Abstract
A new approach for surface modification of hydrophilic and water-soluble polymeric nonwovens has been investigated. Here, the challenge was to prepare a highly water repelling material from a water-based system. This paradox was resolved by a novel concept of coating chemically reactive water soluble polymers with fluorinated silyl ethers, which was probed with poly(vinyl alcohol) (PVA) nanofibers. Key for getting high water repellence was the discovery that PVA had to be preheated to 180°C prior to silanization before coating with a mixture of tetraethyl orthosilicate (TEOS): n-decyltrimethoxysilane (DTMS): ethanol: H2O: HCl. As a result of this surface modification and additional annealing of electrospun PVA nanofibers, highly water repelling nonwovens were obtained which also showed very low roll angles and consequently high dimensional integrity upon contact with water. The nonwovens were characterized by means of thermogravimetric analysis, wide angle x-ray analysis, electron microscopy, capillary flow porometry, contact angle measurement, and energy-dispersive X-ray spectroscopy to gain in-depth insight into the mechanism of the surface transformation.
Keywords
Introduction
Electrospinning has become the most versatile methods for the preparation of polymer sub-micrometer nonwovens.1,2 It has found applications in several fields such as textiles, tissue engineering, wound dressing, drug delivery, filtration, and agriculture.3–6 Electrospinning can be done from solution, dispersions, and from melts. In general, solution electrospinning results in better control of fiber diameter and fiber morphology. However, the harmful nature of solvents could make the electrospinning process itself dangerous and alter properties of the resulting nonwovens. Electrospinning of aqueous polymer dispersions avoids harmful solvents, but it requires carefully tailored additives and is basically limited to specialty polymers only.7,8 In order to avoid harmful organic solvents for electrospinning, water-soluble polymers such as poly(acrylic acid), poly(ethylene oxide), poly(vinyl pyrrolidone), and poly(vinyl alcohol) (PVA) could be used, but nonwovens of water-soluble polymers disintegrate immediately upon contact with water. It is well known that PVA can be stabilized against water by chemical cross-linking with aldehydes, such as formaldehyde, difunctional aldehydes like glyoxal and glutaraldehyde, boric acid, some dicarboxylic acids like oxalic acid, and maleic acid.9–13 Photo cross-linking of PVA nonwovens has also been carried out to improve their water-stability.14–16 Stabilizations have also been achieved by photo cross-linking during electrospinning of water-soluble polymers as shown for polyethylene imines. 17 It should not remain unmentioned that water-stabilization of PVA has also been accomplished by treatment of PVA fibers with methanol, which increases the crystallinity of PVA and thereby reduces its water solubility. 18 PVA is a chemically reactive polymer which opens the option to further increase the water stability by tuning the surface of electrospun PVA fibers hydrophobic by chemical modification. It would be of outmost interest to generate hydrophobic surfaces from water-borne systems as this combines a highly useful property profile with a sustainable approach.
Large amount of work has been reported in the field of biomimetics to develop hydrophobic surfaces of electrospun materials.19,20 The generation of hydrophobic surfaces is in general possible by coating with low-surface-energy materials such as fluoroalkylsilanes or silicone-based compounds.21–24 Water-stable electrospun PVA nonwovens have been prepared by addition of tetraethyl orthosilicate (TEOS) to spinning solutions. 25 This approach has been used recently by Vanithakumari et al. 26 for fabrication of superhydrophobic and self-cleaning PVA-silica fiber coatings on stainless steel surfaces. However, this route requires careful control of viscosity with time due to the progress of sol–gel method. When using this approach, it has been proved that the electrospun PVA nonwoven mats can become hydrophobic by silanization. The results indicate that water repulsion, governed by the advancing contact angle/receding contact angle of the treated PVA nonwovens, depends significantly on the way of silanization. 27 Although an advancing contact angle of up to 160° has been obtained by silanization with fluorinated silanes, no rolling of water droplets have been observed at different positions of the PVA nonwoven plane. In contrast, PVA nonwovens electrospun with TEOS and then treated with fluorinated silanes showed a significant receding contact angle and thereby water rolling. Again, this procedure complicates the electrospinning process due to the increase in viscosity with time and the large increase in the rigidity of the mat due to the silanization, which is difficult for textile applications.
