Abstract
Uniform monoclinite bismuth phosphate (BiPO4) nanorods were fixed on commonly used cotton fabric using two-dip–two-nip technique. Chitosan and acetic acid were applied to act as cross-linking agent in preparing the finishing solution. In this article, BiPO4 nanorods modified cotton fabrics for UV-blocking, as well as photocatalytic degradation of RhB has been studied. This coated fabric was characterized by X-ray diffraction, field-emission scanning electron microscopy equipped with energy-dispersive X-ray spectrometer, X-ray photoelectron spectroscopy, Fourier transform infrared spectroscopy, and UV–Vis diffuse reflection spectroscopy. Furthermore, UV-blocking and photocatalytic properties were also tested. The coated fabric showed better properties compared to uncoated fabric.
Introduction
Photocatalytic materials and technology have drawn much attention due to their potential use, degradation of dyes and contaminants, absorption of UV light, and optoelectronic1–5 equipment. As one of the most typical photocatalysts, TiO2-based materials can efficiently oxidize pollutants, such as toxic and dye waste water into CO2 and H2O.6–9 Because of the high recombination of photo-induced electron-hole pairs and low utilization of visible light, TiO2-based materials still have a lot of problems in actual 10 applications. Developing new type of photocatalysts with high photocatalytic activity is an effective solution to the problem. As an oxoacid salt photocatalyst, BiPO4 was first found to have excellent photocatalytic activity by Pan and Zhu’s 11 group in 2010. Later, different morphologies and phases of BiPO4 such as nanoparticles, nanorods, needle-like, hexagonal phase, and monoclinic phase were successfully synthesized by different research12–16 groups. However, there are few studies on the application of BiPO4 for the photocatalytic degradation of organic dyes and UV-blocking fabric finishing agents.
Protective cotton fabric against UV radiation is an important material for outdoor textiles. Excessive exposure to ultraviolet light causes many diseases such as sunburn, skin cancer, and allergies.17,18 Therefore, many researchers have coated fabrics using various techniques.19,20 Usually ultraviolet shielding agent is divided into two types, one is organic ultraviolet shielding agent and the other is inorganic ultraviolet shielding agent. Compared with organic UV screening agents, inorganic UV screening agents are widely used because of their non-toxic, odorless, non-irritation effects; thermal stability; and safety. 21 Nanoparticles of metal oxides such as titanium oxide (TiO2), cupric oxide (CuO), and zinc oxide (ZnO) are the most commonly used materials for functional coating. 22 For example, Prabu’s group utilized in situ chemical polymerization method to prepare the polypyrrole-zinc oxide and polypyrrole-zinc oxide-carbon nanotube (ppy-ZnO-CNT). 23 After using pad-dry-cure technique to coat the cotton fabric, it endowed the fabric with excellent flame-retardant and UV-protection properties. Compared to TiO2 nanomaterials, the newly emerged BiPO4 shows higher photocatalytic activity which may be due to its unique crystal structure.24–29 Hexagonal BiPO4 has great potential applications in future functional textiles as a finishing agent. As we know, little research has been conducted on this. So in this work, we used a facile hydrothermal method to synthesize uniform BiPO4 nanorods. Then, we applied the two-dip–two-nip technique to coat the fabric with BiPO4 nanorods. UV protection factor (UPF) test and photocatalytic degradation of RhB results showed that the coated fabrics had an excellent UV protection and photocatalytic activity with a good washing fastness.
Experimental section
Synthesis of BiPO4 nanorods
The synthesis was performed via a facile hydrothermal method. 1.5 mmoL of Bi(NO3)3·5H2O and 5 mmol of NaH2PO4·2H2O were added into 60 mL of deionized water under vigorous stirring at room temperature for 1 h. Then, the solution was sealed in a Teflon-lined stainless-steel autoclave, which was maintained at 160°C for 16 h, followed by cooling down to room temperature. The obtained products were washed four times with ethanol and deionized water, respectively. Then, the powder was dried in vacuum oven at 60°C for 24 h.
