Abstract
TESTOPEAK™ (KGC08EA; Korea Ginseng Corporation, Daejeon, Korea) has been developed to improve the symptoms of andropause. This 8-week, randomized, double-blind, parallel-group, placebo-controlled study was conducted to assess its efficacy and safety in improving the symptoms of andropause in Korean men. The eligible subjects were randomized either to the trial group or the control group, followed by an 8-week treatment period. The subjects of the trial group (n = 48) and those of the control group (n = 48) were orally given a tablet of TESTOPEAK™ and its matching placebo, respectively, at a dose of 500 mg/day once daily in the morning within 30 min after meal during an 8-week treatment period. Efficacy outcome measures include differences in changes in the Aging Male’s Symptoms (AMS) scores, the Androgen Deficiency in Aging Male (ADAM) scores, the International Index of Erectile Function (IIEF) scores, hormone biomarker levels, lipid biomarker levels and body compositions at 8 weeks from baseline between the two treatment arms. Incidences of adverse events (AEs) served as safety outcome measures. There were significant differences in changes in the AMS scores, the ADAM scores, the IIEF scores, total testosterone, free testosterone and sex hormone binding globulin (SHBG) levels, total cholesterol, high-density lipoprotein (HDL) cholesterol and low-density lipoprotein (LDL) cholesterol levels and triglyceride levels and visceral and total fat area at 8 weeks from baseline between the two groups (p < .05). Moreover, there were no cases of AEs in each treatment group. TESTOPEAK™ is an effective, safe natural compound in alleviating symptoms of andropause in middle-aged or elderly men.
Introduction
The rapid economic growth and advancements in medical technology in modern society have greatly contributed to prolonging the average lifespan of humans, leading to a growing interest in elderly health due to population aging. It has been of significant importance to treat health-threatening diseases and to improve the quality of life. With aging of the society, it has been of increasing interest to maintain healthy lives in middle and old age people, which is essential for preventing or delaying the aging. This has been a great concern for younger generations. Of the common aging-related symptoms in men and women, menopause deserves special attention (Araujo et al., 2007; Buvat et al., 2010; Singh, 2013) and is commonly considered a symptom that is unique to women. Unlike menopause in women, however, men do not experience a sudden decline in reproductive gland function. Men are vulnerable to symptoms of andropause because of an aging-related decline in the synthesis of male hormones (Martelli et al., 2021; Schwarz et al., 2011).
Andropause is referred to as the clinical and biochemical syndrome; it characterized by typical symptoms, such as erectile dysfunction, decreased sexual vigor, passive attitude and osteoporosis, and accompanied by decreased serum testosterone levels (Bhasin et al., 2010; K. S. Kim & Kang, 2011; C. Wang et al., 2008). In 1946, Werner A, defined andropause as a complex of symptoms in men, similar to menopause in women, involving endocrine changes, such as sensitivity, depression, memory impairment, attention deficit, easy fatigue, insomnia, facial flushing, periodic sweating and decreased sexual vigor (Werner, 1946).
The decline in male hormones occurs gradually, unlike the abrupt onset of menopause in women. Andropause also varies in timing as compared with menopause in women, and its symptoms are not uniformly experienced by all men (Bhasin et al., 2010; K. S. Kim & Kang, 2011; C. Wang et al., 2008). The results of the Massachusetts Male Aging Study, which surveyed 1,156 men aged 40 to 70 years, showed that there were decreases in total testosterone levels by 1.6%, free testosterone levels by 2.8%, albumin-bound testosterone levels by 2.5%, sex hormone binding globulin (SHBG) levels by 1.3% and prolactin levels by 5.3% at one year (Derby et al., 2006). According to a study conducted by the Korean Society for Sexual Medicine and Andrology, the prevalence of the symptoms of andropause was 24.1%, 28.7%, 28.1% and 44.4% in men in their 40s, 50s, 60s, and 70s or older, respectively (M. G. Park et al., 2007). The effects of decreased male hormones include osteoporosis, loss of muscle mass and increased body fat, resulting in decreased physical function, facial flushing, erectile dysfunction, lipid metabolism-related cholesterol issues, decreased vitality, depression, sleep disorders, attention deficit and systemic fatigue (Araujo et al., 2007; Bassil et al., 2009; Snyder et al., 2016).