Recently, superhydrophobic poly(vinyl alcohol-
We report here our finding that hydrophobic PVA nonwovens can be obtained by simple techniques like heat-treatment, thereby eliminating the use of toxic chemicals, followed by silanization of as spun PVA nanofibers. Water-borne electrospun PVA nonwovens can strongly become water-repelling by coating with a special mixture of silanes, which has been further developed to a novel concept for hydrophobic stabilization of water-borne electrospun PVA nonwovens. This straightforward concept for the preparation of hydrophobic electrospun nonwovens could be utilized for a wide variety of polymer materials with protic hydrogens and will thereby open many new options for advanced functional materials for applications in filters, membranes, textiles, implants, tribology, and release applications.
Experimental
Materials
Poly(vinyl alcohol) (Mw ∼ 195,000, degree of hydrolysis ∼98–99%), Sodium dodecyl sulphate (SDS), n-decyltrimethoxysilane (DTMS, CH3(CH2)9Si(OCH3)3)(Alfa Aesar, 98%), tetraethyl orthosilicate (TEOS, Si(OCH2CH3)4)(Aldrich, 99.999% metals basis), ethanol (distilled), hydrochloric acid (37%), and distilled water.
Preparation of PVA nonwovens
PVA nonwovens were prepared by electrospinning. The solution used for electrospinning was 10 wt.% PVA in water, which was prepared by vigorous mixing and constant heating at 90°C for 12 h. SDS was added as a surfactant in the electrospinning solution. Electrospinning setup consisted of a syringe and needle (diameter = 0.55 × 25 mm), a ground electrode, and a high voltage supply. 10 wt. % PVA solution containing SDS was electrospun at 20 kV positive voltage, 21 cm tip to collector distance (the distance between the needle tip and the ground electrode), and 3 mL/h solution flow rate. The average thickness of the PVA nonwovens was 0.082 mm. The PVA nonwovens prepared by electrospinning were treated in the oven at 180°C for 3 min. The solubility of these annealed fibers was analyzed by water treatment for 24 h.
Characterization of the PVA nonwovens
The morphology and the diameter of the fibers of the PVA nonwovens was determined using JSM-7500F Scanning electron microscope (SEM).
The percentage weight loss of the PVA nonwovens after water treatment for 24 h was calculated as follows
Thermogravimetric Analysis
The thermal analysis of the samples was done by using the Mettler thermal analyzer having 851 Thermogravimetric (TG) module from the company Mettler Toledo. The thermal stability of the heated PVA nonwovens was determined by recording TG traces in nitrogen atmosphere (flow rate = 80 mL/min). The measurement was carried out at a fixed temperature of 180°C, and a sample size of 9–10 mg was used for the experiment.
Wide angle X-ray diffraction (WAXD)
X-ray diffraction patterns of the non-heated and the heated 10 wt. % PVA nonwoven mounted on an aluminum cantilever were recorded on a Siemens D-5000 wide-angle diffractometer (Siemens, Germany) equipped with DiffracPlus 3.0 software (BrukerRheinstetten, Germany) at room temperature. Nickel filtered Cu-Kα radiation was used as an X-ray source. WAXD scans were obtained to observe the change in crystallinity before and after heating.
Pore size measurement
The pore size of the PVA nonwovens was determined using the capillary flow porometry (Porous Materials Inc., CFP-1200-AEXL). The sample was saturated with a wetting liquid, Galwick (surface tension = 15.9 dynes/cm). The wetting liquid spontaneously fills up all the pores of the sample. A pressurized non-reacting gas is allowed to displace the liquid from the pores and permit gas flow. The wetting liquid is emptied from largest to smallest pores as increasing gas pressure is applied to one side of the sample. The resulting flow of gas is measured until all pores are emptied. Analysis is then repeated without wetting. The pore size distribution is subsequently calculated from the wet and dry curves according to the Washburn equation. The measurement and analysis of the results were done using the Capwin software.
Contact angle measurements
The water contact angle of the samples was measured using a Contact Angle Measuring System G10 from the Kruess Company. Drop Shape Analysis Software was used for the measurement. An average of at least five droplets of 3 mg of distilled water was taken.
Roll angle measurements
The roll angle refers to the critical angle at which the water droplet of certain volume begins to slide down an inclined plate. The roll angles were measured by varying the volumes of the water droplet on the DTMS-coated PVA nonwovens and measuring the angle at which the droplet slides down an inclined plane.
