Abstract
3-chloro-2-hydroxypropyl acrylate obtained by the esterification of epichlorohydrin with acrylic acid in the presence of zirconium silicate as a catalyst was used for grafting of the polyester fabric in order to improve some of its properties. The grafting was performed in the presence of (NH4)2S2O8 as a polymerization initiator. The grafted samples were characterized by Fourier transform infrared spectroscopy, scanning electron microscopy, and energy-dispersive X-ray spectroscopy analyses. The effect of grafting on the comfort indices (hygroscopicity and vapor permeability) and the tinctorial properties (color intensity and dyeing fastness) were analyzed. Further addition of AgNO3 in the final reaction mixture used for grafting of polyester fabric confers antibacterial effect to the treated samples. The properties of the polyester fabric were highly improved by grafting with 3-chloro-2-hydroxypropyl acrylate.
Introduction
Polyester fibers are one of the most important synthetic fibers used in the textile industry. The materials made from classical polyester fibers exhibit a wide range of uses due to high strength to wearing, dimensional stability, and chemical stability. These materials raise certain problems (low hydrophilicity, charge with static electricity, dyeing capacity limited to a single class of dyes) due to crystalline structure and missing of the polar groups from the fiber surface. Because of these problems, other researchers have tried to modify the surface of the polyester fibers and, therefore, various treatment techniques were reported: alkaline treatment, treatment with polyvinyl alcohol in alkaline medium, and lipase and sericin application on the polyester fabrics surface.1–8 Atmospheric plasma techniques have also been investigated and used for surface modifications.9–12 Many studies concerning the grafting of various vinyl monomers such as acrylic and methacrylic acids, methyl acrylate, acrylamide, and vinyl pyridine onto the polyester fabrics using different chemical initiators or electron beam pre-irradiation initiation have been published.13–24 Since the traditional polyester fibers are dyed only with dispersion dyes, dyes that have lower extinction molecular coefficients and do not exhibit brilliant shades, a number of studies have been published concerning the dyeing of polyester with other classes of dyes. Chemical modification of polyester fibers is an important method to improve the dyeing ability.25–29
The aims of this work were (1) increasing the hygroscopicity of the polyester fiber materials (a necessary requirement for the garments to exhibit adequate hygienic and functional properties) and (2) dyeing these materials with cationic dyes. For this purpose, the samples were grafted with 3-chloro-2-hydroxypropyl acrylate (CHPA), obtained as a main product by reaction of the acrylic acid with epichlorohydrin catalyzed by zirconium silicate beads. Ammonium persulfate was used as a polymerization initiator.
In contrast to acrylic and methacrylic acids, CHPA is preferred because its reaction with the –OH groups of the polyester proceeds easily (the epoxy functional group formed by dehydrochlorination of CHPA is highly reactive and forms chemical bonds with the –HO polyester groups).
Experimental part
Materials and methods
The nonionic surfactant Tween 80 and NaOH were purchased from Merck, LAVOTAN DSU from Bezema, the cationic dye ASTRAZON BLUE BG (C.I. Basic Blue 3) from Dye Star Company, and AgNO3 was obtained from SC CHIMREACTIV SRL. All the chemical products were used without previous purification. The polyester fabric 100% was provided by SC Majutex SRL.
Synthesis of CHPA
CHPA used for grafting of the polyester fabrics was obtained by the reaction of acrylic acid with epichlorohydrin (molar ratio of acrylic acid/epichlorohydrin: 1/1.15; reaction time: 180 min at 85°C) in the presence of zirconium silicate as a catalyst. 30 The reaction of obtaining CHPA is shown in Scheme 1.
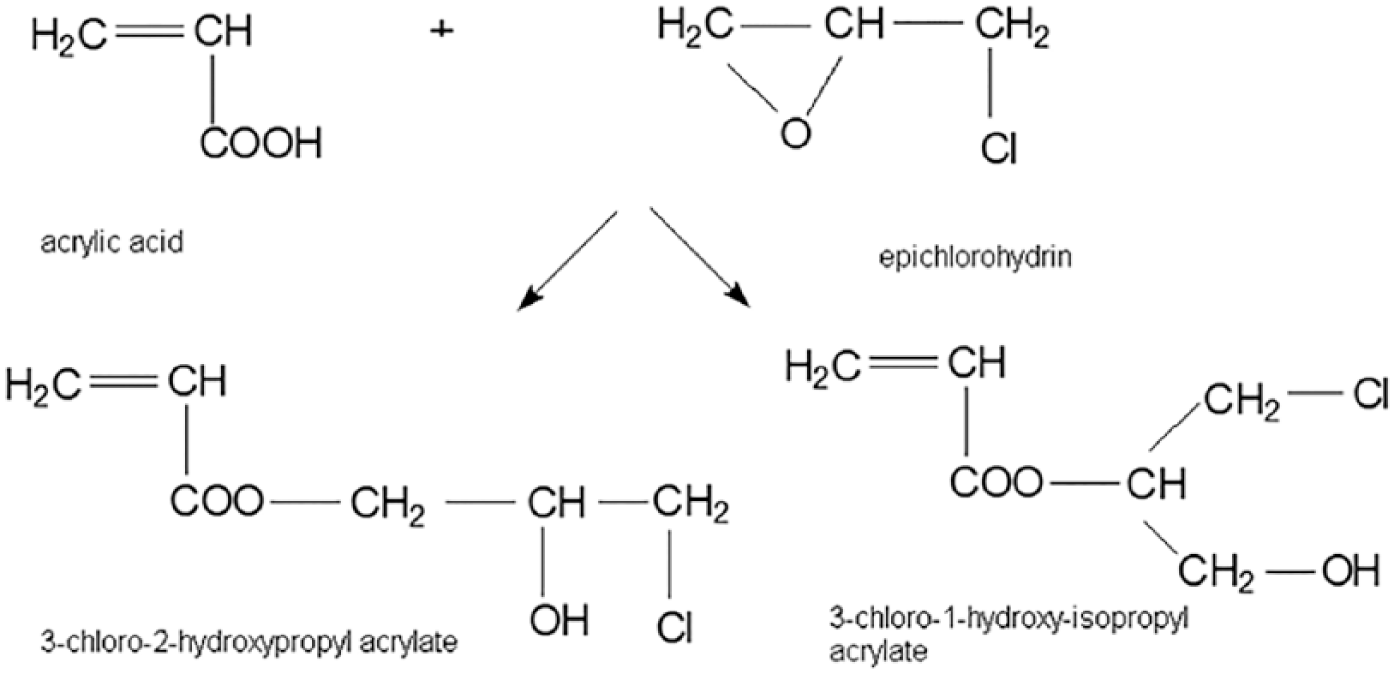
