetric analysis (TGA) was conducted to evaluate the degradation rate and nanoparticle content of the samples. Samples were heated at a controlled rate of 10°C/min from 25°C to 600°C under a nitrogen atmosphere using a Hitachi STA 300 instrument. The weight loss of the samples was continuously monitored to determine the thermal stability and composition of the materials.
Thermal imaging measurement
Thermal imaging, using a FLIR-C P200 thermography camera, was employed to visualize and measure heat transfer through the fabric samples. The samples were placed on a hot plate with a surface temperature of 50°C within a controlled test chamber. Infrared images were captured from a distance of 15 cm at 10-s intervals to monitor temperature changes. The average surface temperature was calculated using the OLIP ThermoCam Series instruction manual and Infrared Analyzer Software. This analysis allowed for a quantitative evaluation of heat transfer through the fabric samples.
Morphological characteristics
Morphological analysis of the samples was conducted using a Field Emission Scanning Electron Microscope (FES-EM, TESCAN MIRA2). Elemental composition was determined using Energy-Dispersive X-ray Spectroscopy (EDX) attached to the same microscope. Fourier Transform Infrared Spectroscopy (FTIR) with Attenuated Total Reflectance (ATR) mode was employed to analyze the fabric’s chemical composition using a Bruker TENSOR 27 spectrometer. X-ray Powder Diffraction (XRD) analysis was performed using a Philips PW 1510 instrument to investigate the crystal structure and phase composition of the samples.