Fabrication of BiPO4/cotton fabric
The finishing agent solutions were prepared with four different contents of BiPO4 nanorod powder such as 0.5, 1.5, 2.5, and 3.5 wt%. Then, 2 mL of acetic acid, 1 g of chitosan, and different amounts of BiPO4 nanorods were added into 50 mL of deionized water followed by ultrasonic dispersion for 30 min to obtain the finishing solution. The cotton fabric was treated before immersing into the finishing solution. The cotton fabric was soaked in a 10% NaOH solution at room temperature for 30 min, washed several times with distilled water, and dried in an oven at 60°C for 16 h. Then, the cotton fabric was dipped in the finishing solution for 1 h, using the two-dip—two-nip technique to reach an average pickup of 80%. The padded fabrics were first dried at 90°C for 30 min and after that curing was performed at 120°C for 30 min. The prepared fabrics were denoted, respectively, as blank cotton, BiPO4/cotton 0.5, BiPO4/cotton 1.5, BiPO4/cotton 2.5, and BiPO4/cotton 3.5.
Characterization
The morphology of BiPO4 nanorods and the surface of coated and uncoated fabrics were observed by scanning electron microscope (SEM, Supra 55, ZEISS) equipped with an energy-dispersive X-ray (EDX) spectrometer annex. Transmission electron microscopy (TEM) and high-resolution TEM (HRTEM) images were obtained using JEOL JEM-3200 transmission electron microscope to further see the BiPO4 crystal structure. The X-ray photoelectron spectroscopy (XPS) was recorded by ESCALAB 250 Xi. Powder X-ray diffraction (XRD) patterns were collected by a diffractometer with Cu-Kα radiation (λ = 0.15406 nm).
UV properties of coated and uncoated fabrics were measured according to the American standard (AATCC 183-2010) with a Varian Cary 300 UV-visible spectrometer. The diffuse reflectance Fourier transform infrared spectra were recorded using Vertex 70 Fourier Transform Infrared Spectrometer. The UV–Vis absorption spectrum and the UPF of the coated fabrics were measured by a UV spectrophotometer (Shimadzu, UV2550). To determine the durability of the coated fabrics, the obtained samples were subjected to laundering test method. We used 2 wt% soap and 2 wt% Na2CO3 to prepare the 1:30 solid-liquid-ratio washing solution. And the samples were washed at 40°C for 15 min and then cleaned with deionized water.
Results and discussion
Structure and composition analysis
The crystal structure of the obtained BiPO4 nanorods was characterized by XRD, as shown in Figure 1. Seven pronounced peaks can be clearly observed in the spectrum, which could be well indexed, respectively, to (–111), (111), (200), (120), (012), and (–202) facet diffractions of monoclinic BiPO4 at 2θ value of 21.3°, 25.3°, 27.1°, 29.1°, 31.2°, and 34.5°(JCPDF No. 15-0767). No characteristic diffraction peaks of other phases can be observed. The energy dispersive X-Ray spectroscopy (EDS) test was also performed to further determine the composition of the samples. As shown in Figure 2, we can see that the obtained materials contain bismuth, oxygen, and phosphorus, which are evenly distributed. In addition, we also tested the XPS spectrum of the obtained BiPO4 nanorods to further see the structure and binding energy of elements. In Figure 3, the peaks at 133.09 and 530.95 eV can be attributed to P2s and O1s in BiPO4, respectively. The peaks at 164.97 and 165.11 eV are ascribed to Bi4f in BiPO4. Therefore, all the results clearly confirm the formation of BiPO4 nanorods.

XRD pattern of the BiPO4 nanorods. The red lines in the figure represent the standard peaks of BiPO4 crystal for comparison.
