Abstract
To improve the photocatalytic property of titanium dioxide, titanous sulfate as the titanium source, cobalt nitrate hexahydrate as the cobalt source, undoped and Co2+-doped titania nanotubes were prepared via coprecipitation method combined with hydrothermal synthesis. Based on the degradation rate of methylene blue, the effects of the codoped rate on photocatalytic activities of the sample were researched. The result showed that doping cobalt could improve the photocatalytic performance of the titanium dioxide nanotubes under the ultraviolet light. The composition and structure of the samples were characterized by energy-dispersive X-ray spectroscopy, X-ray diffraction, transmission electron microscopy, and selected area electron diffraction. Conclusions indicated that the low-concentration Co2+ has no influence on the crystal structure of titanium dioxide and no new crystals were formed in the whole process.
Introduction
In modern society, environmental pollution and the energy crisis have been more and more serious. Photocatalytic degrading organic pollutants as an economical and environmental friendly technique offer great potential for environmental protection and saving energy. 1,2 Titanium dioxide, owing to relatively inexpensive, high chemical stability, and the highly oxidized photogenerated holes 3 , is one of the most widely used photocatalyst at present. However, titanium dioxide has a fatal flaw, that is, it can only able to absorb photons energy in the near ultraviolet (UV) range (λ < 387 nm). 4 To improve its photocatalytic performance under the UV light and the visible irradiation, metal-doped titanium dioxide has been widely studied over the past decades. 1 Titanium dioxide has been doped with different metal ions like alkaline earth (Ca2+, Sr2+, and Ba2+), 5 Fe3+, 6 Cr6+, 7 Co2+, 8 Mo5+, 9 and rare earth cations (La3+, Ce3+, Er3+, Pr3+, Gd3+, Nd3+, Sm3+) 10 via different preparation technologies. Lee et al. 11 reported that compounding cobalt oxide nanoparticles on the surface of titanium dioxide powders via liquid phase plasma method can improve the photocatalytic performance of titanium dioxide under visible light but reduce the photocatalytic performance of titanium dioxide under the UV light. Hamadanian et al. 12 demonstrated that the photocatalytic performance of Co–titanium dioxide was found to be worse than that of undoped titanium dioxide under the UV irradiation, and Co-doped titanium dioxied (0.5%) had the highest photocatalytic performance under visible light irradiation (samples prepared by sol–gel method). According to the aforementioned articles, the photocatalytic performance of titanium dioxide-doped Co usually increased in the visible light and reduced under the UV light. Therefore, in this work, the main purpose was to improving the photocatalytic activities of Co–titanium dioxide under the UV light. As the most promising photocatalyst, 13 –16 nanostructures of titanium dioxide including nanoparticles, nanorods, nanowires, and nanotubes primarily vary with the preparation methods. 17 There are a lot of methods for the synthesis of titanium dioxide nanostructures, such as sol−gel method, 18 micelle and inverse micelle methods, 19 sol method, 20,21 hydrothermal method, 22,17 and so on.
In this study, undoped and Co-doped titanium dioxide nanotubes were prepared via coprecipitation method combined with hydrothermal synthesis and researched the photocatalytic properties of Co-doped titanium dioxide nanotubes under the UV irradiation. The Co-doped titanium dioxide was synthesized using sodium hydroxide (NaOH) hydrothermal treatment. In order to further research on the mechanism of the titanium dioxide nanostructures formation in NaOH solution 23 and establish the crystal structure of the nanotubes, 24 a large number of researchers have done lots of research, since the method was introduced in 1998. 25 The hydrothermal temperature of nanostructures formation was 130°C. Titanium dioxide has a lot of phases such as anatase, rutile, 26 brookite, titanium dioxide (B), or amorphous titania. X-ray diffraction (XRD) patterns showed that its structure was anatase at 500°C. When the temperature increased up to 700°C, anatase begins to transform to rutile, anatase transform to rutile completely, at 900°C.
Materials and methods
Materials
Titanium(IV) sulfate (Ti(SO4)2), (content: [Ti(SO4)2] ≧ 96.0%) as the titanic source, was purchased from the Tianjin Fine Chemical Research Institute (China).