Physical properties
• • • • •
Results and discussions
Characterization of SiO2 nanoparticles
In order to analyze the morphology and structure of silica nanoparticles (SiO2), FE-SEM images, XRD and FTIR spectra were prepared. In Figure 1(a), the FE-SEM image of nano-particles show particles that are nearly uniform and spherical in shape. Characterization of SiO2 nanoparticles, (a) FE-SEM images (b) XRD spectra.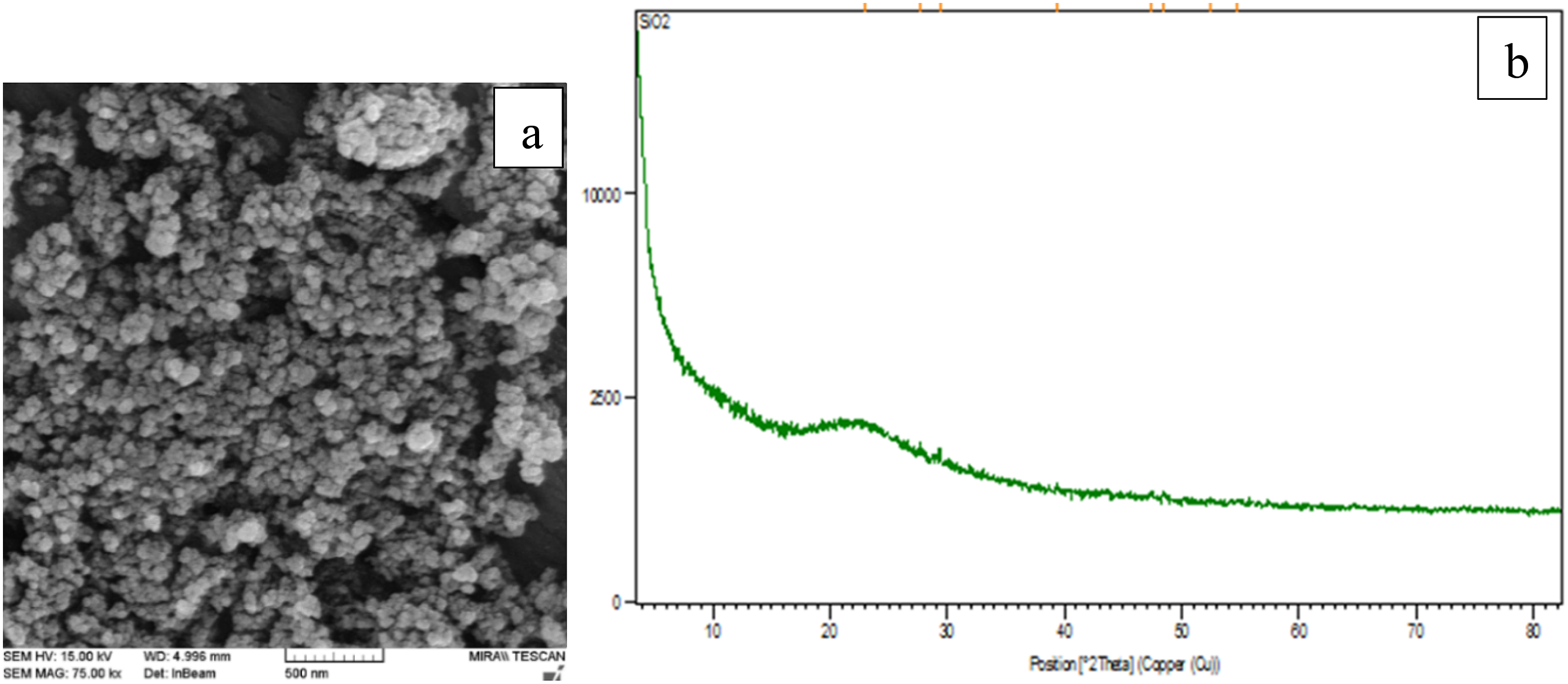
Figure 1(b) displays the XRD spectrum corresponding to the silica nanoparticles. SiO2 are typically analyzed in the range of 2θ = 5–80°. The peaks observed at angles 22.92° correspond to the planes is the main peaks of silica nanoparticles. 18 These peaks, as shown in the XRD pattern obtained for MIL-125 (Ti)-NH2, confirm the successful synthesis. The experimental values are very close to the literature (JCPDS No. 47-0715).
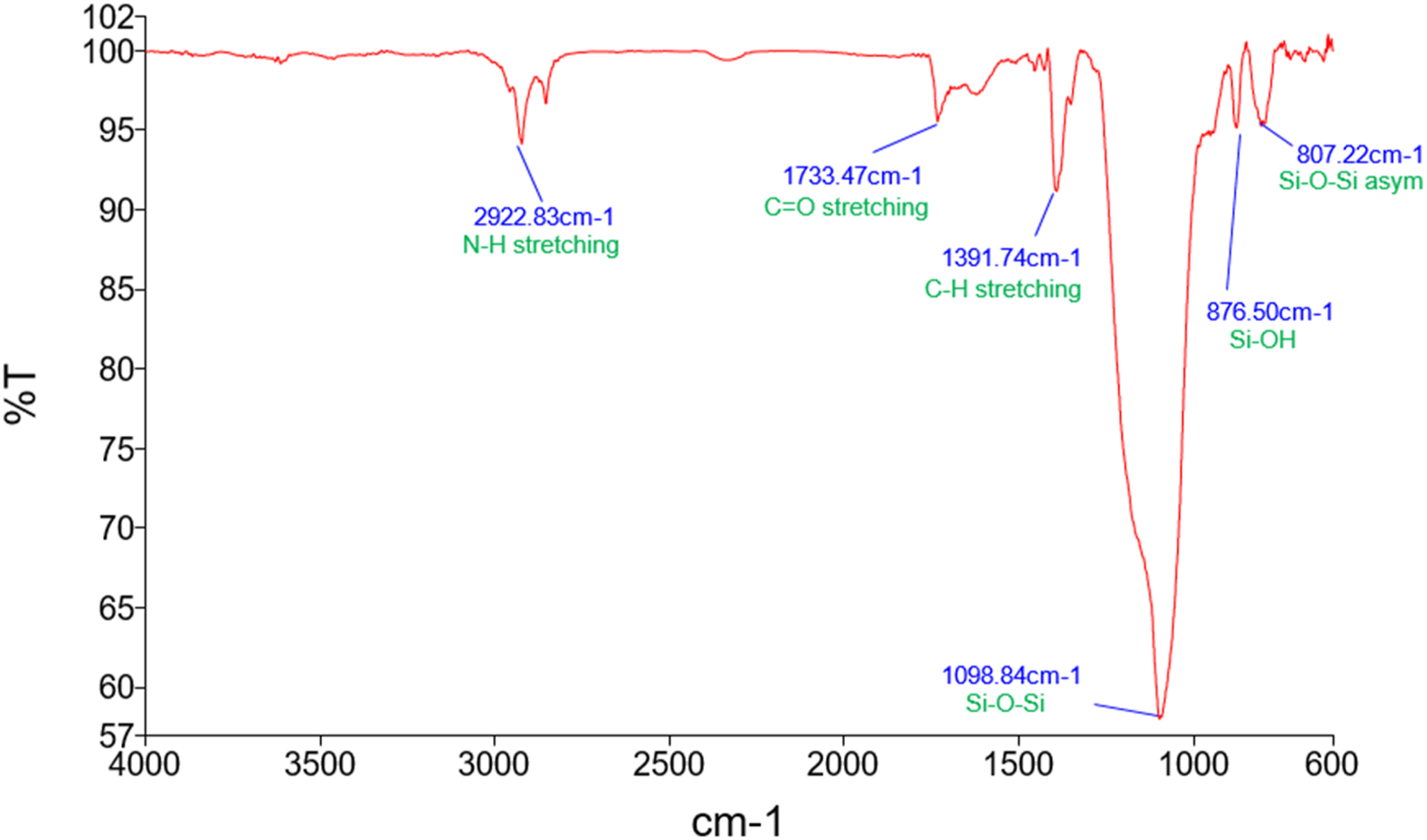
The FTIR spectrum of the synthesized SiO2 nanoparticle powder, shown in Figure 2, exhibits characteristic peaks indicative of silica formation. Peaks observed between 500 cm−1 and 1400 cm−1 correspond to Si-O-Si symmetric vibrations (1032–1056 cm−1) and Si-O stretching (804 cm−1). Additionally, peaks at 467 cm−1, 1872 cm−1, 1934 cm−1, 1500 cm−1, and 3000 cm−1 are associated with O-Si-O bending, C=O and C-O stretching, and OH stretching (adsorbed water), respectively. These findings confirm the successful synthesis of SiO2 nanoparticles. FTIR spectra of SiO2 nanoparticles.
Characterization of coated fabrics with SiO2 nanoparticles