The reaction of obtaining 3-chloro-2-hydroxypropyl acrylate.
Pretreatment of the polyester fabric with NaOH
The polyester samples were washed with a solution of nonionic surfactant LAVOTAN DSU (1.5%) for 60 min at 60°C, Hm = 1:50, rinsed with warm water, then with cold water, and finally dried. Half of the samples used in experiments were soaked in 100 g/L NaOH solution (squeezing degree = 100%), rolled, wrapped in polyethylene foil, and stored for 12 h at room temperature, after which they were energetically rinsed with warm and cold water for removing the NaOH excess. Pretreatment with NaOH (presented in Scheme 2) involves an alkaline hydrolysis reaction, by which it increases the number of OH terminal groups on the surface of the fiber.

Saponification reaction of polyester.
Degradation of the polyester fabric by alkaline hydrolysis reaction was insignificant taking into account that the resulted mass losses (calculated as the average of three measurements) were 4%.
Grafting of the polyester fabric
The saponified and unsaponified polyester samples were treated with a 2% (w/v) ammonium persulfate solution for 30 min at 70°C, squeezed (squeezing degree = 70%), and grafted, where the squeezing degree is the amount of solution retained by the polyester fabric reported to the absolutely dry material, calculated gravimetrically by the following relationship
where m1 is the mass of the fabric soaked solution (g) and m2 is the initial mass of the dried material (g).
The grafting of the samples was carried out in the following conditions:
A set of samples were soaked in an emulsion containing water, 2% (v/v) Tween 80 emulsifying agent, and the final reaction mixture resulted from the reaction of acrylic acid with epichlorohydrin catalyzed by zirconium silicate macrospheres (for the reaction conditions mentioned previously, the yield in CHPA determined by chromatographic analysis was 70%); another set of samples were soaked in an emulsion which contains 90 g/L CHPA, 2 mL AgNO3 0.1 N, and 5 mL aqueous glucose solution (40%).
Squeezing (squeezing degree = 100%)–rolling–wrapping first in polyethylene foil and thereafter in aluminum foil was performed and maintained at 90°C for 60 min.
Soaking in a 20 g/L NaOH solution–rolling–wrapping in polyethylene foil was performed, then stored for 12 h at room temperature, dried at 70°C, and cured for 5 min at 140°C.
After treatment, the samples were washed with a 2% (v/v) LAVOTAN DSU solution for 60 min at 95°C in order to remove the homopolymers and other compounds from the reaction mixture which do not exhibit affinity for the polyester fiber. Finally, the samples were rinsed with warm and cold distilled water, respectively. Because CHPA is attached to the polyester fiber by chemical bonds, it is not removed in the subsequent treatments.
The grafting degree was calculated using equation (2)
where DG is the grafting degree (%) and W1 and W2 are the masses of the ungrafted and dried grafted polyester samples, respectively.
Characterization of the samples
Fourier transform infrared spectroscopic analysis
For the spectroscopic studies, a Bruker FTIR Vertex 70 spectrometer equipped with a diamond ATR (attenuated total reflection) device (Golden Gate; Bruker) and ATR-FTIR (attenuated total reflection Fourier transform infrared) was used. The ATR-FTIR spectra were measured in the spectral range of 800–4000 cm−1, by accumulation of 64 scans.
Energy-dispersive X-ray spectroscopic analysis
The elemental composition of the samples was determined using an EDAX AMETEK energy-dispersive X-ray (EDX) spectrometer attached to the FEI Quanta 3D FEG scanning electron microscope.
Scanning electron microscopy
The scanning electron microscopy (SEM) studies were performed on samples fixed on aluminum supports. The surface morphology of the uncoated samples was examined using a SEM-type Quanta 200 (FEI), operating at 20 kV with secondary electrons in low vacuum mode (large-field detector (LFD)), the magnification being indicated on the micrographs.
