Abstract
The formulated inks with desirable physical and chemical properties are of significance for inkjet printing, to endow the fabric with expected image quality and color performance. Here, various kinds of water-soluble alcohols are preferably selected and introduced into reactive ink with an objective to improve the physical and rheological properties of ink and print quality of cotton fabric. In this study, the effects of alcohols with different boiling points on the rheology properties, drying behavior, and storage stability of ink were extensively investigated. Moreover, the print image performance, including color strength, fixation rate, ink penetration, and outline sharpness, was evaluated. The results indicated that the inks containing glycerol, diethylene glycol, and triethylene glycol possessed slower evaporation rates, but the storage stability and the dye fixation value of these inks would be decreased. The addition of alcohols into the reactive dye ink could increase the ink penetration and reduce the line image quality. The ink containing 1,2-propanediol with a medium boiling point can effectively satisfy various properties of the reactive ink and improve the fixing rate and printing quality onto cotton fabrics. Furthermore, these findings shed light on the propriate selection of reactive ink with alcohols and provide valuable information for technicians and researchers working in the printing industry.
Keywords
Introduction
Textile inkjet printing is one of the most cutting-edge technologies in the textile industry because it has provided several economic and environmental benefits, such as speed, flexibility, competitiveness, eco-friendliness, high print resolution, and rapid response to frequent style and color change with minimum downtime for change over.1,2 In recent years, with the significant improvement in printing speed and image quality of inkjet printers, printhead manufacturing, and demands in the market for digitalization of print designs, textile printing has attracted increasing attention.
During the printing process, tiny ink droplets are ejected from a small printhead and precisely impact specific locations of the fabric. To acquire satisfactory inkjet printing products, many factors need to be considered, such as the inkjet printhead, inks, fabric pretreatment, and posttreatment technologies. Park et al. 3 discussed the effects of parameters on the inkjet print quality of lines printed on polyester fabric and found that the image quality depended on the fabric construction, finishing, and ink type. Liu et al. 4 revealed the relationship between fabric pretreatment to ink drop spreading and demonstrated that the pretreatment agent enhanced the ability of sodium alginate to control the spreading of ink droplets, thereby improving the color performance. Yuen et al.5,6 studied the influences of factors such as the content of pretreatment paste and the steaming time of four colors. The results showed that not only the individual factors affect the final color yield but also the interaction effects of the factors play a role.
In addition, ink formulation is another challenge in designing digital inkjet printing systems. Ink design is influenced by jetting performance, operation reliability, and storage stability. The ink is usually prepared according to various parameters determined by the head design. 7 As the diameter of the nozzle is normally small and the resolution requirement is high, the viscosity, surface tension, concentration, and containing impurities (salt) of the ink have a high demand. Other factors that must be controlled include the conductivity, pH, and drying speed. Furthermore, textile inks must have good storage stability and excellent print quality on the fabric. Therefore, various types of additives, such as surfactants, a pH buffer, an anticlogging fungicide, and water-soluble polymers, have been used in the ink jet ink for textiles in order to meet the above performance of the ink. Among textile inkjet printing inks, reactive inks are one of the most popular inks due to their excellent water solubility, relatively low price, high wash and crock fastness, and beautiful brightness. Many researchers have explored the effect of reactive ink composition on the properties and color performance of inkjet printing. Soleimani-Gorgani and his colleagues8,9 noted that the structure of the reactive dye in the ink formulation plays an important role in the percentage of dye absorbed on printing cotton. Park et al.10,11 elucidated the interaction of dyes with a water-soluble polymer in ink jet ink for textiles and investigated the relationship between the dye/additive interaction and inkjet ink droplet formation. The physical properties of the ink determined ink droplet formation and printing quality,12,13 and the additives of ink have a great influence on droplet formation and printing accuracy. 14
To prevent potential clogging and to adjust the ink rheology, solvents with miscibility in all proportions, low vapor pressure, good dye solution, and water holding capacity properties were added to the reactive dye ink.15,16 First, the addition of alcohols will affect the viscosity and surface tension of the ink. Viscosity is the most difficult parameter to adjust, and its optimum value varies depending on the printhead technology. High viscosity will cause a problem in droplet formation; low viscosity can lead to nonuniform velocity. 17 The surface tension is another critical factor for printer ink. It plays an important role in the drop formation process and the stability of the droplet during its travel from the orifice to the substrate. Second, when using alcohols with a low boiling point, there is an advantage in that the drying properties of the ink is favorable, the ink should dry as quickly as possible without spreading or feathering to prevent a blurred image when it lands on the fabric, and the definition and color brilliance of the image can be improved; however, the drying speed of ink adhered near the nozzles of the head is increased, and clogging of the nozzles is likely to occur. Typically, for drop on demand (DOD) printheads, the ink surface area defined by the size of the nozzle is small, and the ink will gradually dry up to form a crystalline crust composed of dyes if the idle time between drops is long. Finally, for reactive inks, one common problem is that the reactive group will be susceptible to hydrolysis. The alcohols in the ink formulation that contain hydroxyl groups will react with the part of the reactive dye during storage, as well as during the reaction and fixation steps, which occur under heating and alkaline conditions. If the part of reactive dye is hydrolyzed or undergoes alcoholysis, then the colorant will not effectively fix to the fabric and the color of the printed image will not be optimal. 18 Thus, the water-soluble alcohols must be selected cautiously to achieve stable drop formation and reliable and stable ink, with excellent end-use properties.