Traditionally, both Eucommiae Cortex (Eucommia ulmoides Oliver [EU]) and Achyranthis Radix (Achyranthes japonica Nakai [AJ]), the root of Achyranthes bidentata, have been used as herbal drugs and foods in Asia, such as Korea, Japan and China (S. K. Kim et al., 2020; Marcone et al., 2003; C. Wang et al., 2008). Major constituents of EU include chlorogenic acid, rutin, quercetin, iridoid and α-linolenic acid (Li et al., 2019); its pharmacological actions, such as the improvement of insulin resistance (Jin et al., 2010), that of Alzheimer’s disease through neuroprotection (Kwon et al., 2013), the prevention of bone loss (Zhang et al., 2014) and anti-inflammatory and anti-osteoarthritic effects (Xie et al., 2015), have been described in the literature. Moreover, major constituents of AJ include 20-hydroxyecdysone, inokosterone, ecdysterone, oleanolic acid, bisdesmoside and saponins (He et al., 2017); its pharmacological actions, such as anti-oxidative (J. H. Park et al., 2013), anti-bacterial (Jung et al., 2007) and anti-inflammatory and anti-osteoarthritic effects (S. G. Lee et al., 2012), have been well documented.
Given the above background, TESTOPEAK™ (KGC08EA; Korea Ginseng Corporation, Daejeon, Korea) has been developed using a mixture of EU and AJ in an effort to improve the symptoms of andropause. TESTOPEAK™ is a mixture of 30% ethanol extracts of EU and AJ at a ratio of 3:1, and its active ingredients include pinoresinol diglucoside and 20-hydroxyecdysone on high-performance liquid chromatography (Kwon et al., 2023; J. Y. Lee et al., 2022). Kim S, et al. conducted an in vitro study, thus showing that TESTOPEAK™ caused a significant concentration-dependent increase in testosterone levels up to 34 times as compared with the control group at concentrations of 1 to 500 μg/mL (S. K. Kim et al., 2020). Lee JY, et al. conducted another in vitro study using aged Sprague-Dawley (SD) rats, thus reporting that TESTOPEAK™ caused an increase in testosterone levels by elevating bioavailable testosterone, SHBG and enzymes involved in testosterone synthesis at a concentration of 40 mg/kg. These authors showed that TESTOPEAK™ was effective in improving physical and testicular functions at a concentration of 80 mg/kg in an SD rat model of aging (J. Y. Lee et al., 2022). There is a paucity of clinical data suggesting its efficacy and safety in the context of andropause. Therefore, along the continuum of the above previous literatures, this study was conducted to assess the efficacy and safety of TESTOPEAK™ in improving the symptoms of andropause in Korean men.
Methods
Study Subjects and Setting
The current 8-week, randomized, double-blind, parallel-group, placebo-controlled study was conducted at a university hospital in Korea between September 1, 2020 and February 23, 2021.
Inclusion criteria for the current study are as follows:
Korean adult men aged ≥ 40 years old.
The subjects with total testosterone levels < 3.5 ng/mL or Aging Males’ Symptoms (AMS) scores ≥ 27 points.
The subjects who were informed of details of the current study and submitted a written informed consent for study participation.
Exclusion criteria for the current study are as follows:
The subjects with a past history of receiving pharmacological therapies for the treatment of andropause symptoms (e.g., phosphodiesterase-5 inhibitor, anti-androgen, testosterone replacement therapy, statin, fibrate, niacin, steroid, fish oil, anti-obesity drug, ezetimibe, cholestyramin, omega-3-fatty acid, bile acid sequestrant, fiber-based laxative, phytosterol margarine and anti-erectile dysfunction agent).
The subjects with a past history of taking diuretics, β-blockers, anti-coagulants and anti-platelet agents.
The subjects with a past history of hypertension, coronary artery disease, myocardial infarction, angina pectoris, heart failure, ischemic heart disease, atherosclerosis, arrhythmia, cerebral infarction, stroke, diabetes mellitus and thyroid disease.
The subjects presenting with symptoms of psychogenic andropause.
The subjects having serum levels of hepatic parameters, such as aspartate aminotransferase (AST), alanine aminotransferase (ALT), total bilirubin and alkaline phosphatase, of ≥ 2.5 upper limits of normal (ULN).
