Abstract
Nipple discharge is a common and problematic symptom reported by many women seeking evaluation in primary care practices, surgery clinics, and breast radiology facilities. Discharge has been cited as the second to third most commonly reported breast symptom, next to palpable lumps and tenderness.1–3 Numerous groups have discussed management strategies regarding nipple discharge, and there has been a significant evolution away from surgical intervention. For the radiologist, nipple discharge can be a dilemma for many reasons. For academic medical centers with the availability of a network of clinical breast care personnel, subspecialty trained breast radiologists, and breast surgeons, a comprehensive evaluation and management strategy is more realistic. In the community setting, managing a patient with nipple discharge can be challenging due to limited time and resources. Fortunately, the vast majority of patients presenting with nipple discharge can be appropriately triaged with a thorough history and physical exam. For those requiring imaging, mammography and ultrasound are widely available for initial work-up. Magnetic Resonance Imaging (MRI) has also become more commonplace in recent years and has become an important tool in the evaluation process of nipple discharge. Ductography, which is decreasing in popularity, may continue to play a specific role in managing nipple discharge; however, the availability of skilled practitioners may be limited. Our aim in this review is to discuss features that differentiate physiologic from pathologic nipple discharge, provide a literature summary to guide imaging recommendations, and introduce a flow chart as an overview for step-by-step management.
Physiologic versus pathologic nipple discharge
When evaluating a patient with nipple discharge, one of the first steps in a management approach is obtaining a clinical history as well as performing a thorough focused physical exam.1–3 In many radiology practices, this is limited given the demands of daily workflow. A clinical office visit may not occur before imaging evaluation, and the patient’s history may not be routinely available upon presentation. One option for radiology practices could be to elicit specific information on a history sheet from each patient presenting with discharge to help guide management. An example of a patient survey is provided, which outlines several of the salient clinical questions pertinent to work-up (Figure 1). Nipple discharge questionnaire example form.
The definitions of physiologic and pathologic nipple discharge pertain to processes that happen within epithelial cells that line the ductal system. Fluctuations in hormone levels including prolactin, estrogen, and progesterone impact the production of fluid secretions. In addition to hormone stimulation, the natural process of cell death with continuous sloughing of the epithelial lining contributes to such secretions. These processes are responsible for physiologic discharge and account for the non-malignant etiologies. 4
Features of physiologic nipple discharge that are commonly discussed throughout literature include the following: bilaterality, nonbloody (yellow, green, milky, and gray) color, multi-duct location, and nonspontaneous expression. 5 The consensus for work-up of this type of discharge makes the imaging algorithm fairly simple for the radiologist. Once discharge is identified as physiologic, the likelihood of malignancy significantly diminishes. 6 In addition, this type of discharge frequently resolves over the span of 1–6 months. 6 Standard imaging evaluation typically involves ensuring that the patient has undergone recent mammographic evaluation and performing a target ultrasound if requested; however, the American College of Radiology (ACR) appropriateness criteria does not recommend any imaging for this type of discharge. 7 For physiologic discharge, the color may direct subsequent management. For instance, lab work may be indicated in the setting of milky (prolactin level) or purulent (CBC) discharge. Discharge that potentially meets these benign criteria may still cause unpleasant symptoms and warrant evaluation and treatment.
Pathologic discharge is discussed throughout literature as being serous or bloody, spontaneous, unilateral, and single duct.4,5 There have been numerous studies discussing the features of discharge and factors predictive of malignancy. Bloody discharge has been demonstrated as a significant predictor of malignancy. 8 In addition, the presence of single duct discharge is also known to have a significant association with malignancy. 9 While such factors may prompt more vigorous work-up, discharge that is persistent or copious without single duct or bloody characteristics may still raise clinical concern.
Mammography and ultrasound
According to the ACR appropriateness, the management of nipple discharge depends on numerous factors including patient age (age under 30 or over 40), biological gender, characteristic of discharge (pathologic or non-pathologic), and availability of advanced imaging modalities. 7 The first specification of the appropriateness criteria separates physiologic discharge from the management tree. No imaging is recommended in the setting in physiologic discharge. Because patients are often sent for evaluation without a thorough assessment of the nature of discharge and since these patients commonly have not undergone recent mammography, they may need work-up in a diagnostic clinic to discern the type of exam needed. If possible in a breast imaging center, the ideal scenario would involve a discussion with these symptomatic patients, triaging those in need of imaging. Furthermore, physical exam may confirm discharge characteristics or redirect management strategy. This ideal setting is less common than the typical scenario which involves a patient presenting with no provided information regarding discharge characteristics and no reported physical exam findings. For this reason, mammography is often performed in females over the age of 40 who have not undergone mammographic imaging for the past year. With the supportive information of physiologic discharge, this should be a screening mammogram. 7
In the setting of pathologic discharge, the recommendation is to begin with diagnostic mammography and/or ultrasound in patients over the age of 30. Under the age of 30, the recommendation supports ultrasound as the initial imaging modality. 7 Although mammography is not the most sensitive modality in the assessment of nipple discharge, normal mammography and ultrasound imaging combined with an otherwise normal physical exam (no palpable abnormality) has been shown to be predictive in confirming benignity with only one out of 287 malignancies found by Sabel et al. 10 In this study, the one malignancy was found in a patient who had a history of breast cancer. Excluding this history resulted in no malignancies diagnosed in patients with normal diagnostic mammosonography. 10 Gray et al. had a similar experience in 204 patients, finding that abnormal mammograms and ultrasounds were predictive of malignancy. In addition, suspicious discharge (in this study, spontaneous, bloody, unilateral, or serous) in the setting of a negative mammogram resulted in an overall risk of malignancy of 3%. When combined with a subareolar ultrasound, the risk was 0%. 11
Ultrasound has also been widely used in the assessment of nipple discharge with studies showing increased cancer detection when added to mammography. 12 During routine investigation of discharge, ultrasound plus mammography significantly increases sensitivity. 13 Elastography has been investigated as a tool to better characterize intraductal masses. In a study by Zhu et al., supplemental elastography increased both specificity and accuracy in comparison to ultrasound alone. 14 Numerous studies have also documented the benefit of ultrasound in conjunction with galactography15,16 as well as combining ultrasound with galactography and MRI. 17
Imaging with mammography in cases of nipple discharge, whether due to benign or malignant causes, is often normal. Several mammographic findings have been described including the following: mass, duct ectasia, focal asymmetry, solitary dilated duct, and microcalcifications. Pathologic causes of nipple discharge manifest on ultrasound as masses (sometimes intraductal, typical of papillomas or DCIS), duct ectasia, or cysts. Calcifications can also be seen on ultrasound, either within a mass, outside of a mass, or inside the ductal system. A fluid collection can also be seen in cases of nipple discharge. For example, cases of nipple discharge with infectious causes may detect an abscess and cases of postoperative discharge may find seromas or hematomas. Characteristics of malignant ultrasound-detected masses have been reported more commonly as hypoechoic with irregular margins. Benign characteristics tend to favor hypoechoic masses with circumscribe margins. Heterogeneity and complex cystic solid features can be seen in both benign and malignant cases but tend to be more common in the malignant category.