The aim of this work was to investigate the influence of the water-soluble alcohols with different boiling points on the ink properties and the image quality of cotton printed fabrics. To achieve this, we selected diethylene glycol, triethylene glycol, 1,2-propanediol, 1,3-butanediol, glycerol, hexylene glycol, and isopropyl alcohol to formulate the ink. Among them, isopropyl alcohol is a low boiling point (<100°C) alcohol, while diethylene glycol, triethylene glycol, and glycerol have high boiling points (>240°C), and the other three alcohols have medium boiling points. First, eight inks, with and without seven types of alcohols, were formulated, and the physical properties, rheological behaviors, and storage stability of the inks were measured. Second, the image quality (color performance, fixation rate, ink penetration, outline sharpness, and color fastness) of cotton printed fabric was evaluated. Understanding the types of water-soluble alcohols on the ink properties and the color appearance of printed fabrics provides valuable information for technicians and researchers working in industry.
Experiment
Materials
Different alcohols include diethylene glycol, triethylene glycol, 1,2-propanediol, 1,3-butanediol, glycerol, hexylene glycol, and isopropyl alcohol. They were supplied by Sinopharm Chemical Reagent Co., Ltd, China. The main physical properties are detailed in Table 1.
The physical properties of the inkjet ink ingredients (20°C).
C.I. Reactive Red 3:1 was selected for the inks. The dye was purified and supplied by Everlight Advanced Chemicals Ltd, China. Other chemicals used for the formulation of inks were triethanolamine (TEA) as a pH-adjusting agent and polyvinylpyrrolidone K30 (PVP, average molecular weight of 40,000) as a viscosity modifier. TEA and PVP were purchased from Shanghai Sincere Biotech Co., Ltd, China. Surfynol 465 is composed of 65 wt% ethylene oxide and surfynol 104. It is a nonionic surfactant containing ethylene oxide and was used as a surfactant agent. A commercial product of surfynol 465 was purchased from Shenzhen Vtolo Industrial Co. Ltd, China. Unless otherwise stated, all chemicals used were analytical reagent grade and completely dissolved in aqueous solutions.
A plain weave cotton fabric that was 100% singed, desized, scoured, and bleached (88.9 ends/in. (warp) × 78.74 picks/in. (weft), weight of 191.5 g/m2) was used, which supplied by Shanghai Textile Industry Institute of Technical Supervision, China.
Ink formulation preparation
For all the inks, 10% dye was used, which is an acceptable dye concentration for reactive inks. The ink with 20% concentration of seven alcohols was prepared using the recipe in Table 2. All percentages were based on weight/weight. The mixture of ingredients was stirred for 30 min at room temperature, and the magnetic stirrer speed was adjusted to approximately 600 r/min. Then, the ink was filtered through a 0.45-µm filter and a 0.2-µm filter to prevent clogging of the nozzles before measurement and print. The ink formula numbers for adding diethylene glycol, triethylene glycol, 1,3-butanediol, hexylene glycol, isopropyl alcohol, glycerol, and 1,2-propanediol were A1, A2, A3, A4, A5, A6, and A7, respectively.
Chemical composition and properties of inks with different alcohols.

A1: diethylene glycol; A2: triethylene glycol; A3: 1,3-butanediol; A4: hexylene glycol; A5: isopropyl alcohol; A6: glycerol; A7: 1,2-propanediol; PVP: polyvinylpyrrolidone K30.
Inks properties characterization
The viscosity and the rheological properties of the ink were measured using a Rheolab QC rheometer with concentric cylinder geometry (DG42; Anton Paar, Austria). The measurements of viscosity were repeated three times at a controlled shear rate of 400 s−1. The surface tension of the inks was measured using an automatic surface tension analyzer (BZY-1; Shanghai Hengping Instrument and Meter Factory, China) and repeated three times. The pH and conductivity of the inks were measured using a digital pH meter (SevenMulti™, Mettler Toledo AG, Switzerland). All the above tests were done at a temperature of 20 ± 2°C.
Ink evaporation loss (drying behavior) was simulated in the lab by keeping 2 ± 0.01 g ink in a culture dish (diameter of 35 mm) at a constant temperature of 20 ± 2°C and a humidity of 65 ± 2% and observing the weight periodically over 120 h. Another observation made from the ink evaporation test was the physical form of the residual dried ink. After storing the ink for 14 days at 20 ± 2°C and 65 ± 2%, photos of the physical form of the dried inks were taken and the remaining weight of each ink was also tested.
Monitoring a formulation over a period of time is a widely accepted method for evaluating storage stability of the ink. 19 The ink was kept in the room at a constant temperature of 20 ± 2°C and a humidity of 65 ± 2%.