Electron microscopy
The scanning electron microscopy images (SEM) were obtained using a JSM-7500F (JEOL) scanning electron microscope, with a voltage of 2–4 kV. The samples were directly mounted on the sample holder using a conductive tape. They were then coated with a layer of gold to increase the conductivity of the sample and to get a better image on the detector.
Fiber diameter measurement
Images obtained by SEM were used to determine the fiber diameter of the electrospun PVA nonwovens. The software ImageJ was used to calculate the fiber diameter. In ImageJ software, scale was set depending upon the scale of the SEM image, and the fiber diameter was measured by drawing a straight line between the two ends of the fiber. The value of fiber diameter thus obtained was noted down from the software. An average of at least 50 values was taken for each sample to get the average fiber diameter of that sample.
Energy-dispersive X-ray spectroscopy (EDX)
Small fragments of the sample were mounted on a standard sample holder by conductive adhesion graphite-pad (Plano) and examined with a Zeiss LEO 1530 (FE-SEM with Schottky-field-emission cathode; in-lens detector, SE2 detector or Back Scattered Detector) using an accelerating voltage of 3–10 kV. The samples were vapor coated with carbon (using a Balzers Union MED 010) prior to SEM imaging.
Preparation of sol–gel solution
As spun PVA nonwovens were coated by a sol–gel solution containing tetraethyl orthosilicate (TEOS) and n-decyltrimethoxysilane (DTMS) for improvement of their water stability. The sol–gel solution was prepared with the molar compositions of TEOS:DTMS:ethanol:H2O:HCl = 0.5:0.1:20:11:0.008. 24 The reaction involved hydrolysis and polycondensation of TEOS and DTMS. The solution was then strongly stirred at room temperature for 24 h. The PVA samples heated at 180°C for 3 min were dip-coated once with the DTMS sol–gel solution by immersing the samples into the solution for 2 min followed by drying in air for 24 h.
Results and discussion
The concept for the preparation of hydrophobic PVA nonwovens was to (1) electrospin PVA, (2) to anneal the PVA nonwoven, and (3) to react the surface of the annealed PVA nonwoven with fluorinated silanes by sol–gel treatment. To gain an in-depth understanding of the complete process, we have investigated all steps in detail which are reported below.
