Abstract
Given the increasing demand for multifunctional products with programmable and biological functionalities, this study aims to develop bioactive cellulose materials using a cost-effective, eco-friendly process. Plant extracts are well-suited for textile processing due to their accessibility, non-toxicity, biocompatibility, and eco-friendly nature. In this study, viscose fabrics were treated with aqueous solutions of plant extracts from five selected species: industrial hemp (Cannabis sativa L.), mountain germander (Teucrium montanum L.), herb-robert (Geranium robertianum L.), lady’s mantle (Alchemilla viridiflora Rothm.), and pomegranate (Punica granatum L.). Periodate oxidation and chitosan deposition were used to enhance fabric properties. The antimicrobial activity of viscose fabrics treated with plant extracts was assessed using the agar diffusion method on Gram-positive microorganisms: Staphylococcus aureus (clinical isolate and strain ATCC 25923), as well as Gram-negative microorganisms: Escherichia coli strain ATCC 25322 and a clinical isolate of Pseudomonas aeruginosa. All samples treated with plant extracts exhibited antimicrobial activity, evidenced by inhibition zones or contact activity against the tested microorganisms. Samples treated with pomegranate peel and hemp extracts exhibited the highest efficacy against Gram-positive bacteria (inhibition zone of 20 mm), while those treated with pomegranate peel extract showed the highest activity against Gram-negative bacteria (inhibition zone of 17.5 mm). The antioxidant activity was evaluated using the 2,2-diphenyl-1-picrylhydrazyl reagent; the results indicated significant antioxidant effects across all viscose fabrics functionalized with plant extracts. The highest antioxidant activity was recorded in samples treated with extracts of lady’s mantle at 80%, teucrium montanum at 76%, and pomegranate peel at 70%. Interactions between hemp and pomegranate extracts were further investigated using in silico methods to gain insights into the types of interactions and to identify key functional groups/atoms involved in complex formation between chitosan and extract constituents. Results indicate that the examined plant extracts could serve as suitable replacements for synthetic agents in textile functionalization.
Keywords
Introduction
The development of multifunctional textile materials is a significant trend in the modern textile industry, with increasing interest in materials that provide additional properties such as antimicrobial and antioxidant protection. 1 Multifunctional textiles enhance usability, safety, and health beyond their primary function. Traditional antimicrobial protection for textiles has historically involved the use of chemical substances, such as various inorganic salts, phenolic compounds, antibiotics, iodine and formaldehyde derivatives, nitro compounds, and amines. The primary disadvantage of these compounds is their toxicity and poor biodegradability, which are unacceptable from an environmental protection standpoint. 2 With the increasing emphasis on ecological and sustainable chemical technologies, there is growing interest in cellulose-based textile materials and cellulose itself, which is a renewable polymer. 3
The unique chemical structure of cellulose, along with its properties such as hydrophilicity, biocompatibility, biodegradability, non-toxicity, and the reactivity of its hydroxy groups, makes it an excellent base material for various applications. 3 Viscose fibers, regenerated from cellulose, are especially appealing due to their adaptability and compatibility with eco-friendly finishing processes. 4
Oxidation of cellulose fibers is a crucial step in textile preparation, activating reactive functional groups on the fiber surface that significantly increase the binding capacity for bioactive compounds like chitosan. This process creates aldehyde groups on cellulose fibers, which then form stable covalent bonds with the amino groups in chitosan, ensuring long-lasting functionalization. 5
Chitosan, a natural polymer abundant in amino and hydroxy groups, is increasingly valued in the textile industry for its biological activities, which enable the creation of textiles with strong antimicrobial properties. Its versatility as a functionalizing agent becomes especially clear when combined with oxidized cellulose materials like viscose, where it forms durable chemical bonds that enhance and extend the protective qualities of the fabric. This makes chitosan an excellent carrier for plant extracts, promoting their stable incorporation into textile fibers. Chitosan’s unique structure, with its hydroxy, amino, and N-acetyl groups, not only provides bioactivity but also allows it to form strong molecular interactions with cellulose.6,7 When applied to oxidized viscose fabrics, chitosan enhances the fiber’s surface with functional groups, enabling the formation of Schiff bases through covalent bonds. This stabilizes chitosan within the fabric, enhancing its durability and antimicrobial effectiveness over time. 7 The adsorption of chitosan, chitosan nanoparticles, and iodine-incorporated chitosan nanoparticles on unmodified and oxidized viscose fabric was also studied. Satisfactory antioxidant and antimicrobial activity against Staphylococcus aureus, Escherichia coli, and Candida albicans was achieved on fabric samples with incorporated iodine. 5 The antimicrobial properties of chitosan have been investigated for medical applications. Its effectiveness has been successfully demonstrated in the wound healing process, reducing discomfort, and preventing complications in infected skin areas of individuals with diabetes.8,9
Ecological textile finishing is increasingly moving toward techniques that incorporate natural biopolymers and plant extracts, aiming to reduce environmental impact and produce safer consumer products. These textiles provide an eco-friendly alternative to traditional synthetic antimicrobial agents, offering promising multifunctional properties that suit a broad range of applications.10,11
Plant extracts have attracted significant research interest for their potent antimicrobial and antioxidant properties, largely due to their high polyphenolic content. 12 The structural diversity of polyphenols allows them to disrupt bacterial cells, altering their structure and metabolism. 13 By incorporating extracts from plants such as aloe vera (Aloe vera (L.) Burm.f.), eucalyptus (Eucalyptus globulus Labill.), basil (Ocimum basilicum L.), turmeric (Curcuma longa L.), and tulsi (Ocimum tenuiflorum L.), cellulose-based textiles have demonstrated strong antimicrobial properties while also preventing oxidative damage on fabric surfaces. 14 To improve the stability of these extracts on cellulose, pretreatments like oxidation are applied, which enable stronger covalent bonding and enhance the durability of the functional properties. 15