Printing process
The fabric pretreatment chemicals included sodium alginate, urea, sodium bicarbonate, sodium sulfate, and a resist agent, which were all industrial grade and purchased from Sinopharm Chemical Reagent Co., Ltd., China. All cotton samples were padded (with 70% pick up) in a pretreatment print paste using a padding machine, manufactured by Rapid Precision Machinery Co., Ltd, China. The paste solution contained 20 g/L sodium alginate, 80 g/L urea, 10 g/L sodium bicarbonate, 20 g/L sodium sulfate, 10 g/L resist agent, and deionized water. After padding the samples with the pretreatment liquor, they were dried at 100°C for 5 min in an oven. Before printing, the samples were conditioned for 24 h at a humidity of 65% and a temperature of 20°C.
The ink jet printing was carried on an inkjet printer (Epson Stylus Photo R330) at 1200 dpi and used the ink formulation shown in Table 2. The printer was a micro-piezoelectric DOD type and was manufactured by Seiko Epson Corporation, Japan.
The posttreatments were as follows: samples were allowed to air dry for 5 min and steamed at 100°C for 15 min with a laboratory DHE steamer (Mathis, Switzerland). A washing procedure to remove the unfixed and hydrolyzed reactive dyes was performed. This involved an initial cold wash, a hot wash at 70°C for 5 min, a hot wash at 90°C for 15 min in an aqueous solution of the soaping agent (2 g/L), a warm rinse, and finally a cold rinse. The samples were then dried for assessment.
Printing quality evaluation
The color strength (K/S) of the printed samples was evaluated using a HunterLab UltraScan PRO (Hunter Associates Laboratory, Inc., USA) reflectance spectrophotometer (illuminant D65, 10 standard observer, d/8° sphere geometry). Each sample was folded twice to obtain a four-fold thickness, and an average of four readings was taken. The color strength of the printed fabric was expressed by the K/S value of the face of the printed sample at the wavelength of maximum absorption (λmax = 545 nm).
Ink penetration was expressed as the penetration factor (PF) and was calculated based on equation (1), which was determined by calculating the ratio of the K/S value obtained from the back of the print to the corresponding value from the face of the print

where (K/S)b is the K/S value of the back face of the print and (K/S)f is the K/S value of the front face of the print. 20
The fixation rate was measured based on the absorbance values of the inks extracted from the cotton fabric before and after fixation. Two square printed fabrics with a size of 5 × 5 cm2 were used to estimate the percentage of fixation. One fabric was washed immediately after printing to obtain the total amount of dye printed on the fabric, and the solution was diluted with deionized water to 250 mL. The other fabric was printed and steamed, followed by washing and soaping to obtain the total amount of dye without fixation on the fabric, and the washing solution and the soaping liquor were diluted with deionized water to the same volume of 250 mL. Then, the maximum absorbance was determined using the ultraviolet (UV)-2550 spectrophotometer (Shimadzu, Japan) at the wavelength of the maximum absorption (λmax = 535 nm). The percentage of absorbed dye fixation was determined according to equation (2) 21
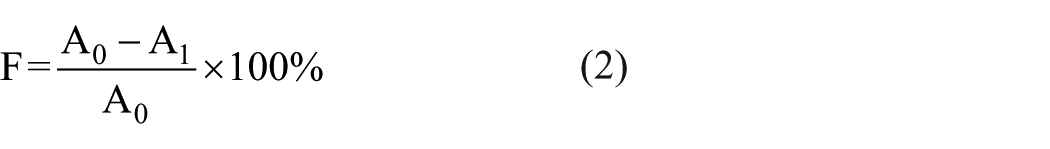
where A0 is the absorbance of the ink solution extracted from the fabric (5 × 5 cm2) before fixation and A1 is the absorbance of the ink solution extracted from the fabric (5 × 5 cm2) after fixation, including the absorbance of the first wash-off solution and the absorbance of the soaping liquor.
To compare the outline sharpness of the printed fabrics, a patch of eight lines with widths of 0.2 × 15 mm2, 0.35 × 15 mm2, and 0.5 × 15 mm2 was measured as width. The line width was defined as the width of the line measured normal to the line from one edge threshold to the other edge threshold (Figure 1). The width of the printed pattern line in the warp and weft two directions was measured using a VHX-1000 super-high magnification lens zoom 3D microscope (KEYENCE, Japan) with a magnification of 100.