The subjects with a past history of renal failure or having serum levels of renal parameters, such as creatinine, of ≥30% ULN.
The subjects having prostate-specific antigen (PSA) levels ≥4.0 ng/mL.
The subjects with positive hepatitis B surface antigen.
The subjects with body mass index (BMI) ≥35 kg/m2.
The subjects with alcohol addiction or a past history of drug abuse.
The subjects with a past history of cancer.
The subjects with a past history of hypersensitivity to study treatments.
The subjects who are at a great risk of serious drug interactions due to concomitant drugs.
The subjects who are prohibited to receive study treatments for medical and psychiatric reasons.
The subjects who are not allowed to participate in the current study for legal reasons.
The subjects who participated in other clinical studies within 6 months prior to the study entry.
The subjects who may not have a cooperative attitude toward the current study or are deemed to be ineligible for study participation according to the investigator’s judgment.
Rationale of Sample Size Estimation
To verify the superiority of the effects of TESTOPEAK™ to the placebo in improving andropause symptoms, considering a non-evaluable rate of 5%, a total of 96 subjects (48 of the trial group and 48 of the control group) will be recruited for this study. This is based on a previous published study showing that the AMS scores were 26.00 ± 10.40 points in the control group and 20.85 ± 3.81 points in the trial group (Udani et al., 2014).
Trial Methods
Once determined to be eligible for study participation, the subjects were evaluated for clinical examinations at baseline. At Visit 2, they were randomized either to the trial group or the control group, followed by an 8-week treatment period. The study schema is illustrated in Figure 1.

Study Design.
The subjects of the trial group were orally given a tablet of TESTOPEAK™ at a dose of 500 mg/day once daily in the morning within 30 min after meal during an 8-week treatment period. The subjects of the control group were also orally given its matching placebo at a dose of 500 mg/day once daily in the morning within 30 min after meal during an 8-week treatment period.
Randomization and Blinding
Based on the randomization table, provided by the contract research organization (CRO), the subjects were randomized to either of two treatment arms using a block randomization. According to the uniform distribution, computer-generated lists of random numbers (RNs) were created using Random Number Generators of SPSS software (IBM Corp., Armonk, NY, USA). At the time of study entry, the principal investigator was provided with a randomization envelope from the CRO. This is followed by allocation of RNs to all the eligible subjects who passed the screening test according to the order of study treatments.
The subjects of each treatment arm were provided with study treatments whose specification and formulation are identical. Both the principal investigator and the subjects would be unaware of allocation of a certain subject to a specific treatment unless a randomization envelope is opened. Thus, the double-blind status was maintained until study treatments have been completed.
Assessment Criteria and Outcome Measures
Baseline characteristics of the subjects include age, height, weight and BMI.
For efficacy assessment, the subjects were assessed for the measurement of the AMS, Androgen Deficiency in the Aging Male (ADAM) and International Index of Erectile Function (IIEF) scores, as previously described (Ku et al., 2005; D. S. Park et al., 2008). Moreover, they were also assessed for hormone biomarker levels, lipid biomarker levels and body compositions. Then, both full analysis and per-protocol analysis were performed, for which full analysis set (FAS) and per-protocol set (PPS) were provided. Efficacy outcomes were finally assessed based on the FAS.
Primary efficacy outcome measures include differences in changes in the AMS scores at 8 weeks from baseline between the two treatment arms. Secondary efficacy outcome measures include differences in changes in the ADAM scores, IIEF scores, hormone biomarker levels, lipid biomarker levels and body compositions at 8 weeks from baseline between the two treatment arms.
For safety assessment, any adverse events (AEs) were categorized by the system organ class and then coded by preferred terms using the Medical Dictionary for Regulatory Activities (MedDRA) version 19 (Yakoot et al., 2017). Incidences of treatment-emergent AEs (TEAEs) and serious AEs (SAEs) served as safety outcome measures.