The SiO2 nanoparticle coating percentage was determined to be 5.3% through gravimetric analysis. FESEM images in Figure 3 compare the untreated fabric with the SiO2 nanoparticle-coated fabric. The hydroxyl groups present in cotton and silica nanoparticles facilitated the formation of covalent bonds between the two materials, resulting in a durable coating. The use of ultrasound during nanoparticle synthesis contributed to the formation of nanoparticles within the fabric’s intramolecular chain, further enhancing the binding to cellulose fibers. FE SEM image of (a) uncoated fabric (b) NaOH treated fabric (c, d) coated fabric with SiO2 nanoparticles: 5200 nm.
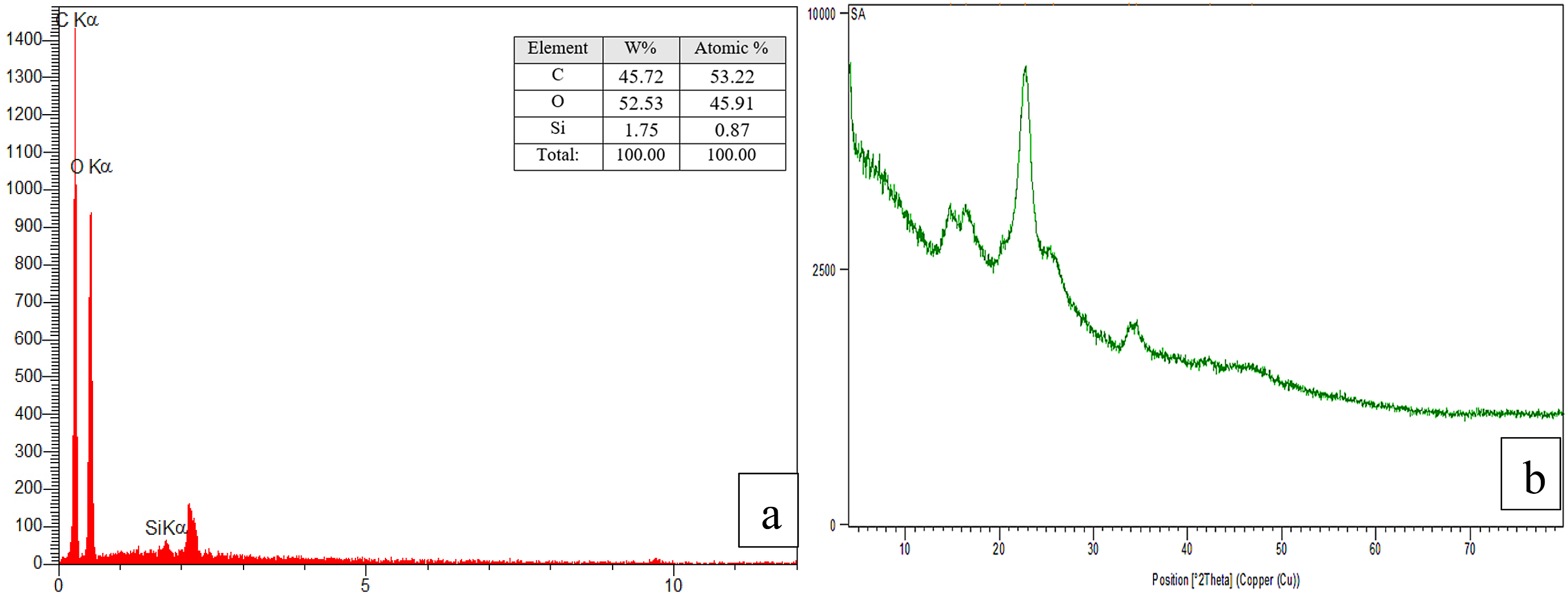
Elemental analysis using EDX confirmed the presence of C, O, and Si elements in the SiO2 nanoparticle-modified fabric (Figure 4). XRD analysis (Figure 5) revealed characteristic diffraction peaks at 14.83°, 16.66°, 22.74°, and 34.49°, indicating the successful in-situ synthesis of SiO2 nanoparticles on the fabric. FTIR-ATR analysis (Figure 6) revealed the presence of O-Si-O stretching (807 and 1098 cm−1), Si-OH stretching (876 cm−1), C-H stretching (1391 cm−1), C=O stretching (1733 cm−1), and O-H stretching (2922 cm−1). These results confirm the successful coating of the fabric with SiO2 nanoparticles and the introduction of new functional groups.
18
Therefore, based on the obtained data, it can be concluded that the fabrics have been effectively coated with SiO2 nanoparticles. Morphological analysis: EDX analysis of coated fabrics with SiO2 nanoparticles. XRD spectra of coated fabrics with SiO2 nanoparticles. FTIR-ATR spectra of coated fabrics with SiO2 nanoparticles.