Cobalt(II) nitrate hexahydrate
Hydrochloric acid (concentration: 36–38%) and NaOH (content: [NaOH] ≧ 96.0%) were purchased from Beijing Chemical Plant (China).
Methylene blue (MB; content: [C16H18ClN3S·3H2O] ≧ 96.0%) was used to study the photocatalytic property and was purchased from Sinopharm Chemical Reagent Co. Ltd. Distilled water was used in all experiments.
Synthesis of titanium dioxide and Co-doped titanium dioxide
At room temperature, a certain amount of Co(NO3)2·6H2O was added to 20 ml (1 mol/L) Ti(SO4)2 solution to form solution A. About 20 mL (4 mol/L) NaOH solution was named solution B. Solution A and solution B were dropped into a beaker at the same rate, which results in precipitation. The precipitates were washed with distilled water for several times to get neutral solutions. In the next synthetic process, 1.0 g precipitate was mixed with 30 ml (10 mol/L) NaOH aqueous solution, and then the mixture was transferred into a teflon container which is enclosed in a stainless steel autoclave maintained at 130°C for 24 h. The samples prepared by the hydrothermal method were washed using distilled water for several times to get neutral solutions. After filtrating, drying at 60°C, and grinding, the final products were obtained. The samples were calcined at 450°C in muffle for 3 h. Pure titanium dioxide sample was prepared in the same way. Here, different molar rates of Co2+/Ti4+ were set at 0, 0.1, 0.3, 0.5, 0.7, 1.0, 1.3, 1.5, 1.7, 2.0, 2.3, and 2.5%.
X-ray diffraction
XRD patterns were recorded with copper (Cu) K α radiation using an automated X-ray diffractometer (Bruker AXS, Germany). XRD patterns of the samples were collected in the range of 20°–70°(2θ) with a step size of 0.02°, the scanning speed of 4° (2θ)/min, and step time of 8 s.
Transmission electron microscopy
The morphologies of the nanotubes were evaluated by transmission electron microscopy (TEM). TEM observations were conducted on a JEM-2100F microscope (JEOL Ltd.) operated at 200 kV. The samples for TEM measurements were suspended in ethanol and dropped onto holey carbon films supported on Cu grids for imaging.
Photocatalytic performance test
The photocatalytic degradation of MB was carried out at room temperature in a 50-mL beaker containing 20 mg sample and 20 mL (20 mg/L) MB aqueous solution. The beaker was placed in a dark environment with vigorous stirring for 30 min, and subsequently, 6 ml mixture was measured and centrifuged for 3 min at a speed of 11,000 r/min. Then, the photoreaction was conducted in the irradiation of UV light. In the process of photochemical reaction, the 6 ml samples were taken every 5 min and then centrifuged in the same way. The supernatant was taken to measure the absorbance in the end. The MB was analyzed by a UV spectrophotometer (detection wavelength of 664 nm) to determine the degradation yield. All the photocatalytic results were checked at least twice to ensure the accuracy of the data.
Results and discussion
Figure 1 showed the XRD patterns of undoped and Co2+-doped titanium dioxide nanotubes (0.5% and 1.3%). The characteristic peaks appeared in the 2θ angles of 25.4, 37.1, 37.8, 38.6, 48.2, 54.0, 55.1, 62.8, 68.9, 70.3, and 75.2, and corresponding diffraction rings were identified as (101), (103), (004), (112), (200), (105), (211), (204), (116), (220), and (215), respectively, and the results can be readily indexed to anatase phases. By contrasting with the three graphs in Figure 1, it was easy to draw the conclusion that low concentration of Co2+ doping has not changed the crystal structure of titanium dioxide and no new crystals were formed in the whole process. However, the sharp degree of diffraction peak increased with the increase of doping concentration. This was mainly because Co had smaller atomic radius than Ti. When Co2+ was inserted into the interstice of titanium dioxide crystal or took the place of Ti4+, it would decrease the interplanar spacing which was in accordance with the Scherrer equation

XRD patterns of undoped and Co-doped titanium dioxide nanotubes. XRD: X-ray diffraction.