Statistical Analysis
All data was expressed as mean ± standard deviation or the number of the subjects with percentage, where appropriate. In each treatment arm, changes in outcome measures at 24 weeks from baseline were analyzed using the Wilcoxon signed-rank test. Moreover, differences in efficacy and safety outcome measures between the two treatment arms were analyzed using independent two-sample t-test or Wilcoxon rank sum test, if applicable. Statistical analysis was performed using the Statistical Analysis Software Version 9.4 (SAS Institute Inc, Cary, North Carolina, USA). A p-value of < 0.05 was considered statistically significant.
Results
Baseline Characteristics of the Subjects
A total of 96 subjects (n = 96) were finally enrolled in the current study, who were equally randomized to the trial group (n = 48) and the control group (n = 48). The disposition of the study subjects is illustrated in Figure 2.
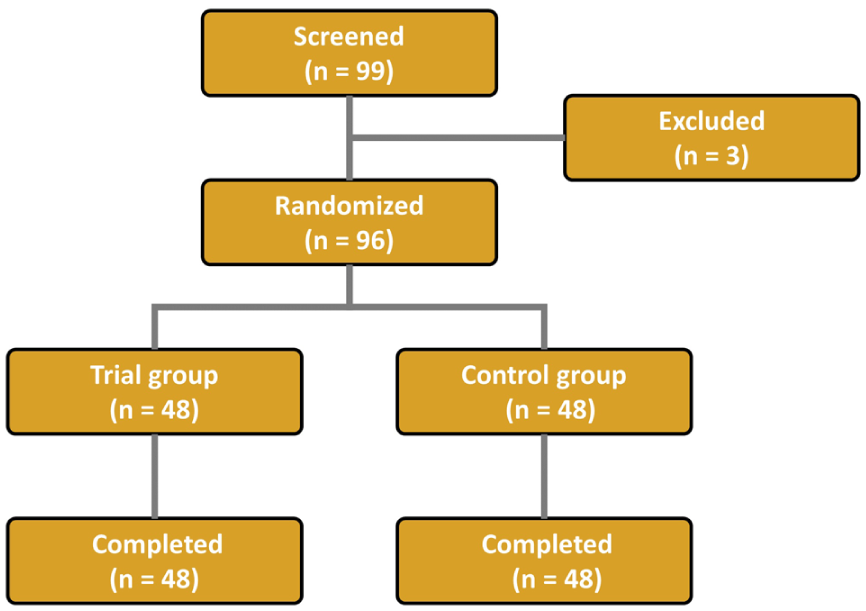
Disposition of the Study Subjects.
There were no significant differences in the age, height, weight and BMI between the two treatment arms (Table 1).
Baseline Characteristics of the Subjects (n = 96).

Note. BMI, body mass index. Values are mean ± standard deviation with the range.
Efficacy Outcomes
In the current study, there were no subjects who were ineligible for the PP analysis; there were no subjects who were dropped out of the current study, used prohibited concomitant drugs or foods, had a ≤ 80% compliance with study treatments or seriously violated the protocol. There was no difference in the number of the subjects between the FAS and the PPS. Therefore, the FA was solely performed in the current study.
There were significant differences in changes in the AMS scores, the ADAM scores and the IIEF scores at 8 weeks from baseline between the two groups (p < .001) (Figures 3–5). Of hormone biomarker levels, total testosterone, free testosterone and SHBG showed significant differences in changes at 8 weeks from baseline between the two groups (p < .001) (Table 2).