Characterization of silica aerogel
Figure 7(a) depicts the spherical, uniform, and dispersed morphology of silica aerogel particles as observed by FE-SEM. The XRD spectrum in Figure 7(b) exhibits sharp, symmetrical, and intense peaks at low 2θ angles, indicative of a highly crystalline structure. The primary peaks associated with silica aerogel are located at 14.74°, 16.48°, 20.42°, and 33.78°. Characterization silica aerogel, (a) FE-SEM images (b) XRD spectra.
Characterization of coated fabric with silica aerogel
The silica aerogel coating percentage was determined to be 5.1% through gravimetric analysis. FESEM images in Figure 8 illustrate the successful placement of silica aerogel nanoparticles on the fabric surface. EDX analysis (Figure 9(a)) confirmed the presence of C, O, and Si elements, indicating the presence of silica aerogel. XRD analysis (Figure 9(b)) revealed characteristic diffraction peaks at 14.78°, 16.43°, 22.75°, and 33.79°, further supporting the successful coating of silica aerogel onto the fabric. These findings align with previous research in this area.
35
FE SEM image of coated fabric with silica aerogel. Morphological analysis of coated fabrics with silica aerogel: (a) EDX analysis, (b) XRD spectra.
FTIR-ATR analysis of the SiO2 nanoparticle-coated fabric (Figure 10) revealed characteristic peaks at 1056 cm−1, 1608 cm−1, and 2922 cm−1, corresponding to Si-O stretching, C-O stretching, and O-H stretching vibrations, respectively. These results confirm the successful coating of SiO2 nanoparticles onto the fabric and the introduction of new functional groups.