Studies have also explored combining chitosan with various plant extracts, such as rosemary (Salvia rosmarinus Spenn.), galangal (Alpinia galanga (L.) Willd.), 16 and onion peel (Allium cepa L.), 17 as well as with essential oils, revealing synergistic effects on antimicrobial activity. In addition to interacting with cellulose, chitosan also binds with compounds from plant extracts, with the nature and binding affinities of these interactions determining the therapeutic properties of these textiles. For instance, chitosan-curcumin functionalized fabrics exhibited an additive effect, characterized by enhanced antimicrobial properties compared to fabrics functionalized solely with chitosan or curcumin. Furthermore, all chitosan-curcumin functionalized fabrics demonstrated significant antioxidant activity, in clear contrast to chitosan-functionalized fabrics, which displayed only minimal antioxidant activity. 18 Moreover, viscose fabrics treated with ethanol-based extracts from plants like anise (Pimpinella anisum L.), fennel (Foeniculum vulgare Mill.), lavender (Lavandula latifolia Medik.), and sage (Salvia officinalis L.) exhibited strong antimicrobial effects against S. aureus and C. albicans, alongside substantial antioxidant activity. 19 Furthermore, grape seed and skin extracts have shown antimicrobial and antioxidant effects when applied to cotton knits. 3
In this study, viscose fabrics were treated with aqueous extracts from five selected plants: industrial hemp (C. sativa), mountain germander (T. montanum), geranium (G. robertianum), lady’s mantle (A. viridiflora), and pomegranate peel (P. granatum), all of which have demonstrated biological activity. The plants used to obtain extracts for the functionalization of viscose fabric were selected due to their easy availability and well-documented antimicrobial and antioxidant properties.20 –25 Geranium, 26 lady’s mantle, 25 and mountain germander are naturally available in their native habitats, while the harvesting of mountain germander on Mount Ozren in Bosnia and Herzegovina has been included on the UNESCO World Heritage List. 27 Hemp 28 and pomegranate 23 are cultivated on private properties, and thus their availability is not limited to wild habitats. Pomegranate peel is a by-product of fruit consumption, which is another reason why its extract was used for the functionalization of viscose fabric. Since hemp has so far primarily been used for the production of textile fibers, this research demonstrates an additional application of this plant. 28
To enhance biological activity, the fabrics were pretreated by combining sodium periodate oxidation followed by chitosan deposition. Periodate oxidation of cellulose is characterized by the specific cleavage of the C2-C3 bond in the glucopyranose ring, resulting in the formation of two aldehyde groups per monomer unit. 29 This reaction creates additional sites for covalent bonding of chitosan through the formation of a Schiff base.19,30 A significant advantage of periodate over other oxidizing agents is that it minimizes degradation, thereby preserving the mechanical and morphological properties of the initial material. 29 Molecular docking simulations were conducted first to evaluate binding affinities and interactions between chitosan and compounds from selected plant extracts. Theoretical modeling aligned well with in vitro results in sorption processes, 29 suggesting that similar in silico approaches could be used to estimate binding affinities of ligands from plant extracts. Antioxidant activity was then assessed using the diphenyl-picrylhydrazyl (DPPH) reagent. Finally, the antimicrobial activity of viscose fabric treated with plant extracts was determined using the agar diffusion method on Gram-positive microorganisms, including a clinical isolate S. aureus and the strain ATCC 25923, as well as on Gram-negative microorganisms, specifically the E. coli strain ATCC 25322 and a clinical isolate of P. aeruginosa. Our objective was to create a fabric suitable for use in medicine, specifically for wound healing and for single use. 31
Experimental
Materials
Acetic acid 99,8% (Lach:ner, Czech Republic), Sodium-periodate (Acros Organic 99% ), sodium acetate (Centrohem, Serbia), chitosan (powder, low molecular weight, 75%–85% deacetylated), methanol, 2,2 diphenyl-1-picrylhydrazyl (DPPH), Mueller–Hinton agar, sulfuric acid, boric acid, hydrochloric acid, copper (II) sulfate pentahydrate, sodium carbonate (Na2CO3) and Folin-Ciocalteu (FC) reagent (Sigma Aldrich).
Bleached, plain weave viscose (regenerated cellulose) fabric provided by IGR Agence Slovenia, was used as experimental material. The cellulose fabric structure was: yarn fineness (warp/weft) – 9,6/9,9 tex, warp/weft count – 40/35 cm−1, and surface mass – 80 g m−2.
Pre-treatment of viscose fabric (oxidation and chitosan deposition)
The viscose fabric samples underwent pre-treatment via oxidation using a 0.4% (w/v) sodium periodate solution at pH 4 in an acetate buffer. 32 Oxidation was carried out in the dark with constant stirring, for 60 and 120 min, with a liquor-to-fabric ratio of 1:50 (w/v). Upon completion of the oxidation time, the samples were rinsed with ice-cold water to stop the process and dried at room temperature. The oxidized viscose fabric samples were then immersed in a 1% (w/v) solution of low molecular weight chitosan, with a liquor ratio of 1:50 (w/v). Chitosan was dissolved in 2% acetic acid under stirring for 1 h at 60°C. The samples were processed in a water bath for 1 h at 60°C, then rinsed with 500 ml of distilled water and dried for 30 min at 60°C.
Preparation and application of plant extracts
The selected plant species were collected through various means: lady’s mantle (A. viridiflora) and geranium (G. robertianum) were sourced from natural habitats, pomegranate peel (P. granatum) and industrial hemp (C. sativa) from private farm fields, and mountain germander (T. montanum) from the commercial supplier “Galenfarm” (Gradiska, Bosnia and Herzegovina). Plant materials were ground and extracted in methanol (1:10 ratio) for 24 h at 20°C using ultrasound. Extracts were filtered, concentrated to a smaller volume using a rotary vacuum evaporator, and dried in an airstream at room temperature.
A dry extract weighing 1 g was dissolved in 100 ml of distilled water and used for further treatment of viscose fabric at a ratio of 1:50. The plant extract was applied to the chitosan-treated viscose fabric samples using a direct impregnation method for 120 min at a temperature of 20°C ± 2°C with constant stirring. Afterward, the samples were rinsed with distilled water and dried at room temperature. The sample labels are provided in Table 1.