Analysis of the comfort indices
In order to analyze the comfort indices, we determined the hygroscopicity and permeability to water vapor.
Hygroscopicity was determined according to the Standard EN ISO 12571:2000 using the following relation
where H is the hygroscopicity; Wu is the average mass (g) of four samples with 50 × 50 mm2 dimensions, kept for 24 h in an atmosphere with a relative humidity of 100%; and Wc is the average mass (g) of the conditioned samples for 24 h in a standard atmosphere (Φ = 65%).
The relative water vapor permeability (Pwv) was determined according to ISO 11092:1993 on a PERMETEST (Sensora) apparatus using equation (4)
where Us is the heat loss of the free wet surface and Uo is the heat loss of the wet measuring head with a sample.
Dyeability with cationic dyes and color measurement
The ungrafted and grafted polyester samples were dyed with ASTRAZON BLUE BG (C.I. Basic Albastru 3) cationic dye. The dyeings were performed with 1.5% dye and 2% acetic acid for 60 min at 100°C on a Mathis Polycolor 2002 digital apparatus. After having been dyed, the samples were washed with 2 g/L nonionic surfactant at 60°C for 20 min, rinsed with warm and cold water, and then dried. The color intensity of the dyed samples (K/S) was determined using the light reflectance technique on Spectraflash SF 300 (Datacolor) equipped with a Micromach 2000 software.
Dyeing fastness
The color fastness to washing was carried out using the ISO 105-C10:2006 standard. The rubbing fastness (dry and wet) was evaluated using a “crock-meter” with 10 strokes of rubbing according to the AATCC Test Method 8-2007. The color fastness to light was determined by the ISO 105-B04 standard.
Antibacterial analysis
Antimicrobial sensitivity was tested in vitro against two bacterial species: Gram positive (Staphylococcus aureus ATCC 25923) and Gram negative (Pseudomonas aeruginosa ATCC27853) according to the Kirby–Bauer disk diffusion method. 31 Round samples of 2 cm diameter were disposed on the surface of a Mueller Hinton solidified agar medium and were incubated in a thermostat at 37°C, the results being interpreted after 24 h.
Results and discussion
Grafting mechanism
In the first step, the polyester fabric pretreated with the polymerization initiator (NH4)2S2O8 was grafted with the CHPA monomer. During the thermic treatment, the polymerization reaction and the formation of polymer on the fiber surface, respectively, occurred.
In the second step, in the presence of NaOH as a catalyst, etheric bonds between the free –OH groups from polyester and –OH groups from CHPA formed (Scheme 3).
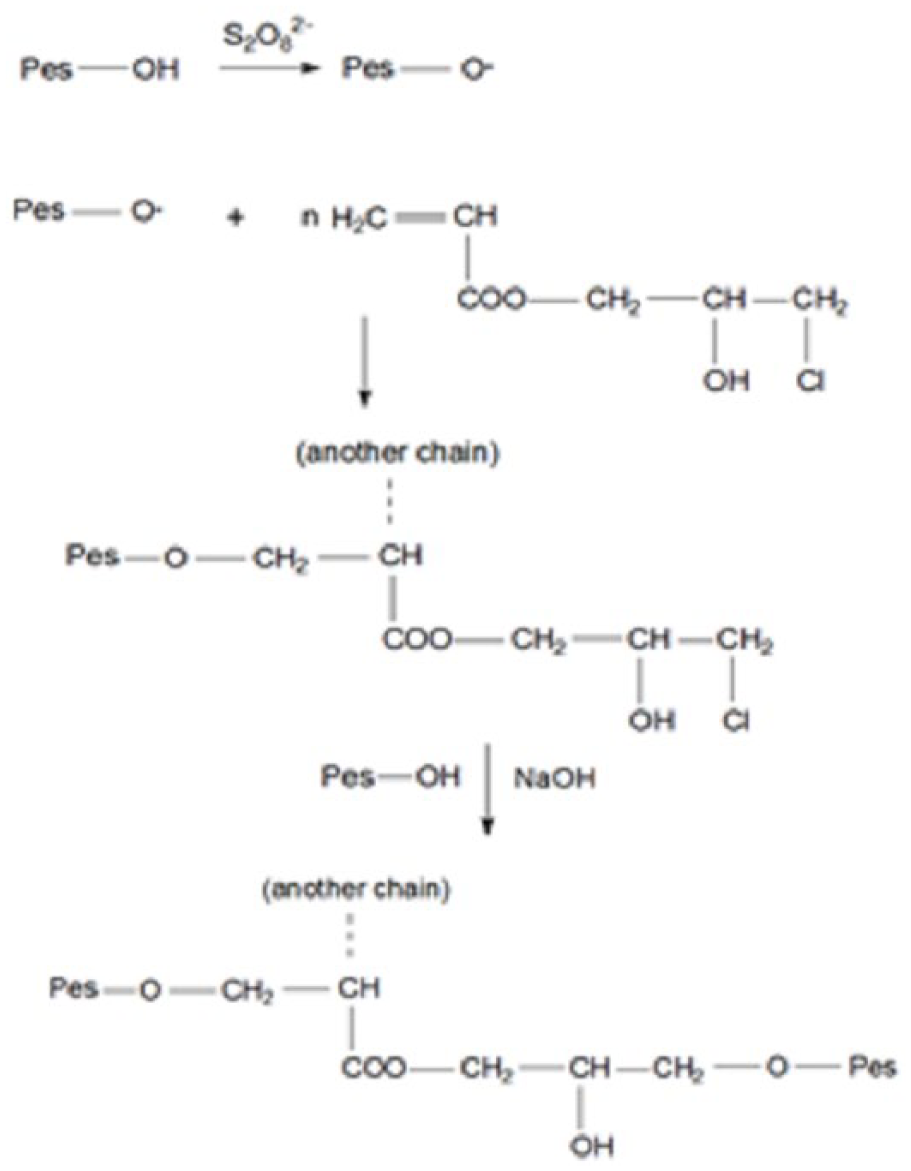
The proposed mechanism for the grafting of polyester fabric with CHPA.