PVA nonwovens were obtained by electrospinning and annealed at 180°C for 3 min for enhancement of their water stability. The average fiber diameter of as spun fibers was found to be 530 nm and decreased to 488 nm upon annealing (Figure 1(a) and (b)). Water treatment of as spun and annealed PVA fibers for 24 hours at 20°C showed immediate solubility of as spun fibers while the annealed electrospun PVA fibers were found to be insoluble in water at room temperature (Figure 1(c)). SEM images of (a) as spun PVA, (b) annealed (180°C/3 min) fibers, and (c) annealed PVA fibers after water treatment (20°C/24 h).
The thermal stability of PVA nanofibers was analyzed using a thermogravimetric analyzer (TGA) (Figure 2). Isothermal TGA of the electrospun PVA fiber mat was carried out at a temperature of 180°C for 3 min to observe the effect of heating on weight loss. Temperature-induced cross-linking of PVA by condensation of the hydroxyl groups can be ruled out for the enhanced water stability after annealing, as isothermal TGA at 180°C for 3 min showed a weight loss of only 0.05%. Isothermal TGA of PVA fiber mat at 180°C for 3 min.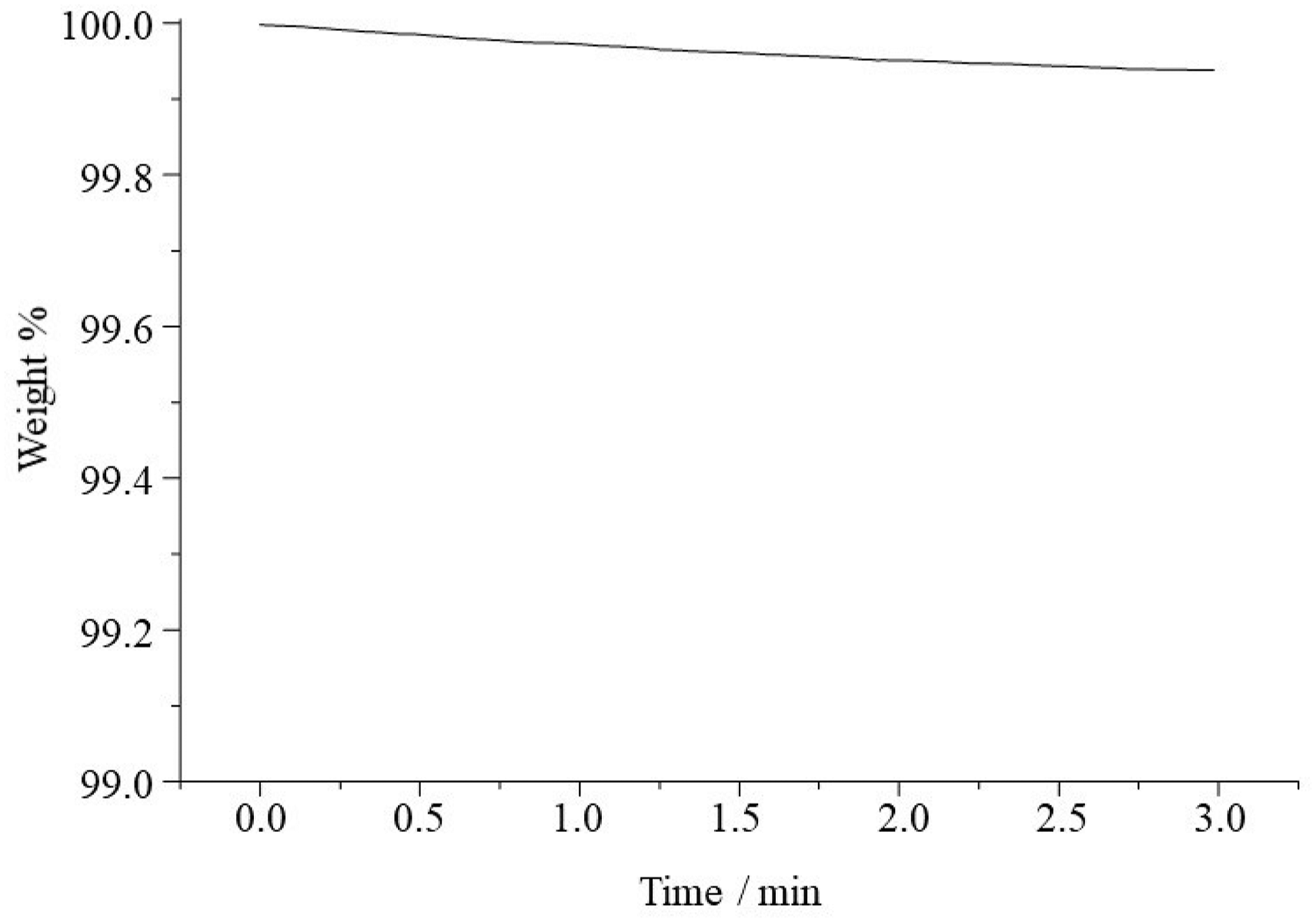
Wide angle X-ray diffraction analysis of PVA fibers (as spun and annealed at 130° and 180°C) showed indeed a significant increase of the crystallinity (Figure 3). The WAXD pattern for PVA fiber mat heated at 130°C showed greater increase in peak intensity as compared to the PVA fiber heated at 180°C. This is attributed to the fact that heating removes the residual water within the fibers. As a result, PVA-water hydrogen bonding is replaced by intermolecular hydrogen bonding between the hydroxyl groups resulting in additional crystallization. However, the intensity of the peak decreased as the heating temperature was increased. The decrease of crystallinity by annealing at higher temperatures (here 180°C) could be attributed to the onset of decomposition of PVA.
30
X-ray diffraction patterns of (A) electrospun PVA nonwovens, (B) PVA nonwoven annealed at 130°C for 3 min, and (C) PVA nonwoven annealed at 180°C for 3 min.