Notation of the prepared samples.

Characterization methods
Determination of total polyphenol content for selected plant extracts and treated viscose fabric samples
A 100 μl sample was mixed with 750 μl of 10-fold diluted Folin-Ciocalteu reagent, vigorously shaken, and after 5 min, 750 μl of Na2CO3 solution was added to each sample. The mixtures were vigorously shaken again and incubated for 90 min in the dark at room temperature. Absorbance was measured at 725 nm against a blank. The blank was prepared identically, using 100 μl of solvent instead of the sample. Polyphenol content was expressed as the amount of gallic acid (GA) in mg/g of dry extract. 26
Fabric samples, cut to 25 × 25 mm, were placed in test tubes and soaked with 1.50 ml of 10-fold diluted Folin-Ciocalteu reagent. The samples were vigorously shaken, and after 5 min, 1.50 ml of Na2CO3 solution was added to each. Following shaking, samples were incubated for 90 min in the dark at room temperature. After incubation, the fabric was removed, and the absorbance of the remaining solution was measured at 725 nm against a blank. The blank contained a 1:1 mixture of Folin-Ciocalteu reagent and Na2CO3 solution. This procedure was based on a modified method from Ilić et al. 26 Polyphenol content was expressed as the amount of gallic acid (GA) in mg/g of fabric.
Determination of total nitrogen content
Nitrogen content in chitosan and the analyzed viscose fabrics was determined by the Kjeldahl method, 18 following ISO 937:2007. Digestion of the ground viscose fabric sample (1 g) was performed with concentrated sulfuric acid using copper (II) sulfate pentahydrate and potassium sulfate as catalysts. Following digestion, the free ammonia was distilled with an excess of boric acid solution. Nitrogen content in the analyzed sample was determined by titrating the resulting solution with hydrochloric acid in the presence of an appropriate indicator.
FTIR analysis
FTIR analysis was conducted in absorption mode, using attenuated total reflectance (ATR) on a Tensor 27 spectrometer by Bruker, across a spectral range of 400–4000 cm⁻¹ with a resolution of 4 cm⁻¹.
Antimicrobial activity
Antimicrobial activity was tested by the agar diffusion method. 19 Bacterial isolates of S. aureus (clinical isolate, ATCC strain 25923) were used as representatives of Gram-positive bacteria, while E. coli strain ATCC 25322 and a clinical isolate of P. aeruginosa represented Gram-negative bacteria. Bacterial isolates were inoculated onto Mueller-Hinton agar at a concentration of 0.5 McFarland, and fabric samples of 10 × 10 mm were applied. Samples were incubated for 24 h at 37°C, after which the inhibition zone for the analyzed fabrics was measured.
The minimal inhibitory concentrations (MIC) of pomengranate peel (PP) and hemp (H) extracts were determined by the broth microdilution method (according to the European Committee on Antimicrobial Susceptibility Testing, breakpoint tables for interpretation of MICs and zone diameters; version 14.0 (EUCAST) recommendation). Briefly, serial dilutions of PP and H were prepared in 99.5% methanol in concentrations from 1.25 to 0.001 mg/ml. Bacterial inoculum of S. aureus ATCC 25923 and E. coli ATCC 25322 was prepared in the final concentration of 5 × 105 CFU/ml. Muller Hinton broth (Liofilchem, Italy) was used as the testing medium. The MIC is defined as the lowest concentration of antimicrobial agent that inhibited growth at 35°C ± 2°C after 24 h of incubation in aerobic conditions. We also performed MIC testing for disolvent (99.5% methanol) to distinguish methanol influence to bacterial killing. Methanol was diluted in concentration from 25% to 0.05%.
Antioxidant activity
The neutralization of DPPH was evaluated using methods previously described in the literature. 19 The fabric samples being tested (with adsorbed chitosan, with chitosan and extracts, and the control, untreated sample) of appropriate dimensions (25 × 25 mm) were placed in test tubes and covered with 3.5 ml of freshly prepared diphenyl-picrylhydrazyl (DPPH) reagent in methanol (1.0 × 10⁻⁴ mol l⁻¹). The reaction was allowed to proceed for 30 min at room temperature (20°C) in the dark. After this, the absorbance of the samples was measured at 517 nm using methanol as the blank. The inhibition activity of DPPH radical formation was calculated using the formula:
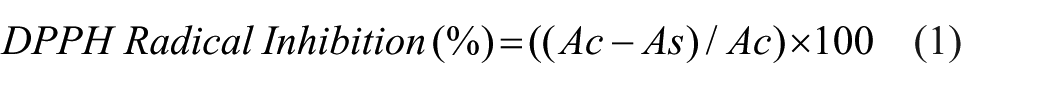
where: Ac is the absorbance of the control (untreated fabric sample),
As is the absorbance of the fabric sample with the adsorbed extract.
Computational simulation
The structures of punicalin, punicalagin, cannabidiol, and cannabidiolic acid were obtained from the PubChem database and optimized using Yasara Structure software to ensure stable conformations for docking. The chitosan (high T) extended structure was downloaded from PolySacDb 33 and underwent energy minimization in Yasara Structure to achieve a stable conformation suitable for docking. Molecular docking studies were then conducted using Yasara Structure software, employing the Vina algorithm in default mode, which utilizes standard parameters provided by Yasara Structure. 34 The binding site for docking was defined based on the known or predicted active site of the chitosan structure, and the grid box dimensions were set to cover the entire active site region to explore all potential binding modes. Each compound (punicalin, punicalagin, cannabidiol, and cannabidiolic acid) was docked individually to the chitosan (high T) extended target, with multiple docking runs performed to identify the most favorable binding conformations and ensure reproducibility. The docked complexes were analyzed using Discovery Studio software to investigate interactions between the compounds and the chitosan target, identifying hydrogen bonds, hydrophobic interactions, and other non-covalent interactions. Binding affinities and docking scores obtained from Yasara Structure were evaluated to determine the most potent inhibitors, and the docking scores and interaction profiles were compared among the tested compounds to understand the key interactions responsible for binding affinity. 35
Results and discussion
Functionalization of viscose fabric with plant extracts
The fabric samples treated with plant extracts exhibited a noticeable color change compared to the untreated white fabric, as can be seen in Figure 1, and emitted a pleasant scent.