16
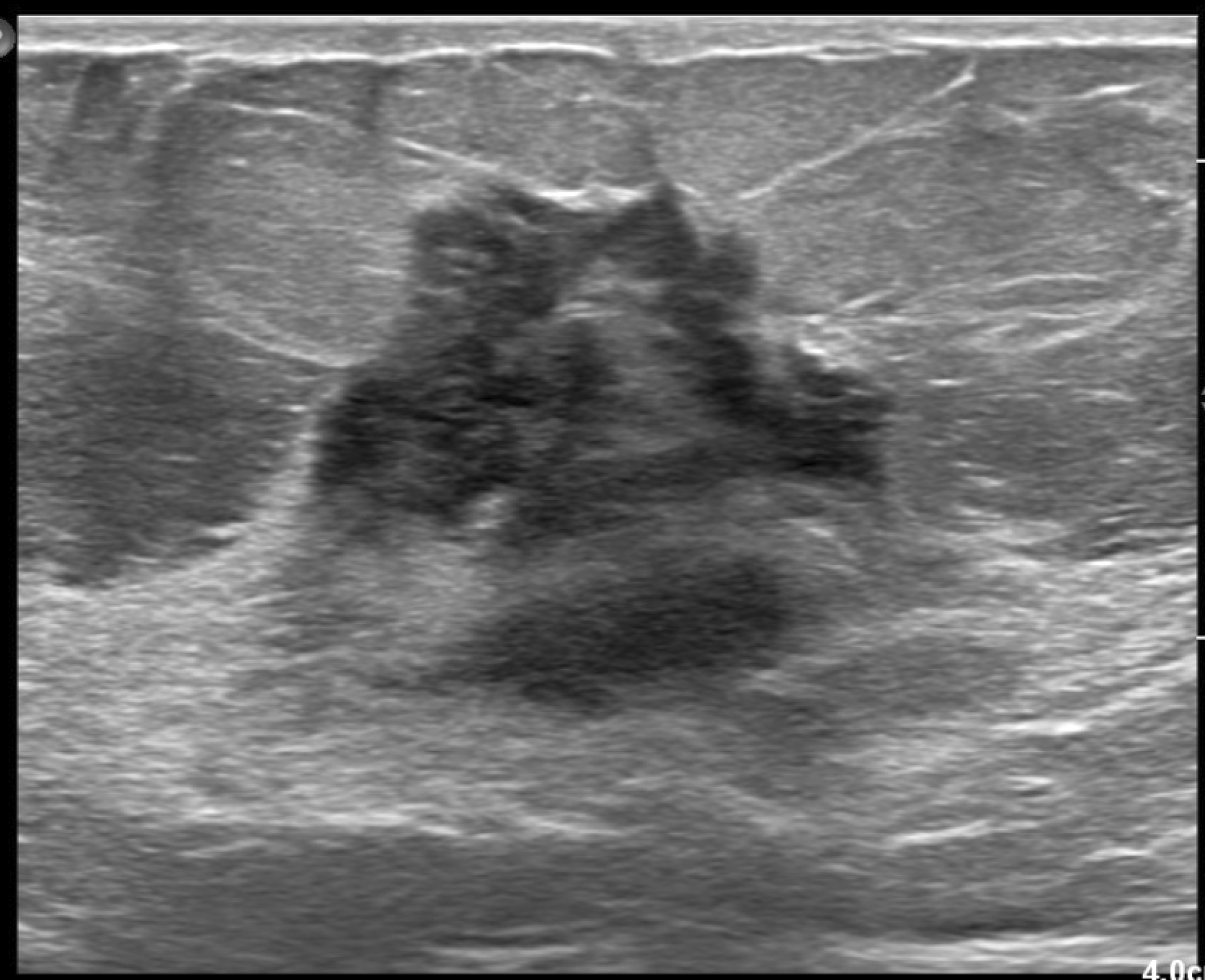
Figures 2–14 demonstrate mammographic and sonographic findings from initial work-up of three different cases of pathologic discharge, one due to a papilloma (Figures 2–5), one due to high-grade DCIS (Figures 6–8), and one case of low-grade DCIS (Figures 9–14). Each of these cases also underwent MRI evaluation (Figures 4, 7, and 14). Left CC view full-field mammogram demonstrates a solitary dilated duct (oval) in the central breast at anterior depth. Patient is a 66-year-old female with a history of right breast ER-, PR- DCIS diagnosed 6 years prior. She presented with new clear single duct left nipple discharge from a central/12 o’clock orifice. Targeted B-mode ultrasound of the subareolar region of the left breast in the transverse plane demonstrates an intraductal mass (oval) corresponding to the mammographic finding. Note that the intraductal mass is primarily in the more proximal (left-hand side of image) portion of the duct, but nondependent solid tissue could be seen in the more distal (right-hand side of image) portion of the duct. Patient is a 66-year-old female with a history of right breast ER-, PR- DCIS diagnosed 6 years prior. She presented with new clear single duct left nipple discharge from a central/12 o’clock orifice. Bilateral T1-weighted contrast-enhanced subtraction breast MRI in the axial plane and first post-contrast phase demonstrates linear non-mass enhancement in the left central breast at anterior depth (oval). Ultimately, biopsy performed under MRI guidance demonstrating a papilloma. Patient is a 66-year-old female with a history of right breast ER-, PR- DCIS diagnosed 6 years prior. She presented with new clear single duct left nipple discharge from a central/12 o’clock orifice. Full-field ML mammogram from Savi scout localization procedure demonstrates the Savi device (arrow) just posterior to the biopsy clip (circle), demarcating the lesion location. No upgrade was noted at the time of excision. Patient is a 66-year-old female with a history of right breast ER-, PR- DCIS diagnosed 6 years prior. She presented with new clear single duct left nipple discharge from a central/12 o’clock orifice. Right breast spot CC tomosynthesis demonstrates architectural distortion in the central breast anterior depth (circle). Patient is a 66-year-old female who presented with right single duct bloody nipple discharge. Right breast targeted B-mode grayscale ultrasound images in the transverse plane demonstrate an irregular mass at 12 o’clock in the subareolar location. Patient is a 66-year-old female who presented with right single duct bloody nipple discharge. Bilateral T1-weighted contrast-enhanced subtraction breast MRI in the axial plane, first post-contrast phase, demonstrates an irregular mass with spiculated margins, spanning 11 o’clock and 12 o’clock at anterior and middle depth. Pathology of biopsy demonstrated estrogen receptor positive and progesterone receptor positive (ER + PR+) high-grade ductal carcinoma in situ (DCIS). On mastectomy, no upgrade was noted. Patient is a 66-year-old female who presented with right single duct bloody nipple discharge. Right full-field MLO (Figure 9) and left full-field MLO (Figure 10) mammography demonstrate a right breast MLO view asymmetry in the superior breast at middle depth. Right MLO (Figure 9) compared to left (Figure 10) demonstrates an overall increase in tissue density in the superior breast middle depth. Finding is only seen in MLO view (CC not