Abstract
This study aimed at producing the biodegradable composite from lignocellulose nanofibers (LCNFs) and Pebax® thermoplastic elastomer. For this purpose, LCNFs at different levels of 0, 1, 3, and 5% were considered. The LCNFs were prepared by benzyl alcohol and then mixed with Pebax®. The liquid phase of the LCNFs and soluble polymer was prepared and then the masterbatches were mixed in an internal mixer (Model 815802, Brabender, Germany). The mixtures from the internal mixer were put into a hot press and test samples were compress-molded. The physical properties results indicated that water absorption and thickness swelling decreased by the addition of more amount of LCNF. By the addition of LCNFs to polymer, the tensile strength and modulus and impact strength were increased compared to samples without LCNF. No regular trend of enthalpy changes was observed as the content of LCNF changed. When the LCNF concentration was increased to 5%, the crystallization temperature was increased. As the LCNF concentration increased to 3%, the glass transition temperature (
Introduction
The interest in nanocomposites has increased markedly in recent years due to the great potentials associated with this relatively new group of materials. Nanocomposites are defined as composites with reinforcement in the nanometer range (<100 nm) in at least one dimension. 1,2 Nanocomposites can be divided into three groups, depending on the geometrical shape of the nanoreinforcement. One-dimensional nanocomposites are polymers reinforced with sheets with a thickness in the nanometer range. Examples of nanoreinforcement in this group are different kinds of clays and graphite. Two-dimensional nanocomposites are reinforced with tubes or whiskers with a diameter in the nanometer range; examples are carbon nanotubes, cellulose, and chitin whiskers. The third group, three-dimensional nanocomposites, is reinforced by spherical particles in the nanometer range. Examples of nanoreinforcement in this group are nanosized metals, metal oxides, silica, and carbon. 3
Using nanomaterial, it is possible to produce composites with higher wear and abrasion strength, good dimensional and thermal stability, and better mechanical strength compared to conventional composites. 4 –6
Pebax® is polyether-b-amide elastomeric thermoplastic which confers good specific mechanical properties such as a high elongation at breaks. Moreover, it has a melting point below 200°C helping to prevent wood fibers degradation and a hydrophilic characteristic which enables it to interact with natural fibers. Pebax® copolymer consists of rigid polyamide segments, acting as physical cross links, and flexible polyether segments; its properties depend on the polyether/polyamide ratio. 7 –11 Pebax® is plasticizer-free engineered thermoplastic elastomer and comes from renewable resources. This is a good point as our final objective is to elaborate a composite mainly issued from natural resources: bio-based composites.
Sliwa et al. 12 studied the thermal stability of a new family of wood polymer composites which use a thermoplastic elastomer matrix (Pebax® copolymers). These polymers present a hydrophilic characteristic being capable to interact with the wood fibers. They showed a spectacular improvement of thermal stability of the composites under air atmosphere, as opposed to measurement performed under nitrogen atmosphere. The presence of wood fiber in Pebax® hinders the thermo-oxidation in air by the formation of char residue in the earlier stages of degradation.
Recently, the use of nanocellulose materials in applications like biodegradable polymer composites has been explored extensively. Lignocellulose nanofiber (LCNF) is an unpurified form of cellulose nanofibers (CNFs) containing lignin and insoluble hemicellulose along with cellulose fibers. Because of the presence of these fibers besides cellulose, the hydrophobicity and also mechanical stiffness of LCNF would be more than CNF. 13 The LCNF also can be produced by mechanical and chemical pretreatments followed by high-pressure homogenization from the pulp of woods and wastes of agricultural crop. 14 These nanofillers exhibit excellent properties such as good barrier and transparency properties, desirable crystalline structure, high specific surface areas, convenient access, being renewable, having low thermal expansion coefficient, and compatibility with most biopolymers. 14,15 It has been shown that nanocellulose/montmorillonite composite films were prepared from oxidized cellulose nanofibrils showing ultrahigh mechanical and oxygen barrier properties through the nanolayered structures. 16,17 The mechanical performance improvement of polysaccharide-based biocomposite fibres which used alginate fibers that employed cellulose nanocrystal (CNC) and its oxidized derivative as the reinforcing nanofillers was reported. 18
Numerous papers have been published about the improving effect of CNF on the barrier and mechanical properties, 15,19,20 as well as the release controller of additives 21 in biodegradable edible films, but there are few reports on the use of LCNF and its effects on polymer or biopolymer composites.
The purpose of this study was to develop a method for producing Pebax® nanolignocellulose composites. In order to investigate the effect of the LCNF concentration on physical, mechanical, and thermal properties of the nanocomposites, samples with varying amount of LCNF were produced by the internal mixer and compress molded machine. It was also interesting to evaluate whether the LCNF contents within the composite affected the thermal behavior of composites or not. Further, Fourier transform infrared (FTIR) analysis was used to study how the chemical bonds of composites were affected by an increased amount of LCNF.