Fabric samples before and after treatment with plant extracts (sample labels are provided in Table 1).
The color change observed in the fabric treated with plant extracts (Figure 1) serves as a visual confirmation of the interaction between cellulose, chitosan, and the plant extract. Plant extracts are rich in bioactive compounds, with polyphenols being the most prevalent class of secondary biomolecules. These molecules are characterized by the presence of at least one aromatic ring, substituted with at least one free or modified (ether, ester, or glycosidic) hydroxy group. Polyphenols are classified by structural complexity and biosynthetic origin, and based on the number of phenolic rings and the elements linking them, they are categorized into flavonoids, stilbenes, coumarins, tannins, and phenolic acids. 36 The chemical structures of the most abundant compounds identified in the tested samples are presented in the Supplemental Material (Figures S1–S5).
The total polyphenol content of the selected plant extracts is presented in Figure 2; the highest value was obtained for pomegranate peel extract (PP) and mountain germander extract (MG), with 12.48 mg gallic acid equivalents (GAE) per gram of dry extract. Hydrolyzable tannins are the most abundant polyphenols in pomegranate, including ellagitannins, gallotannins, and esters. These compounds, concentrated in the peel and membranes of pomegranate, contribute significantly to its antioxidant activity, with punicalagin and punicalin being the most important hydrolyzable tannins. 37 The antioxidant activity of pomegranate peel extract increases linearly with its total phenol content. 38 Bioactive components identified in mountain germander include polyphenolic compounds, especially phenolic acids, and flavonoids, as well as terpenoids, which are responsible for the antioxidant, antimicrobial, and other biological properties of its extracts and essential oils.39,40

Content of total polyphenols in selected plant extracts and viscose fabric impregnated with plant extracts.
The total polyphenol content of geranium (G) was found to be 9.64 mg GAE/g of dry extract. Herb geranium extract contains tannins, pectin, choline, flavonoids, phenols, and essential oils. The primary tannin in geranium species is geranin, and plants in this genus contain both hydrolyzable and condensed tannins. It has been shown that aqueous geranium extract exhibits antioxidant and antibacterial effects. 21 The polyphenol content of hemp extract (H) and lady’s mantle extract (LM) was found to be 7.48 and 8.06 mg GAE/g of dry extract, respectively. Besides cannabinoids, the most well-known and studied compounds in hemp, it synthesizes other secondary metabolites, including terpenes, hydrocarbons, nitrogen-containing compounds, carbohydrates, flavonoids, fatty acids, non-cannabinoid phenols, simple alcohols, aldehydes, ketones, acids, esters, lactones, and others. 41 Lady’s mantle contains tannin, bitter glycosides, saponins, flavonoids, and essential oils in its chemical composition. 42
Results of the total polyphenol content in viscose fabric samples treated with plant extracts (Figure 2) showed the highest value in the fabric sample treated with pomegranate peel extract, which had undergone a periodate oxidation pre-treatment for 60 min (sample PPCV60). Lower polyphenol levels were recorded in samples with hemp extract (samples HCV60 and HCV120; however, as with the pomegranate peel extract samples, a 60-minute oxidation pre-treatment was more favorable for polyphenol binding. For fabrics treated with mountain germander (samples MGCV60 i and MGCV120), lady’s mantle (samples LMCV60 and LMCV120), and geranium (samples GCV60 and GCV120) extracts, higher polyphenol values were achieved with a 120-min oxidation period. These findings suggest that pomegranate peel and hemp extracts are better suited for treating viscose fabric, as shorter oxidation times result in higher polyphenol binding from the plant extracts.
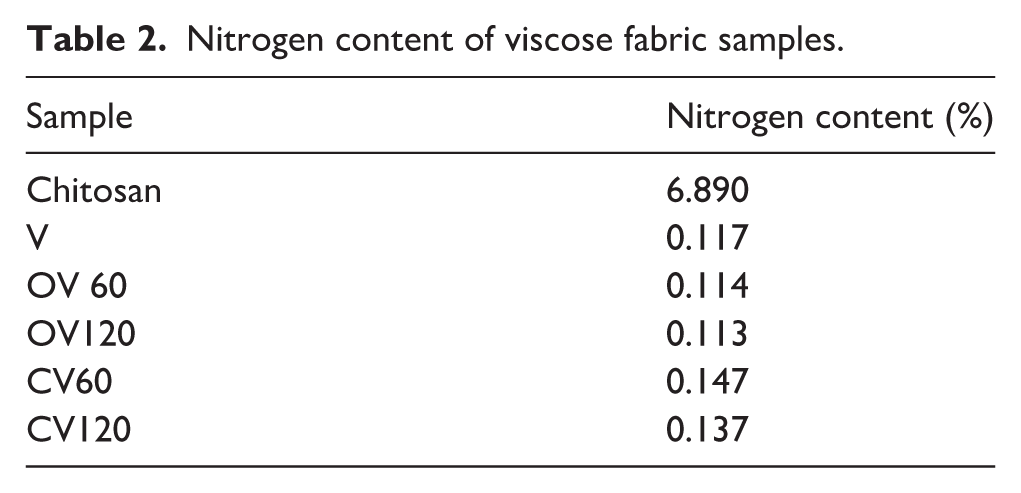
The pre-treatment of the examined samples involved periodate oxidation of the viscose fabric to create active sites for chemical bonding with the amino groups of chitosan. 5 Since nitrogen is present in chitosan, its content in the viscose fabric provides insight into the adsorption of chitosan in the treated textile samples. 18 The nitrogen content values (Table 2) indicate that a 60-minute periodate oxidation pre-treatment of the viscose fabric is more effective for chitosan binding. Higher nitrogen content obtained for the shorter oxidation time can be attributed to the difference in the size of the periodate ion and the chitosan molecule. With prolonged oxidation, the small size of the periodate ion allows it to penetrate the interior of the cellulose fibers and oxidize the glucose units both inside and on the surface of the cellulose fiber. In contrast, large chitosan molecules cannot access the aldehyde groups formed in the fiber’s small pores. Additionally, it is also possible that individual chitosan molecules react with multiple aldehyde groups, limiting the amount of chitosan bound despite the availability of free aldehyde groups capable of reaction. 43
Nitrogen content of viscose fabric samples.