shown), making it an asymmetry. Patient is a 49-year-old female who presents with persistent two duct discharge in the upper outer nipple duct system, involving ducts at approximately 10 and 11 o’clock. She underwent mammographic imaging followed by ultrasound evaluation. Right full-field MLO (Figure 9) and left full-field MLO (Figure 10) mammography demonstrate a right breast MLO view asymmetry in the superior breast at middle depth. Right MLO (Figure 9) compared to left (Figure 10) demonstrates an overall increase in tissue density in the superior breast middle depth. Finding is only seen in MLO view (CC not shown), making it an asymmetry. Patient is a 49-year-old female who presents with persistent two duct discharge in the upper outer nipple duct system, involving ducts at approximately 10 and 11 o’clock. She underwent mammographic imaging followed by ultrasound evaluation. B-mode grayscale targeted ultrasound in the right breast at 12 o’clock 2 centimeters from the nipple in the transverse (Figure 11) and sagittal (Figure 12) planes demonstrates an irregular hypoechoic not parallel mass with indistinct margins (oval). There is posterior shadowing. Associated abnormal duct changes are noted in a segmental distribution (arrows). Power Doppler imaging in the sagittal plane demonstrates internal vascularity involving the mass (Figure 13). Patient is a 49-year-old female who presents with persistent two duct discharge in the upper outer nipple duct system, involving ducts at approximately 10 and 11 o’clock. She underwent mammographic imaging followed by ultrasound evaluation. B-mode grayscale targeted ultrasound in the right breast at 12 o’clock 2 centimeters from the nipple in the transverse (Figure 11) and sagittal (Figure 12) planes demonstrates an irregular hypoechoic not parallel mass with indistinct margins (oval). There is posterior shadowing. Associated abnormal duct changes are noted in a segmental distribution (arrows). Power Doppler imaging in the sagittal plane demonstrates internal vascularity involving the mass (Figure 13). Patient is a 49-year-old female who presents with persistent two duct discharge in the upper outer nipple duct system, involving ducts at approximately 10 and 11 o’clock. She underwent mammographic imaging followed by ultrasound evaluation. B-mode grayscale targeted ultrasound in the right breast at 12 o’clock 2 centimeters from the nipple in the transverse (Figure 11) and sagittal (Figure 12) planes demonstrates an irregular hypoechoic not parallel mass with indistinct margins (oval). There is posterior shadowing. Associated abnormal duct changes are noted in a segmental distribution (arrows). Power Doppler imaging in the sagittal plane demonstrates internal vascularity involving the mass (Figure 13). Patient is a 49-year-old female who presents with persistent two duct discharge in the upper outer nipple duct system, involving ducts at approximately 10 and 11 o’clock. She underwent mammographic imaging followed by ultrasound evaluation. Sagittal T1-weighted contrast-enhanced subtraction MRI of the right breast demonstrates clumped segmental non-mass enhancement in the right breast at 12 o’clock middle depth. Pathology demonstrated ER + PR + DCIS with micropapillary and cribiform patterns. Patient is a 49-year-old female who presents with persistent two duct discharge in the upper outer nipple duct system, involving ducts at approximately 10 and 11 o’clock. She underwent mammographic imaging followed by ultrasound evaluation.