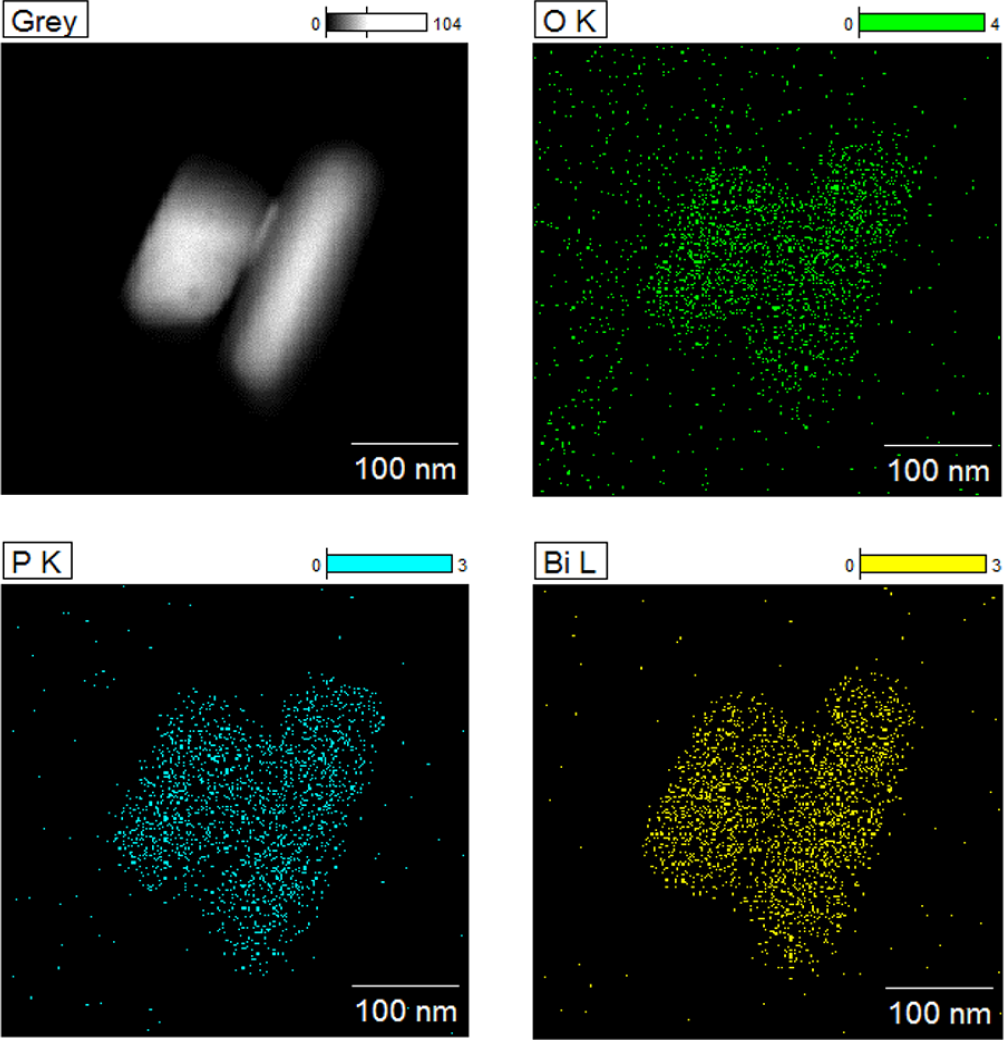
EDX mapping information of obtained BiPO4 nanorods. Different colors stand for the existence of different elements in the image. And the contrast scale of gray stands for the whole element scanning. And the contrast scale represents the brightness of each element.

(a) XPS spectrum of pure BiPO4 nanorods, (b) Bi4f XPS spectrum, (c) O1s spectrum, and (d) P2p XPS spectrum.
Morphological characteristics
TEM was used to see the morphology of the obtained BiPO4 nanorods and the BiPO4/cotton samples. From Figure 4(a) and (b), we can see that the BiPO4 nanorods have a clearly uniform rod shape. The length of the nanorods is about 500 nm with an average diameter of 74 nm.

(a) and (b) TEM images of pure BiPO4 rods and (c) and (d) SEM images of BiPO4 rods.
