Abstract
This study compared the efficacies of single-dose ceftriaxone therapy at doses of 250 mg, 500 mg, and 1 g for gonococcal paraurethral duct infection in men. Forty-eight men with gonococcal paraurethral duct infection were assigned to Groups A, B, and C based on their order of arrival in the clinic; each group comprised 16 patients. Groups A, B, and C received single intramuscular doses of ceftriaxone at 250 mg, 500 mg, and 1 g, respectively. The mean times to resolution of purulent discharge from the paraurethral duct were 1.87 ± 0.81 days in Group A, 1.06 ± 0.25 days in Group B, and 1.13 ± 0.34 days in Group C. The mean times to resolution of erythematous swelling at skin lesions were 4.75 ± 1.69 days in Group A, 3.44 ± 0.89 days in Group B, and 3.31 ± 1.25 days in Group C. The mean times to resolution of tenderness at skin lesions were 5.25 ± 1.00 days in Group A, 3.06 ± 0.10 days in Group B, and 2.38 ± 1.03 days in Group C. The mean times to closure of the paraurethral duct orifice were 9.88 ± 2.92 days in Group A, 6.88 ± 1.71 days in Group B, and 6.38 ± 1.86 days in Group C. The resolution times significantly differed between Group A and Groups B and C (all p < .05); however, there were no significant differences between Groups B and C (all p > .05). The study shows that all three ceftriaxone single-dose regimens effectively cured gonococcal paraurethral duct infection in men. A single 500 mg dose may represent a more suitable treatment option.
In men, the paraurethral ducts are small blind channels lined with columnar epithelium that run parallel to the terminal part of the urethra for varying distances and open near or within the lips of the external meatus (Harkness, 1948; King & Nicol, 1969). These ducts appear to be embryological remnants and are not visible to the naked eye (Gilhooly & Hensle, 1984). Neisseria gonorrhoeae can invade the paraurethral ducts through their openings, leading to gonococcal paraurethral duct infection in men (Fan, 2010; Fan & Zhang, 2012; Fan, Zhang, Fan, et al., 2018; Fan, Zhang, & Ye, 2016; Fan, Zhang, Ye, et al., 2018).
This infection typically manifests as local erythematous swelling at the external urethral orifice; a pinhead-like ostium at the center marks the opening of the paraurethral duct. Application of pressure to the glans penis can produce purulent exudate from this ostium. Gonococcal paraurethral duct infection in men can arise as a localized complication of gonorrhea (Fan, 2010; Fan & Zhang, 2012; Harkness, 1948; King & Nicol, 1969) or as an independent primary infection (Fan et al., 2019). Currently, no established guidelines exist for treating gonococcal paraurethral duct infection in men. To evaluate the efficacy of different ceftriaxone doses in treating this condition and to lay the groundwork for standardized treatment protocols, we administered various ceftriaxone doses to male patients with gonococcal paraurethral duct infection, based on treatment guidelines for uncomplicated urethral gonorrhea.
Patients and Methods
The study protocol was approved by the Medical Ethics Committee of our hospital (approval number 2020-csyy-05), and all participants provided written informed consent.
Patients
We enrolled male patients with gonococcal paraurethral duct infection treated at our hospital from March 2020 to August 2024.
Inclusion criteria: Eligible patients presented with local erythematous swelling at the external urethral orifice, with a pinhead-like ostium at the center. Application of pressure resulted in purulent exudate from the ostium. The pathogen was identified as N. gonorrhoeae, and concurrent urethral gonorrhea was identified. Patients had no prior history of gonococcal paraurethral duct infection or urethral gonorrhea and had not received antibiotic treatment since the infection.
Exclusion criteria: Patients were excluded if other pathogens were detected at skin lesions, if multiple or non-penetrating lesions were present, or if there was a previous history of paraurethral duct gonococcal infection. Patients with redundant prepuce or phimosis, and those who did not provide informed consent, were also excluded.
Method
Grouping
Patients were assigned to Groups A, B, and C in a cyclical manner based on their order of arrival at the clinic. Each patient was assigned to the next group in the sequence (A, B, C), ensuring a balanced and unbiased distribution across the groups.