Effect of the monomer concentration on the grafting degree
The untreated and the alkaline-treated polyester samples were grafted with emulsions that contained different monomer concentrations. As can be noticed from Figure 1, the grafting degree increases with the increase of the CHPA concentration used in the grafting experiments.
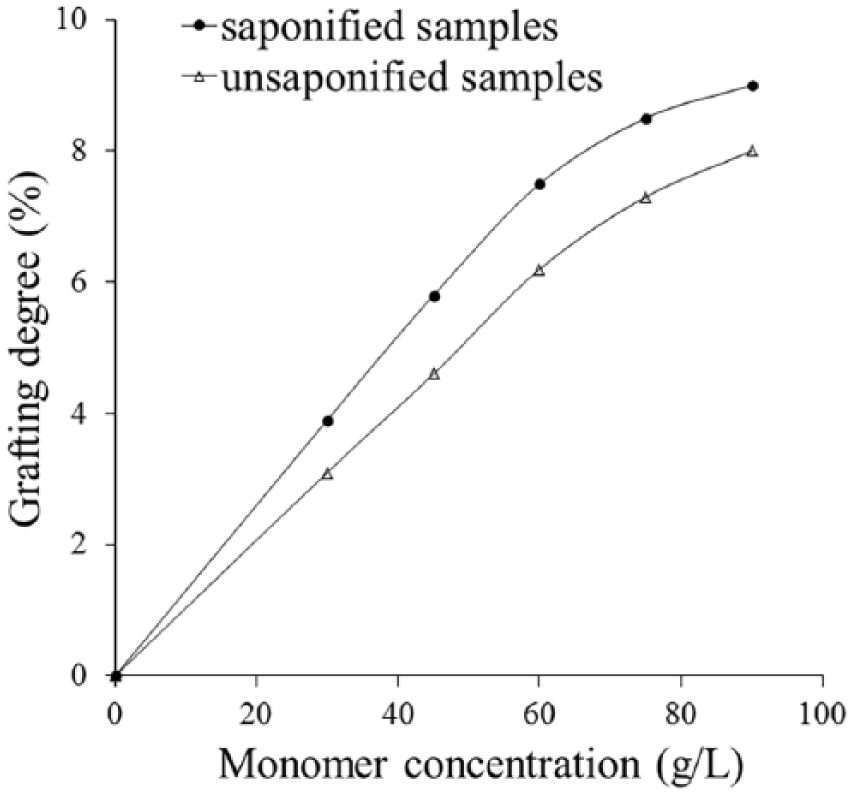
Effect of the monomer concentration on the grafting degree.
Higher grafting degrees were obtained for the saponified samples due to the higher number of active sites formed on the surface of the polyester fiber as a result of the alkaline hydrolysis reaction.
Characterization of grafted materials using Fourier transform infrared spectroscopic analysis
The comparative Fourier transform infrared (FTIR) spectra of polyester, grafted polyester, and Ag-loaded grafted polyester are shown in Figure 2. Increasing the intensities of the peaks located in the 2800–3000 cm−1 regions attributed to the stretching vibrations of the CH2 and CH groups, respectively, confirms the presence of the polymer on the fiber surface. The higher peak that appears at 3417 cm−1 in the FTIR spectrum of the polyester grafted with CHPA suggests the presence of the –OH groups introduced on the surface of the polyester fabric by the grafting. The difference is not very high because, while a number of –OH groups are introduced by grafting of the monomer onto the surface of the polyester fiber, some –OH groups disappear due to their involvement in the formation of etheric bonds with CHPA. It is a very small difference between the intensity of the peak at ~1700 cm−1 of the grafted samples, compared to the ungrafted sample, probably due to the overlap of the C=O group from the polyester with the C=O group from CHP.