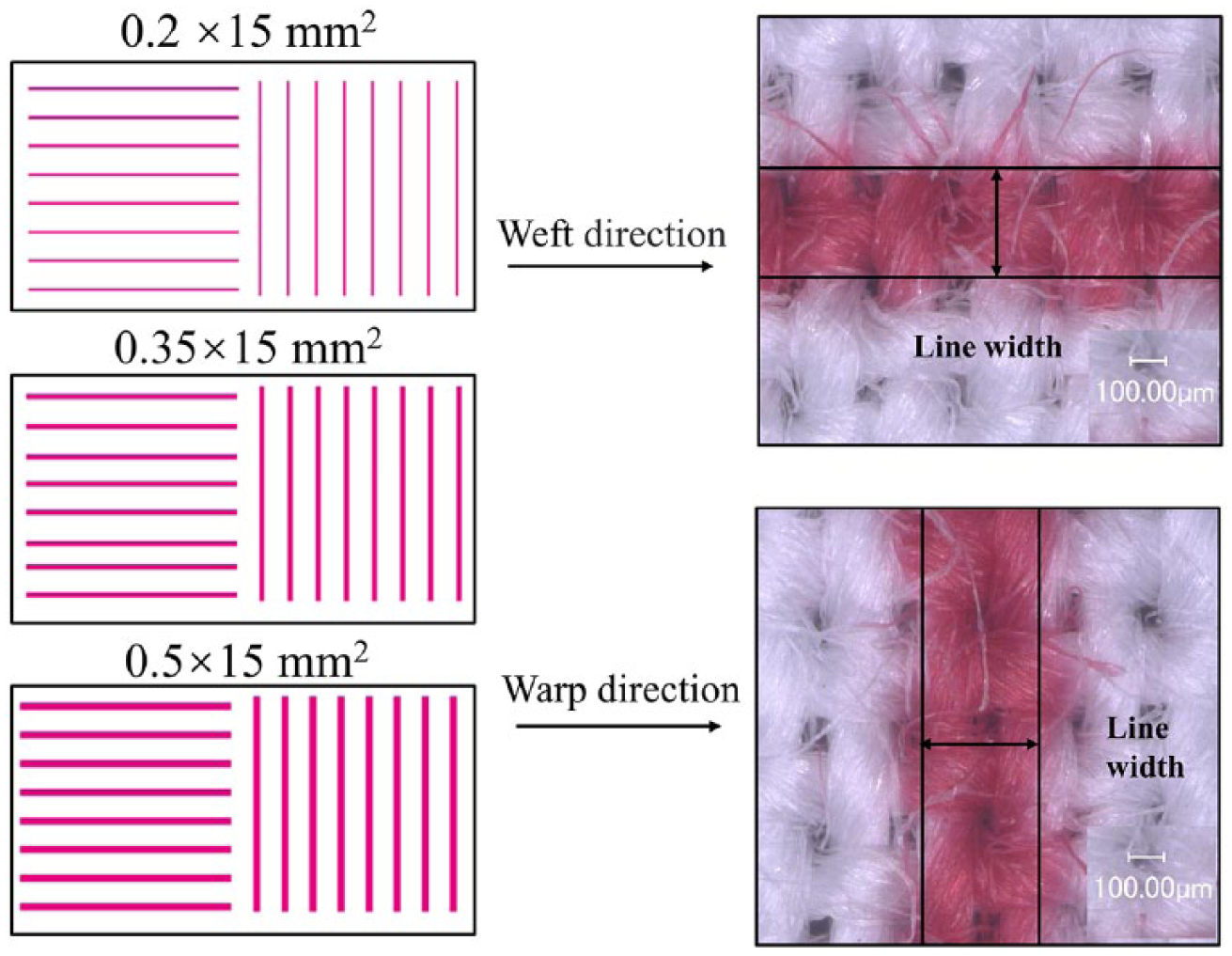
The pattern and principle of measuring printed line quality.
The L*(lightness), a* (redness–greenness value) and b* (yellowness–blueness value) color coordinates, and ΔE values of the printed samples were also evaluated using the UltraScan PRO reflectance spectrophotometer.
The wash fastness of the printed fabrics was determined using a Wash Tec-P fastness tester (Roaches International, England) using the standard test method ISO 105-C06:2010 A1S. The rubbing fastness was measured on a Y571B rubbing fastness instrument (Nantong Hongda Experiment Instruments Co., Ltd, China) according to ISO 105-X12:2016. The color fastness to light was evaluated using an Atlas XenoTest Alpha (SDL Atlas, USA) lightfastness tester according to ISO 105-B02:2014 (method 3) with exposure condition A1.
Results and discussion
Properties of inks with different types of alcohols
For all inks, 10% dye was used. The compositions and properties of the inks containing seven different types of alcohols are summarized in Table 2. The effects of alcohols on the conductivity, pH value, viscosity, and surface tension of the ink were examined.
The seven alcohols have different effects on the viscosity of the ink with the same concentration. These differences can be related to the molecular weight of the alcohols, the polarity of the molecule, the molecular structure, molecular shape, and so on. The viscosity of initial 10% dye solution without any additives was 1.55 mPa·s. The ink A6 has the minimum viscosity value, because glycerol has the highest polarity and the smallest difference in polarity from water molecules, so it has the least effect on the viscosity of the mixed system. On the contrary, hexylene glycol molecule has lower polarity and larger molecular weight. It is easier to form hydrogen bonds with dye macromolecule in solution, which makes the ink A4 have a maximum viscosity value. Therefore, the viscosity of the ink can be adjusted by selecting different alcohols to meet the ejection requirements of the printhead.
The surface tension of the initial 10% dye solution and with 1% surfynol 456 were 68.2 and 34.5 mN/m, respectively, compared with pure water, which was found to be 72.5 mN/m at 20°C. The alcohols added in the ink is not the main factor affecting the surface tension of the ink. However, it can be seen from Table 2 that different alcohols have different effects on the surface tension of the ink, which is mainly related to the polarity, hydrophilicity of alcohol molecules, and the surface tension of the solvent itself.