The results in Table 3 show that the highest nitrogen content was obtained for fabric samples treated with pomegranate peel extract. For the sample labeled PPCV60, the nitrogen content reached 0.162%, while for PPCV120, it was 0.149%. The increase in nitrogen content on the fabric treated with pomegranate peel extract, compared to fabric samples treated only with chitosan (CV60 and CV120), is likely due to the presence of nitrogen-containing compounds from the plant extract. It is hypothesized that these compounds in the plant extract contributed to the increased nitrogen content in the fabric treated with lady’s mantle extract (LMCV60) as well, compared to fabrics treated with chitosan alone. For all other samples, the nitrogen content on the viscose fabric after treatment with plant extracts was lower than that in the samples treated only with chitosan. This decrease is likely due to processing in aqueous plant extract solutions, where chitosan reacts with poly/phenolic compounds in the solution and desorbs from the fabric, as it was initially physically deposited on the surface. 18
Nitrogen content of viscose fabric samples treated with plant extracts.

ATR-FTIR analysis was conducted on differently treated viscose fabrics to investigate changes in the surface chemistry of viscose/cellulose fibers and postulate possible interactions between cellulose, chitosan, and plant extracts. On the FTIR spectra of the samples (Figure 3(a) and (b)) of unmodified and oxidized viscose fabrics, characteristic peaks of cellulose can be observed. The peak at 890 cm−1 corresponds to the C-O-C stretching vibration of the glycosidic ring or deformation at the C1 atom in cellulose II. The peak at 1018 cm−1 corresponds to the C-O stretching of cellulose II, while the peak at 1156 cm−1 is attributed to the asymmetric stretching of the C-O-C glycosidic bond in the cellulose molecules. 44 At 1366 cm−1, a peak is recorded, originating from the symmetric C-H bending vibration. The peak observed at 1638 cm−1 results from the presence of adsorbed water, and the appearance of a “shoulder” in the spectra for the OV60 and OV120 samples can be explained by the presence of aldehyde groups formed during the oxidation process. The spectrum clearly shows that the presence of the “shoulder” is more pronounced in the OV60 sample, confirming that oxidation for 60 min is more effective in terms of aldehyde group content. The peak at 2890 cm−1 represents C-H stretching in cellulose II and amorphous cellulose. The interval between 3000 and 3600 cm−1 is attributed to the presence of OH groups involved in hydrogen bonding within cellulose. 30

FTIR spectra of viscose fabric samples at all stages of treatment for: (a) oxidation time 60 min and (b) oxidation time 120 min.
Due to the structural similarities between cellulose and chitosan, the FTIR spectra of viscose fabric treated with chitosan show peaks characteristic of both polymers with minor modifications. The shift at the 890–894 cm−1 wavenumber originates from the presence of chitosan. The band at 989 cm−1 corresponds to the C-O stretching vibration of the C6 atom, while the shift to 995 cm−1 (Figure 3(b)) indicates that the OH group on the C6 atom of the cellulose molecule interacts with chitosan during deposition, likely through hydrogen bonding. Characteristic chitosan peaks are observed at 1560 cm−1 (stretching vibration of amino groups), 1590 cm−1 (N-H bending vibration of amide II), and 1651–1654 cm−1 related to amide I. 32 These peaks are difficult to distinguish because they are located in the spectral region where the broad peak of adsorbed water at 1640 cm−1 overlaps their signal. For the CV60 and CV120 samples, changes in the appearance of the “shoulder” at 1640 cm−1 are observed compared to the OV60 and OV120 samples, due to the formation of hydrogen bonds between cellulose and chitosan. Similar changes in the “shoulder” appearance were identified for samples treated with plant extracts. These changes are most pronounced for the GCV60 and PPCV120 samples and are a result of the reaction between chitosan and phenolic groups from the plant extract (aromatic ring vibration corresponds to the 1500–1600 cm−1 interval). Furthermore, peaks originating from polyphenolic compounds may overlap with cellulose signals, as they appear in the same regions as cellulose. It can be assumed that the most efficient binding of the plant extract to the viscose fabric occurred in the GCV60 and PPCV120 samples, as confirmed by the excellent antimicrobial performance results (Table 4) for both samples. The FTIR spectral band in the wavenumber range of 3000–3500 cm⁻¹ is attributed to O–H stretching vibrations, which in this case originate from phenolic compounds and flavonoids present in the plant extract. Given that the spectra of the GCV60 and PPCV120 samples lack peaks within this region, it is assumed that hydrogen bonding has occurred between the hydroxy groups and the amino groups of chitosan. 45 The formation of hydrogen bonds is also observed in the 3000–3600 cm−1 range, corresponding to O-H and NH2 stretching vibrations, which are slightly shifted to higher wavenumbers and show lower peak intensity.17,19,46 The fact that interactions between the viscose fabric treated with chitosan and the plant extract are based on hydrogen bonding is further confirmed by computational simulations in this study.
Antimicrobial activity of fabric samples at all stages of treatment.

CI – inhibition in contact.