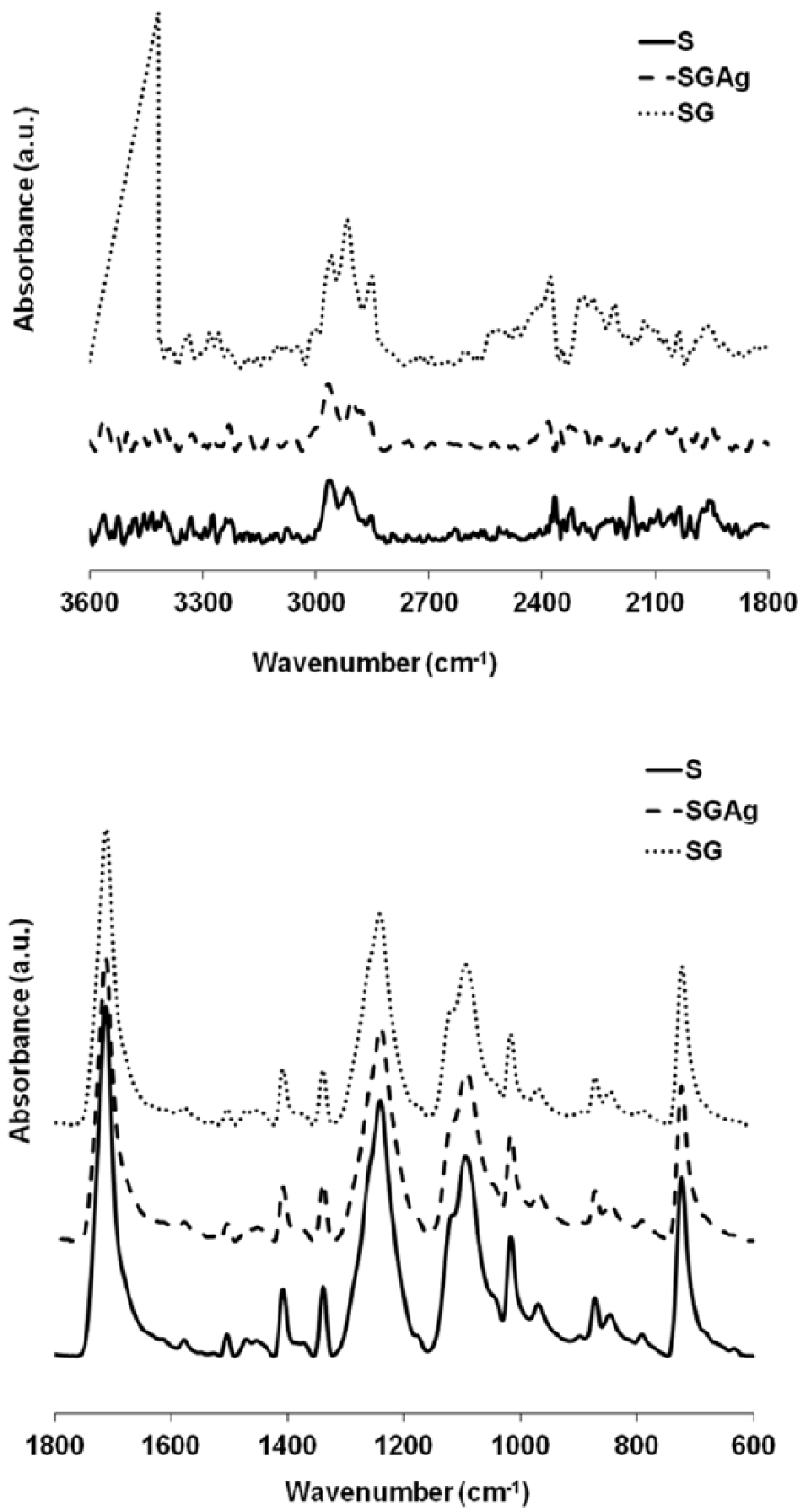
FTIR spectra of the saponified polyester (S), grafted saponified polyester (SG), and Ag/grafted polyester (SGAg).
EDX analysis
The percentage composition of the elements for the studied samples and the corresponding EDX spectra are shown in Figure 3. The values represent the average of three experiments. Because the differences between the mean values and the corresponding values of the three determinations were not greater than ±0.4%, we have selected for each case the graphic representation that illustrates the closest values to the experimentally determined mean value.
