Abstract
This current study proposes a novel, successful method for multi-finishing polyacrylonitrile fabric. The performance of the polyacrylonitrile (PAN) fabric was successfully modified using functional finishing with hybrid composite (chitosan-titania NPs-organic UV absorber). PAN was activated to increase the number of reactive sites available for functional finishing. The effect of the finishing process on the fabric properties and the interaction between the activated PAN fabrics with the hybrid composite was demonstrated. Dyeing with reactive dye was done to monitor the reactive groups formed after alkali hydrolysis of PAN fabric and finishing and has been estimated as the starting point to determine the appropriate finishing bath formulation. The results revealed that the hybrid composite is a multifunctional agent in the fabrication process of bacterial and ultraviolet resistances. The maximum bacterial reduction was 95% E. coli and 92% for S. aureus and the maximum UPF value (108) and excellent protection category against UV radiation. The surface characteristics of the fabrics were investigated by Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), and energy-dispersive X-ray spectroscopy (EDX). The UPF and antibacterial properties show a minor decrease after five wash cycles. The reduction might be attributable to the unfixed and physically connected finishing agents.
Introduction
The most significant reasons for textile functionalization are enhancing the parent properties of the synthetic fibers such as polyacrylonitrile (PAN) and developing new products. There is an industrial prerequisite to improve PAN fibers’ hydrophilicity alongside maintaining the fibers’ mechanical properties. PAN fibers claim a prominent role in the synthetic fiber category and replace wool in many respects in clothing and the related industry. They are used primarily for the manufacture of bulk yarns and sweaters. In recent years, wide varieties of products for PAN have been available for different applications, including water absorbent and antimicrobial fibers. They are the most used material in the textile industry and related industries by combining desirable characteristics such as high outdoor and chemical tolerance, outstanding elasticity, and aesthetical properties. However, due to its hydrophobic character, PAN fibers often exhibit undesirable properties such as uncomfortable hand, static charge accumulation, and. These disadvantages could be overcome through chemical means. The application of solid chemical agents is crucial for creating a character similar to natural fiber, improving performance, and altering the polymer surface properties. 1
Reaction conditions need to be designed to achieve the required functionalities in the fiber. Alkali hydrolysis overcomes low water absorption problems and softness so that it acquires the same properties as natural fibers. 2 During the alkaline hydrolysis reaction of PAN, the alkali directly affects the surface and then penetrates within the fiber structure. The chemical alteration of the hydrolyzed PAN gives carboxylic and amide groups in the backbone of the polymer. Further hydrolysis leads to converting nitrile groups into the corresponding amides and carboxylic groups. Consequently, the surface of PAN alters precisely, and such reactive sites are susceptible to wet processing such as functional finishing and dyeing.1,3
Chitosan (CS) is a naturally occurring polymer that results from chitin deacetylation and, it is harmless, poly-cationic character, and biodegradable. It is antibacterial, antifungal, and biocompatible with organic and inorganic substances via free amino or hydroxyl functional groups, which react with other functional groups through electrostatic forces, hydrogen bonds, or by a compound in the polymeric matrix. Therefore, mechanical and biological properties have improved. Biopolymer properties like mechanical, physical, and biological can be enhanced by TiO2 (crosslinking agent), ideal for applications in different fields like food, medicine, antimicrobials, and renewable energy.
Nanostructured materials, particularly metal oxide nanoparticles, have received widespread interest in recent years due to their unique physical, chemical, and biological characteristics and future uses. TiO2 is widely utilized as an antibacterial agent against various bacteria, viruses, and fungi. TiO2 is a stable photocatalyst, cost-effectiveness, ultraviolet protection, safe manufacturing, and biocompatibility.4,5 The combinations of natural polymers such as chitosan (CS) and inorganic materials (TiO2) are obviously of particular concern to develop hybrid composites (CS – TiO2) that have beneficial properties for a wide range of industrial uses. 6
The hybrid composite polymer system shows the ability to facilitate an enormous number of potential functions, increasingly requested by textile manufacturing sectors. The tendency of TiO2 NPs to agglomerate is one of the main drawbacks. The interactions between titania NPs and chitosan biopolymers have been reported to minimize the aggregation that improves composite functions. 7 It should be mentioned that structural properties of chitosan are improved via doping of TiO2 NPs at the same time in uniform dispersion of NPs on chitosan surface films. 8
Organic or inorganic UV absorbers have a high UV absorption, which converts electronic vibrational energy into heat energy and behaves as scavengers of single oxygen and radicals, along with their ability to dissipate the absorbent energy as long-wave radiation.9,10 The organic UV blockers are usually referred to as chemical filters because of the chemical mode of action in their molecules, preventing UV radiation from penetrating the skin. On the other hand, the inorganic UV blockers have been referred to as physical blockers because their skin safety mode is related to biological phenomena, such as UV radiation scattering and reflection.11,12
Numerous materials fabricate high-UPF products with different modifications and topical applications for nearly all polymer fibers types. Most industrial products have been established to be compatible with textile dyes and finishing agents. They can be applied using various techniques, such as padding, exhaust methods, pad-thermal, and pad-dry-cure. The fabric gains ultraviolet protection by shielding it from harmful radiation by ultraviolet blocking agents, where the fabric surface reflected part of the incident radiation and transmitted other parts. The transmitted part consists of waves passing through surface layers of the textiles and the scattered waves directly interacting with the fabric layer. Another amount of radiation absorbed and penetrated the fabric and converted into another form of energy. The part passing into the skin is called the transmission portion. 13
Generally speaking, modification of polyacrylonitrile fabric with TiO2 NPs could be summarized into three approaches based on previous researches. The first one is conventional finishing, where self-cleaning polyacrylonitrile fabric could be gained by embedding commercial TiO2 NPs (Degussa P-25) and using polysiloxane softener (stabilizer) on the fabric surface to provide soft handling hydrophilic and self-cleaning properties, using conventional finishing technique pad-dry-cure. The self-cleaning characteristic has been substantially enabled to absorb light with wavelengths less than 400 nm and enhance the discoloration of C.I. Reactive Yellow 1. 14
Moafi et al. 15 prepared anatase nanocrystals effectively and coated them onto polyacrylonitrile fibers with photocatalytic self-cleaning activity, utilizing the sol-gel method at low temperatures. The photocatalytic activity was determined by monitoring the degradation of adsorbed Eosin Yellowish dye. The photoactivity of titanium dioxide-coated fibers is considerably greater than that of untreated fibers. The photocatalytic self-cleaning capabilities of the treated fibers were entirely preserved after multiple cycles of photodegradation.
In another research paper, the authors compared TiO2 NPs activities and ZrO2 NPs produced onto PAN fibers using the sol-gel method at low temperatures. The photocatalytic activity was determined by measuring the degradation of methylene blue and Eosin Yellowish dye. TiO2 NPs 10–20 nm coated polyacrylonitrile fibers have more excellent activity for dye degradation than ZrO2-coated fibers upon exposure to UV–Visible light. 16
Secondly, wet spinning was used to produce polyacrylonitrile porous fiber-containing TiO2 NPs and is considered a potent approach for manufactured photocatalytic active fiber on a wide scale. The polyacrylonitrile solution as spin dope was produced using dimethyl sulfoxide (DMSO) as a solvent at different concentrations to manufacture continuous wet spun fiber from TiO2 NPs (Aeroxide® P25)-filled polyacrylonitrile solution. The viscosity influences the wet-spinnability of the TiO2 NPs-filled PAN/DMSO solution. The PAN/TiO2 fiber was continually stabilized in the air at temperatures ranging from 250°C to 300°C and carbonized in N2 at 800°C. The rheology of the PAN solution was considerably impacted by the type of dope solvent used. Because of the increased physical interaction between PAN and DMSO, PAN/DMSO solutions have a higher viscosity than PAN/DMF solutions. The diameters of the carbonized PAN/TiO2 fiber and the stabilized PAN/TiO2 porous fiber are 15 and 10 μm, respectively. After 3 h of UV irradiation, the wet spun PAN/TiO2 porous fiber with 0.5 weight percent TiO2 destroyed methylene blue at 50%; however, the fiber without TiO2 did not demonstrate photocatalytic degradation. 17
Kizildag et al. 18 used wet spinning to create nanocomposite polyacrylonitrile filaments containing TiO2 and silver NPs to produce multifunctional filaments with antibacterial activity, photocatalytic activity, and electrical conductivity. The nanocomposite filaments (shape, composition, nanoparticle dispersion, tensile characteristics, crystallinity, conductivity, thermal properties, photocatalytic activity, and antibacterial activity) were studied. The nanoparticles were found to be evenly distributed. The conductivity of the nanocomposite filaments was on the order of 104 S/cm, indicating that they were semi-conductive, and the nanocomposite filaments demonstrated antibacterial as well as photocatalytic activities. The research demonstrated the feasibility of creating multifunctional filaments by incorporating various nanoparticles into the filament structure.
Thirdly, the electrospinning process created polyacrylonitrile and polyacrylonitrile-TiO2 nanofibers with variable TiO2 NPs loading. The latter was formed by changing the calcination temperatures in the sol-gel technique. The absorption and emission spectra demonstrated the production of TiO2 NPs, with smaller particles exhibiting an increase in absorption band margins. Different TiO2 NPs loadings enhanced electro-spinnability and morphology while decreasing nanofiber size. These nanofiber fibers are air and moisture resistant, as well as thermally stable. 19
Another research paper used TiO2 powder for PAN-supported TiO2 with different TiO2 to PAN ratios for indoor air cleaning applications was investigated. PAN-TiO2 threads with varying ratios were created by combining PAN, N,N-dimethylformamide as a solvent and TiO2 (anatase powder) and were mixed in a specific weight ratio to make a polymer solution. The decomposition efficiencies of PAN-TiO2 fibers improved with the proportion of PAN: TiO2 N,N-dimethylformamide for all target aromatic compounds. More than 3 h of photocatalytic operation, the decomposition efficiencies of the PAN-TiO2 fibers for the target compounds were close to 100% for the highest TiO2 ratio (1:1:9). Overall, when operational circumstances are adjusted, PAN-TiO2 fibers may be employed successfully for indoor air aromatic compound cleaning. 20
It should be mentioned that PAN is particularly interesting because it can be stabilized and carbonized after electrospinning, resulting in conductive carbon nanofiber mats that may be utilized in a wide range of applications. It is worth noting that, TiO2 is one of the most intriguing semiconductors in manufacturing dye-sensitized solar cells. The embedding of TiO2 in PAN nanofiber mats was discovered to allow semiconductor dyeing without the need for sintering the TiO2 layer, which would be required on glass or macroscopic textile surfaces. That enables TiO2 on textiles that are not heat-resistant enough to allow sintering at the usual temperature of 500°C.21,22
Last but not least, the multilayered composed of chitosan and clay nanoplatelets coatings of acrylic fabric using the layer-by-layer production process as a sustainable and effective way to impart flame retardant characteristics to the textile fabric was achieved. Compacted chitosan/montmorillonite complexes are produced by directly mixing an oppositely charged solution/suspension using a doctor-blading technique. The 20% add-on had the most outstanding results, self-extinguishing the flame within a few seconds of the first flame application (Table 1). 23
A literature survey was carried out related to the research paper.