For DOD printers, the conductivity of reactive inks should be as small as possible to minimize the salt content. High salt content more than 1.0% has a negative effect on the printing behavior, and blocking of nozzles also occurs with a high salt content. When the salt content is higher than 1%–1.5%, the conductivity of 16.8–26.5 ms/cm negatively affects the printing performance. 16 The conductivity of ink A0 without alcohol was 16.22 µs/cm, and the dyes were highly purified. With the addition of alcohol, the electrical conductivity of the ink decreased. Since the polarity strength of water is higher than that of alcohol, the addition of alcohol causes the electrical conductivity of the dye ions to decrease and thus the conductivity of the solution decreases. 22 Various alcohols have different effects on the conductivity of the solution, mainly because the alcohols have different polarity strengths. The stronger the polarity strength of alcohol molecules, the stronger the hydrogen bonding with dyes molecules, which causes the dye to be more easily ionized in solution, so the conductivity of the solution is greater.
The pH value of water-based inks is important because it can affect dye solubility and cause corrosion. Strong alkaline or acid inks may attack and corrode metals if they are allowed to come in direct contact with the printing head. To avoid these problems, the pH of water-based inks is usually adjusted to be neutral or weakly alkaline. Furthermore, if the pH of the reactive ink is too high, then the ink would have an extremely limited storage life, as the dye would rapidly hydrolyze to a nonreactive form, leaving behind a precipitate that will block nozzle and decrease the color depth of printed fabrics. As seen from Table 2, after adding 0.5% TEA buffer solution to the eight inks, the pH value of all the inks was in a similar range from 6.9 to 7.2.
Rheological behavior of the ink
An ideal ink has to show Newtonian rheological behavior in order to be reliable for printing. The ink should exhibit a fairly steady viscosity under increased shear rate values, while the relationship between the shear stress and shear rate has to follow a linear form with a steady slope as close as possible to 1. Examining the shear stress and viscosity of the inks under various shear rate provides clear information about the printability of the inks. The rheological measurements of the different formulations are shown in Figure 2. It can be seen that all seven inks in this research exhibit a clear Newtonian behavior for both the flow curve and the shear stress versus shear rate relationship. This result indicates that these seven types of alcohols can be successfully formulated for ink.

The flow curves and viscosity (inset) for the seven inks.
Evaporation property of the ink
Ink evaporation is considered a crucial characteristic of water-based inkjet inks, and it can be changed by adding a humectant to the formulation. The evaporation rates of the eight inks are illustrated in Figure 3(a). The weight loss of the ink after being placed in the same environment was used to evaluate its moisturizing performance. The weights of A0(water) and A5(isopropyl alcohol) decreased significantly faster than those of the other inks. This result may be attributed to the lower standard boiling point of water, and isopropyl alcohol accelerates the drying of the ink during test; the use of glycerol, diethylene glycol, and triethylene glycol as a humectant in A6, A1, and A2 led to lower weight loss than for 1,2-propanediol, 1,3-butanediol, and hexylene glycol. This phenomenon occurs because glycerol, diethylene glycol, and triethylene glycol have higher standard boiling points, and their chemical structure contains more hydroxyl groups that can form hydrogen bonds with water. The result was that the amount of free water in the ink decreased, which slowed the evaporation. The stronger the effect of the alcohols in the ink solution, the less weight the solution loses. The evaporation of the ink is affected not only by the interaction between various alcohol molecules and water in the ink but also by the interaction between alcohol molecules and dye molecular chains.

Evaporation property of the ink: (a) the weight loss of the eight inks at a constant temperature and (b) the physical form of the residual dried inks.
Another observation made from the ink evaporation test is the physical form of residual dried ink. After storing the ink for 14 days at 20°C and 65%, the photos of physical form of the dried inks were taken, as illustrated in Figure 3(b). The physical forms of A0 and A5 inks were crusty, while a viscous liquid was observed for the A1, A2, and A6 glycerol inks. These different drying phenomena may be attributed to the different boiling points of the alcohols in the ink during test. The drying mechanism of water aqueous ink was the process of free water and organic volatiles gradually evaporating from the ink. The inks A3, A4, and A7 were observed to be sticky residues or powder. A large part of the water and alcohol volatilized, and the dye underwent condensation or cross-linking with small amounts of alcohol and water in the residues, forming a wet and sticky ink film.
The pristine weights of the eight inks from A0 to A7 were 1.9993, 2.0077, 2.0025, 2.0022, 2.0036, 1.9988, 2.0044, and 2.0056 g, and the residue weights of the inks from A0 to A7 were 0.2298, 0.7775, 0.7577, 0.6962, 0.5526, 0.2322, 0.8050, and 0.6538 g, respectively. The residue weights of A0 and A5 were the smallest and approximately equal to the initial amount (0.2 g) of dye added into the formulation, while the residue weights of A6, A1, and A2 were relatively the most in eight inks. It is indicated that the ink to which alcohol is not added or alcohol with lower boiling point is added has poor moisturizing performance, and almost all of the water inside the ink is evaporated. However, glycerol, diethylene glycol, and triethylene glycol, which have high boiling point and contain more hydroxyl groups in the molecule, can form hydrogen bonds with water, and the amount of free water in the ink increases and is not easily volatilized. In summary, A0 and A5 are not suitable for ink jet printing due to poor moisture performance, and other alcohols can be selected as additives to satisfy the moisturizing performance of the ink.