Experimental materials
The polymer matrix used in this study was Pebax® 55R53 SP 01 (polyether-b-amide) with a density value of 1.03 g cm−3. Its melting temperature was 167°C and was supplied by Arkema Colombes Cedex, Co. France. The benzyl alcohol used in this work was purchased from Merck, Germany. In this study, LCNF was purchased from Nano Novin Polymer Co. Iran. The X-ray diffraction and transmission electron microscope (TEM) images of LCNFs gel taken by Bruker Advance D8 (Karlsruhte, Germany) and Zeiss Leo 912 AB, Germany, and its apparent characteristics were given in Figure 1.
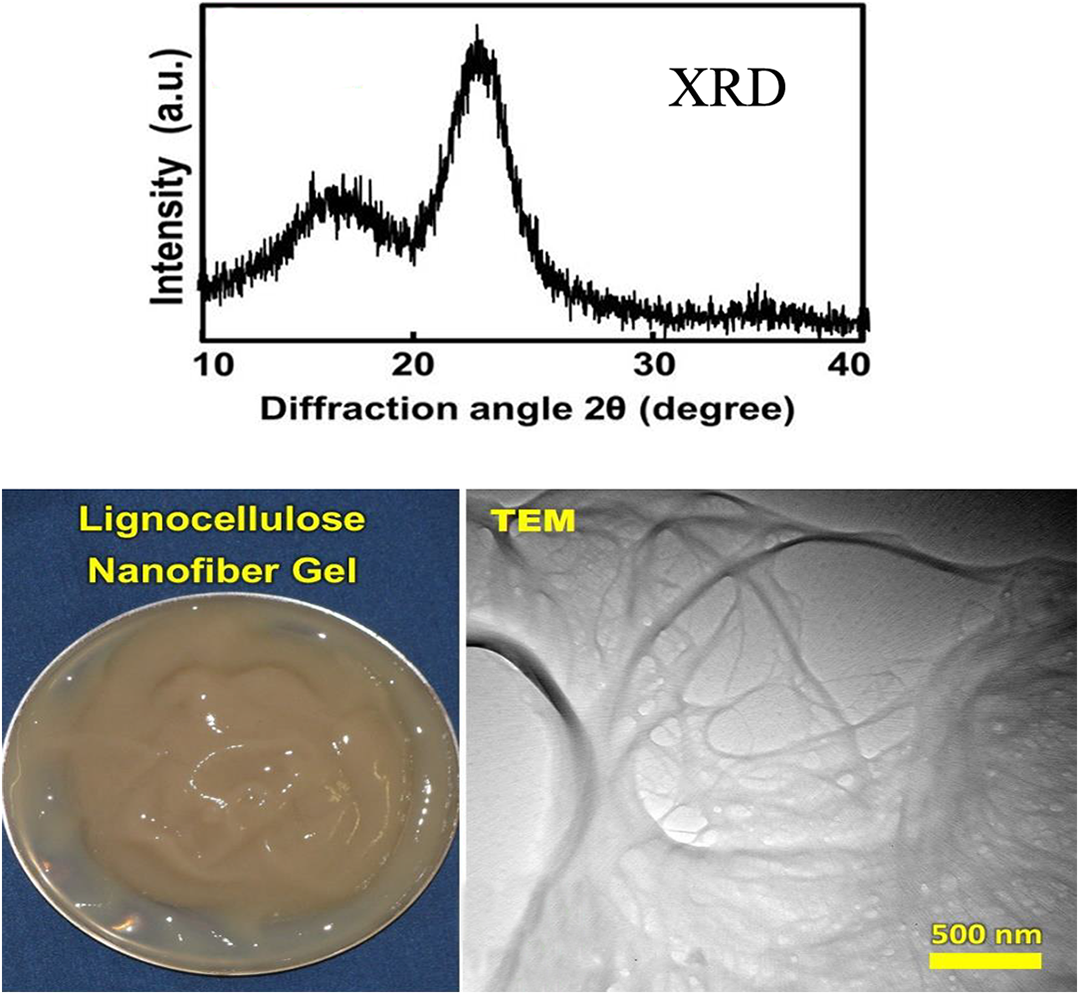
XRD, TEM, and the apparent color of lignocellulose nanofibers gel produced by Nano Novin Polymer Company.
The morphological evaluation was performed by a TEM). Samples for the morphological phase study were first cryo-fractured under liquid nitrogen atmosphere and then sputter-coated with a thin gold layer on the fractured surface before TEM observation.