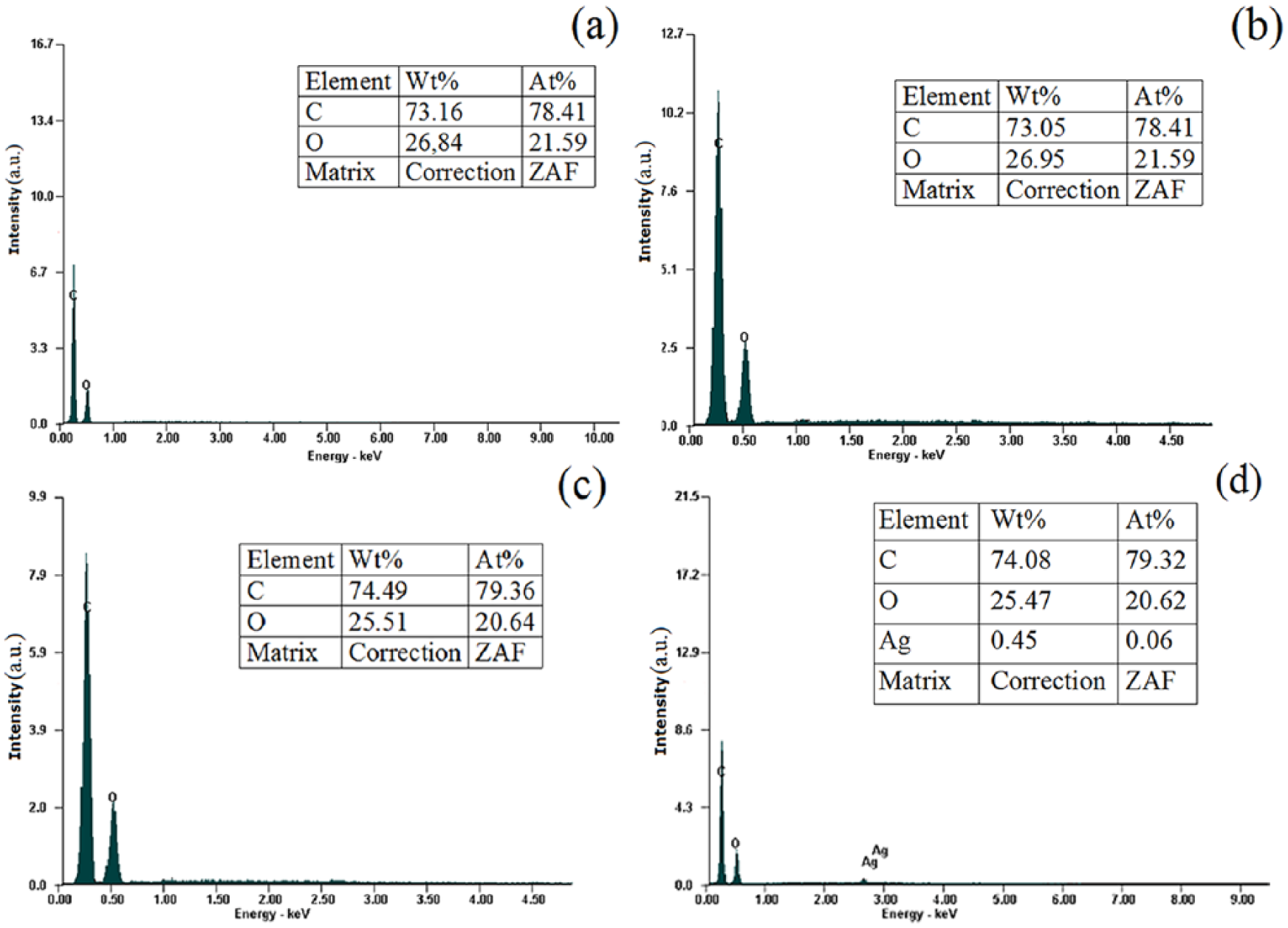
EDX elemental analysis of the polyester samples: (a) untreated, (b) saponified, (c) saponified and grafted with CHPA, and (d) saponified and grafted with CHPA and AgNO3.
The EDX analysis of the untreated sample indicates only the presence of C and O elements (Figure 3(a)). By treating with NaOH, the percentage composition of the elements is not modified significantly (Figure 3(b)). In the case of the grafted sample, an increase in the carbon content can be noticed. Changing the value of the C/O ratio in favor of carbon is due to the higher number of C atoms on the fiber surface, resulting from grafting with CHPA.
SEM analysis
SEM images for the untreated and treated polyester fabrics are presented in Figure 4.

SEM images (5000×) of the polyester samples: (a) untreated, (b) saponified, (c) saponified and treated with CHPA, and (d) treated with CHPA and AgNO3.
By cold treating with a 100 g/L NaOH solution for 12 h (Figure 4(b)), no essential changes of the surface were noticed compared to that of the untreated polyester fiber (Figure 4(a)). In Figure 4(c), the polymer deposition on the fiber surface can be noticed. The presence of silver on the surface of polyester fibers is highlighted in Figure 4(d).
Effect of grafting on the comfort properties of the polyester fabrics
The effect of the monomer concentration used for grafting on the properties of the polyester fabric (keeping unchanged the other working parameters: initiator concentration, temperature, treatment duration—presented previously) was investigated. The obtained results are listed in Table 1.
Effect of the chemical treatments on the comfort parameters of the polyester fabric.