MRI
Although for over 20 years the role of MRI has been studied in the assessment in nipple discharge, 18 many radiologists still debate its utility. The added value of MRI to the work-up of nipple discharge is supported by multiple studies, including a 2011 study by Lorenzon et al. who showed better sensitivity and specificity compared to mammography and ultrasound. 19 A 2015 study by Bahl et al. showed that in patients with benign sonomammography, negative/benign/probably benign MRI assessments had 100% sensitivity and 100% negative predictive value (NPV). 20 In 2017, a retrospective review by Bahl et al. confirmed these findings (those with MRI of BI-RADS 1, 2, or 3) with demonstrating a less than 4% risk of malignancy in women with negative or inconclusive mammograms. 21 Zacharioudakis et al. discussed the use of MRI in the management of nipple discharge in a prospective study involving 82 patients over a 9-year period of time who underwent mammography and ultrasound exams with the detection of malignancy in 14 patients who had normal mammographic and sonographic evaluation. MRI was performed on all patients as a part of standard protocol and demonstrated sensitivity, specificity, positive predictive value (PPV), and negative predictive value of 85.71%, 98.53%, 92.31%, and 97.1%, respectively. 22 In 2019, a similar study by Zaky et al. reported these measures as 100%, 83.3%, 63.6%, and 100%, respectively. 23
The ACR appropriateness criteria address the use of MRI in the evaluation of nipple discharge; however, for the variants listed in the appropriateness criteria, MRI is listed as a “usually not appropriate” radiologic procedure. This rating is given because each variant addresses only the initial imaging examination for the given scenario. The literature summary in the ACR appropriateness criteria goes on to discuss MRI as a valuable tool. 7
MRI has been compared to numerous other imaging modalities. In 2003, Nakahara et al. reported that compared to galactography and ultrasound, MRI better demonstrated the imaging features of ductal carcinoma in situ (DCIS) in patients presenting with bloody nipple discharge. 16 In 2015, Manganaro et al. concluded that MRI had higher sensitivity and specificity compared to galactography in those with clear or bloody discharge. 24 Berger et al. performed a review of MRI compared to galactography, published in AJR in 2017. They reviewed 10 studies involving 921 patients. Their findings support the use of MRI over galactography in the setting of negative mammosonography. 25 In 2014, Lubina et al. reported a prospective study of patients with nipple discharge performed using a 3 Tesla (3T) MRI and compared findings to galactography. Based on their results, the recommendation was replacing galactography with 3T MRI due to the improved correlation with lesion size. 26
On MRI, various features have been described to correlate with suspicious nipple discharge. Non-mass enhancement (previously described as non-mass like enhancement) was most common with a segmental distribution, heterogeneous internal enhancement, and plateau kinetics. Additional malignant appearance included clustered ring enhancement.
27
Benign features responsible for pathologic discharge may include nonenhancing proteinaceous debris within the ducts, duct ectasia, cysts, and fluid collections (similar to ultrasound). Other common MRI findings that have been reported in patients presenting with nipple discharge include masses which vary in appearance from those with irregular margins to those with circumscribed margins. Diffuse enhancement has also been noted, which is by far a benign characteristic.
16
Figures 4, 7, and 14 are selected images from MRIs performed in patients experiencing nipple discharge. Characteristics include linear non-mass enhancement (Figure 4) corresponding to a papilloma, an irregular mass (Figure 7) corresponding to high-grade DCIS, and clumped segmental non-mass enhancement (Figure 14) corresponding to low-grade DCIS. In Figures 15–23, the use of MRI is illustrated in both benign and malignant scenarios. In Figure 17, MRI was utilized for management purposes in a 23-year-old female patient who underwent biopsy of a papilloma (producing nonspontaneous single duct unilateral bloody nipple discharge) with vacuum-assistance. Figure 15 (ultrasound) and 16 (ductogram) provided depiction of the lesion of interest, which was the intraductal mass (papilloma). MRI (Figure 17) changed management when numerous bilateral similar-appearing enhancing masses were seen, any of which could represent additional papillomas. Discharge had resolved after biopsy and no enhancement was seen at the biopsy site. Therefore, surveillance with MRI was chosen instead of surgical excision. Power Doppler imaging in the sagittal plane demonstrates an intraductal mass in the right breast 1 o’clock subareolar location showing internal vascularity. Patient is a 23-year-old female with a history of unilateral one-duct spontaneous discharge initially nonspontaneous. She presented for evaluation which was first performed with ultrasound. Right CC ductogram demonstrates an intraductal filling defect (arrow) at 1 o’clock anterior depth. Biopsy demonstrated intraductal papilloma without atypia. Patient is a 23-year-old female with a history of unilateral one-duct spontaneous discharge initially nonspontaneous. She presented for evaluation which was first performed with ultrasound. Bilateral T1-weighted contrast-enhanced subtraction breast MRI in the axial plane, first post-contrast phase, demonstrates multiple bilateral oval enhancing masses (circles). No discrete enhancement could be seen at the site of right 1 o’clock biopsy (not shown). Ultimately decision was for surveillance due to the number and similarity of lesions. A follow-up MRI in 6 months demonstrated stability. Discharge did not recur after biopsy. Patient is a 23-year-old female with a history of unilateral one-duct spontaneous discharge initially nonspontaneous. She presented for evaluation which was first performed with ultrasound. Right ML ductogram demonstrates poor filling and opacification of the central duct system with duct cut-off (arrow). Patient is a 66-year-old female with a history of right breast single duct brown nipple discharge. Sagittal T1-weighted contrast-enhanced subtraction MRI of the right breast demonstrates linear clumped non-mass enhancement in the right central breast at middle depth (oval). This abnormal enhancement initiates at the site of duct cut-off seen on galactogram (a), and biopsy was performed under MRI guidance. Pathology yielded papillary DCIS. Patient is a 66-year-old female with a history of right breast single duct brown nipple discharge. Current (Figure 20) versus 3-year prior (Figure 21) right MLO view full-field mammogram demonstrates a developing asymmetry (oval) at 12 o’clock middle depth. There is also increased trabecular thickening and overall shrinking of the breast. Patient is a 50-year-old female who presented with new unilateral right spontaneous green nipple discharge. She had a history of right breast invasive ductal carcinoma (IDC) and DCIS, triple positive, treated with lumpectomy and radiation 13 years ago. Current (Figure 20) versus 3-year prior (Figure 21) right MLO view full-field mammogram demonstrates a developing asymmetry (oval) at 12 o’clock middle depth. There is also increased trabecular thickening and overall shrinking of the breast. Patient is a 50-year-old female who presented with new unilateral right spontaneous green nipple discharge. She had a history of right breast invasive ductal carcinoma (IDC) and DCIS, triple positive, treated with lumpectomy and radiation 13 years ago. Targeted B-mode ultrasound of the 12 o’clock subareolar region of the right breast in the transverse plane demonstrates a fluid collection with indistinct margins (arrows) corresponding to the mammographic finding. Patient is a 50-year-old female who presented with new unilateral right spontaneous green nipple discharge. She had a history of right breast invasive ductal carcinoma (IDC) and DCIS, triple positive, treated with lumpectomy and radiation 13 years ago. Sagittal T1-weighted contrast-enhanced fat-subtraction MRI of the right breast demonstrates the lumpectomy bed with fluid collection and irregular enhancing thick rim. Biopsy of her lumpectomy bed ultimately showed recurrent IDC. Patient is a 50-year-old female who presented with new unilateral right spontaneous green nipple discharge. She had a history of right breast invasive ductal carcinoma (IDC) and DCIS, triple positive, treated with lumpectomy and radiation 13 years ago.