Functional properties
Plant polyphenols exhibit antimicrobial action through interactions with bacterial cell wall proteins, energy mechanism inhibition, DNA damage, changes in cytoplasmic function and membrane permeability, and inhibition of nucleic acid synthesis in bacterial cells. The inhibitory mechanism depends on the polyphenol structure and the bacterial type. 13 To achieve the desired biological activity, bioactive compounds must be present at an appropriate concentration, and a sufficient number of active sites available for the chemical reaction should be present. 36 Previous research shows that chitosan has antimicrobial effects, 30 and it has been found that adding substrates such as plant extracts enhances synergistic action, especially against G− bacteria, as chitosan alone is more effective against G+ bacteria. 47
To reproduce the condition of the fabric application in wound treatment, we selected the agar diffusion method to assess the antimicrobial efficacy of the extracts. It is crucial to demonstrate that the extract molecules can diffuse from the fabric into wounds in bactericidal concentrations. 31
For testing the antimicrobial activity of treated viscose fabric samples, bacterial isolates of S. aureus (clinical isolate, strain ATCC 25923 as a representative of G+ bacteria), as well as E. coli strain ATCC 25322 and a clinical isolate of P. aeruginosa (representing G− bacteria), were used. The results (Table 4) indicate that viscose fabric samples with chitosan exhibit antibacterial action in the form of contact inhibition against both G+ and G− bacteria. The most intense antimicrobial effect was observed in samples with pomegranate peel extract, showing a clear inhibition zone against G+ bacteria (an inhibition zone of 20 mm, Figure 4) and G− bacteria as well (an inhibition zone of 17.5 mm, Table 4). This result aligns with the high total polyphenol content from the pomegranate peel extract in these samples (Figure 2). The pretreatment duration of viscose fabric had no significant impact on antimicrobial activity. Additionally, samples with hemp extract (Figure 4), geranium (GCV60 and GCV120), and mountain germander (MGCV60) also showed strong antimicrobial activity with inhibition zones against G+ bacteria, as shown in Table 4. Conversely, samples with lady’s mantle extract exhibited the weakest antimicrobial effect through contact inhibition, as did mountain germander extract samples (MGCV120). Observed clear inhibition zones are indicators of bioactive agents’ diffusion, which are particularly important for effectively targeting bacteria embedded within biofilms. 48
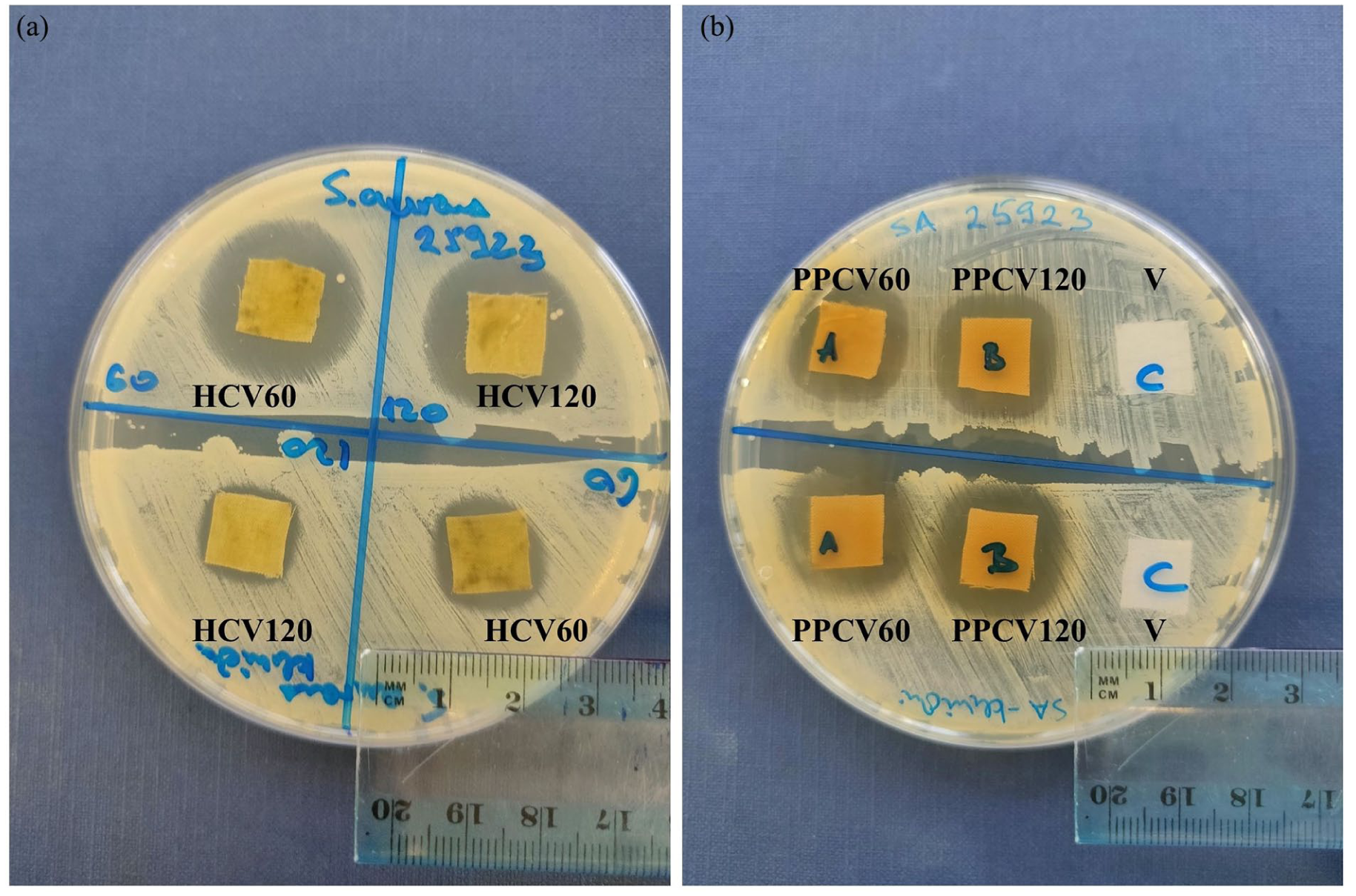
Antimicrobial activity of viscose fabric samples treated with plant extract: (a) HCV60, HCV120, and (b) PPCV60, PPCV120) on G+ bacteria S. aureus strain ATCC 25923 (upper row) and clinical isolate S. aureus (bottom row).