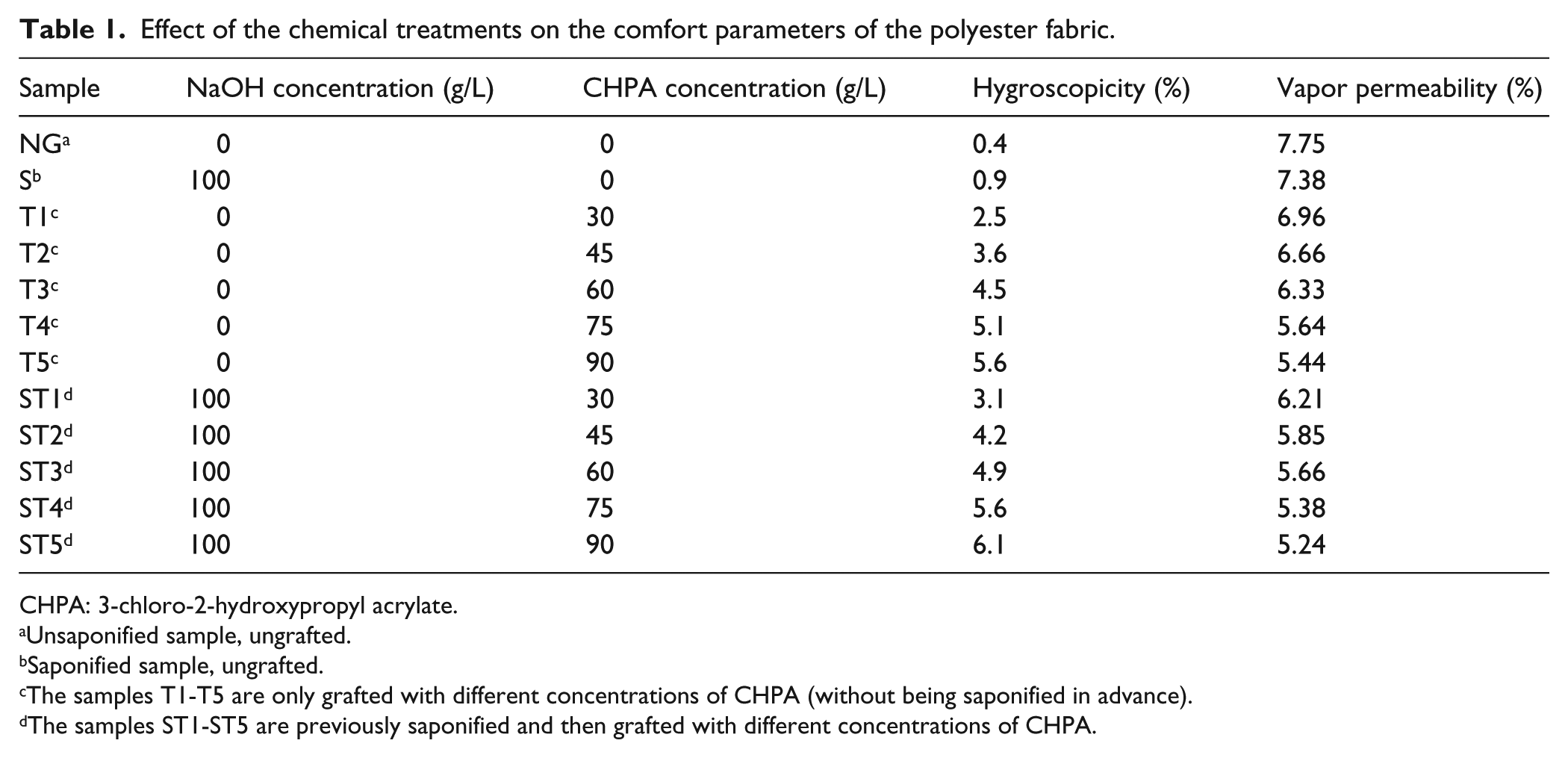
CHPA: 3-chloro-2-hydroxypropyl acrylate.
Unsaponified sample, ungrafted.
Saponified sample, ungrafted.
The samples T1-T5 are only grafted with different concentrations of CHPA (without being saponified in advance).
The samples ST1-ST5 are previously saponified and then grafted with different concentrations of CHPA.
Hygroscopicity
Hygroscopicity is an important physical parameter, which can significantly influence the functional behavior of the polyester materials. Higher hygroscopicity is a necessary requirement for various garments (dress, shirts, socks) to be comfortable to wear and therefore to exhibit appropriate hygienic functional properties. The values for hygroscopicity increase by the increase of the monomer concentration used for treating the polyester fabric due to the higher number of polar –OH groups inserted on the fiber surface by grafting with CHPA. The higher values of hygroscopicity, for the polyester samples pretreated with NaOH (saponified samples) compared to the unsaponified ones, can be explained by the higher number of grafting sites existing on the surface of the saponified fiber.
Vapor permeability
By increasing the concentration of CHPA used for grafting, the number of –OH groups increases and thus the fibers retain a larger amount of water vapor, which prevents their transfer to the outside and therefore decreases the water vapor permeability for the grafted polyester fabrics.
Effect of grafting on dyeing
By grafting of the polyester with CHPA, new –OH groups are inserted on the surface of the polyester fiber, which act as dyeing sites for bonding of the ASTRAZON BLUE BG cationic dye. The results presented in Table 2 indicate a magnification of the dyeing intensity with the increase of the CHPA concentration due to the higher number of dyeing sites formed on the fiber surface.
Effect of grafting on the tinctorial characteristics.