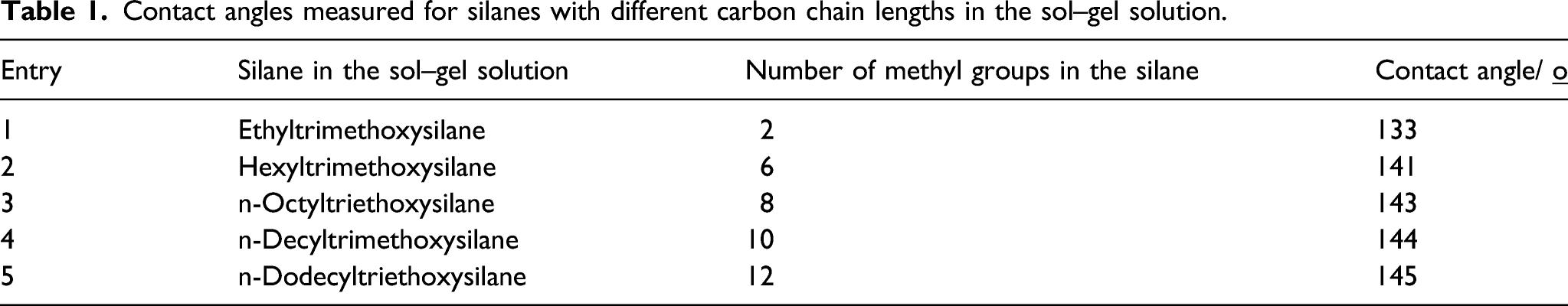
Although the electrospun and annealed PVA nonwovens (180°C/3 min) showed good water stability, they absorbed water easily (see movie 1 in supporting information). The hydrophobicity of the electrospun PVA nonwovens increased significantly when the nonwovens were subjected to a silanization by a sol–gel treatment with different silanes (Scheme 1). The DTMS-coated PVA nonwovens showed a contact angle of 144°. Figure 4 shows a set of water droplets on the surface of a DTMS-coated PVA fiber nonwoven which corresponds to entry 1 of Table 1. The contact angles decreased slightly with shorter carbon chain length of the silanes in the sol–gel solution (Table 1). This proves that the methylene groups present in the silanes were responsible to bring about the hydrophobicity on the surface of the PVA nonwovens. Chemical structures of different silanes (a) ethyltrimethoxysilane, (b) hexyltrimethoxysilane, (c) n-octyltriethoxysilane, (d) n-decyltrimethoxysilane, and (e) n-dodecyltriethoxysilane used in the sol–gel process. Photograph of water droplets on a DTMS-coated PVA fiber nonwoven. Contact angles measured for silanes with different carbon chain lengths in the sol–gel solution.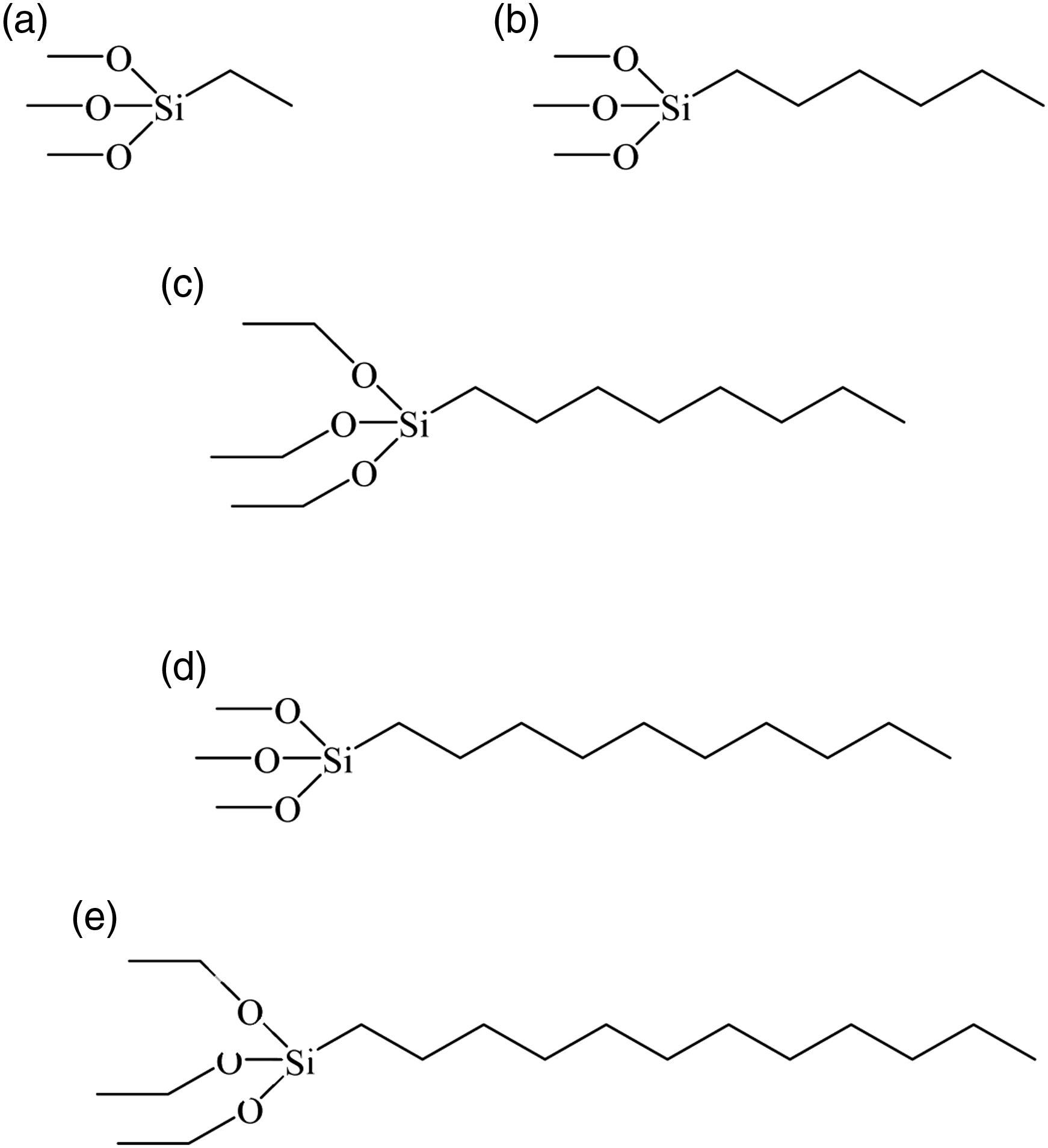

A proposed schematic diagram of sol–gel reaction on the surface of heated poly(vinyl alcohol) fiber mat is shown in Scheme 2. A proposed schematic diagram of sol–gel reaction on the surface of heated PVA fiber mat.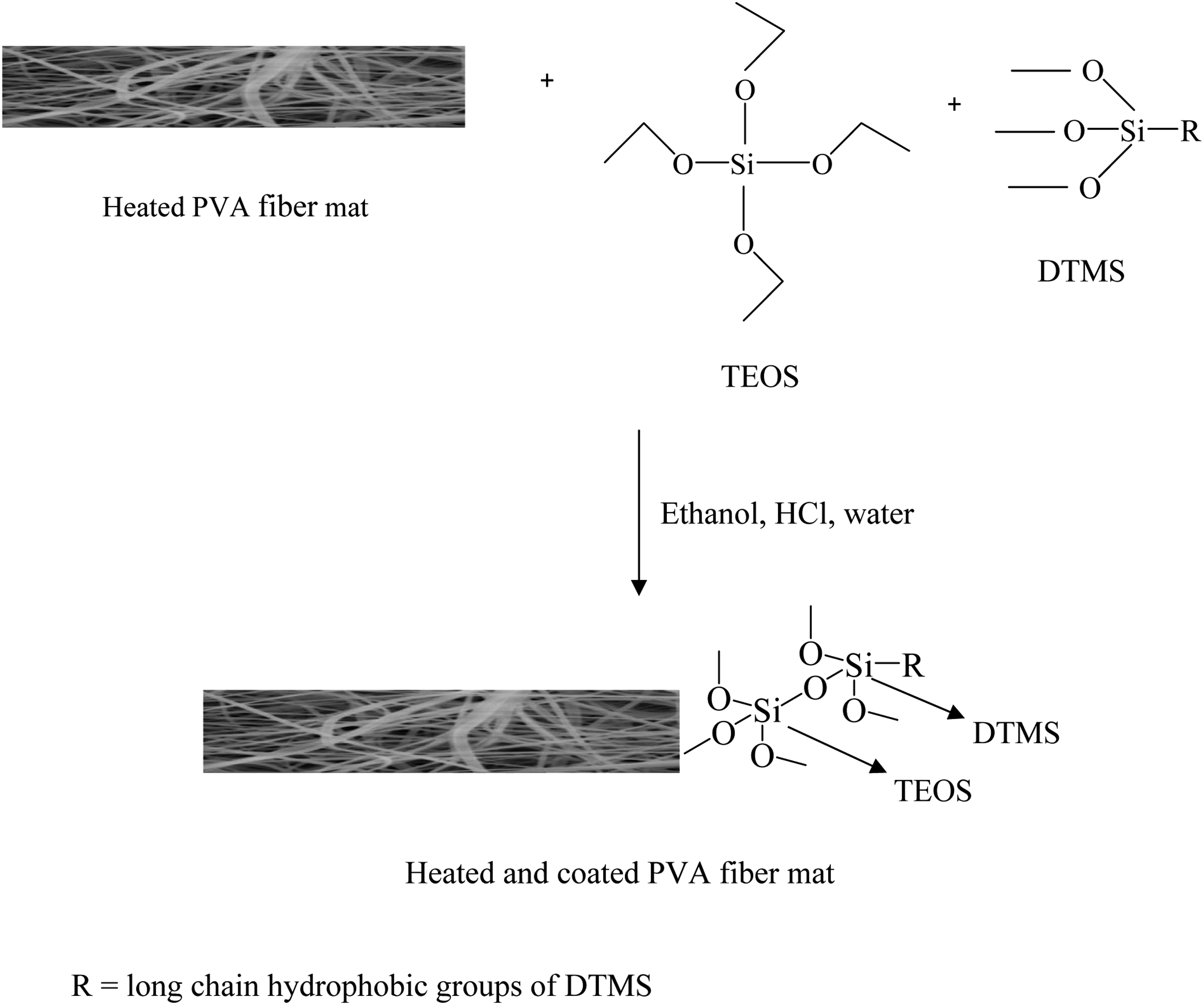