Figure 1 shows the SEM image of the fractured cross section of the neat LCNF, which clearly depicts the lamellar structure and voids throughout the film are believed to be due to the fact that the fibrils tend to form clusters of layered sheets leaving voids. This finding agrees with other studies where the nanofibrils were found to be organized in layers with their principal axis in the plane of the film. 22,23
LCNFs (with an average diameter of about 55 nm, an average length of about 2–5 μm, a specific surface area of 106 m2 g−1, and 99% purity) were produced from unbleached hardwood pulp through mechanical and acid treatments by a super-grinding procedure. 24 At first, Neutral Sulfite Semi Chemical (NSSC) wood pulp was rinsed with distilled water three times and then was placed in a 5% concentration of potassium hydroxide solution for 1 h at 80°C under the mechanical stirring. After this alkaline treatment, NSCC suspension with a 1% concentration was prepared and then passed three times through the super-grinding disk machine (MKCA6-3; Masuko Sangyo Co., Ltd., Kawaguchi, Japan) to produce LCNF. The super-grinding disk machine consisted of a static and a rotating grinder disk. The grinding stone was silicon carbide and its diameter was 6 in. The time and speed of grinding were 40 g h−1 and 1800 r min−1, respectively. The energy consumption of the grinder was 25 kWh kg−1 and the nano-size fibers in gel form were achieved. The specific Young’s modulus, the specific tensile strength, and strain at failure of the neat LCNF films were reported as 7.6 GPa g cm−3, 46.8 MPa g cm−3, and 1.9%, respectively. 23
Methods
Masterbatch preparation
Pebax® has a high chemical resistance to various chemicals and solvents because of its structure as a block-copolymer of polyamide and polyether. After applying various chemicals, benzyl alcohol proved to be a good solvent for Pebax®. Specific amount of Pebax® was mixed with benzyl alcohol (at concentrations of 6-6) at 100°C for 3.5 h and then was stirred at room temperature until it was dissolved and a homogenous solution was prepared. After that, nanolingocellulose–ethanol solution was substituted with benzyl alcohol by applying the centrifuge technique. With the aim of producing an LCNF solution that was able to combine with polymer solution, at first, the liquid phase of LCNF was substituted by the ethanol. To achieve this, LCNF gel was centrifuged at 10000 r min−1 at 20°C to remove water and then ethanol was substituted. This process was repeated three times.
The LCNF and polymer solution were mixed and stirred at 60°C. The stirring time was changed between 5 min and 10 min based on the viscosity of the solution. The mixture was poured into the laboratory Petridis gently to prevent the creation of air bubbles. The gel-like masterbatches were put into a laboratory oven for approximately 3 days and the solvent was evaporated.
Nancomposite sample preparation
The Pebax® granules and masterbatches were oven-dried at 75°C for 8 h to remove the absorbed water before the preparation of samples. The production of nanocomposites was carried out in an electrically heated internal mixer (Model 815802, Brabender Technologie GmbH & Co. KG, Duisburg, Germany). The temperature and rotor speed of the internal mixer were fixed at 180°C and 60 r min−1, respectively, for 10 min.
The mixing process was started by melting Pebax® granules into the mixer and after 2 min the prepared masterbatches were added into the internal mixer and the mixing process took 10 min and then the mixture was cooled and milled. The mixtures from the internal mixer at different LCNF concentrations were put into a mold and then were compress molded with a Nautilus hot press (Villeurbanne, France) into the test samples at 190°C and 15 MPa constant pressures for 15 min. Then, the press temperature was reduced to 80°C and samples were cooled under atmospheric pressure. The formulations of the samples and abbreviations used for the respective mixtures prepared are given in Table 1.
Composition of evaluated formulations.
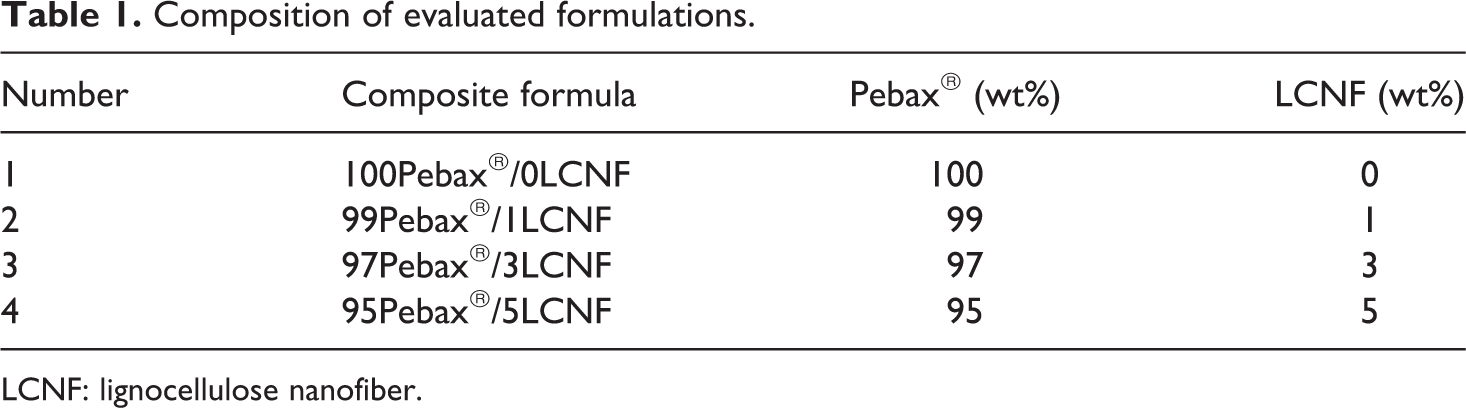
LCNF: lignocellulose nanofiber.