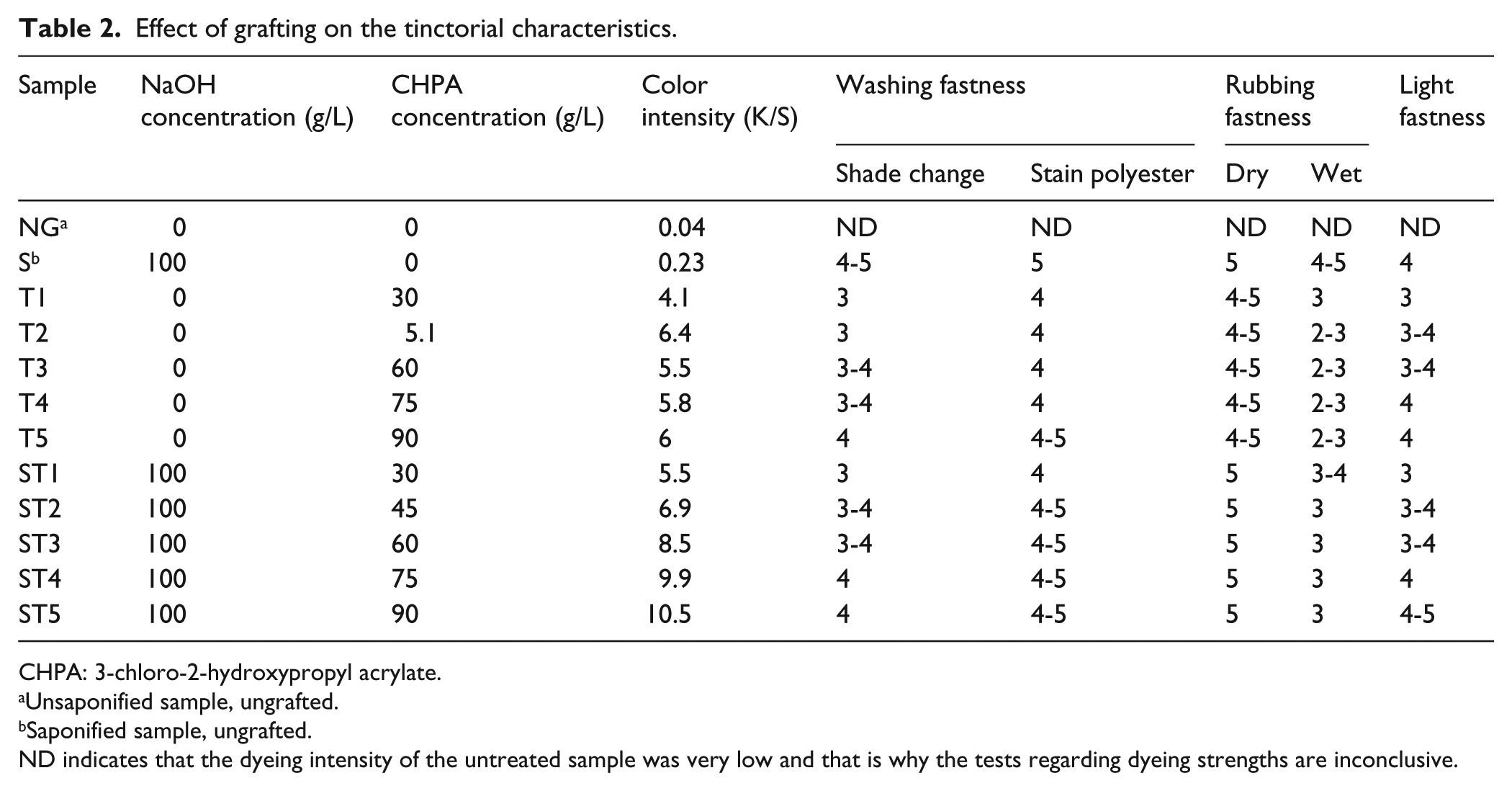
CHPA: 3-chloro-2-hydroxypropyl acrylate.
Unsaponified sample, ungrafted.
Saponified sample, ungrafted.
ND indicates that the dyeing intensity of the untreated sample was very low and that is why the tests regarding dyeing strengths are inconclusive.
The dyeing fastness is also improved by the increase of the monomer concentration used for grafting. The increase in the values of light strength is due to the fact that a higher amount of dye is adsorbed on the grafted fabric surface.
Antibacterial analysis of the polyester samples
Figures 5 and 6 present the results of the antimicrobial activity for the ungrafted and grafted samples. This activity is proved by the appearance of an inhibition zone around the tested samples.

Antibacterial activity of the polyester samples against Staphylococcus aureus: (a) ungrafted sample, (b) sample grafted with CHPA, and (c) sample grafted with CHPA and AgNO3.

Antibacterial activity of the polyester samples against Pseudomonas aeruginosa: (a) ungrafted sample, (b) sample grafted with CHPA, and (c) sample grafted with CHPA and AgNO3.
A better antibacterial activity, demonstrated by a higher inhibition area, was obtained against Gram-negative bacteria P. aeruginosa.
Conclusions
The hygroscopicity, color intensity, dyeing fastness, and antibacterial activity were greatly improved by grafting the polyester fabric with CHPA. The hygroscopicity values of the samples increased from 0.4% to 6.1% for the saponified samples and from 0.4% to 5.6% for the unsaponified samples.
Grafting with CHPA made possible the dyeing of the samples with cationic dyes. The color intensity value (K/S) of the polyester fabric dyed with cationic dye was significantly increased (from 0.04 to 10.5) by increasing the monomer concentration used for grafting, with higher values being obtained for the samples previously treated with NaOH and then grafted with CHPA. The samples treated with the emulsion additionally containing AgNO3 exhibits antibacterial activity against the S. aureus and P. aeruginosa bacteria.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the TUIASI research grant entitled “Macrospherical hierarchical porous metallosilicate materials obtained by mono or multi-templating technique: synthesis, characterization and application” (project no. 0199/2018), awarded by the “Gheorghe Asachi” Technical University of Iasi.