Sol–gel coating of PVA fiber nonwovens had other consequences as well which were investigated in more detail with DTMS-coated samples. The PVA nonwovens coated by DTMS showed an increase of the average diameter of the fibers to 780 nm which marked an increase by about 250 nm in diameter (Figure 5(a)). After water treatment of the DTMS-coated PVA fibers, an average diameter of 810 nm was obtained which indicates that the fiber structure was retained during water treatment (Figure 5(b)). The presence of silane on the surface of the DTMS-coated PVA nonwoven was proved by the elemental analysis of the nonwoven by energy-dispersive X-ray spectroscopy (EDX). Evaluation of the nonwoven by EDX proved the presence of Si on the surface as shown in Figure 6. SEM image of (a) DTMS-coated PVA nonwoven and the same nonwoven (b) after water treatment (20°C/24 h). EDX spectra of DTMS-coated PVA nonwoven shows the presence of Si on the surface.

It appeared from Figure 5 that the DTMS coating of the nonwovens resulted in film formation and thereby a loss of the breathability. However, capillary flow porometry as given in Figure 7 showed that the maximum pore size increased after DTMS coating. The as spun PVA nonwovens were found to have a pore size of 1.3 µm (Bubble point pressure = 0.466 bar) while that for the annealed and DTMS-coated PVA nonwovens was 3.6 µm (Bubble point pressure = 0.177 bar). This can be attributed to the adhesion of fibers after coating, thereby increasing the pore diameter. The pressure required to blow