This study focuses on the novel synergetic effect of titania NPs, chitosan, organic UV absorbers through the application of hybrid composites polymers with variant mixtures. Such as (titania NPs-chitosan), (organic UV blocker-chitosan), (titania NPs-organic UV blocker), and (titania NPs-chitosan-organic UV blocker) in wet processing operations of PAN finishing to convey sustainable multifunctional finishing with versatile properties on the surfaces of acrylic fabric. Citric acid is used as a crosslinking agent to strengthen NPs’ and chitosan binding. This study suggested a novel route to manufacture a durable surface functionalization with versatile properties on the surfaces of acrylic fabric. Simple conventional finishing techniques such as pad-dry–cure and exhaustion were used. The dyeing process is used as an indirect estimation for the finishing processes.
Experimental work
Materials
PAN single jersey, number of wales is 8/cm, number of courses is 11/cm, loop length is 0.5 cm, the fabric weight is 150 g/m2, scoured, and half bleached was used in this study.
Chemicals and dyes
Chitosan (low molecular weight) and Titanium dioxide NPs (30% in H2O) colloidal dispersion were purchased from Sigma–Aldrich. Glacial acetic acid, hydrochloric acid, citric acid, sodium hypophosphite, sodium hydroxide, sodium sulfate, sodium carbonate, nonionic detergent Hostpal®CVL-EL, peptone, and beef extract were laboratory-grade chemicals. Huntsman, formerly Ciba Specialty Chemicals, generously provided UV-SUN® a reactive anionic UV absorber based on oxalanilides. Novacron® Red LS-B (C.I. Reactive Red 270) bis-mono fluoro-s-triazine. In addition, C.I. Basic Violet 14 was supplied by European Color Plc, Stockport, England.
C.I. Basic Violet 14
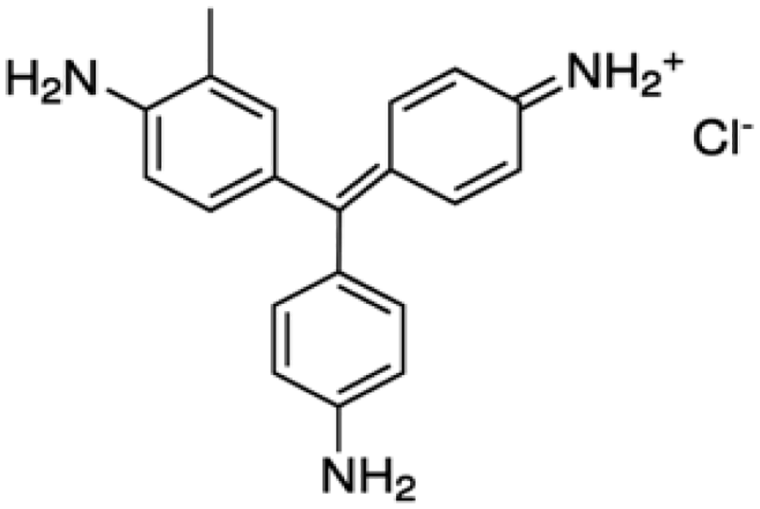
Microorganisms and growth conditions
The antibacterial potential of PAN fabric was tested using Gram-negative bacteria Escherichia coli (E. coli) and Gram-positive bacteria Staphylococcus aureus (S. aureus). Bacterial strains were cultured overnight at 35 ± 2°C in nutrient broth (NB). These cultures were used as inoculums in the antimicrobial test.
Methods
Alkaline treatment
PAN was subjected to alkaline hydrolysis with sodium hydroxide 1 M at 80○C for 120 min and material to liquor ratio M:LR (1:20). After hydrolysis, samples were neutralized with 1% HCL acid, then washed with deionized water, and dried in the air (PAN1).
Fabric finishing
Chitosan and TiO2 NPs finishing by Pad-dry-cure method
Chitosan 0.5%–4% w/v, TiO2 NPs 1.5% w/v, citric acid 10%, sodium hypophosphite 7 g/l, and glacial acetic acid 0.5% were used. Samples were soaked at 80°C for 30 min, then padded with 2-dips and 2-nips to a wet pick-up ~100%, dried at 80°C for 5 min, and cured at 140°C for 3 min.
Washing
The treated acrylic fabric samples were washed after finishing with nonionic detergent 2 g/l at 40°C for 20 min to remove unfixed particles and materials. The treated fabrics were rinsed several times and then dried at an ambient temperature.
UV absorber finishing by exhaust method
UV-SUN® 2% o.w.f, sodium sulfate 15 g/l, and sodium carbonate 10 g/l, samples were finished for 30 min at 70°C and M:LR 1:40. The samples were washed after finishing with nonionic detergent 2 g/l at 40°C for 20 min, then rinsing and drying at ambient temperature.
Dyeing with reactive dye
Untreated, alkali-treated, and/or finished PAN fabric samples were dyed with Novacron® Red LS-B dye (C.I. Reactive Red 270) 2% o.w.f, sodium sulfate 15 g/l, sodium carbonate 10 g/l, M:LR 1:50. The dyeing process was carried out as described elsewhere. 24 All dyed samples were washed with nonionic detergent 5 g/l at 60°C for 20 min before testing and evaluation.
Dyeing with basic dye
Untreated, alkali-treated, and/or finished PAN fabric samples were dyed with Basic dye (C.I. Basic Violet 14) 2% o.w.f shade, sodium sulfate 10 g/l, pH = 4.5, M:LR 1:50, raising the temperature to 95○C for 40 min. All dyed samples were washed with nonionic detergent 5 g/l at 60°C for 20 min before testing and evaluation. The alkali-activated and finished polyacrylonitrile samples’ abbreviations are described in Table 2.
Abbreviation description of the finishing type of acrylonitrile fabric.