Ductography
Ductography, or galactography, involves injection of a contrast agent into the discharging duct followed by mammography. The utility of ductography has been questioned in recent years with several publications documenting the improved performance of other imaging modalities. Ductography has been utilized for decades in the assessment of nipple discharge. While it may not be needed for many cases, galactography provides anatomic information that can guide surgical management.
A clear benefit of galactography is the concept of isolating a specific ductal system. This has been supported throughout literature, for example, in 1983 by Tabar et al., demonstrating the less invasive surgical measures needed with use of ductography. 28 In assessing the use of galactography in unilateral discharge, Florio et al. in 1999 noted an improved detection of malignancy and high-risk lesions, 29 and in 2001 Hou et al. showed similar benefits. 30 As more studies investigated the utility of galactography, metrics were found to be less impressive, showing a sensitivity and specificity of 31.2% and 97.4%, respectively, according to Dinkel et al. 31 Additional investigation in Breast in 2003 described certain features on galactography that had varying predictors, with the following sensitivity/specificities: filling defect 55.6% and 62.1%, duct ectasia 22.2% and 94%, filling stop (termination of the duct) 5.6% and 77.6%. In contrast, a normal ductogram was 93% specific for absence of disease but only 78% sensitive. 32 A similar description of such imaging features of galactography was supported by Kim et al. in 2008. 33 More recently in 2018, Istomin et al. sought to revisit the role of galactography in the evaluation of pathologic nipple discharge in 146 patients. These patients underwent standard imaging as well as breast MRI, and the calculated sensitivity and specificity of those tests included 77.4%, 75.7% (galactogram) versus 85.7% and 71.4% (MRI). 34
Galactography can isolate the site of interest specifically; however, internal features of the causative lesion itself are not assessed. Galactography does assess the changes of the ductal system that occur due to mass effect. Reported galactogram findings include most commonly a solitary filling defect. Other reported findings include irregular appearance of the duct wall, multiple filling defects, and a duct cut-off. Normal ductography as well as a limited or incomplete galactogram is also reported.
16
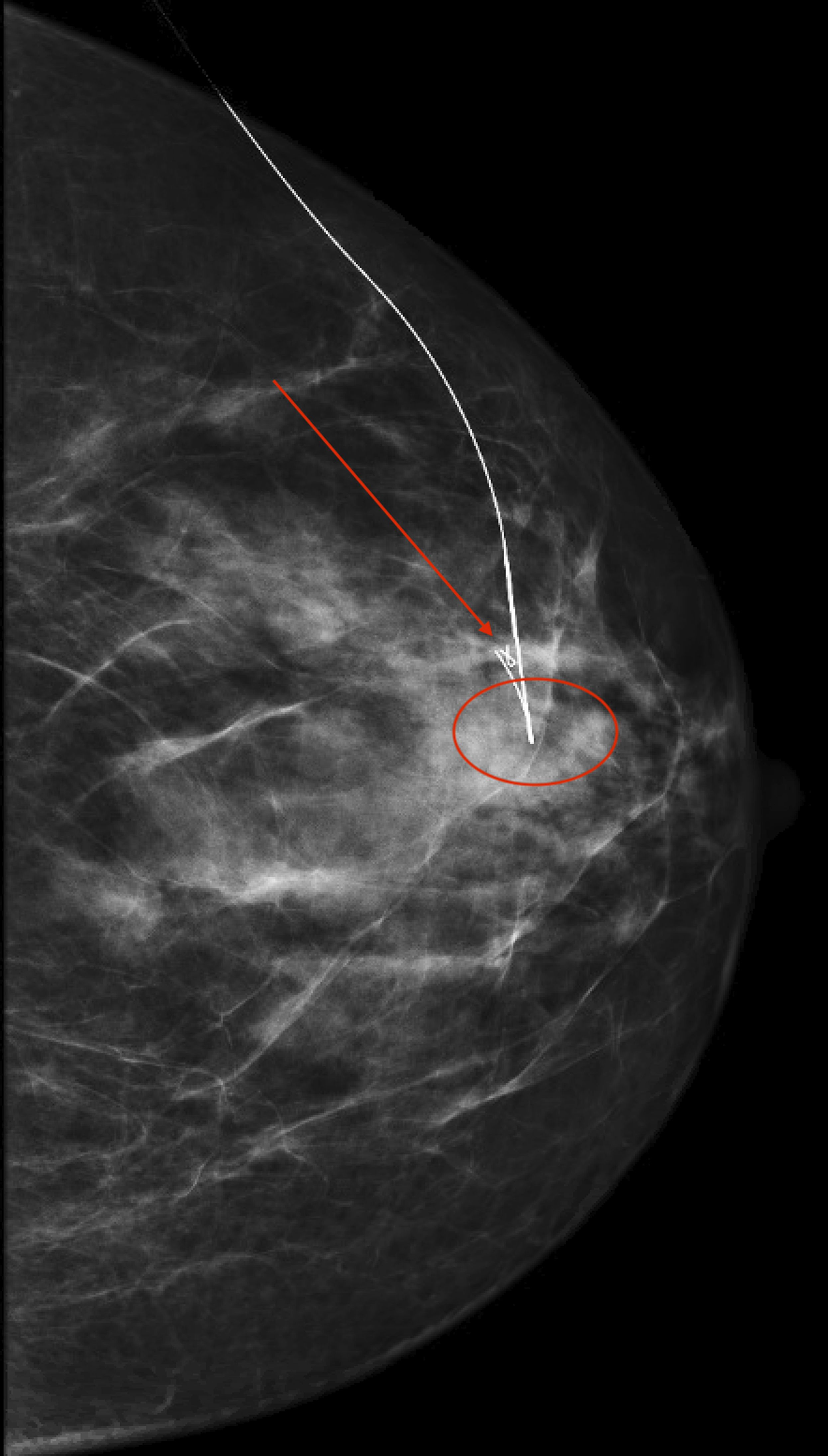
Figures 16, 18, 24, and 25 demonstrate ductography findings in patients with pathologic nipple discharge. The pathologies include papilloma, papillary DCIS, atypical ductal hyperplasia (ADH) with other high-risk lesions (radial scar, complex sclerosing lesion, and papilloma), and papilloma with atypia, respectively. Figures 24–27 demonstrate a work-up of unilateral single duct bloody nipple discharge, for which mammography and initial ultrasound were negative. After ductogram (Figure 24), ultrasound (Figure 27) was able to appropriately identify a small intraductal mass corresponding to a filling defect. Ductogram also aided in localizing the abnormal ductal system for surgery. Figures 27–29 provide a similar management technique. Neither case underwent MRI evaluation, which may have impacted management. Left CC full-field mammogram obtained after the administration of intraductal contrast. An intraductal filling defect and be seen in the central breast (arrow) at anterior depth along with an overall irregularity of the ductal system (oval). Intraductal