Figure 2 shows the TEM images of the Co2+-doped titanium dioxide, which displays that the microstructure of the sample was one-dimensional and the length of the nanotubes could reach 30–70 nm. The high-resolution transmission electron microscopic images clearly showed the lattice fringes and layered structure. As shown in Figure 2(a) and (b), TEM observations were conducted on a 250,000× magnification microscope operated at 200 kV. Figure 2(a) shows the pure titanium dioxide and Figure 2(b) shows the Co2+-doped titanium dioxide. From the figure, there were nearly no obvious microstructure differences between the Co2+-doped and undoped titanium dioxide nanotubes. The as-grown titanium dioxide nanotubes were hollow. Figure 2(c) shows the TEM image that was recorded by a 300,000× magnification microscope operated at 200 kV. Figure 2(c) shows that the titanium dioxide nanotubes’ outer diameter was 10–15 nm, inner diameter was 5–10 nm, and thickness was 5.9 nm. The characterization of the prepared samples indicated that both ends of the nanotubes were opened and the walls were like that many sheets crimped along an intrinsic axis except the diameter and thickness, which was mainly caused by reaction time and reaction temperature. The conclusions were in accordance with the article reported by Yang et al. 27

TEM images of the Co-doped titanium dioxide nanotubes. TEM: transmission electron microscopic.
Figure 2(d) shows the selected area electron diffraction images of the Co2+-doped titanium dioxide nanotubes. The interplanar spacing was almost the same with anatase titanium dioxide, which demonstrated our former view. Based on the analysis shown earlier, we speculated that the Co atoms mainly took the place of partial Ti atoms or inserted into the octahedral spaces.
Figure 3 shows the data of the temporal degradation of MB when different titanium dioxide photocatalysts were used. It could be seen that all the samples not only titanium dioxide but also Co-doped titanium dioxide had efficient photocatalytic rate. The concentration of Co2+ dopants in titanium dioxide nanotubes directly affected photocatalytic activities under the UV light. As shown in Figure 3, the photocatalytic activities of Co-doped titanium dioxide in low concentration were higher than the undoped titanium dioxide samples. When the concentration of Co-doped exceeds 2.5%, the photocatalytic rate was lower than undoped titanium dioxide. This was because that excess dopants would lead to photon scattering. Along with the photocatalytic reaction, the degradation rate reached up to 97.2% when the Co content was 1.3%, which was superior to undoped titanium dioxide with the degradation rate of 80.6%. The figure shows that the best doping concentration of Co was 1.3%.

The photocatalytic property of different catalysts.
Figure 4 shows the energy-dispersive X-ray spectroscopic images of undoped titanium dioxide and Co2+-doped titanium dioxide nanotubes. Figure 4(b) shows that the Co ion successfully inserted into the lattice of titanium dioxide.