Measurements
Mechanical testing
The tensile and notched impact strengths were conducted using Santam STM-So (Iran) and Zwick Z250 (Germany), respectively. The tensile and impact strength of five samples were measured for each treatment, respectively, based on ASTM D638-10 and ASTM D256-10 standards. The tensile tests were performed at a crosshead speed of 5 mm min−1.
Physical properties
The 24 h water absorption and thickness swelling of samples were measured based on ASTM D-7031-04 standard. Five specimens of each nanocomposite were selected and dried in an oven for 24 h at 102 ± 3°C.
The weights of dried specimens were measured with the precision of 0.001 g using a high precision weighing machine. The specimens were then placed in distilled water and kept at room temperature. The specimens were removed from the water and the surface water was wiped off using blotting paper. The weights of the specimens were measured at different times with a high precision weighing balance.
The values of the water absorption were calculated in percentage using equation (1)

where
A similar procedure for water absorption was applied to determine the thickness swelling of the samples. The specimen thicknesses were determined by taking a measurement at a specific location, the diagonal crosspoint, on the sample with digital calipers. The values of the thickness swelling in percentage were calculated using equation (2)
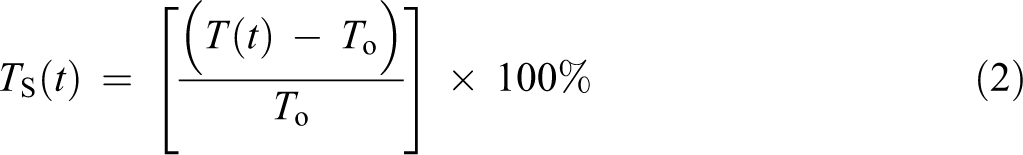
where
Differential scanning calorimeter
The technique of differential scanning calorimeter (DSC) was employed to measure the enthalpy (
FTIR spectroscopy
The chemical bonds on the polymer matrix were detected by FTIR (Equinox 55, Bruker, Germany). The samples were scanned in the frequency range from 500 cm−1 to 4000 cm−1 at a resolution of 4 cm−1. Thin films of nanocomposite were prepared for the FTIR analysis.
Statistical analysis
Statistical analysis was conducted using SPSS software (version 16) as analysis of variance. Also, the Duncan multiple range test was used to test the statistical significance at