administration of a water-soluble iodinated contrast (Iohexol, Omnipaque 350) is required for obtaining galactography imaging. A lidocaine/prilocaine (EMLA) cream is applied to the nipple followed by a warm compress prior to procedure. The nipple is cleansed and the duct is cannulated with a 30-gauge sialogram catheter. Approximately 1 mL of contrast is injected and mammography images are obtained, typically first with fulfilled imaging followed by magnification views. Patient is a 48-year-old female presenting with history of bloody left nipple discharge. Initial mammographic and ultrasound imaging were non-contributory. Left full-field CC ductogram demonstrates irregularity of the 12 o’clock ductal system with termination of the ductal system. Patient is a 57-year old female who presented with unilateral spontaneous clear nipple discharge. Yellow-tinged discharge elicited from one duct upon physical exam. Target ultrasound of the left retroareolar region at the level of the nipple demonstrates an intraductal mass (arrow). Images obtained with color flow which does not demonstrate discrete suspicious findings. Intraductal administration of a water-soluble iodinated contrast (Iohexol, Omnipaque 350) is required for obtaining galactography imaging. A lidocaine/prilocaine (EMLA) cream is applied to the nipple followed by a warm compress prior to procedure. The nipple is cleansed and the duct is cannulated with a 30-gauge sialogram catheter. Approximately 1 mL of contrast is injected and mammography images are obtained, typically first with fulfilled imaging followed by magnification views. Patient is a 48-year-old female presenting with history of bloody left nipple discharge. Initial mammographic and ultrasound imaging were non-contributory. Ductogram localization in the CC projection demonstrating filling defect (arrow) again in the central breast anterior depth. The posterior extent of the irregular ductal system (oval) is marked by the thick portion and hook of the wire. Pathology after duct excision demonstrated atypical ductal hyperplasia (ADH), radial scar, complex sclerosing lesion, and papilloma. Intraductal administration of a water-soluble iodinated contrast (Iohexol, Omnipaque 350) is required for obtaining galactography imaging. A lidocaine/prilocaine (EMLA) cream is applied to the nipple followed by a warm compress prior to procedure. The nipple is cleansed and the duct is cannulated with a 30-gauge sialogram catheter. Approximately 1 mL of contrast is injected and mammography images are obtained, typically first with fulfilled imaging followed by magnification views. Patient is a 48-year-old female presenting with history of bloody left nipple discharge. Initial mammographic and ultrasound imaging were non-contributory. Targeted B-mode ultrasound of the 12 o’clock subareolar region of the left breast in the sagittal plane demonstrates an intraductal mass (oval) corresponding to the ductogram finding. Biopsy under ultrasound guidance ultimately demonstrated a papilloma with atypia. Patient is a 57-year old female who presented with unilateral spontaneous clear nipple discharge. Yellow-tinged discharge elicited from one duct upon physical exam. Full-field CC mammogram from needle and wire localization procedure demonstrates the ribbon clip (arrow) at site of biopsy and hook of wire (circle) at the site of irregular ducts. No upgrade was found with excision. Patient is a 57-year old female who presented with unilateral spontaneous clear nipple discharge. Yellow-tinged discharge elicited from one duct upon physical exam.