Characterization and testing
Color strength (K/S)
Dyeing performance was characterized in terms of color strength value (K/S) value was assessed using the Kubelka–Munk equation;

Where K, S, and R are the absorption coefficient, scattering coefficient, and reflectance, respectively, measuring C.I. Reactive Red 270 at 575 λmax and C.I. Basic Violet 14 at 485 λmax.
UV protection ability
The ultraviolet protection factor (UPF) of polyacrylonitrile samples was calculated according to the Australian New Zealand standard test method 135-2000 by measuring ultraviolet radiation transmission (UVR) using UV-JASCO V-750 Spectrophotometer. The following equation based on the percent UVR through the specimen was measured three times. Depending on the UPF value, the protection categories are excellent with the UPF values are 40–50, <50 and very good with the UPF values in the range of 25–39, and good with the UPF values in the range of 15–24. 24

Where Eλ is the relative erythema spectral effectiveness, Sλ is solar spectral irradiance in W/cm2/nm; Δλ is the wavelength interval of the measurements; Τλ is the spectral transmittance of the fabric (measured). The percentage blocking of UVA (315–400 nm) and UVB (315–290 nm) was calculated from the transmittance data.
Antibacterial activity
Turbidity method
The antimicrobial activity of acrylic fabric samples was determined by the turbidity method.25,26 Briefly, 30 µl of the tested microorganism (106 colony-forming units (CFU)/ml were inoculated into 3 ml of NB or peptone diluted broth (PDB) medium in standard test tubes containing tested textiles (2 × 2cm2). Test tubes were incubated at 35 ± 2°C for 24 h under shaking conditions. Antimicrobial activity was evaluated by measuring the Optical density (OD) at 600 nm and comparing it to the blank sample. The microbial reduction percentage was calculated from the following equation.