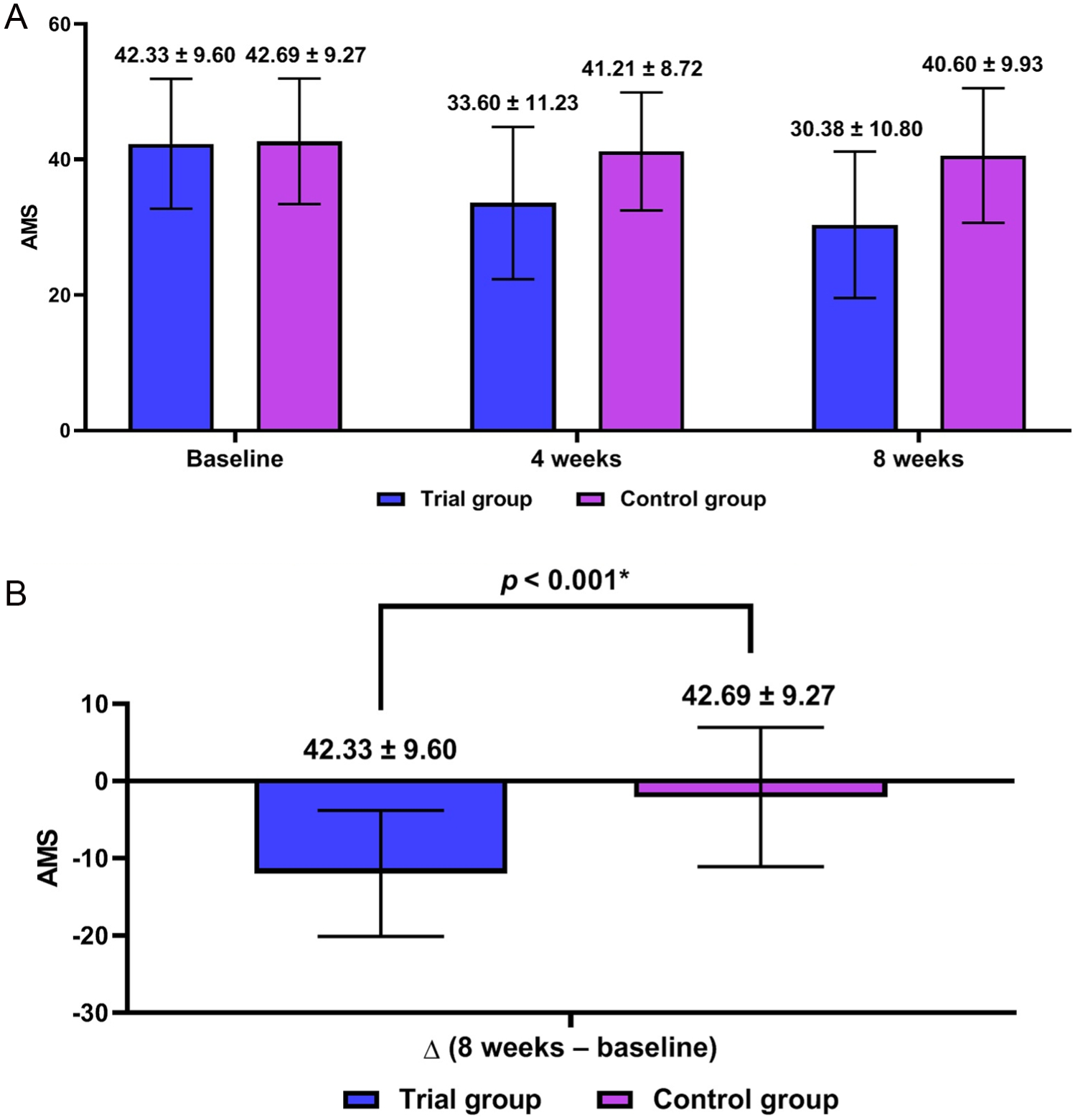
Aging Male’s Symptoms (AMS) Scores (n = 96).