UV-shielding performance of BiPO4/cotton samples
The UV-shielding properties of different obtained samples were evaluated by the UPF value using equation (1)
where E (λ) is the relative erythema action spectrum, S (λ) is the spectral irradiance (W m−2 nm−1), T (λ) is average spectral transmittance of fabric, dλ is bandwidth, and λ is wavelength. The Ultraviolet Radiation A (UVA, with wavelength between 320 nm and 400 nm) and Ultraviolet Radiation B (UVB, with wavelength between 290 nm and 320 nm) transmittance was calculated by equations (2) and (3), respectively
In Table 1, we can find the percentage UVA and UVB radiation blocking and UPF of different samples. As the mass concentration of BiPO4 increases from 0.5 to 3.5 wt%, the UPF value increases from 7.54 to 68.94. On one hand, the BiPO4 can effectively absorb the UV light and further generate the photo-induced carriers, which could reduce the transmittance of UV light through the fabric. On the other hand, the increased BiPO4 nanorods on fabric could scatter a large amount of UV light, these all will lead to a higher UPF values of samples.
UVA and UVB transmittance and UPF values of samples.

UPF: UV protection factor.
Photocatalytic properties of obtained BiPO4/cotton samples
UV–Vis transmittance spectrum of the uncoated and coated fabrics is presented in Figure 5. When the content of BiPO4 nanorods increased from 0.5 to 3.5 wt%, about 98% of UVA and UVB light could be shielded. After 10 cycles of washing with soap (0.3 wt%) solution, theses coated fabrics showed a slight decrease in UPF values, which may be due to the removal of BiPO4 nanorods. To further evaluate the UV-blocking properties of the samples, we performed the test of photocatalytic degradation of RhB. As shown in Figure 6, the photocatalytic activity of the BiPO4/cotton 3.5 sample was evaluated by the degradation of RhB in aqueous solution under visible light irradiation. The 2 cm length and width of obtained cotton fabric was placed on the bottom of the beaker and then 100 mL of RhB solution with a concentration of 5× mol L−1 was added. It could be easily found that the RhB was almost completely degraded after 60 min. On the other hand, the blank cotton showed none photocatalytic ability. This result revealed that the as-prepared sample can effectively absorb the UV light and generate electrons and holes with oxidation-reduction ability which can degrade the RhB solution.
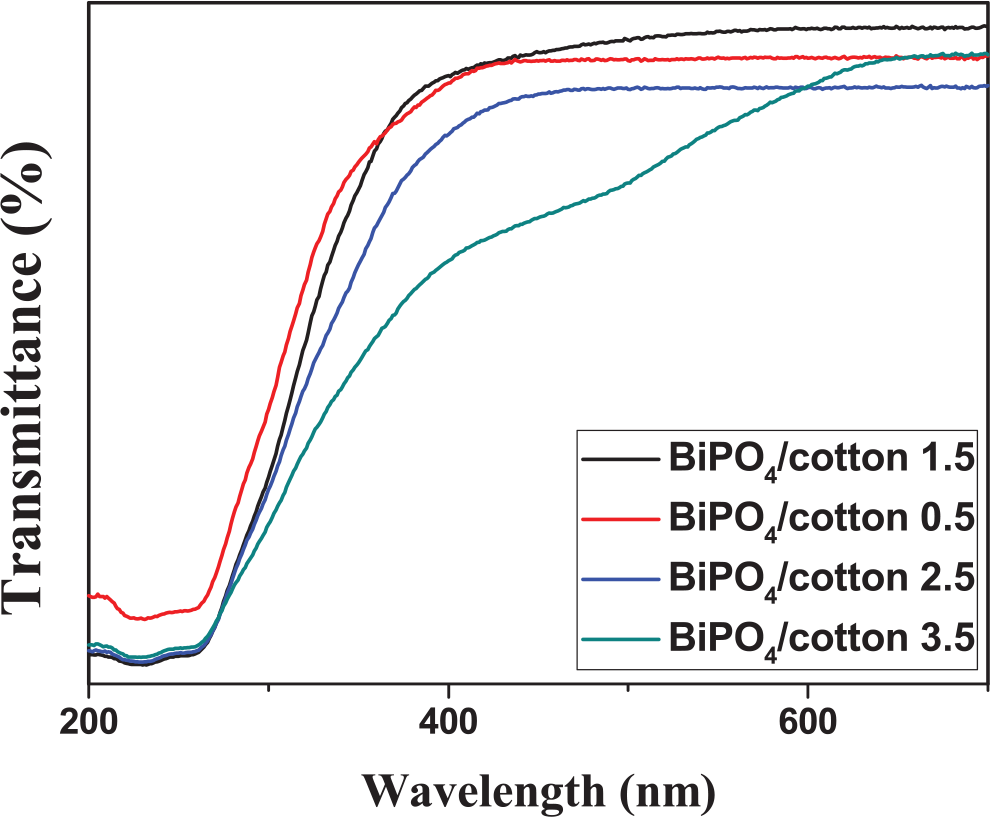
UV–Vis light transmittance spectra of different amounts of BiPO4 coated on the fabrics.