Storage stability of the ink
The structure of the dye, the pH value of the ink, the type and concentration of the additive, and the storage environment all affect the stability of the reactive ink during use. Under the condition of constant temperature and dye concentration, it is closely related to the type of organic solvent added. In this study, the pH value, conductivity, surface tension, and viscosity of the ink jet ink formulations were monitored over a period of 180 days (Figure 4).

Storage stability of ink formulations: (a) pH value, (b) viscosity, (c) surface tension, and (d) conductivity.
The degree of hydrolysis reaction of the active ink solution during storage can be indirectly reacted by the change of the pH value of the solution. As the hydrolysis increases, the pH value of the solution decreases. The pH values (Figure 4(a)) for A5, A7, A3, and A4 remained relatively stable over the period of 180 days, and especially, the ink formulation A5 with isopropanol has the smallest change in pH values. However, the pH values of inks for A1, A3, and A6 decreased significantly over the period of 180 days. This indicates that glycerol, diethylene glycol, and triethylene glycol will accelerate the hydrolysis of the reactive ink when they are added to the ink during storage. Therefore, these three alcohols should not be preferred in order to obtain an ink having better storage stability.
The effect of the seven alcohols on other properties such as the viscosity, surface tension, and conductivity values are shown in the Figure 4(a) to (c). The conductivity values of several ink formulations decrease slightly over the period of 180 days. This may be due to the aggregation of dyes in the storage. Reactive dyes can dissociate into anions and cations in water, so the degree of aggregation and ionization of reactive dyes are related to the electrical properties of the solution to a certain extent. The viscosity and surface tension of the inks remain relatively stable over the period of 180 days. For changes of these three performances of reactive inks during storage process, the impact of alcohols is not very great.
Printing quality of printed cotton fabrics
Color strength, dye fixation, and ink penetration
The color strength (K/S), dye fixation (F), and ink penetration (PF) of the fabrics printed with the eight inks are summarized in Figure 5. It can be seen from the fixation data that A6, A1, and A2 were obviously lower than that of other formulations. The main reason is that glycerol, diethylene glycol, and triethylene glycol can preferentially react with the reactive dye in the reaction and fixation steps carried out by steaming in a high temperature and alkaline environment, which causes part of the dyestuff to lose the ability to react with the fabric and reduces the dye fixation of the printed fabric.23,24 The competitive hydrolysis, alcoholysis, and dye fixation reactions of the reactive dye are illustrated in Figure 6. The boiling points of these three alcohols are the highest, and the ink is less volatile. More alcohols easily remain on the fabrics during storage after printing or steaming process. The reactive dye is more likely to undergo alcoholysis and less effective in terms of fixation to the fabric. Therefore, the dye fixation of the printed fabric with inks added with these three alcohols will decrease. Compared with A0 and A5, the dye fixation of printed fabric with A3, A4, and A7 inks was slightly higher. This may be due to the better ink penetration of A3, A4, and A7 than A0 and A5, which facilitates more diffusion of dyes into the fiber and subsequent fixation.

The color strength, dye fixation, and ink penetration of the printed fabric.

Competitive hydrolysis, alcoholysis, and dye fixation reactions of the reactive dye.
It is also apparent from Figure 5 that the K/S value of the fabric is related to the penetration of the fabric. The K/S values decreased in the order of A0 > A5 > A7 > A3 > A4 > A6 > A1 > A2 and the PF value in the order of A1 > A2 > A6 > A4 > A3 > A7 > A5 > A0. The addition of alcohol obviously increased the penetration of the ink into the fabric. In addition, the eight printed fibers showed a distinct decrease in color strength, while the ink penetration increased. Although the fixing rate of A0 and A5 is not the highest, due to the poor penetration of ink, more dyes are concentrated on the surface of the fabric, which makes the color strength of the fabric the highest. Thus, higher color strength from ink jet printing, in this particular case, does not necessarily result from higher fixation. For inks A1, A2, and A6, the fixation rate on fabrics is lower and the permeability is higher, which eventually results in a lower K/S value of the fabric. Therefore, it can be concluded that the color strength of printed cotton fabrics is affected by two factors: the fixation and penetration of ink in the fabrics. When the fixation rate is similar, the color strength of printed cotton fabrics is inversely proportional to the penetration. The ink with the 1.2 propylene glycol can achieve higher fixation rate and higher K/S value.
Outline sharpness
Since each fabric received the same pretreatment, the variations in print quality can be exclusively attributed to the ink formulation. The printed line width on the cotton fabric in both the warp (vertical) and weft (horizontal) directions are illustrated in Figure 7.