Where A and B are the optical density of the microbial culture at 600 nm in the presence of blank and finished fabric samples, respectively.
Bursting strength
Bursting strength of untreated, hydrolyzed, and treated fabric samples were evaluated according to ASTM D3786-87 by Mullen tester, USA.
Air permeability
The air permeability of the fabric was determined according to ASTM procedure D737-04-2008.
Surface roughness (SR)
Surface roughness was measured according to JIS B0031-1994 standard test method, using surface roughness measuring instrument SE-1700, Japan.
Wettability properties
Wettability properties of acrylic fabric were evaluated before, after hydrolysis, and after finishing according to ISO 4920-1981 standard test method.
Durability properties of the finishing to washing
The finished fabrics were repeatedly washed for five wash cycles according to the standard AATCC test method 61-1989 to assess the durability of PAN finished fabric efficiency.
Light and wash fastness properties
The colorfastness of dyed PAN fabrics to light and wash was tested using ISO 105-B02:2014 (Colorfastness to artificial light: Xenon arc fading lamp test) and ISO 105-C02:1989, respectively.
Scanning electron microscopy (SEM) and energy disperse X-ray spectroscopy (EDX)
A JEOL-Model JSM T20 scanning electron microscopy (SEM) operating at 19 kV was used to obtain photomicrographs of the fiber surface.
FTIR Analysis
Fourier Transform Infrared Spectroscopy (FTIR) of the untreated, treated samples were recorded using an FTIR spectrophotometer (JASCO 4700, Japan) in the region of 4000–400 cm−1 with a spectral resolution of 4 cm−1.
Results and discussions
FTIR spectroscopic characterization
Fourier-transform infrared analysis was investigated for tracking the structural changes of polyacrylonitrile fabric surface and evidence of hydrolysis and finishing process. The description of samples’ abbreviation was mentioned in Table 2. Figure 1 shows the spectra of hydrolyzed and hydrolyzed then finished PAN fabrics. The hydrolyzed fabric displays characteristics absorption peaks at ~3420 cm−1, and 1711 cm−1 could be assigned for N-H stretching and carbonyl of amide groups, respectively. These changes are consistent with the transformation of nitrile groups of polymer backbone into amide groups due to hydrolysis. 27

FTIR spectra of hydrolyzed PAN, hydrolyzed PAN → chitosan, hydrolyzed PAN → TiO2 + chitosan, and hydrolyzed PAN → chitosan + TiO2 + UV absorber.
The activated PAN and finished with chitosan display two weak absorption bands characteristics for primary amine at ~3535 cm−1. The other one is near 3419 cm−1, assigned to asymmetrical and symmetrical N-H stretching vibration. These bands are shifted to longer wavelengths by hydrogen bonding with the functional groups on the surface of hydrolyzed PAN. The band at ~ 1382 cm−1 assigns to C-H symmetric bending vibration, and the band at 1091 cm−1 assigns to C–O stretching. 28
The hydrolyzed PAN then finished with hydride composite chitosan- titania NPs shows bands in the region 3620–3200 cm−1 merged into one wideband. Because of intermolecular and intramolecular hydrogen bonding between amide and carboxyl, the bands for N-H of amide groups and carboxyl groups likely combined to generate this peak broadening. Besides, the band at ~3443 cm−1 assigned to –NH2 and –OH stretching vibrations in chitosan, which confirms that titanium ions bonded onto amino and hydroxyl groups of chitosan; as a result, electrostatic interaction of (N-H-O-Ti). It is clear from the literature that this electrostatic attraction is due to the interaction between the electron pair on the nitrogen atom of amino groups and the oxygen atom of hydroxyl functional groups of chitosan serve as reactive sites for adsorption titanium which promotes the composite formation bonds. In addition, these bands may be attributed to the probabilities of interactions between composite reactive groups and hydrolyzed PAN fabric reactive groups and enhancing the composite fixation via a citric acid crosslinking agent.29,30 Other researchers suggested that the stretching vibration of C-O, NH2, and O-H groups is firmly attached to titania NPs consequently enhancing the composite formation.7,31 The band’s appearance at ~1000 cm−1 could be attributed to Ti-O-C, proving that chitosan chemically attached with titania not only adsorbed. The band at ~3300 cm−1 combines peaks of OH and NH2 group stretching vibrations. Incorporation of titania NPs into chitosan may occur in the amorphous part; this led to strengthening the hydrogen bond in the composite.31,32 The aggregation of NPs is prevented by the chitosan effect on van der Waals forces on titania NPs.31,33
The peak at 2963 cm−1 was assigned to asymmetric and symmetric vibration of C-H in CH2 and CH3 groups. The peak at 1712 cm−1 was attributed to N-H scissoring from the primary amine of chitosan (interaction between NH2 and Ti4+) and merged with the carbonyl group of hydrolyzed PAN. The peak at 600–900 cm−1 attributed to Ti–O (Ti–O–Ti) asymmetric stretching mode due to the immobilization of titania NPs onto chitosan. The peaks at 400–600 cm−1 attributed to Ti–O–C interaction of the Ti4+ Lewis site with amino groups of chitosan. 28 The peaks at 2400–2200 cm−1 assigned to C≡N disappear that may be due to the chemical interaction of the hydrolyzed PAN with the composite function groups. 34
Functional properties
The most significant reason for the textile fabric functionalization is improving the parent properties of the textile fabric and creating a new one/s. This study aims to assess hybrid composite for modification PAN fabric properties and enhance antibacterial, anti-ultraviolet, and dyeing properties. Alkali hydrolysis of acrylic fabric was carried out for activation and creating of reactive sites. Treatment with sodium hydroxide transformed the nitrile groups into amide and/or carboxylic groups, as shown in reaction scheme 1, which improved the hydrophilic property of PAN and made it more accessible to additional functional finishing and dyeing.
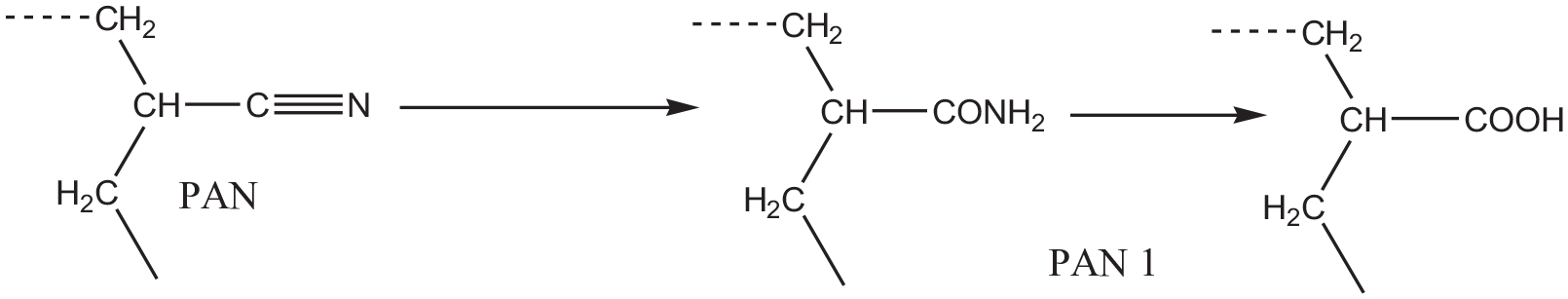
The proposed reaction of PAN nitrile groups under alkaline hydrolysis.
Color strength (K/S)
The dyeing process has four stages: the transfer of dye molecules onto fiber surface, adsorption and diffusion into the fibers, and finally, the interaction between them. The dyeing process with reactive dye was studied to monitor the reactive groups formed after alkali hydrolysis of PAN fabric. Figure 2 shows the effect of chitosan concentration on the color strength of dyed PAN1 fabric. The results revealed that increasing the color strength with increasing chitosan concentration. This results due to increased reactive sites available for interaction with the dye molecules. Finishing PAN1 with chitosan enhanced the color strength of the dyed fabric. PAN1 has carboxylic and amide groups because of the hydrolysis process, which may undergo esterification with hydroxyl groups of chitosan and the formation of salt linkage between those carboxylic groups and amino groups of chitosan; consequently, the dye uptake enhanced significantly. Citric acid is used as a crosslinking agent for chitosan biopolymer through the esterification reaction between the carboxylic group(s) of citric acid and the primary hydroxyl groups of chitosan. 35 In addition to the reaction between the hydroxyl group of citric acid and the carboxylic group of PAN1 and the formation of salt linkages between the carboxylic and amino groups of the chitosan biopolymer. 36