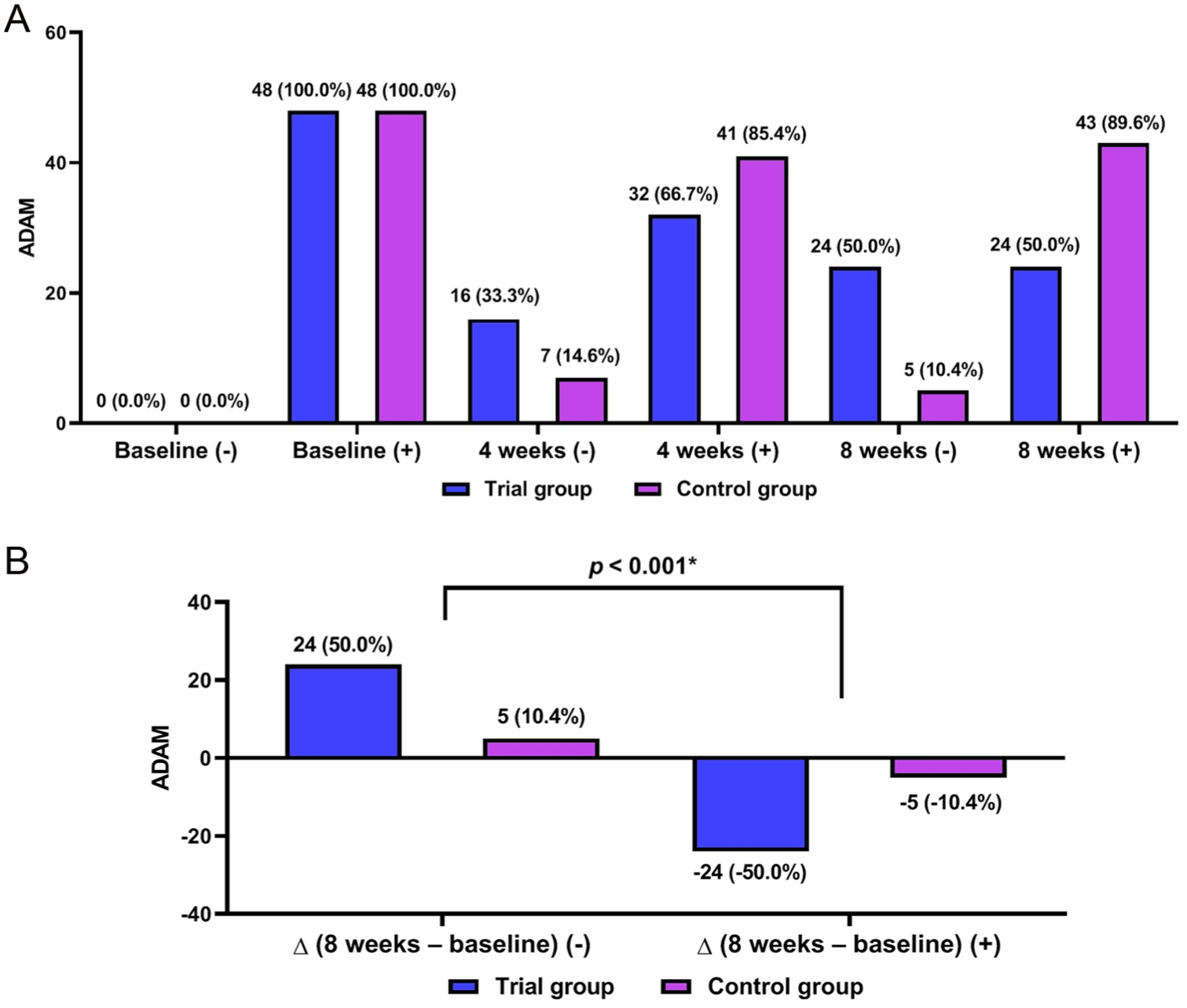
Androgen Deficiency in Aging Male (ADAM) Scores (n = 96).

International Index of Erectile Function (IIEF) Scores (n = 96).
Time-Dependent Changes in Hormone Biomarker Levels (n = 96).

Note. N/A, non-applicable. SHBG, sex hormone binding globulin. Values are mean ± standard deviation.
Statistical significance at p < .05.
There were significant differences in changes in total, high-density lipoprotein (HDL) and low-density lipoprotein (LDL) cholesterol and triglyceride (TG) levels at 8 weeks from baseline between the two groups (p = .004, .024, .003, and .010, respectively) (Table 3).
Time-Dependent Changes in Lipid Biomarker Levels (n = 96).

Note. N/A, non-applicable. HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Values are mean ± standard deviation.
Statistical significance at p < .05.
There were significant differences in changes in visceral and total fat area at 8 weeks from baseline between the two groups (p = .014 and .017, respectively) (Table 4).
Time-Dependent Changes in Body Compositions (n = 96).

Note. N/A, non-applicable. Values are mean ± standard deviation.
Statistical significance at p < .05.
Safety Outcomes
There were no cases of TEAEs and SAEs in each treatment group.
Discussion
Symptoms of andropause occur as a result of decreased androgen levels; testosterone levels are annually decreased by 1% to 2% (Matsumoto, 2002). Testosterone is synthesized in the mitochondria through several cascades of steroid hormone synthesis from cholesterol as a raw material. Cholesterol is converted into pregnenolone via P450scc and StAR, which is converted into progesterone via 3β-HSD2. Progesterone is converted into androstenedione via CYP17A1, and it is synthesized into testosterone via 17β-HSD3 (Payne & Hales, 2004). With aging, however, there is an impairment in the function of the Leydig cells, accompanied by a decrease in the expression of P450scc and StAR (Midzak et al., 2009).