(a and b) Photodegradation curves of RhB solutions by BiPO4/cotton 3.5 sample and blank cotton fabric.
Chitosan is a commonly used textile finishing agent. It is an amino-polysaccharide, which is normally obtained by partial N-deacetylation of chitin. 30 Also, because of the chemical nature, chitosan and cellulose have a very similar structure, with the same β-glycosidic linkages. There are a lot of literature on the chemical bonds between chitosan and cellulose. For example, Orden’s group used diffuse reflectance spectroscopic techniques (UV–Vis and Fourier-transform infrared spectroscopy (FTIR)) and fluorescence spectroscopy to study the chemical interactions between cellulose and chitosan. 31 They found that the existence of chemical interactions in chitosan-treated celluloses had been demonstrated by the appearance of new UV–Vis absorption and fluorescence emission bands. And the new bands can be assigned to conjugated imines, produced in the condensation between chitosan amino groups and carbonyl groups. And also, many other amino compounds have the similar reaction with chitosan.32–35 In our study, we applied chitosan and acetic acid as the finishing agent to improve the attachment between the BiPO4 nanorods and cotton fabric. We also tested the UV–Vis and FTIR spectrum (Figure 7(a) and (b)) of the coated cotton fabric, in which we found some new peaks, which may be due to the chelation between the BiPO4 and chitosan. Figure 7(a) shows the diffuse reflectance UV–Vis spectra of pure cotton fabric and cotton fabric treated with chitosan in dilute acetic acid solution. The spectrum of the treated cotton fabric shows the characteristic chitosan reflection at 217 nm, however, this peak apparently shifts, which suggests the possible chemical interactions between cellulose and chitosan. Also, the new band at 350 nm, which do not appear in the spectra of the starting materials, indicates the existence of a chemical reaction between these two materials. In order to verify the existence of above chitosan–cellulose interactions, the FTIR spectra of chitosan, cotton fabric, and chitosan-treated cotton fabric were recorded. Figure 7(b) and (c) shows the significant features of the FTIR spectrum of the materials. The most characteristic absorption of the chitosan is the band at 1591 cm−1, which may be assigned to the amino groups of chitosan. 36 After treated with chitosan, the above band at 1591 cm−1 disappears, which reveals the gradual disappearance of the chitosan amino groups in a chemical reaction. Also, the appearance of a new absorption at wavenumber of 1708 cm−1 further confirms the existence of the chemical reaction between these materials.