open a larger pore is lower than the pressure required to blow open a smaller pore. This is evident from the fact that the pressure required to blow open the largest pore in as spun PVA nonwovens (bubble point pressure) is higher than that in the case of DTMS-coated PVA nonwovens. The pore size distribution and the average diameter of (a) as spun and (b) annealed and DTMS-coated PVA nonwovens.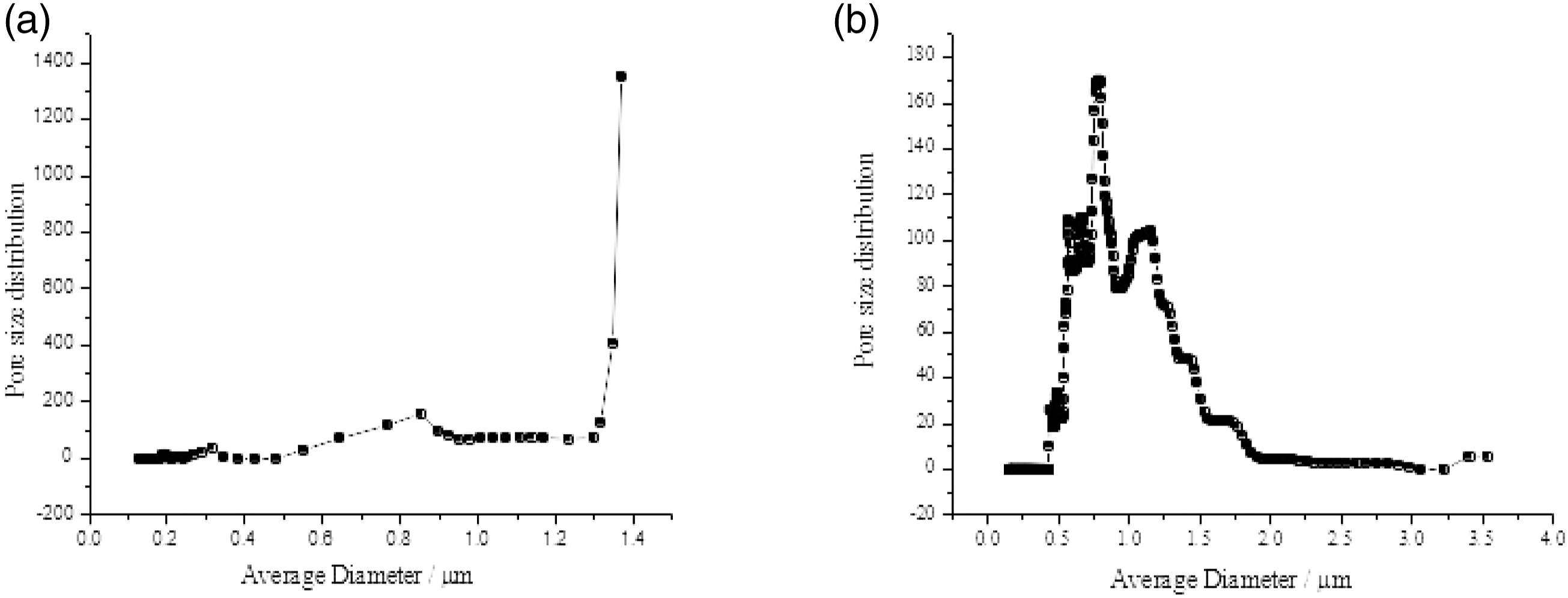
The roll angle for the DTMS-coated nonwovens was found to decrease as the volume of the droplet was increased from 5 µL to 1000 µL as indicated in Figure 8. The maximum droplet size showed a roll angle of 10°. Because of the low water roll angle, the nonwovens were strongly water repelling which can be seen in movie 2 (supporting information). In contrast, as spun or PVA nonwovens showed no roll angle for water by soaking water instantaneously. Roll angle for different volumes of droplet for the DTMS-coated PVA nonwoven.
Conclusion
The contradiction and thereby challenge to obtain hydrophobic nonwovens from water-borne electrospun nonwovens has been accomplished by electrospinning of PVA nonwovens from aqueous solutions and their conversion to highly water stable and hydrophobic nonwovens by simple annealing and sol–gel treatment with fluoroalkylsilanes using sol–gel dip coating method. PVA nonwovens were prepared by electrospinning and were rendered water insoluble by heat treatment at 180°C for 3 min. WAXD analysis and isothermal TGA of the annealed PVA sample confirmed the water stability was due to increased crystallinity. Silanization of the heat-treated PVA fibers by sol–gel treatment using different silanes was carried out to improve their water repellency. With the increase in the length of the carbon chain in the silane, the contact angle increased due to higher hydrophobicity induced by methylene groups present in the carbon skeleton. The fiber structure of the PVA fibers was retained after heating as well as coating which was evident from the SEM images. High contact angles and low water roll angles were observed while the breathability was modified but also retained. It is obvious that the concept present here offers a large potential for future electrospinning based material solutions with even better hydrophobicity and breathability. A major challenge based on the present work could be the direct preparation of superhydrophobic nonwovens from aqueous systems. Electrospinning offers here numerous options for materials improvement which is the topic of presently running investigations.
Supplemental Material
Supplemental Material
Footnotes
Acknowledgements
The author would like to thank Prof. Dr. A. Greiner from Department of Macromolecular Chemistry, University of Bayreuth, Germany and Prof. Dr. Seema Agarwal from Department of Macromolecular Chemistry, University of Bayreuth, Germany for their valuable guidance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author is indebted to DFG for financial support.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