Results and discussion
Fourier transform infrared
Figure 2 shows the spectra for nanocomposite samples containing 1, 3, and 5% LCNF and neat Pebax®. At pure Pebax® samples, the highest intensity of absorbance was observed at wave numbers from 1692 cm−1 to 1749 cm−1 which were related to carbonyl groups (C=O). The peaks at wave numbers from 1540 cm−1 to 3670 cm−1 were related to compounds containing nitro like N–O and hydroxyl groups, respectively, and showed stretching frequencies. 25
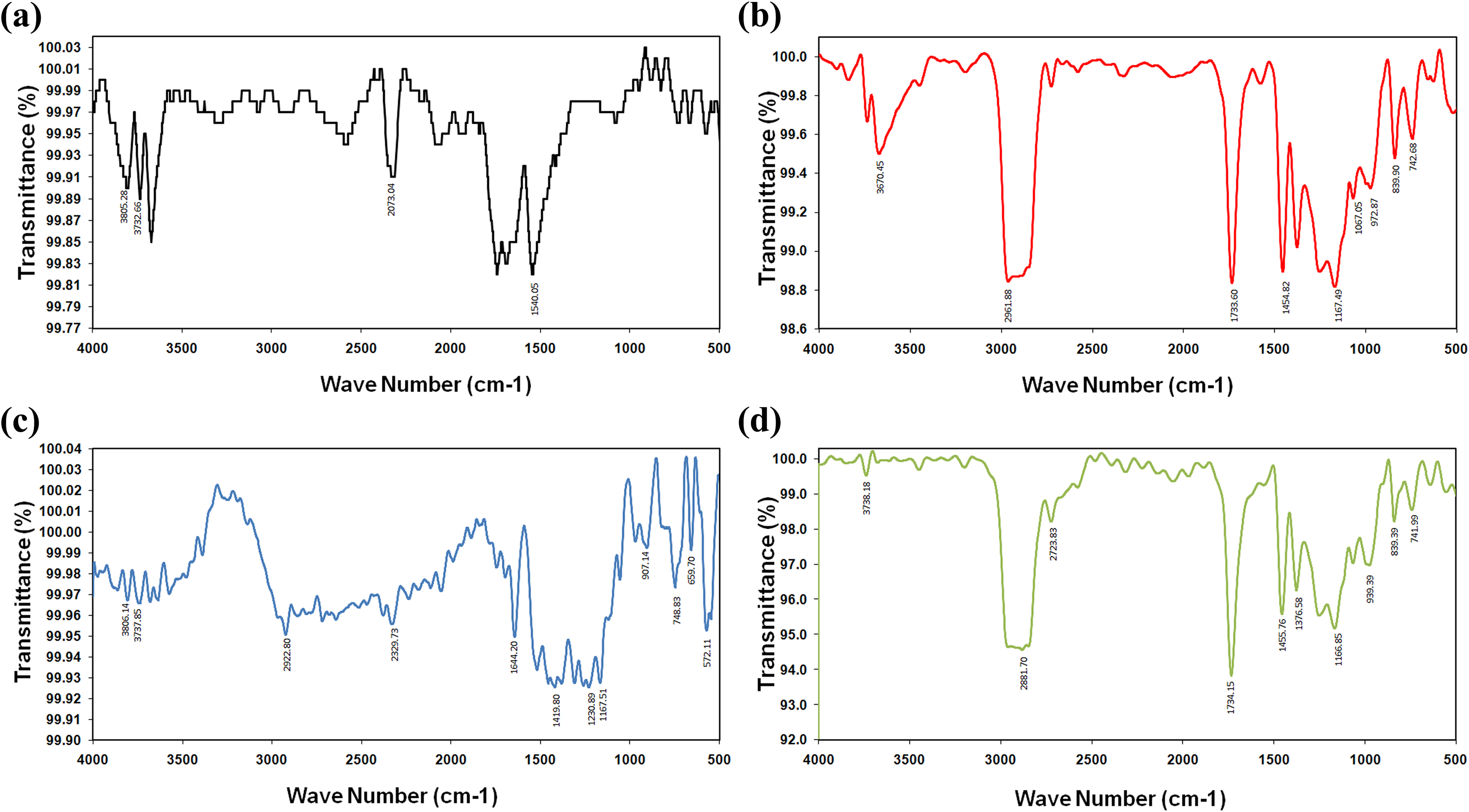
FTIR spectra of (a) Pebax®, (b) Pebax® with 1% LCNF, (c) Pebax® with 3% LCNF, and (d) Pebax® with 5% LCNF.
The spectrum of composites, containing 1% of LCNF, showed higher intension of absorbance at wave numbers from 1167 cm−1 to 1733 cm−1. These intense absorbencies were related to ether (C–O) stretching frequencies. Moreover, peaks at wave number of 2961 cm−1 related to C–H bonds were distinguished. 26 Weaker peaks from around 1400 cm−1 to 1500 cm−1 observed were probably related to C–C bonds at rings of glucose in LCNF.
In the samples containing 3% of LCNF, the most intense absorbance was observed at wave numbers of 1230 and 1419 cm−1 that were related to C–H and C–C bonds at glucose, respectively. 27 Also, the presence of ether bonds (C–O) was detected at lower peaks at 1251 cm−1. The samples containing 5% of LCNF showed peaks at 1455.67 cm−1 that was related to the saturated carbon bonds. 28 In these samples, peaks at wave number of 1734 cm−1 was detected that was related to carbonyl groups. A peak at wave number of 1166 cm−1 was distinguished which was related to C–O bonds. 18
The result of the samples containing 1% of LCNF showed the peaks of C=O bonds at 1733 cm−1 which proved the presence of polyamide groups at Pebax® polymer. Moreover, hydroxyl groups were detected at higher frequencies (3670 cm−1) that proved the presence of the benzyl alcohol and revealed that it was not completely removed from composite that contains 1% of LCNF.