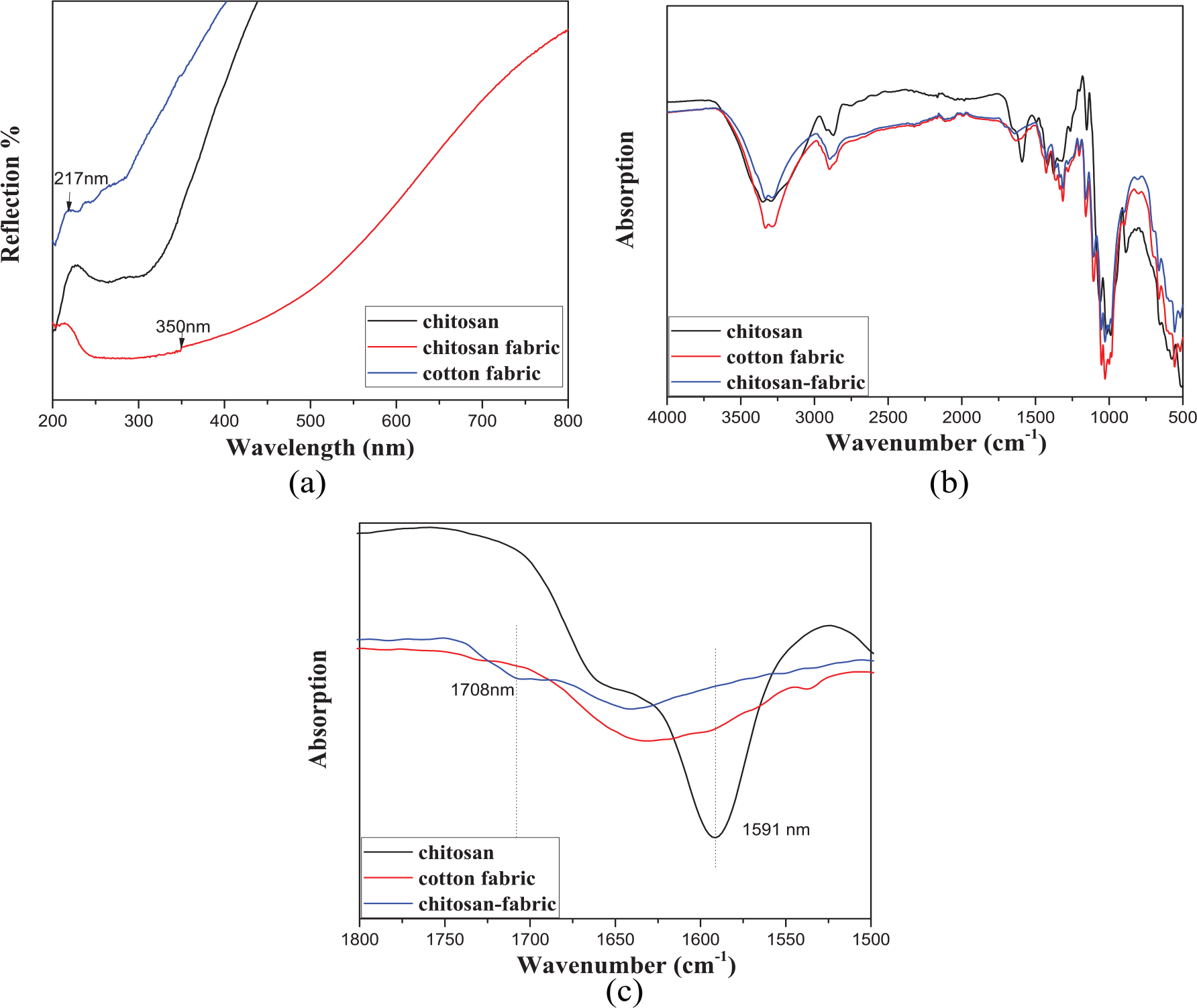
(a) UV–Vis spectra of cotton fabric, cellulose-treated cotton fabric (chitosan-fabric), and pure chitosan; (b) FTIR spectra of cotton fabric, cellulose-treated cotton fabric (chitosan-fabric), and pure chitosan; (c) FTIR spectra of cotton fabric, cellulose-treated cotton fabric (chitosan-fabric), and pure chitosan of wavenumber between 1500 and 1800 cm−1.
Thanks to these chemical interactions, the BiPO4 nanorods could be fixed on the cotton fabric. When the ultraviolet light is irradiated onto the cotton fabric, part of it is absorbed and converted to photo-generated carriers by BiPO4 nanorods on the cotton fabric, and the other part is scattered out. All these lead to reduced transmittance of ultraviolet and increased UPF values of coated fabrics (Scheme 1).

The mechanism of the UV-blocking properties of coated fabric.
Conclusion
In summary, we successfully prepared a functional cotton fabric with excellent UV-blocking property and photocatalytic activity by coating the fabric with uniform BiPO4 nanorods. With chitosan and acetic acid as the capping agent, the BiPO4 nanorods could be fixed on the cotton fabric, which further enhances the scattering rate of the UV light and the washing fastness of the fabric. The resultant functional fabric may become a potential UV-shielding material for outdoor products.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Program for Innovative Research Team of Huizhou University (IRTHZU).