Phenolic acids form a complex with chitosan via positively charged amino groups, 49 as explained in the section Computational results. This complex subsequently interacts with the negatively charged bacterial cell membrane and exhibits antimicrobial activity against S. aureus, P. aureginosa, and E. coli. 49 Inhibition zones for G− bacteria were recorded only for the fabric samples PPCV60 and PPCV120, while contact inhibition was observed for all other samples. MICs for PP were also the same for both tested bacteria, 0.15 mg/ml, while hemp extract MIC was 0.07 mg/ml for S. aureus strain ATCC 25923, and higher than 1.25 mg/ml for E.coli strain ATCC 25322. These results are similar to the results of the agar diffusion method. Methanol had an effect on bacterial inhibition, but it was less pronounced than the combination of plant extract diluted in methanol. The methanol MICs for both examined bacteria was 7.5%. The weaker effect of polyphenols from plant extracts on G- bacteria is attributed to the presence of an outer membrane in these bacteria, which necessitates a higher deposition of hydroxy groups from polyphenols to damage the membrane and inhibit the growth and development of G− bacteria. 13
The antimicrobial activity of viscose fabric samples treated with plant extract solutions, without chitosan (Figure 5), was significantly lower compared to the samples treated with both, chitosan and plant extracts. In the case of G+ bacteria (S. aureus strain ATCC 25923), inhibition zones measuring 15 mm were observed in samples treated with lady’s mantle extract (LMV) and geranium extract (GV), whereas samples treated with extracts of mountain germander (MGV), hemp (HV), and pomegranate peel (PPV) exhibited only contact inhibition. All treated samples demonstrated contact inhibition against the G− bacterium (E. coli strain ATCC 25322).

Antimicrobial activity of viscose fabric samples treated with plant extract without chitosan on G+ (SA – S. aureus strain ATCC 25923) and G− bacteria (EC – E.coli strain ATCC 25322).
Plant extracts, due to their high content of polyphenols and hydroxy groups capable of donating hydrogen to free radicals, 47 exhibit remarkable antioxidant properties by preventing the formation of free radicals, destroying those already formed, or neutralizing cell damage caused by radical activity. 50 Chitosan itself does not exhibit antioxidant effects, as evidenced by the results of antioxidant activity analysis (DPPH), which showed values of 0.35% for CV60 and 0.17% for the CV120 sample. This limitation is effectively overcome by the addition of plant extracts, 7 as fabrics subsequently treated in an aqueous solution of plant extract demonstrate significant antioxidant activity.
The results of antioxidant activity in fabrics treated with chitosan and selected plant extracts revealed that the fabric treated with lady’s mantle extract displayed the highest antioxidant activity value at 80% (Figure 6) while exhibiting the weakest antimicrobial activity (Table 4). Fabric with mountain germander extract also demonstrated an exceptional antiradical potential, with 76%, followed by fabrics treated with pomegranate peel and geranium extracts. Viscose fabric samples with hemp extract had the lowest antioxidant activity value but exhibited strong antibacterial effects. It is noteworthy that pretreatment time did not significantly impact antioxidant activity, except for the fabric with pomegranate peel extract (PPCV60: 70%, and PPCV120: 60%). Textile materials with antioxidant properties protect the skin from the activity of free radicals originating from external influences, decrease the oxidative stress caused by reactive oxygen species and subsequently improve cell viability and skin regenerative potential.3,51 Flavonoids in the plant extract have a therapeutic effect on skin exposed to free radicals, a favorable impact on human health, 52 and an essential role in treating microdamage to the skin and preventing disease. 53
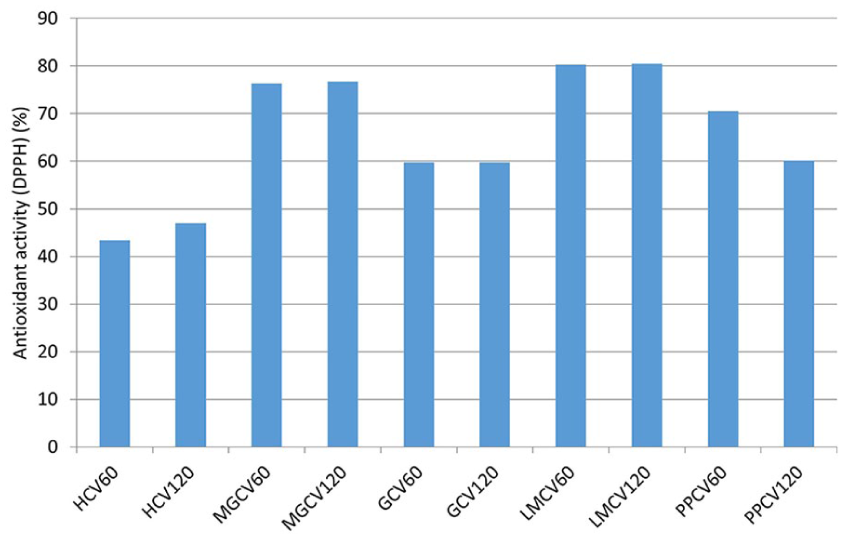
Antioxidant activity of viscose fabric treated with plant extract.
Table 5 presents several previous studies in which plant extracts or a combination of chitosan and plant extracts were used for the finishing of cellulosic textile materials. The results of these studies demonstrated that the used functional finishes exhibited satisfactory antimicrobial and antioxidant properties.
Functional finishing of cellulose textile material with plant extracts.

Computational results
The interactions between the bioactive components of hemp and pomegranate extracts were investigated using in silico methods. These extracts were selected for further analysis due to their proven antimicrobial activity and potent antioxidant properties. Their dual functionality made them ideal candidates for computational modeling and simulation studies aimed at exploring potential synergistic interactions and mechanisms of action. The analysis focused on providing detailed insights into the types of interactions and identifying the key functional groups and atoms involved in complex formation with chitosan and the most significant extract components. For the pomegranate extracts, punicalagin and punicalin were selected due to their high abundance and well-documented biological activities. 37 Similarly, for the hemp extracts, cannabinoids such as cannabidiol (CBD) and cannabidiolic acid (CBDA) were chosen, reflecting their prevalence in the extract and their established roles in modulating various biological processes. 37 These compounds were considered key candidates for understanding the interactions due to their potential contributions to the overall bioactivity of the extracts. The molecular docking results, shown in Table 6, reveal a significant affinity of pomegranate extract components – punicalin and punicalagin – toward chitosan, with complex energies of −5.271 and −6.043 kcal/mol, respectively.