Effect of chitosan concentration on K/S values.
The effect of finishing and dyeing bath formulations on the color strength K/S, antibacterial, and ultraviolet protection was presented in Table 3. The data have shown that the activation of PAN enhances the color strength K/S of dyed PAN1 is (1.6) higher than the color strength of the parent fabric (0.05). These findings may be seen as the first demonstration of surface activation of PAN. It is not typical for the polyacrylonitrile fabric to be dyed with reactive dyes (negative charged) unless the material is activated and modified. Thus, the color strength of the finished PAN1 and dyed with reactive dye indirectly show the reactive groups’ created on the fabric surface.
Effect of finishing condition on the functional finishing properties of acrylic fabric and reactive dyeing.
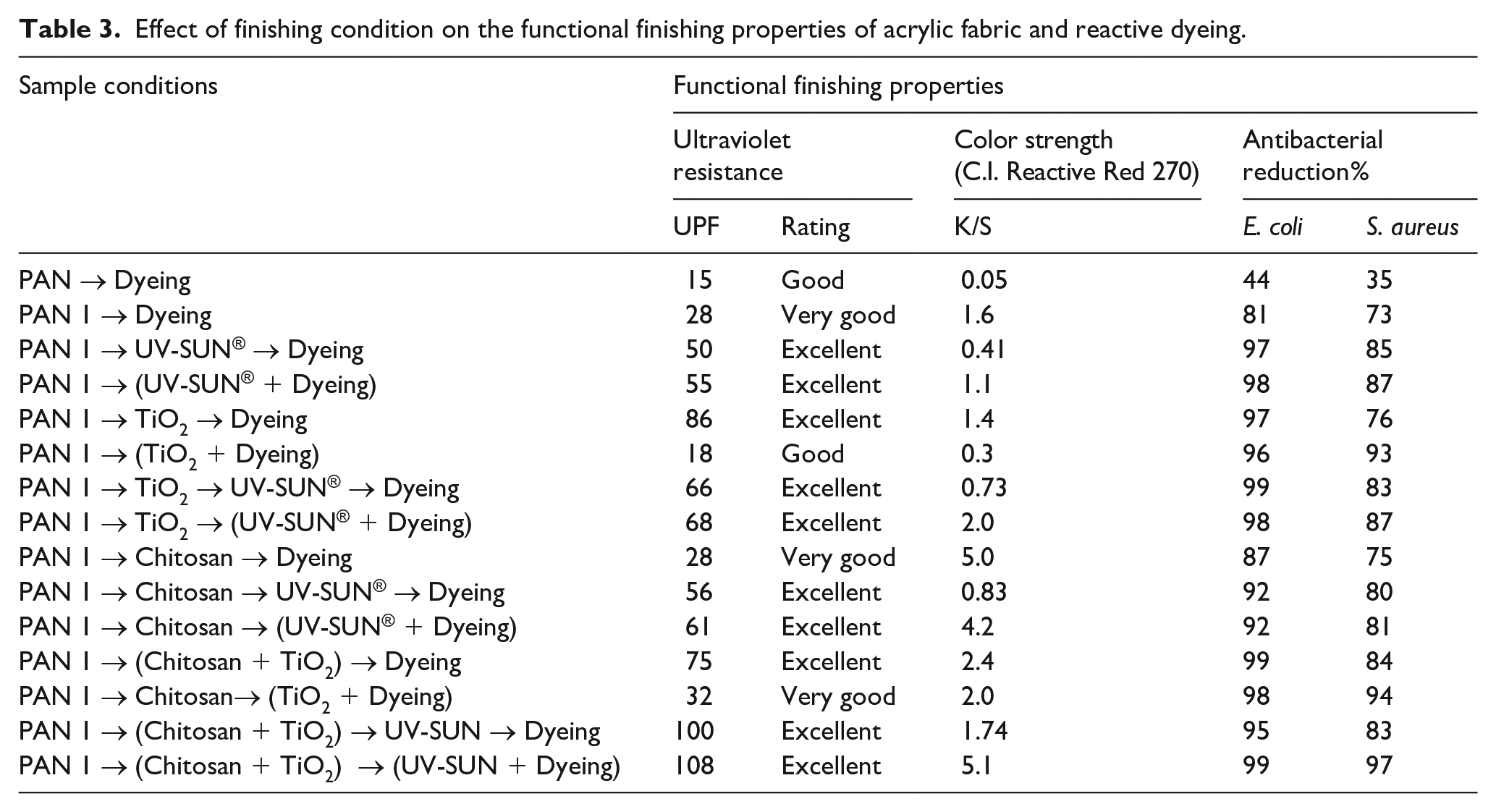
Dyeing PAN1with C.I. Reactive Red 270 post titania NPs finishing (i.e. two steps finishing and dyeing) showed higher color strength than simultaneous dyeing and finishing. The interaction between the reactive sites of titania NPs with the dye molecules in the simultaneous operation decreases the migration of dye molecules to the fabric surface, so the color strength of dyed PAN1 in the simultaneous process is lower than that K/S of dyed post finishes with titania NPs. Since the increased surface contribution of titania terminal hydroxyls increases surface potential through the dispersion in water solution. 34 Figure 3 shows the structure of the UV absorber based on oxalanilides. In the case of UV-SUN®, simultaneous finishing and dyeing improve the color strength of dyed PAN1 fabric slightly when compared to that finished with a UV absorber post dyed, which may be due to lower interaction between the organic UV absorber and dye molecules in the simultaneous process, which enhances the color depth.