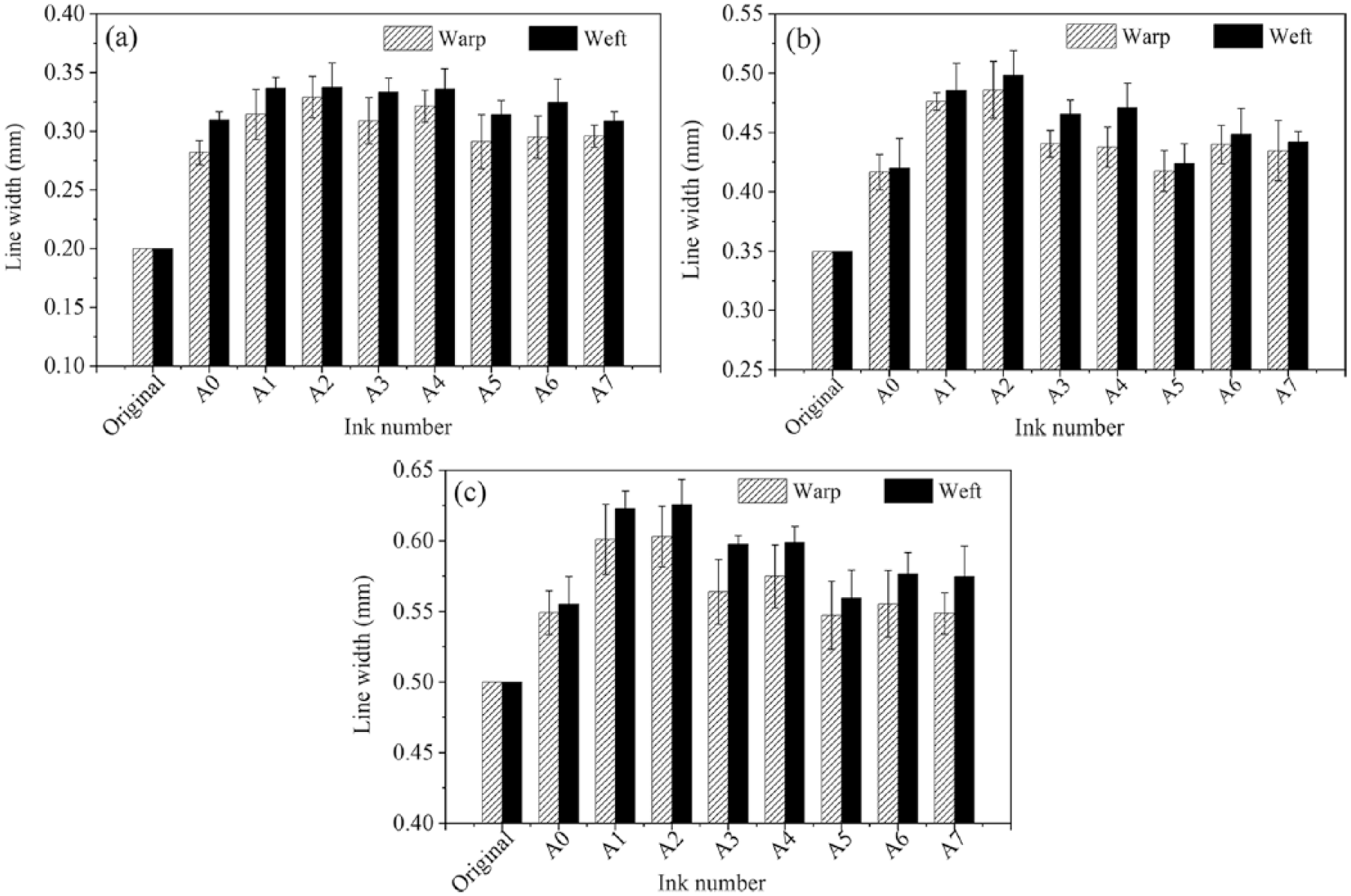
The warp and weft directions line width of printed cotton fabrics: (a) 0.2 mm, (b) 0.35 mm, and (c) 0.5 mm.
For lines of 0.2 × 15 mm2, 0.35 × 15 mm2, and 0.5 × 15 mm2, the line widths (Figure 7) of the eight different inks in the warp direction were all narrower than those in the weft direction. This result is due to the difference in the wicking rates along the warp and weft directions of yarns. 25 The asymmetric fabric structure (88.9 ends/in. for the warp vs 78.74 picks/in. for the weft) caused the higher spreading and wicking of ink in the warp direction because the length of floated yarns in the warp direction was longer than that in the weft direction. Ink was easily wicked by those floated yarns and halted at crossover points, which resulted in the width of the weft direction becoming larger.
When only considering the width of the printed pattern in the weft or warp direction, the widths of the lines of the eight inks showed a similar trend to that seen for the ink penetration data (Figure 5). It is clear from these data that inks A0 and A5 have the lower wettability and permeability on fabrics, so the line widths of the inks A0 and A5 are narrower. Inks A1 and A2 have wider line width due to the high wetting and permeability of ink. For ink A6, the ink permeability is good and the line width is relatively low, because in the ink A6, with a relatively lower viscosity and surface tension, ink droplets formed during printing are smaller and tend to diffuse more into the fabric, improving print definition. In contrast, for ink A4, with a higher ink viscosity, ink droplets tend to diffuse less into the inside of the fabric, and during subsequent processing (air drying/steaming), there is a tendency to spread on the substrate, reducing print definition. The penetration of ink into a fabric is influenced by a range of factors, such as the surface tension and viscosity of the ink, the volatility of ink, and the wettability of ink to the fabric, all of which influence the ink drop spreading property and print quality of fabric.