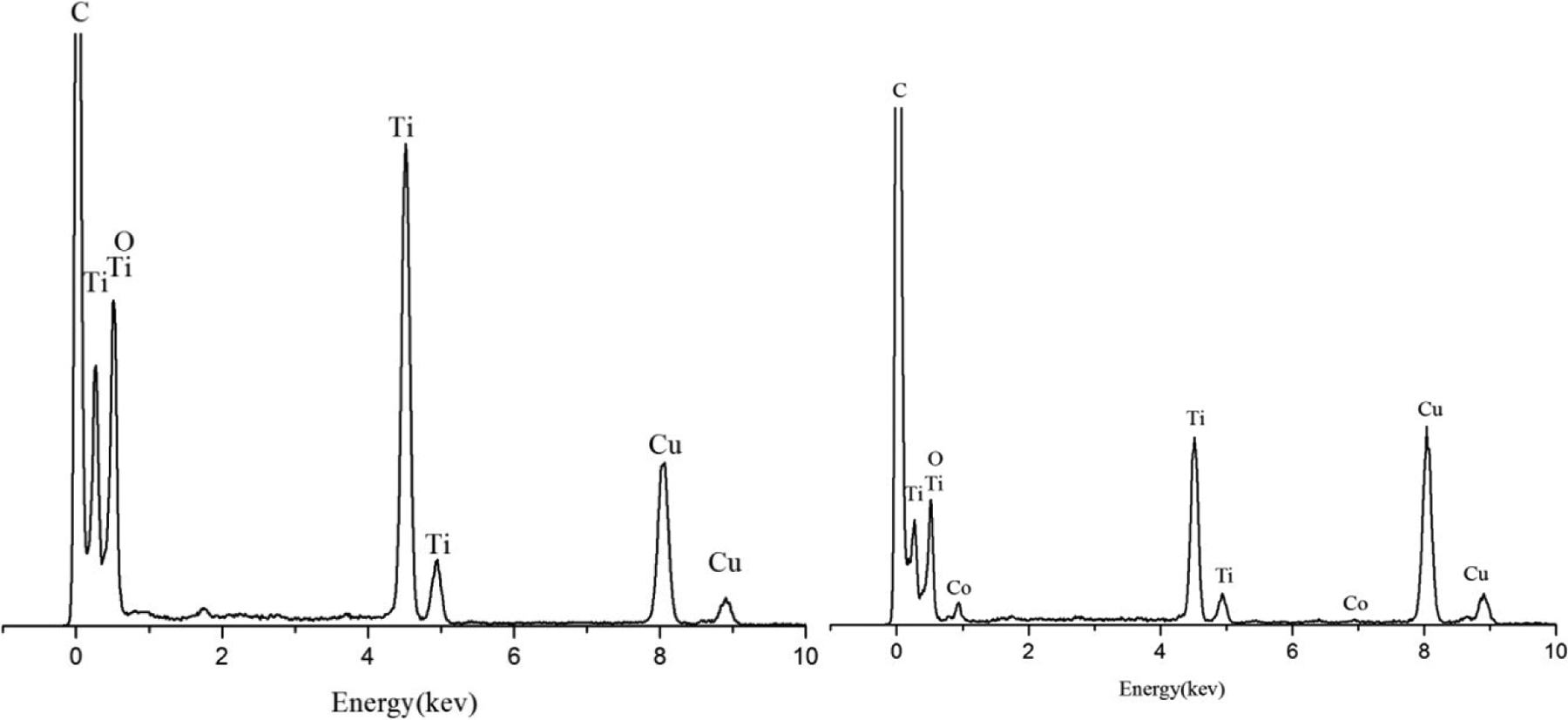
EDS images of the pure titanium dioxide and Co-doped titanium dioxide nanotubes. EDS: energy-dispersive X-ray spectroscopy.
The formation of Co-doped titania nanotubes prepared from hydrothermal synthesis was rarely discussed. Upon NaOH treatment, some of Ti–O–Ti bonds were broken and then an intermediate containing Ti–O–Na, Ti–O–Co, and Ti–OH was formed. This indicated that Co and sodium (Na) atoms would occupy some broken Ti–O bonds of the titanium dioxide precursor, leading to the formation of lamellar fragments that were intermediate phase in the formation process for the nanotube material. These intermediates would proceed with rearrangement to form sheets of edge-sharing TiO6 octahedra with Co2+, Na+, and OH− intercalated between the sheets. The presence of Ni ions in these sheets would probably cause surface heterogeneity, forming different curvatures of nanotubes. This could be attributed to a fact that the Co doping affected the surface characteristics of as-grown nanotubes.
Conclusions
We have successfully synthesized titanium dioxide nanotubes of doping Co2 + ions via coprecipitation method combined with hydrothermal synthesis. The diameters of the obtained nanotubes were about 10 nm and the lengths were approximately 30–70 nm. In the degradation of MB, the photocatalytic performance of the samples was the best when the Co doping content was 1.3% under the UV light. The best degradation rate could reach 97.2%.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Scientific and Technological Project of Jilin Province, Project grant no. 20140204052GX.