Also, some peaks around 742 and 1067 cm−1 were detected, which showed the presence of C=H groups of benzyl alcohol. As previously described, removing solvent from composites at all the masterbatches was done by heating at the same constant temperature. The presence of benzyl alcohol at 1% LCNF samples revealed that compared to samples with higher content of LCNF, these samples needed more heating time to completely wipe out the solvent. The remaining solvent acted as an unfavorable plasticizer and changed
By adding the LCNFs to the polymer (e.g. 1% of LCNF), FTIR peaks were changed. New peaks detected at wave numbers from 1400 cm−1 to 1500 cm−1 were attributed to the C–C linkages of LCNF glucose units. Normally, the band 1454 cm−1 in IR spectra is assigned to C–O bonds, but wave numbers from 1450 cm−1 to 1650 cm−1 are also attributed to the aromatic rings of lignin compounds. 28 The peaks from 1000 cm−1 to 1380 cm−1 were assigned to the C–O ether bonds of LCNF and Pebax® polymer. The peaks from 665 cm−1 to 980 cm−1 were due to the results of the N–H stretching at Pebax® polymer. While no new chemical bonds were created, it seems that LCNFs and Pebax® were attracted to each other by hydrogen bonding and van der Waals forces. In fact, electronegative atoms like the oxygen, nitrogen, and fluorine were attracted by hydrogen atoms by means of hydrogen bonding.
Accordingly, it can be concluded that hydrogen bonds were attributed to the N–H and O–H attractions. The surface of cellulosic chains has enough hydrogen groups that can form inter and intramolecular hydrogen bonds. On the other hand, N–H bonds of polymer also help to create hydrogen bonding. It should be noted that, however, hydrogen bonds are weaker than covalent bonds, but among the intermolecular forces, hydrogen bonding is one of the strongest one.
Thermal properties
Thermal properties of samples are shown in Table 2. As can be seen, no regular trend of enthalpy changes was seen by increasing the amount of LCNF to polymer. The highest amount of enthalpy was related to the samples containing 5% of LCNF. Sliwa et al.
12
and Kumar et al.
29
reported that the crystallization enthalpy (
Thermal properties of Pebax® polymer reinforced by LCNF.
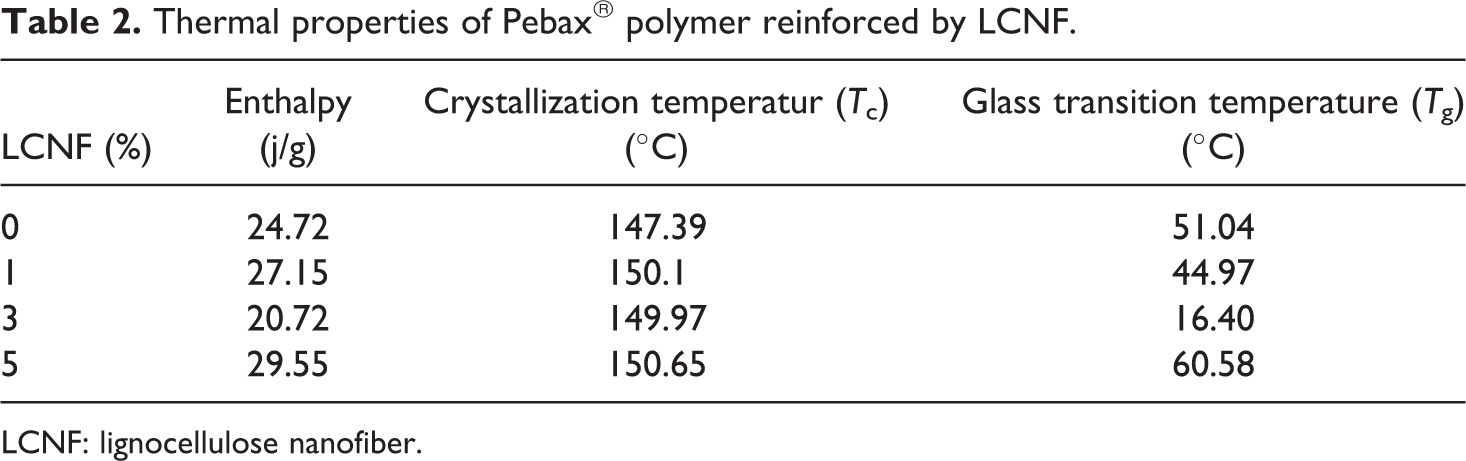
LCNF: lignocellulose nanofiber.
By the addition of LCNFs from 1% to 5%, the
By the addition of LCNF up to 3%,
In the case of miscible polymer with the same homogenous phase, only one
Water absorption and thickness swelling
Table 3 shows the water absorption and thickness swelling of polyether-b-amide composites reinforced with LCNF after 24 h soaking in water.
24 h water absorption and thickness swelling of LCNF/Pebax® nanocomposites.

LCNF: lignocellulose nanofiber.