Testosterone is converted into DHT via 5α-reductase and to estradiol via aromatase. This eventually leads to a decrease in the total testosterone levels (Rasmussen & Ekstrand, 2014). Symptoms of andropause include sexual and erectile dysfunction, weight gain, muscle weakness, insomnia, fatigue and depression (Behre et al., 2012; Rosen et al., 2011). Hormone and hormone-replacement therapies are prescribed for individuals diagnosed with androgen deficiency; they are vulnerable to certain risks, including prostate cancer and prostatitis (Jarvis et al., 2015; Osterber et al., 2014). Potential natural sources to increase testosterone levels and to restore symptoms of andropause should be explored; the recent increase in the awareness of andropause has led to a simultaneous upsurge in studies about natural functional compounds for reducing symptoms of andropause (K. Y. Lee et al., 2022). Because there has been an increase in the number of men presenting with symptoms of andropause according to the prolongation of life span, it would also be mandatory to identify safe natural substances that are effective in reducing symptoms of andropause (Noh et al., 2012).
TESTOPEAK™ deserves special attention; its efficacy and safety in the context of andropause have been confirmed in two recent in vitro studies (S. K. Kim et al., 2020; J. Y. Lee et al., 2022). According to Lee JY, et al., TESTOPEAK™ ameliorated symptoms of andropause symptoms by elevating testosterone levels and enhancing physical functions (J. Y. Lee et al., 2022). This is in agreement with a previous study showing that TESTOPEAK™ restored an aging-related decline in testosterone levels in an SD rat model of aging by modulating the expression of genes involved in steroidogenesis in the testis (S. K. Kim et al., 2020). Bioavailable testosterone is a determinant of the muscle strength and physical function. Over 60% of sex hormones bind to SHBG, which increases inversely with obesity or hyperinsulinemia while the opposite phenomena occur in the context of aging (Muller et al., 2003). In both middle-aged and elderly men, there is strong correlation between body fat mass and low sex hormone levels (Huhtaniemi & Forti, 2011). Presumably, serum TG and epididymal fat might be decreased after the treatment with TESTOPEAK™ (J. Y. Lee et al., 2022).
The current study showed that TESTOPEAK™ was effective in improving the AMS and ADAM scores at 8 weeks as compared with baseline. This suggests that TESTOPEAK™ might be effective in alleviating age-related androgen deficiency-associated symptoms. Both self-assessment questionnaires are used to screen for late-onset hypogonadism in aging men, with the AMS being a more detailed 17-item survey covering psychological, somatic and sexual issues, while the ADAM is a shorter, 10-item screening tool focusing on core symptoms, such as decreased libido and erectile dysfunction (Chen et al., 2013).
Erectile function is closely associated with androgen availability, endothelial integrity and metabolic status (H. Wang et al., 2025; Zhong et al., 2025). Both hormonal and vascular determinants of erectile function are commonly impaired in men with androgen deficiency (Yassin et al., 2011). The IIEF scores are a measure of the severity of erectile dysfunction, assisting clinicians to assess erectile function, desire and satisfaction (Rhoden et al., 2002). Moreover, TESTOPEAK™ was found to elevate total and free testosterone levels as well as SHBG levels in an in vivo experiment using an animal model of late-onset hypogonadism (J. Y. Lee et al., 2022). SHBG plays a pivotal role in regulating androgen bioavailability, particularly in aging men, in whom elevated SHBG is commonly associated with reduced free testosterone despite preserved total testosterone levels (Fan et al., 2024; Mukherjee & Abilash, 2025). From this context, it is noteworthy that the current study showed concurrent changes in SHBG and free testosterone levels at 8 weeks as compared with baseline. Indeed, there is an inverse genetic correlation between SHBG and free testosterone levels, suggesting that genes may influence them in opposite directions, which is consistent with there is an inverse correlation between their circulating levels (Coviello et al., 2011). Taken together, the current study suggests that TESTOPEAK™ may enhance androgen bioavailability rather than merely increasing circulating testosterone.