UV absorber based on oxalanilides.
The hybrid composite chitosan/titania NPs (as shown in reaction scheme 2) in the finishing bath formulation pre-dyeing enhances the color strength of the fabric (2.4) compared with that finished with titania only (1.4), which may be because of the presence of chitosan functional groups. Simultaneous finishing (organic UV absorber) and dyeing for PAN fabric finished with chitosan will enhance the color strength (4.2) compared with that finished with organic UV absorber and dyeing stepwise (K/S 0.83). Finishing PAN fabric with a hybrid composite chitosan-titania NPs then applied simultaneous finishing (UV absorber) and dyeing enhanced the color strength (5.1) significantly. The citric acid was used in the finishing bath as a binder and a crosslinking agent. 36 It is worth mentioning that citric acid is a crosslinking agent for chitosan through the esterification of its carboxylic groups with hydroxyl groups of chitosan. Besides the formation of salt linkage between carboxylic groups of citric and chitosan amino groups and fix titania on the acrylic fabric surface as shown in reaction scheme 3. 36
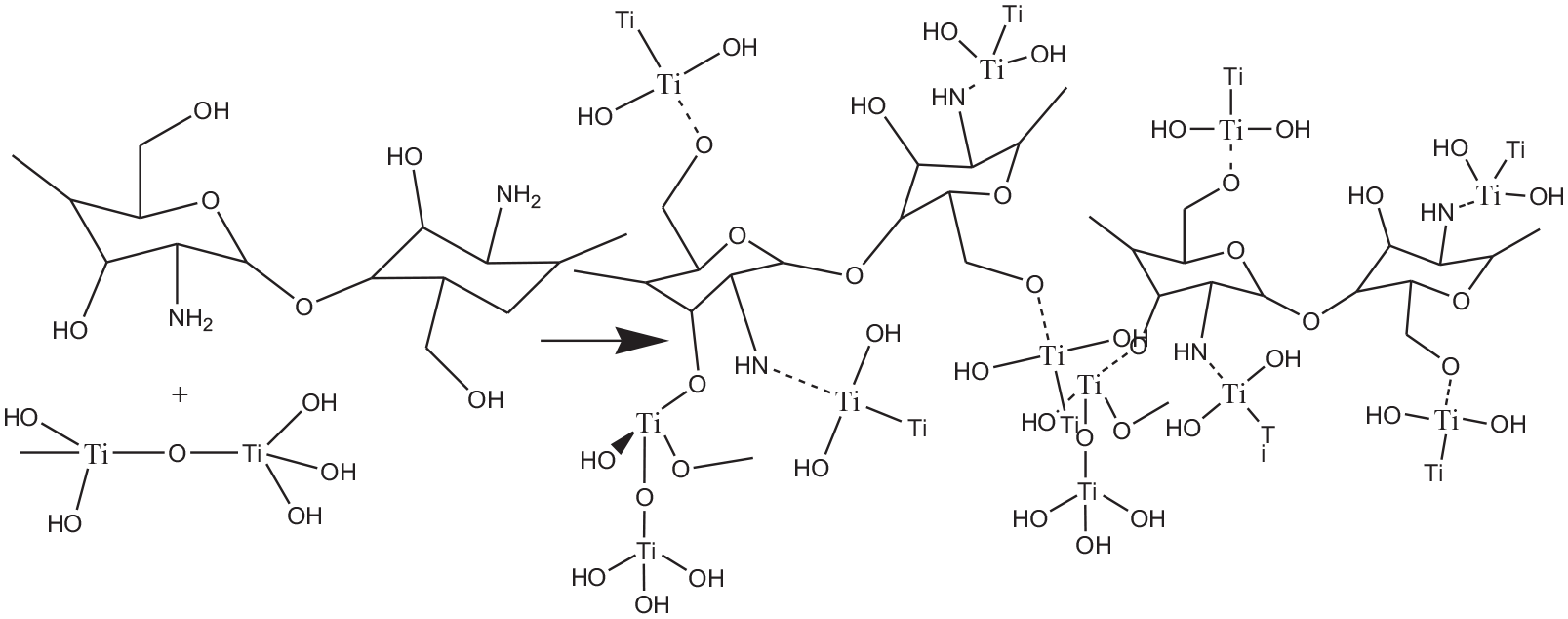
The proposed interaction between the titania NPs and chitosan.

Mechanism of crosslinked chitosan with CA.
The data presented in Table 4 showed the effect of finishing bath formulations on the color strength (K/S) of PAN fabrics dyed with C.I. Basic Violet 14. The affinity of PAN to basic dye results from the electrostatic attraction between the dye positively charge group to the negative charge group of the fabric. Alkali hydrolysis increases the number of negative charge groups due to the transformation of nitrile groups of PAN into amid and/or carboxylic groups, thus enhancing the color strength of the PAN1 (7.2) compared with that of the parent one PAN (1.3). Titania NPs’ finishing enhances the color strength of PAN1 fabric dyed with basic dye (21.1) compared with that finished with chitosan only (11.1). It can be stated that may be a result of the electrostatic attraction for the hydroxyl groups of titania NPs surfaces and the reactive sites of the basic dye. On the other hand, finishing with chitosan only covered the surface with the protonated amino groups, which repulsed the basic dye’s reactive groups, decreasing adsorption on the fabric surface.
Effect of finishing bath formulation on the functional properties of PAN fabric and basic dyeing.