40
Therefore, based on the obtained data, it can be concluded that the fabrics have been effectively coated with silica aerogel. FTIR-ATR spectra of coated fabrics with silica aerogel.
UV protection properties
The UVA/UVB transmittance properties of the uncoated and coated fabrics were evaluated according to the AS/NZ 4399:1996 standard (Figure 11). The UPF values were calculated as follows: uncoated fabric (3.25), SiO2 nanoparticle-coated fabric (39.28), and silica aerogel-coated fabric (38.11). These results demonstrate that both SiO2 nanoparticle and silica aerogel coatings offer excellent protection against ultraviolet radiation, significantly surpassing the uncoated fabric. UVA/UVB transmittance property of the uncoated fabrics and the fabrics coated with SiO2 nanoparticles and silica aerogel according to the AS/NZ 4399:1996. UVA: Ultraviolet (315–400 nm); UVB: Ultraviolet (280–315 nm).
Exposure to UVA and UVB radiation poses significant health risks. UVA can penetrate deep into the skin, while UVB primarily affects the outer layer. Uncoated fabrics often exhibit higher UVA transmittance, reducing their protective capabilities. This study aligns with previous research by Thennarasu et al., demonstrating that silica aerogel coatings can effectively enhance UV protection in textile materials. 41
Figure 11 illustrates the UV absorption properties of the uncoated and coated fabrics. The uncoated fabric exhibited variable absorption behavior, with decreased or increased absorption at certain wavelengths, likely due to inherent fabric limitations. In contrast, the coated fabrics demonstrated increased absorption in the UVA range (360–400 nm). This trend aligns with previous studies, which have observed that UVA transmission generally exceeds UVB transmission in textile materials. 42 In fact, some studies have found that the coated fabric with SiO2 nanoparticles performs very well in blocking UV radiation at around 340 nm. 43 Krzemińska et al.’s study showed that the use of Silica Aerogel in fabric covers increases their resistance to radiation. 23 This feature can enhance the fabric’s ability to reflect solar radiation, thereby reducing heat gain from the environment. This property can be particularly beneficial for specialized applications such as curtains or barriers, where minimizing heat transfer is crucial.
Thermal and optical analyses
Thermogravimetric analysis (TGA) was conducted to assess the thermal stability of the fabric and quantify changes in mass as a function of temperature under controlled conditions. TGA provides insights into dehydration, oxidation, and decomposition processes.
23
Figure 12 depicts the TGA curves of the samples. Both specimens exhibited an initial mass loss of approximately 5–7% within the first 100°C, likely due to the evaporation of moisture, as reported by Pamuk et al.
44
Following the initial moisture loss, the coated fabrics exhibited negligible mass change up to approximately 300°C. Subsequently, both curves showed a gradual weight loss between 388°C and 325°C, likely attributed to the evaporation of residual water and solvents. Thermal degradation began around 300°C and continued up to 450°C. The primary weight loss observed at 362°C for SiO2 nanoparticles and 357°C for silica aerogel is likely due to the breakdown of compound chains and denaturation of cellulose fibers within the cotton fabric. The UV-blocking and refractive properties of the coatings contributed to the enhanced thermal stability of both materials, aligning with findings from previous research.
18
(a) TGA and (b) DTG thermograms of samples treated with SiO2 nanoparticles, silica aerogel.