In aging men, there is an association between dyslipidemia, androgen deficiency and cardiovascular risk, which is characterized by a complex interplay of metabolic shifts. It has been shown that low testosterone often acts as a precursor to or biomarker for unfavorable lipid profiles and increased cardiovascular mortality (Barrientos et al., 2020; Kampka et al., 2026; Traish & Kypreos, 2011). A previous experiment showed that TESTOPEAK™ was found to lower total cholesterol, LDL cholesterol and TG while elevating HDL cholesterol in an animal model of late-onset hypogonadism (J. Y. Lee et al., 2022). These results can be extended to the current clinical trial showing that TESTOPEAK™ was effective in improving total, HDL and LDL cholesterol and TG levels at 8 weeks as compared with baseline.
Previous experimental studies have suggested that TESTOPEAK™ might affect visceral and total fat area via mechanisms underlying testosterone synthesis and lipid metabolism (Kwon et al., 2023; J. Y. Lee et al., 2022). This was confirmed from the current study showing that TESTOPEAK™ was effective in improving visceral and total fat area at 8 weeks as compared with baseline. Testosterone significantly affects visceral and total fat areas by not only directly regulating lipid metabolism, such as lipolysis, but also interacting with androgen receptors that are upregulated in the visceral fat. In men, there is a relationship between low testosterone levels and increased visceral and total fat, although normal testosterone levels help maintain a favorable body composition (Holland et al., 2016; Klaver et al., 2022; Ma et al., 2024).
There were no cases of TEAEs and SAEs in the current study. This is in agreement with a previous experiment showing no acute toxicity and hepatic damages (J. Y. Lee et al., 2022).
To summarize, the current results are as follows:
There were significant differences in changes in the AMS scores, the ADAM scores and the IIEF scores at 8 weeks from baseline between the two groups (p < .001).
Of hormone biomarker levels, total testosterone, free testosterone and SHBG showed significant differences in changes at 8 weeks from baseline between the two groups (p < .001).
There were significant differences in changes in total, HDL and LDL cholesterol and TG levels at 8 weeks from baseline between the two groups (p = .004, .024, .003, and .010, respectively).
There were significant differences in changes in visceral and total fat area at 8 weeks from baseline between the two groups (p = .014 and 0.017, respectively).
There were no cases of TEAEs and SAEs in each treatment group.
But the current results cannot be generalized because there are two limitations of the current study: First, the current study enrolled a small number of the subjects from a single center. The possibility of selection bias could not therefore be completely ruled out. Second, the treatment period of 8 weeks is relatively shorter. According to a review of the previous literatures about the effects of a mixture of EU and AJ, the study period ranged between 6 weeks for an animal experiment and 24 weeks for a clinical trial (Ha et al., 2016; J. Y. Lee et al., 2022). Further long-term randomized clinical trials are therefore warranted to corroborate the current findings. Third, the current study failed to identify a dose-response relationship. But the treatment dose in the current study was based on previous published literatures. That is, there is a synergistic dose-response relationship in the treatment of musculoskeletal and hormonal conditions (Choi et al., 2020a, 2020b; J. Y. Lee et al., 2022).
Conclusions
Based on the current results, it can be concluded that TESTOPEAK™ is an effective, safe natural compound in alleviating symptoms of andropause in middle-aged or elderly men. But further large-scale, long-term multi-center studies are warranted to corroborate the current results.
Footnotes
Acknowledgements
We thank all the research subjects for their participation in the current study.
Ethical Considerations
The current study was approved by the Institutional Review Board (IRB) of Seoul National University Bundang Hospital in Korea (IRB approval # B-2008/628-001) and then conducted in compliance with the relevant ethics guidelines. All the study treatments and procedures described herein were performed in accordance with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The current study was conducted in compliance with the International Conference of Harmonization—Good Clinical Practice (ICH-GCP) and it is registered with the Clinical Research information Service (KCT0009341). A written informed consent to participate was obtained from all of the participants.
Consent for Publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current study was supported by Korea Ginseng Corporation (KGC-MD 20-169).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All the authors are employees of Korea Ginseng Corporation for the current study. The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending or royalties.
Data Availability Statement
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