Organic UV absorber enhances the color strength of PAN1 fabric dyed with basic dye (12) compared with that finished with chitosan only (11.1) as well as PAN1 (7.2). Hybrid composite PAN1 finishing followed by simultaneous UV absorber finishing and dyeing enhance the color strength (18) but is lower than finishing with titania NPs only (21.1). Finishing PAN1 with the hybrid composite followed by finishing with organic UV absorber then dyeing enhances K/S (12.4) compared with that finished with chitosan and UV absorber, but lower than that finished with titania only (21.1). It may be due to the interactions of the chitosan and titania NPs in the finishing step and the UV absorber with the dye in the simultaneous finishing step.
Antibacterial activity
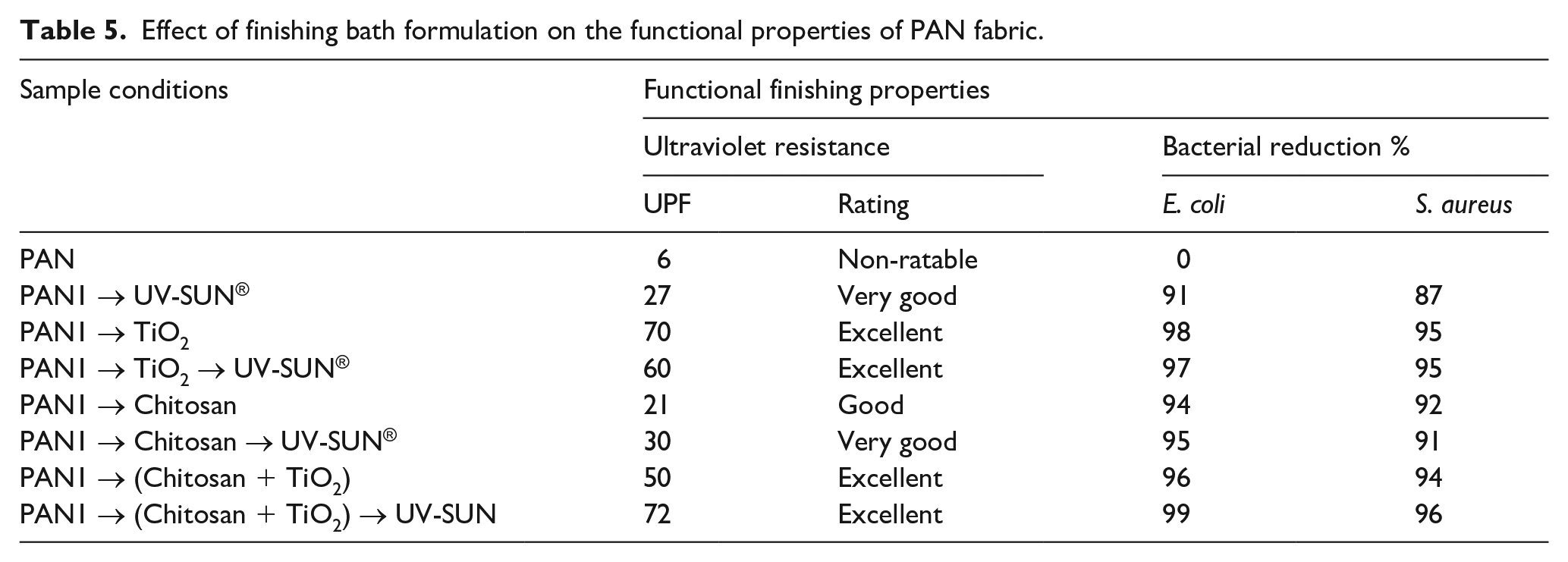
Effect of finishing on antibacterial activities against E. coli and S. aureus presented in Table 5. There is a remarkable enhancement in the percentage of bacterial reduction after finishing with chitosan, titania NPs, and their hybrid composite. Application of a hybrid composite finishing followed by UV absorber significantly enhances the antibacterial activities (99% against E. coli and 96% S. aureus). It may result from the interaction of polyanions on the surface of the bacteria cell and the finishing agents; additionally, surface negativity of Gram −ve bacteria is higher than that of Gram +ve bacteria. 37
Effect of finishing bath formulation on the functional properties of PAN fabric.
Antimicrobial mechanisms of chitosan could be assumed to be analogous to other cationic biocides through six steps: (i) adsorption of the bacteria cell; (ii) diffusion to the cell wall; (iii) absorption to cytoplasmic membranes; (iv) destruction of cytoplasmic membranes; (v) leakage of cytoplasmic components; (vi) death of bacteria cell. There is tremendous controversy about chitosan’s inhibitory activity against various microorganisms. The accepted mechanism could be as follows: primary amino groups’ positive charges interact with the negatively charged microbial cell. This interaction significantly alters the cell surface and cell permeability, leading to intracellular leakage. 38
Different mechanisms were proposed for antimicrobial activities of titania NPs. The first is the electrical attraction between the metal oxide with a positive charge and the microorganism with a negative charge. This attraction oxidized microorganisms and caused their death. 39 The second mechanism is the de-activation of the cellular enzymes and DNA of the bacteria cell due to interaction between the electron-donating groups like thiols, carbohydrates, amides, indoles, and hydroxyls, etc., metal oxide NPs which act as electron-accepting groups. This interaction produces holes in bacterial cell walls, which lead to permeability and death of the cells. The third mechanism is the advantages of the nanoscale of titania NPs which enable the most accessible penetration to microbe cells leading to a rapid deterioration effect on the intracellular system of the microbes. The fourth mechanism and the major one for titania NPs activities are producing the reactive oxygen species (ROS) on its surface when exposed to light at specific wavelengths (photocatalysis). 40 Many studies have shown that Gram-positive bacteria are more resistant to NPs bioactivities. The interpretation of this consequence is assumed mostly by various cell walls.
Antibacterial activities of finished PAN1 were evaluated in terms of the reduction percentage of bacteria growth. The data presented in Table 4 revealed the best results obtained with the hybrid composite followed by simultaneous UV absorber finishing/dyeing. The maximum reduction was 98% E. coli and 95% for S. aureus. The reactive dye used can attain the modified fabric antibacterial activities against E. coli and S. aureus. 41
It is worth mentioning that finishing with a commercial UV absorber attained PAN1 antibacterial activities and UV resistance as there is a noticed improvement in the antibacterial activities of PAN1. Hybrid composite is a promising material for a wide variety of applications such as wound care. It has higher antibacterial activities than individual components; these results agree with other research results. 6
The possible synergistic effect of chitosan and titania NPs indicates that antimicrobial activities are correlated with the electrostatic interactions of cell membranes with the composite and accompanied by a photo-generated reactive oxygen species, resulting in cell death. Another suggestion demonstrates that chitosan and titania NPs films promote bacterial cell destruction, membrane deformation, and significant cell-wall changes. Other research indicated that chitosan-titania film showed moderate antibacterial activity against S. aureus and no activity against E. coli. 41 The final composite exhibited antibacterial activities against E. coli and S. aureus. The bonding between the polymer and nanoparticles depends on the linkage between the interfaces of them. 6
Ultraviolet resistance
Most of the sun protection products in the market are composed of inorganic and organic UV filters. As much as some sunscreen products have intense protection in one particular area of the UV spectrum, small amounts of different UV filters must be combined to provide the required sun protection factor (SPF). 12
The primary function of ultraviolet protection finishing is absorbing dangerous UV radiation and its conversion to harmless thermal energies. This UV protection mechanism refers to organic compounds that can absorb UV energy and turn it effectively into kinetic and thermal energy without photo deterioration. Organic UV absorber is uncolored aromatic molecules with a double bonding absorbing ultraviolet energy in 290–360 nm wavelengths, stimulating the molecules. UV energy is transferred to vibration energy by the UV absorber, and heat energy is released into the surrounding atmosphere when returning to normal conditions. Ultraviolet absorbers shield textile fibers against chain fracture and interconnection reactions caused by photo-oxidation of polymers. 42
The ultraviolet protection factor (UPF) is used as a basis for UV protective textiles. This research is concerned with applications of finishing agents to achieve the desired UV protection. The effect of finishing and dyeing on UPF of PAN fabric is shown in Tables 3 to 5. The results showed that the finishing agents’ as titania NPs, organic UV absorber, and the hybrid composite enhanced the protection against UV. The enhancement in UPF value (70) was remarkable in the case of finishing with titania NPs (protects from damage by scattering, reflecting, and/or absorbing UV radiation). The hybrid composite showed excellent protection against UV radiation and suggested the usage of such composite for that fabric’s direct contact with the body.
Tables 3 and 4 showed that dyeing enhances UPF of the fabrics and this reflected the considerable influence of the dye on the permeability of ultraviolet radiation within textile material. Some factors that affect the UV resistance of the dyes include (type, chemical structure, absorptive groups), color strength, the uniformity of dyed fabric, and the additives used in the dyeing bath. The absorption band of many dyes extends to the ultraviolet spectral region making those dyes act as UV protective agents. 24
In addition, the tables showed that finishing PAN1 fabric with UV absorber followed by dyeing and simultaneous UV absorber finishing/dyeing increased the UPF values for C.I. Reactive Red 270 and C.I. Basic Violet 14. Hybrid composite chitosan-titania finishing followed by simultaneous UV absorber finishing/dyeing enhances UPF significantly (108).
Mechanical properties of the finished fabrics
Data presented in Table 6 showed that alkaline pretreatment enhanced the wettability and the air permeability of the fabric, which could be attributed to the creation of reactive sites on the fabric surface. It allows the fiber to get more wettability and changes in the fiber surface morphology due to such treatment. The finishing increases the air permeability of the fabric as well as wettability. The bursting strength of PAN fabric is affected by alkali pretreatment as well as finishing. The strength properties of the parent PAN decreased, causing some fiber damage through the process. However, even after that reduction in bursting strength of the finished PAN fabric is still has an acceptable level to be used as a textile with suitable mechanical properties.
Effect of processing on the physic-mechanical properties of PAN fabric.