The highest water absorption was achieved when the pure polymer without LCNF was tested. When the LCNF was increased to 3%, the water absorption decreased but higher loading of LCNF from 3% to 5% led to a slight increase in the water absorption.
The highest thickness swelling was related to the polymer samples containing 1% of LCNF (3.01%), and higher loading of LCNF led to a reduction in the thickness swelling so that the lowest thickness swelling was obtained at 5% of LCNF (2.22%).
Due to the hydrophilic nature of LCNF and the presence of functional groups at Pebax® matrix, it was expected that water absorption and thickness swelling of composites were increased by adding the LCNF to the polymer matrix. But the results showed that the addition of LCNFs to polymer led to decrease in water absorption and thickness swelling of samples as compared to neat polymers. The decrease in the water absorption is attributed to the large surface area of LCNFs which may form a good interface bond between the matrix and the fiber as compared to the control composites without LCNF. The improvement in the interface bond is also realized in the mechanical test. These results were in agreement with the results of researches that studied the effects of adding nanocellulose to starch-based composites 33,35 and Nayak’s and Ray’s research about the effect of nano-aluminum oxide filler concentration on moisture absorption kinetics of hydrothermally treated glass fiber which reinforced polymer composites 36
Furthermore, the presence of lignin in LCNF that has higher hydrophobicity could explain the lower Water Absorption (WA)% noticed for the polyether-b-amide composites which were reinforced by LCNF. 23
The results of FTIR and DSC showed that the LCNF has a good interaction with Pebax® matrix, so it seems that the substitution of the hydrophilic hydroxyl groups at LCNF by functional groups of polymer led to a reduction in the water absorption of samples. Also, the reduction of water absorption may be related to the substitution of the hydroxyl groups of LCNFs by inter and intramolecule bonds of LCNFs chains that reduced the distances between LCNFs chains.
The thickness swelling of samples containing 1% of LCNF was slightly increased. This increase could be due to the presence of the benzyl alcohol in nanocomposite. This assumption was proved by the FTIR results. The benzyl alcohol acts as a plasticizer and it should be removed completely at the evaporation stage but the imperfect separation of solvent can negatively affect the final properties of LCNF.
In fact, because of the presence of hydroxyl groups at benzyl alcohol, the interaction between LCNF and polymer was weakened and the reaction position of polymer and reinforcement was blocked by hydroxyl groups of benzyl alcohol. As a result, the physical and mechanical properties of nanocomposite were reduced. With the increment of hydroxyl groups of benzyl, the thickness swelling of final nanocomposite was increased. This could be probably due to the reduction of the interactions of LCNFs three-dimensional network due to the presence of benzyl alcohol which was not evaporated completely.
Mechanical properties
The mechanical properties of polyether-b-amide at different levels of LCNF are shown in Table 4. The tensile modulus of samples was significantly increased as the content of LCNF was increased from 1% to 5% compared to samples without LCNF. The highest modulus level (27.4 MPa) was obtained as the concentration of LCNF was increased to 5%. Also, as the concentration of LCNF increased from 1% to 3%, the average decreased from 24.3 MPa to 23.34 MPa, but this reduction was not significant according to the variance analysis.
Mechanical properties of samples at different levels of LCNF.
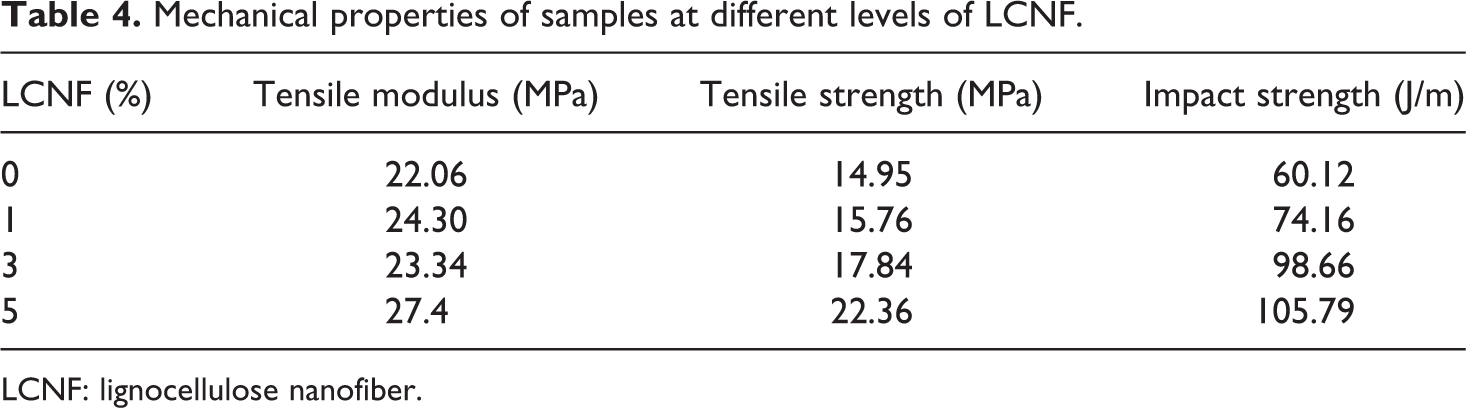
LCNF: lignocellulose nanofiber.