The DTG peaks’ intensity correlates with the slope of the TGA curves, indicating the rate of weight change. Narrower, more intense peaks signify rapid weight loss. By analyzing the peak areas, we can compare the overall mass changes in the samples. SiO2 nanoparticle-coated samples exhibited higher thermal stability than silica aerogel-coated samples. This enhancement is attributed to the formation of a stable network within the cellulose matrix, which inhibits thermal decomposition and makes these materials suitable for high-temperature applications. The presence of Si-OH groups in silica particles facilitates thermodynamic interactions, such as hydrogen bonding and dipole-dipole interactions, with the fibers. The polar chain structure of polyester fibers enhances these interactions, leading to increased adhesion between silica additives and the fabric. The formation of hydrogen bonds between the surface hydroxyls of silica and the fabric contributes to the strong adhesion observed in the samples. 45 Therefore, the higher the percentage of silica additives, the greater the thermal resistance of the samples, as seen in the results of the Xu et al. study. The study showed that the thermal stability of the coated fabric has been greatly improved by the addition of aerogels and SiO2, and the amount of carbon left at 600°C has significantly increased. 46 Therefore, coating aerogels and SiO2 on fabrics and surfaces can be used as thermal insulation. 47
Infrared thermography
Thermal imaging analysis was conducted to evaluate the thermal insulation properties of the coated fabrics. Uncoated and coated samples were placed on a hot plate (50°C) to assess heat transfer. The results demonstrated the superior thermal insulation capabilities of the SiO2 nanoparticle and silica aerogel coatings. After 10 s of contact, the uncoated fabric reached an average temperature of 43.1°C. In contrast, the coated samples attained average temperatures of 35.7°C and 36.4°C Figure 13. Thermal imaging of (a) untreated samples, (b) samples treated with SiO2 nanoparticles, (c) samples treated with Silica Aerogel.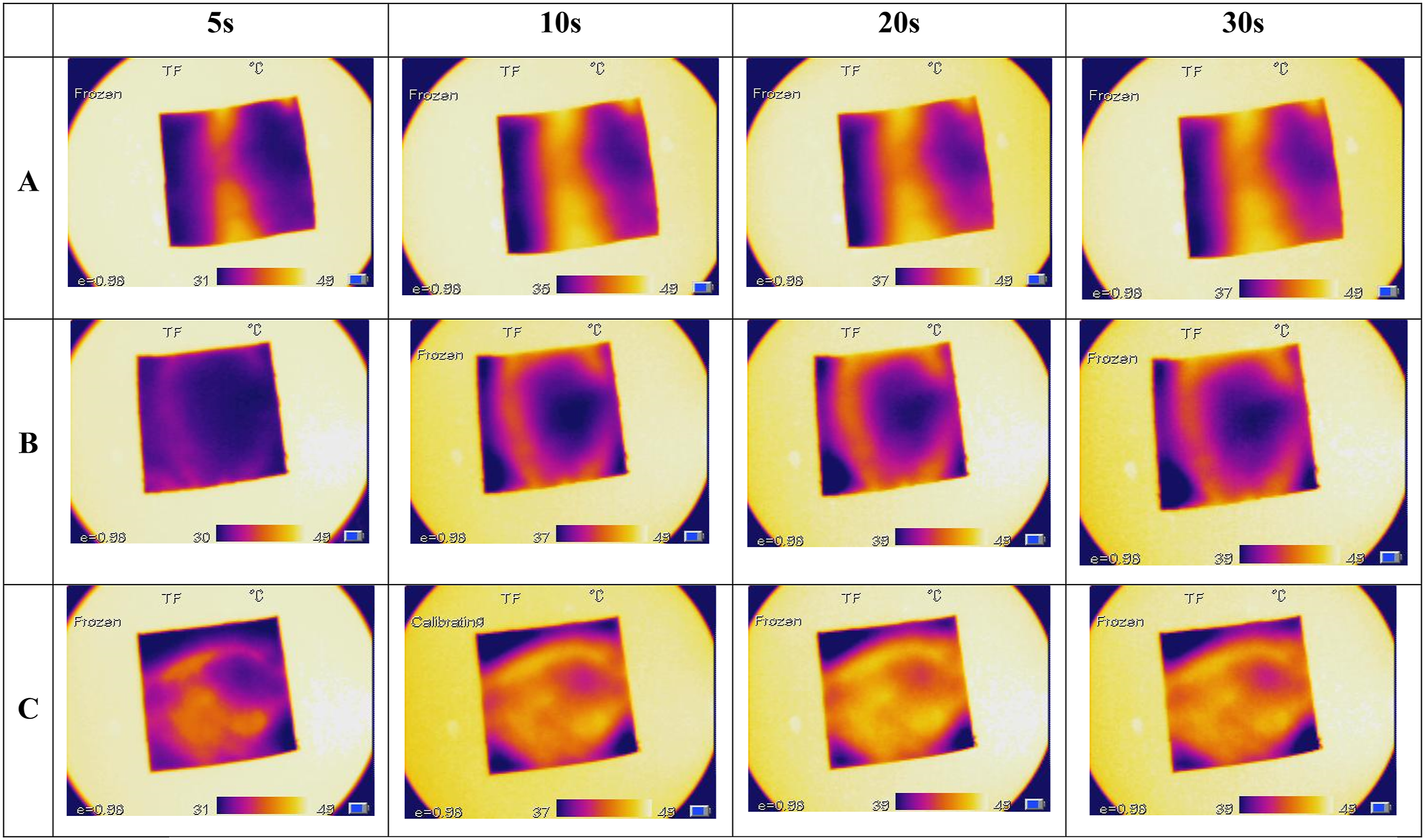
This improved thermal insulation is attributed to the formation of a thin air gap between the fabric and the hot surface, reducing heat conductivity. Additionally, the coated fabrics exhibited slower heat release to the environment compared to the uncoated fabric, indicating their ability to retain heat and thermal energy. These findings highlight the potential of these materials for developing high-performance thermal insulation fabrics.