MR ductography
MR ductography, which uses a heavily T2-weighted sequence to better identify intraductal lesions, has been described by several authors. It can be performed in conjunction with the administration of IV contrast and, subsequently, the T2-weighted and contrast-enhanced images are fused. 35 In 2010, this technique was compared to conventional galactography and was proposed as a comparable alternative by Wenkel et al. 36 In 2015, Nicholson et al. similarly used contrast-enhanced MRI, conventional galactography, and MR galactography to evaluate a small group of patients (n = 20) in a feasibility study. They reported sensitivity, specificity, PPV, and NPV as 65, 33.3, 76.5, and 22.2 for conventional galactography, versus 95, 66.7, 90.5, and 80.8 for contrast-enhanced MRI, and 55, 66.7, 84.6, and 30.8 for MR galactography. 37 As recently as 2020, the use of MR ductography was retrospectively evaluated by a group in Thailand, showing a sensitivity 100%, NPV of 100%, but specificity of 38%. 38 Currently, the use of MR ductography for the evaluation of nipple discharge is not endorsed by the ACR appropriateness criteria. 7
Contrast-enhanced ultrasound (CEUS) galactography
Contrast-enhanced ultrasound (CEUS) galactography has been explored as a diagnostic method in patients with pathologic nipple discharge. For CEUS galactography, ductography is performed with injection of a contrast agent such as sulfur hexafluoride microbubbles. After injection, an ultrasound examination is performed to see the ductal system. Wang et al. investigated the application of this technique and found that CEUS galactography improved diagnostic accuracy compared to traditional ultrasound. It also provided more detailed anatomical information. 39
Jiang et al. demonstrated that CEUS galactography significantly improves the visualization of the ductal system, allowing for more precise identification of underlying pathologies before surgery. This technique not only enhances diagnostic accuracy but reduces the need for certain invasive procedures. Just as with the Wang study, these findings supported CEUS galactography as a pre-surgical tool. 40
Contrast-enhanced digital breast tomosynthesis (DBT) galactography
In addition to CEUS, digital breast tomosynthesis (DBT) with galactography has been investigated as another possibility for evaluating nipple discharge. Tao et al. explored the use of DBT with breast galactography to show a more detailed depiction of the ductal system compared to digital mammography. DBT galactography can improve the identification of intraductal masses and aid in sensitivity. 41
Contrast-enhanced spectral mammography (CESM)
Contrast-enhanced spectral mammography (CESM) has not been widely studied in patients presenting with nipple discharge. Certain causes of nipple discharge, such as papillomas and DCIS, are known to be visualized with CESM. The performance of CESM compared to MRI was evaluated by Hegazy et al. in a 2020 retrospective review of 37 biopsy-proven papillomas. This study reported that CESM was significantly lacking in specificity for papillomas of all sizes and sensitivity for lesions less than 5 mm in size. 42 At this time, CESM is not recommended in the routine evaluation of nipple discharge. 7
CESM has not been utilized for large volume studies in the evaluation of nipple discharge at this time, but findings on CESM have been reported in cases involving patients with nipple discharge, most recently by Hegazy et al. as previously referenced. 42 In studying papillomas where 84% presented with nipple discharge, the contrast mammography findings were most commonly described as non-mass enhancement, followed by no enhancement, and least commonly an enhancing mass. 42 A more recent study demonstrated that CESM provides higher sensitivity in distinguishing benign from malignant causes of nipple discharge, which may lead to more accurate and earlier diagnoses and improve patient outcomes. 43
Other imaging modalities
Specific use of MBI or PEM to evaluate nipple discharge may be available in case reports or pictorial reviews; however, no large studies have been designed to evaluate imaging characteristics of nipple discharge with these modalities. Molecular breast imaging (MBI) and positron emission mammography (PEM) currently are not recommended in the evaluation of nipple discharge per the ACR appropriateness criteria. 7 Both modalities are capable of identifying metabolically active processes within the breast; however, evidence is lacking for their routine use in the evaluation or management of discharge.
Surgery
For many years, central duct excision (CDE or subareolar excision, SAE) was the mainstay of treatment for pathologic nipple discharge. The various imaging modalities previously described, primarily mammography, ultrasound, galactography, and MRI, have been used to localize and guide surgical intervention. Prior to the wide use of MRI, the lack of imaging to provide a consistently high negative predictive value prompted routine CDE due to the rate of malignancy in patients presenting with nipple discharge, up to 12.7% in postmenopausal females according to Lau et al. 44 A review from a 20-year follow-up in patients presenting with pathologic nipple discharge reported outcomes from CDE, citing no missed malignancies. 45 In 2010, Alcock and Layer also retrospectively evaluated patients with pathologic nipple discharge with a final recommendation of either major or minor duct excision as the recommended diagnostic and therapeutic intervention. 46 Morrogh et al. echoed this recommendation after review of 287 cases. 47 Sabel et al. contradicted these recommendations more recently in 2011, only finding one malignancy in follow-up of a group of 142 patients with pathologic nipple discharge after complete imaging work-up and surgical intervention appropriately diagnosed and treated malignancy in seven. 10 One suggested alternative to surgery is the use of interventional ductoscopy in the treatment of nipple discharge; however, this is not as widely reported. Filipe et al. described this procedure involving 215 patients (60 eventually undergoing surgery), and reported no major complications. 48 Recently, CDE has become less favored due to the improved diagnostic capabilities of imaging combined with appropriate triage of symptomatic patients.