More addition of LCNF up to 5% had significant effects on the tensile strength so that the highest tensile strength was yielded at 5% loading of LCNF. The results showed the role of LCNF on improvement of polymer-LCNF matrix interfaces.
Accordingly, the Young’s modulus of samples containing 3% LCNF increased up to 27.4 MPa that was much higher than the control samples.
The results showed that the increased aspect ratio and the nucleation effects of LCNFs, besides the higher modulus of it and the strong interaction between the Pebax matrix and nanocomposite were highly beneficial in terms of mechanical properties improvement. 37 –39 Also, about the significant increase of mechanical properties at 5% loading LCNFs, it should be noted that the greatest improvement of properties will occur when the sufficient amount of reinforcement was added to the polymer matrix. 40 –42 At this condition, a continuous structure is formed and the mechanical properties will improve. 43 In fact, the LCNF is an unpurified form of CNFs containing lignin and insoluble hemicellulose along with cellulose fibers. The bionanocomposite containing 5% of LCNF was more rigid because these nanofibers increase crystallinity of biopolymer films and as a result, the tensile strength and Young’s modulus of nanocomposites were increased. LCNF displayed the most important role on the improvement of mechanical properties of bionanocomposites. This behavior was expected and was attributed to the resistance exerted by the LCNF itself and to the orientation of the LCNF chains. In addition, the stretching resistance of the oriented backbone of the polymer chain in the bonded points by hydrogen interaction with nanofillers also contributed to enhancing the tensile strength. The LCNF acts as a mechanical reinforcement of polyether-b-amide reducing the flexibility of the polymer. The main reason for this improvement in the mechanical properties is the stronger interfacial interaction between the matrix and chains of LCNF due to the vast surface exposure to nanofibers. 44 The observed improving effect on the mechanical properties of biopolymer films by the incorporation of organic nanofibers has already been demonstrated in other researches. 23,45 –47
The results of impact strength showed similar trends like the Young’s modulus, so that by increasing LCNF from 1% to 5%, the impact strength increased up to 105.8 J m−1 which were significantly more than the control samples.
All the mechanical properties of the composites are not related to the properties of reinforced material. These properties also depend on the internal adhesion properties of interfacial phase. Better adhesion at the polymer-LCNF matrix interface led to interfacial stress concentration reduction and as a result stress transfer and impact strength were increased.
The improvement of the interfacial region could be related to the reaction of functional groups of Pebax® and hydrophilic groups of LCNFs which led to the increase in the contact of LCNF particles and Pebax® polymer and as a result the agglomeration of LCNF reduced. This improvement led to an increase in the distribution of LCNF within the polymer prohibiting the creation of stress concentration points and finally crack propagation at polymer interface reduced.
Also, higher aspect ratio (fiber length to diameter) of LCNF could help to improve stress transfer at polymer-nano interface. The promotion of interface adhesion was examined by FTIR and DSC tests and the results proved this improvement.
Conclusion
The present study investigated the effect of LCNF reinforcement on some properties of nanocomposites made from polyether-b-amide polymer.
From the results, it was concluded that:
It is possible to make nanocomposites by mixing thermoplastic elastomer (Pebax®) and LCNFs.
FTIR spectra showed that by adding LCNFs to nanocomposites, some of the functional groups were reduced and new groups were created.
The thermal behavior of nanocomposites showed that by adding LCNFs to the polymer, a slight change of
Adding the LCNFs led to improvement in the water absorption and thickness swelling of the nanocomposite.
Mechanical properties were increased significantly by adding LCNFs, whereas tensile modulus, strength, and impact strength were improved.
The authors have not studied reinforced nanocomposites more than 5% of LCNFs, but based on the results from the point of physical and mechanical properties, in this research, most compounds of materials were obtained when 5% of LCNFs were added to polyether-b-amide polymer.
Footnotes
Authors’ note
This article was extracted from the research project of Sari branch, Islamic Azad University.
Acknowledgements
The authors would like to acknowledge Nano Novin Polymer Company (Iran) for the support provided for LCNF supply and preparation, the Iran Polymer and Petrochemical Institute (IPPI) for providing laboratory equipment used in the manufacture of the nanocomposites, and the Dean of Research, Sari Branch, IAU, for the financial support.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Dean of Research, Sari Branch, IAU.