Physical properties analyses
This study extends beyond thermal and ultraviolet insulation, investigating the effects of the coatings on the intrinsic properties and physical structure of the workwear fabric. Key properties evaluated include air permeability, abrasion resistance, tensile strength, and creep (Figure 14). These properties are essential for ensuring thermal comfort and effective interaction with the environment. The results revealed a decrease in air permeability after coating, with silica aerogel having a more significant impact due to its high porosity and the fabric’s structure. Previous studies have similarly observed a reduction in air permeability after silica gel coating. Furthermore, a correlation between higher air permeability and lower thermal resistance was evident in the silica aerogel-coated fabric.
45
Comparison of the amount of abrasion resistance, tensile, tearing strength and air permeability of uncoated and coated fabric.
The coating with SiO2 nanoparticles enhanced abrasion resistance and tensile strength while slightly reducing tearing resistance. Conversely, silica aerogel coating improved abrasion and tearing resistance but decreased tensile strength. These findings highlight the differential effects of the two coating materials on the mechanical properties of the fabric. A study by Saleemi et al. revealed that coating the fabric with SiO2 nanoparticles improved the tensile and bending strength of the sample while reducing air permeability. 48 Another study by Mazrouei and colleagues demonstrated that combining aerogels with fibers can enhance mechanical and insulation properties. 21 It was also found that Timar Silica Aerogel had no adverse effect on the appearance of fabrics or their strength properties. 45
Conclusion
This study successfully developed innovative workwear fabrics with enhanced thermal and UV protection properties. By employing in situ synthesis methods, we successfully synthesized and coated SiO2 nanoparticles and silica aerogel onto the fabric surface. Morphological analysis confirmed the successful loading and distribution of these materials. The coated fabrics exhibited remarkable thermal insulation and UV protection capabilities. After 5% coating, the UPF values for SiO2 nanoparticle- and silica aerogel-coated fabrics reached 39.28 and 38.11, respectively, demonstrating excellent protection against UV radiation. Furthermore, the coated fabrics demonstrated superior thermal stability compared to the uncoated control. Infrared imaging revealed significantly slower temperature increases in the coated samples, highlighting their ability to trap heat and reduce heat transfer. The in-situ synthesis method facilitated a homogeneous distribution of SiO2 nanoparticles and silica aerogel on the fabric fibers, contributing to the enhanced thermal stability and overall performance. In conclusion, this research demonstrates the potential of using SiO2 nanoparticles and silica aerogel as coatings for workwear fabrics to improve thermal insulation and UV protection. The in-situ synthesis method offers a promising approach for developing innovative textiles with enhanced functionality and safety benefits for workers in various industries.
Footnotes
Acknowledgements
The authors would like to thank the collaboration of laboratories from Laser and Plasma Research Institute, Shahid Beheshti University (Dr Neda Roostaei), Tarbiat Modares University, and Amirkabir University of Technology.
Author contributions
S.F.D. managed and planned the project. S.F.D., E.A.P., S.S.KH. & S.B.M contributed to the conceptualization and methodology. E.A.P., S.S.KH. & S.M.H did the experiments and data analysis. S.F.D., E.A.P. & S.S.KH were a major contributor in in data interpretation and original drafting, reviewing and editing the manuscript. All authors review and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was part of the research projects supported by Shahid Beheshti University of Medical Sciences (Grant no. 43007098). This study was financially supported by Shahid Beheshti University of Medical Sciences.