Risk stratification
Risk stratification of patients has been discussed as a means of potentially averting surgery. A study by Gray et al. in 2007 found that age (≥ 50 years old) and abnormal imaging (mammography and ultrasound) were factors predictive of malignancy. 11 Cytologic examination has also been evaluated and found to have a PPV of 85% when breast imaging was abnormal. 49 Regarding the characteristics of discharge, unilaterality, bloody, single duct discharge all have strong associations with malignancy according to Chung et al. Unfortunately, they found that the lack of such characteristics alone was not sufficient to exclude malignancy. It is important to note that the advanced imaging modalities discussed above were not utilized for work-up in this study. 50
Throughout literature, risk stratification has been discussed when concerning management strategies. In 2015, Dupont et al. found that a prior history of ipsilateral breast cancer, BRCA mutation, or atypia on core needle biopsy carried an association with malignancy. Without these findings, the cancer risk was less than 2%. 51 Li et al. reported that the presence of symptoms (other than nipple discharge) have also been associated with higher risk for breast cancer, and also found that palpable masses were independently associated with malignancy in patients presenting with pathologic nipple discharge. 52
Management algorithm
Considering the data regarding the imaging options discussed, Figure 30, which is geared toward flow chart utilization, provides information for radiologists caring for patients presenting with nipple discharge. Using the questionnaire provided in Figure 1, the characterization of discharge can be categorized into either pathologic or physiologic. It is important to note that if any one of the characteristics of discharge are present (unilateral, bloody, brown, clear, single duct, or spontaneous), the pathologic algorithm should be considered. Mammography and ultrasound are recommended in accordance with ACR appropriateness criteria, and if no lesion is identified, MRI is recommended if available. If a lesion is identified, biopsy should follow, and malignant results should be handled as oncologic guidance deems appropriate. Depending on breast density and patient age, this may include MRI. If no malignancy or culprit for discharge is found or if a papilloma or other high-risk lesion is identified, MRI is suggested. As indicated in the algorithm, MRI should be considered for preoperative evaluation even in non-malignant cases. MRI may identify occult lesions that could potentially need work-up, may delineate greater extent of disease than expected (change surgical planning), or may provide information that could allow for nonsurgical management altogether (in the setting of multiple bilateral similar-appearing masses). In the setting of mammographically and sonographically occult lesions, MRI has an obvious role for evaluation. If the culprit lesion is identified, MRI also provides a method for biopsy. If no lesion is identified, symptoms should be re-addressed and management determined by the clinical scenario. Ductography provides the additional step needed in cases where non-malignant but clinically problematic issues continue (such as surgical localization for cases of persistent yet benign discharge), primarily if there is involvement of only one or two ducts. Unfortunately, clinically problematic cases of multi-duct (greater than two) discharge may still warrant CDE for symptomatic relief. Flow diagram for patients presenting with nipple discharge with steps involving history-taking and imaging suggestions.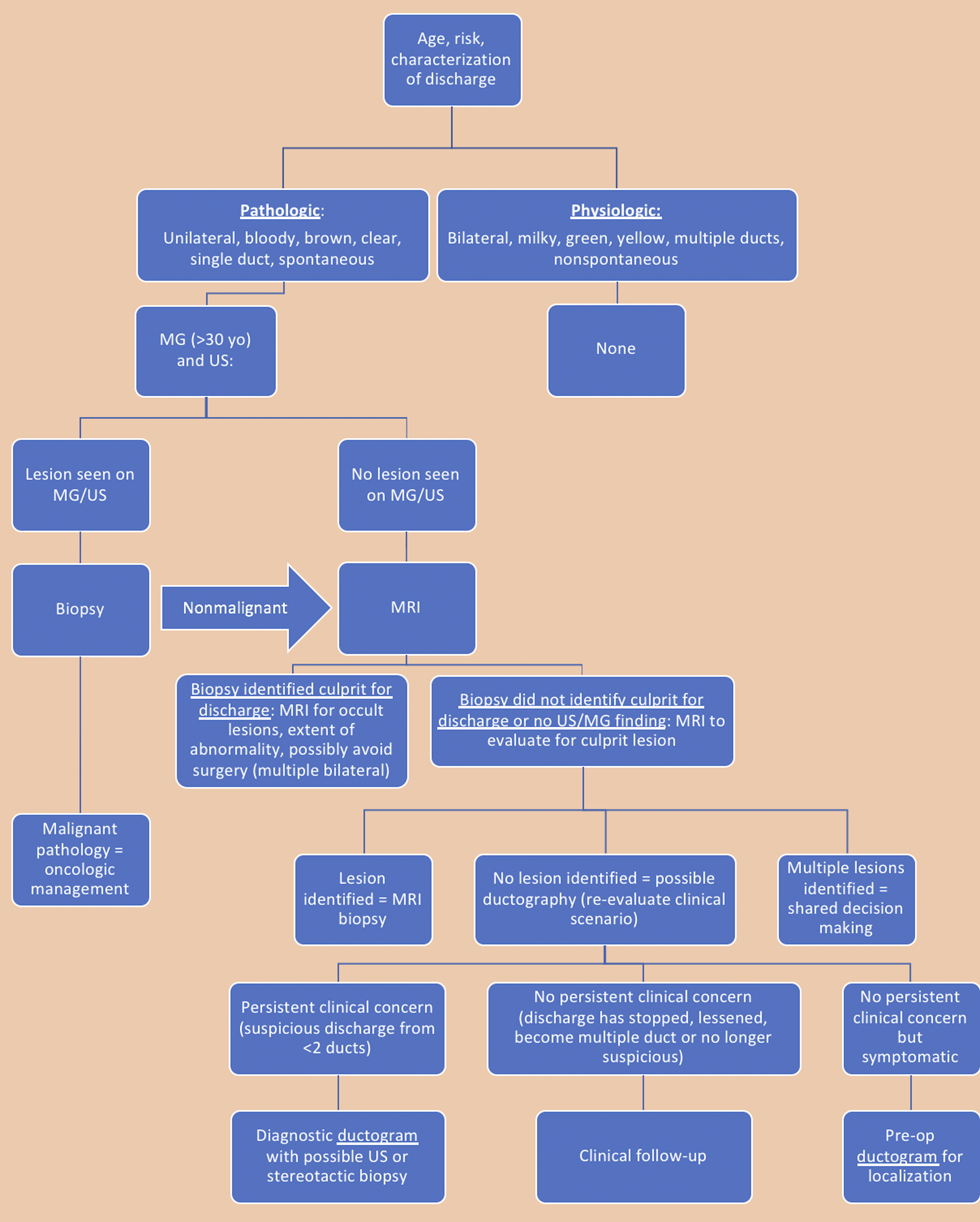
Conclusion
The evaluation, management, and treatment of nipple discharge have evolved significantly to allow for less invasive measures while appropriately identifying those with malignancy. For this strategy to be effective, all elements of work-up need to be addressed, beginning with the basic patient history, including characterization of discharge, personal history of breast cancer, and elevated lifetime risk of breast cancer, and ideally this should be followed by physical examination of discharge. Furthermore, an imaging strategy can be introduced to allow for appropriate triage of patients in need of more advanced imaging. The role of MRI has altered the course of nipple discharge management due to the high negative predictive value in certain cases. For many patients, surgical management can be averted due to MRI’s contribution to the imaging algorithm. For certain types of discharge, primarily involving one or possibly two ducts, galactography can not only minimize surgical intervention but also isolate a specific duct system that may be problematic yet not malignant. For this reason, none of the aforementioned primary imaging modalities should be obsolete when discussing nipple discharge.
Statements and declarations
Footnotes
Acknowledgments
This manuscript was originally published in the Journal of American Osteopathic College of Radiology JAOCR). JAOCR has concluded publication. The article is reproduced with permission.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