Durability properties to washing
The data listed in Table 7 show the effect of repeated washing on the performance and sustainability of the fishing with different formulations on UV blocking and antibacterial properties after five washes. The samples are subjected to the washing test after finishing under different circumstances. The UPF and antibacterial properties show a minor decrease after five cycles. The reduction might be attributable to the removal of unfixed and physically connected finishing agents.
Effect of finishing bath formulation on the functional properties of PAN fabric after five washes.

Fastness properties
The light and wash fastness of dyed PAN1 with C.I. Basic Violet 14 was evaluated in Table 8. The obtained results demonstrate that PAN’s light and wash fastness findings (pre-finished post-dyed) using basic dye are better than those of are unfinished fabrics (control). This might have been because the interaction arises between both the finished fiber and the dye. Moreover, improved fixation was observed in the washing fastness ratings.
Effect of processing on the light and wash fastness properties of PAN fabric dyed with C.I. Basic Violet 14.

Alt: alteration or change in color; SC: staining on cotton; SW: staining on wool; SP: staining on polyester.
SEM and EDX
Scan electron microscopy was used to examine the surface morphology of the PAN fabrics before and after alkali pretreatment and finishing, as shown in Figure 4(a) to (e). The fabric was analyzed elementally using the EDX spectrum technique, as shown in Figure 5(a) to (d). The surface of the blank sample is smooth, and that of the pretreated is rough (Figure 4(b)) indicated that such alkali pretreatment causes cracks and grooves on the fabric surface, getting more accessible to the finishing agents and other wet processes. Chitosan and organic UV absorber display noticeable coating of the fabric surface and no pore void, cracks observed. Introducing nanoparticles in the finishing bath formulation leads to the distribution of the NPs on the surface of the finished fabric, as shown in Figure 5(a) to (d), and the particles were observed and evaluated by EDX.

SEM analysis of acrylic fabric: (a) PAN fabric, (b) PAN → Alkali hydrolysis (PAN1), (c) PAN1 → Chitosan, (d) PAN1 → UV absorber, and (e) PAN1 → (Chitosan + UV absorber).

SEM and EDX analysis of partially hydrolyzed acrylic fabric: (a) PAN1→ Titania NPs, (b) PAN1 → (Chitosan + Titania NPs), (c) PAN1→ (UV absorber + Titania NPs), and (d) PAN1 → (Chitosan + Titania NPs) → UV absorber.
Conclusion
The study sought concurrent UV blocking and antibacterial action to protect different types of apparel (daily use, athletic, medical). This work proposed a unique method for producing a sustainable surface functionalization with versatile properties on the surfaces of acrylic fabric. Nano TiO2 particles are incorporated in a chitosan polymeric matrix during multiple chemical finishing processes and using the organic UV absorber. This multifunctional finishing considerably increases the acrylic fabric’s durability and performance in a variety of applications. The presence of nano TiO2 particles on the surface of the treated acrylic fabrics was confirmed by FTIR spectra, SEM, EDX. PAN pretreated with sodium hydroxide has reactive sites available for functional finishing. Different finishing bath formulations were used to acquire PAN functional properties like UV blocking and antibacterial (essential for sportswear, military uniforms, covering fabric). Characterization results indicated that groups of NH2, COOH, and CONH are involved in the finishing process. For the synergetic purpose, a combination of organic and inorganic UV blockers and bioactive biopolymer and inorganic bioactive NPs as a composite material, in the finishing bath both simultaneous finishing and dying or in a two-step. SEM revealed that the alkali pretreatment causes cracks and grooves on the fabric surface, getting more accessible to the finishing agents and other wet processes. Chitosan and organic UV absorber display noticeable coating of the fabric surface and no pore void, cracks observed. Introducing nanoparticles in the finishing bath formulation leads to the distribution of the NPs on the surface of the finished fabric. On the other hand, finishing PAN1 material with UV absorber followed by dyeing and simultaneous UV absorber finishing/dyeing increased the UPF values to a great extent. Hybrid composite chitosan-titania finishing followed by simultaneous UV absorber finishing/dyeing enhances UPF significantly (108). The finishing increases the air permeability of the fabric material and its wettability, but the strength properties of the parent PAN decreased, causing some fiber damage through the process. However, even after that reduction in bursting strength of the finished PAN fabric is still has an acceptable level to be used as a textile with suitable mechanical properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