Molecular docking results.

The values shown in the table indicate the formation of stable complexes between chitosan and pomegranate peel extract components, highlighting the potential therapeutic application of these biologically active molecules in treating various diseases. Systems based on these complexes could be beneficial as medical materials for skin application, allowing targeted action on surface layers without absorbing active molecules through the skin.
Additionally, the interactions of CBD and CBDA with chitosan were investigated, with complex energies recorded at −2.661 and −3.206 kcal/mol, respectively. Although CBD shows an affinity for chitosan, its interaction is less stable compared to CBDA, implying a potential for faster CBD release, which would be beneficial in therapeutic formulations requiring rapid effects. Conversely, CBDA offers more prolonged release due to its higher complex stability, an advantage for formulations requiring controlled release.
Both the polyphenols from pomegranate peel and the cannabinoids studied have confirmed pharmacological effects, including antimicrobial and antioxidant properties, and may play a key role in the development of innovative biomedical products, such as antimicrobial medical fabrics.22,23 These fabrics can be applied as wound dressings or in transdermal drug delivery.
Analysis of interaction types (Figures 7 and 8) reveals that hydrogen bonds, as well as electrostatic and hydrophobic interactions, are present in molecules from both extracts. Common to all interactions is that hydrogen bonds are formed through hydrogen atoms from amino groups, while nitrogen atoms from chitosan molecules contribute to electrostatic interactions, involving oxygen atoms from phenolic groups.

Interactions between punicalagin and chitosan.

Interactions between CBDA and chitosan.
The interactions between a ligand, such as an antimicrobial agent, and chitosan play a crucial role in creating effective drug delivery systems. This binding allows chitosan to function as a reservoir, providing a controlled and gradual release of the antimicrobial agent, which is essential for maintaining sustained antimicrobial activity at the wound site. Our analysis of binding affinity energy shows that these interactions are favorable, as they keep the ligand securely attached to the carrier while also enabling the necessary disruption of interactions for the release of the ligands into the interstitium. This release mechanism is particularly important for effectively targeting bacteria, especially those embedded within biofilms. By allowing for localized delivery of the antimicrobial agent, the treatment can be effective at lower concentrations compared to topical or systemic applications, which helps reduce the risk of potential toxicity and adverse side effects. Additionally, localized interstitial release minimizes the chance of systemic absorption, making this approach particularly suitable for sensitive areas or for patients with wounds that are prone to infection. 57
Conclusion
In conclusion, the results indicate that plant extracts derived from industrial hemp, mountain germander, geranium, lady’s mantle, and pomegranate peel, could serve as suitable replacements for synthetic agents in the functionalization of textile materials through an economical and environmentally friendly approach. Biologically active cellulose materials were successfully produced by pre-treating viscose fabric through oxidation, which enabled the effective binding of chitosan and plant extracts, resulting in enhanced antimicrobial and antioxidant properties alongside the colorization of the material. A 60-minute oxidation period proved optimal for chitosan binding, while pomegranate peel, mountain germander, and hemp extracts exhibited the most promising biological functionalities. Viscose fabric samples treated with pomegranate peel and hemp extracts exhibited the most effective antimicrobial activity against G+ bacteria, with an inhibition zone of 20 mm. The strongest effect against G− bacteria was observed in samples treated with pomegranate peel extract, showing an inhibition zone of 17.5 mm. Furthermore, all chitosan-plant extract functionalized fabrics, obtained in this research, demonstrated significant antioxidant activity (i.e., being able to prevent cell damage caused by radical activity), in clear contrast to chitosan-functionalized fabrics, which displayed only minimal antioxidant activity (0.35% for CV60 and 0.17% for the CV120 sample). The highest antioxidant activity was recorded in samples treated with extracts of lady’s mantle at 80%, teucrium montanum at 76%, and pomegranate peel at 70%. This study identifies pomegranate peel and hemp extracts as the most promising for further research. Computational analysis of binding affinity energy shows that favorable interactions between these extracts and chitosan keep the extracts’ compound securely attached to the carrier (chitosan) while also enabling the necessary disruption of interactions for the release of the bioactive compounds, providing localized delivery.
The resulting cellulose materials are promising raw materials for the production of biologically active textiles with potential therapeutic applications. However, to increase the scale-up potential and industrial applicability of this environmentally friendly finishing protocol for viscose fabric, future research should investigate the possibilities of fabric functionalization in a shorter processing time and with lower concentrations of chitosan and plant extract. Considering that the intended use of the functionalized fabric is for medical purposes, particularly for wound care and applications involving skin contact, cytotoxicity and acute dermal irritation testing should be carried out on the most promising functionalized fabrics, to assess the biocompatibility and safety of plant extract-functionalized textiles. Furthermore, it is necessary to analyze the long-term durability of the achieved bioactivity, as this is especially important for medical applications. In the case of functionalized fabrics intended for multiple uses, special attention should be paid to evaluating the durability of the obtained properties after multiple washing cycles.
Supplemental Material
sj-docx-1-jef-10.1177_15589250251368306 – Supplemental material for Eco-friendly functionalization of viscose with bioactive plant extracts
Supplemental material, sj-docx-1-jef-10.1177_15589250251368306 for Eco-friendly functionalization of viscose with bioactive plant extracts by Vesna Ivanovic, Relja Surucic, Aleksandra Smitran, Aleksandra Borkovic, Biljana Lazic, Svjetlana Janjic and Mirjana Kostic in Journal of Engineered Fibers and Fabrics
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors V.I., R.S., A.S., A.B., B.L. and S.J. received no financial support for the research, authorship, and/or publication of this article. M.K. disclosed receipt of the financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia (Contract No. 451-03-136/2025-03/200135).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