Colorimetric properties
The colorimetric data L*, a*, b*, C*, h, and ΔE of the fabrics printed with the eight ink formulations are presented in Table 3. The colors of fabrics printed with inks A0, A3, A5, and A7 were darker (lower L* value), brighter (higher C* value), and more reddish (a* value) than those of the other fabrics. This phenomenon may occur because the higher the K/S value of the printed fabrics, the higher the dye concentration on the surface of the fabric and the darker the shade of the color. The difference in color of the eight printed fabrics was accompanied by a change in hue (h values). Comparisons of the colors of the prints with and without (A0) alcohols are also presented in Table 3. As shown, when alcohols were added into the inks, the maximum color differences were obtained for the fabric printed with the ink containing glycerol, with a ΔE of 9.04, and the minimum color differences were obtained for the fabric printed with the ink containing 1,2-propanediol, with a ΔE of 3.84. These results indicated that adding different types of alcohols into the reactive inks affected not only the color strength (K/S value) but also the different color coordinates, as observed for the lightness, chroma, and hue.
Colorimetric data for cotton printed with reactive ink.

A1: diethylene glycol; A2: triethylene glycol; A3: 1,3-butanediol; A4: hexylene glycol; A5: isopropyl alcohol; A6: glycerol; A7: 1,2-propanediol.
Colorfastness properties
The results of the color fastness to washing at 40°C, crocking, and light of the cotton fabrics printed with the eight inks are listed in Table 4. The change in the shade and the degree of staining were assessed visually using a grayscale. As seen from the results in Table 4, the wash fastness of each ink fixed to cotton fabrics was very good, and slight changes were exhibited in the shade of the printed samples, as well as no staining on the adjacent cotton. Furthermore, there was little staining on the adjacent cotton in the case of dry crocking (rating of 4–5) and slightly more staining (rating of 3–4 or 3) on adjacent cotton in the case of wet crocking. The wet rubbing fastness properties of inks A0, A5, and A7 were rated as 3. The main reasons for this phenomenon are that the higher the color strength, the higher the dye concentration on the fabric surface, the greater the dye molecular concentration gradient between the test fabric and the rubbed white cotton, the greater the possibility of transfer of the dyestuff to the white cotton, and the worse the wet rubbing fastness. The lightfastness of each printed fabric was moderate.
Color fastness properties of the printed cotton fabrics.

A1: diethylene glycol; A2: triethylene glycol; A3: 1,3-butanediol; A4: hexylene glycol; A5: isopropyl alcohol; A6: glycerol; A7: 1,2-propanediol.
Conclusion
This study was done to investigate the influence of the water-soluble alcohols with different boiling points on the ink properties and the image quality of printed fabrics. This study has been planned and carried out in a systematic manner, and diethylene glycol, triethylene glycol, 1,2-propanediol, 1,3-butanediol, glycerol, hexylene glycol, and isopropyl alcohol were selected to formulate the ink.
The results indicated that the viscosity and the ink drying ability of the ink can be changed by changing the type of alcohols to the formulation. The inks containing glycerol, diethylene glycol, and triethylene glycol, which have high boiling point as humectants, possessed slower evaporation rates, but the storage stability (pH value) of the ink and the dye fixation value of printed fabric with these inks would be decreased. The addition of alcohols with medium and high boiling point into the reactive dye ink could increase the ink penetration and reduce the line image quality, especially in the weft direction. The inks containing isopropyl alcohol, which has lower boiling point, have the good outline sharpness of printed cotton fabric, but has the poor moisturizing performance.
Therefore, it has been found that if the alcohol with high boiling point was added to the reactive ink, it can enhance the moisturizing properties of the ink, but if its boiling point is too high, such as greater than 240°C, it can affect the storage stability of the ink and reduce the fixing rate of the printed fabric. If the alcohol with a lower boiling point was added to the reactive ink, it is advantageous to improve the print definition of the fabric and reduce the penetration of the ink, but the poor moisture retention of the reactive ink may cause the clogging of the nozzle of the printhead. The use of 1.2 propylene glycol with a medium boiling point can effectively improve various properties of the reactive ink and improve the fixing rate of the ink onto the cotton fabric. The above findings indicate that choosing the right alcohols for reactive inks is very important, which provides valuable information for technicians and researchers working in the industry.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by National Key Technology R&D Program (2017YFB0309800), the Six Talent Peaks Project of Jiangsu Province (JNHB-066), the Applied Basic Research program of “Textile Vision Science” (J201605), China Postdoctoral Science Foundation (2017M621815), and Natural Science Research Projects in Jiangsu Higher Education Institutions (18KJB540004).