Clinical Data Recording
Recorded data included age, body weight, marital status, source of infection, sexual orientation, sexual practices, condom use, incubation period (interval from sexual exposure to the onset of the first symptom of gonococcal paraurethral duct infection), disease duration for urethral gonorrhea (time from symptom onset to treatment), disease duration of gonococcal paraurethral duct infection caused by N. gonorrhoeae, characteristics of skin lesions, subjective symptoms of skin lesions, and lower urinary tract symptoms, such as dysuria, frequent urination, urinary urgency, and urethral pus discharge.
Laboratory Tests
To prevent cross-contamination between discharges from the paraurethral duct and the urethra, the glans penis was rinsed with normal saline for 3 min. Pressure was then applied to the external urethral orifice, and discharge from the paraurethral duct orifice was collected using a sterile cotton swab. After another 3-min rinse with saline, a thin cotton swab was inserted 4 cm into the urethra and gently rotated to collect urethral secretions. Gram staining and the 1000 × oil mirror were performed to identify intracellular Gram-negative diplococci within phagocytes. Specimens were also cultured to detect common bacteria and fungi. Polymerase chain reaction was conducted to detect DNA from N. gonorrhoeae, Chlamydia trachomatis, Ureaplasma urealyticum, and herpes simplex virus types 1 and 2. The reagent kits were purchased from DAAN Gene Co (Sun Yat-sen University, Guangzhou, China). Venous blood samples were analyzed with the rapid plasma reagin test, the Treponema pallidum hemagglutination assay, and a human immunodeficiency virus antibody assay.
Treatments and Observation
Groups A, B, and C received single intramuscular doses of ceftriaxone at 250 mg, 500 mg, and 1 g, respectively. Patients were instructed to abstain from alcohol and sexual activity. They attended daily follow-up visits for 21 consecutive days. The times to resolution of purulent discharge from the paraurethral duct, erythematous swelling at skin lesions, tenderness at skin lesions, and closure of the paraurethral duct opening were recorded for each group, along with treatment efficacy and incidence of any sequelae.
Efficacy Evaluation
Three weeks after treatment, gonococcal paraurethral duct infection was considered “cured” if skin lesions and associated symptoms had resolved. If lesions persisted and/or polymerase chain reaction (PCR) test results remained positive for N. gonorrhoeae DNA, the infection was deemed “uncured.” For patients with urethral gonorrhea, the condition was considered “cured” if symptoms and signs had resolved, with a negative PCR test result for N. gonorrhoeae DNA in urethral secretions; otherwise, the outcome was regarded as “uncured.”
Assessment of Sequelae
After ceftriaxone therapy, local erythematous swelling at the external urethral orifice resolved, and purulent discharge disappeared (Fan et al., 2015). However, the ostium remained open, and pressure application led to the expression of a transparent fluid from the ostium. However, pathogen testing of the transparent fluid returned negative results. The ostium did not close during the 21-day follow-up period, leading to a diagnosis of paraurethral duct dilatation.
Statistical Analysis
Data were analyzed using Statistical Package for the Social Sciences (SPSS) statistical software (version 13.0) with a significance threshold of p < .05. Continuous variables were assessed with t-tests, whereas categorical variables were analyzed using chi-square tests. Analysis of variance was used to compare multiple variables. Tukey’s honestly significant difference (HSD) test was utilized for pairwise comparisons.
Results
Clinical Data
In total, 53 male patients with gonococcal paraurethral duct infection were treated at the Department of Dermatology and Venereology of our hospital from March 2020 to August 2024. Of these patients, 48 met the criteria for inclusion in this study. Patients were cyclically assigned to three groups (A, B, and C; 16 patients per group) based on their order of arrival in the clinic. Data regarding age, body weight, disease incubation period, disease course of urethral gonorrhea, and disease course of gonococcal paraurethral duct infection for the three groups are presented in Table 1. The numbers of married and unmarried patients were as follows: nine (56.25%) and seven (43.75%) in Group A, 11 (68.75%) and five (31.25%) in Group B, and nine (56.25%) and seven (43.75%) in Group C; these numbers did not significantly differ among the groups (p > .05). The three groups were similar in age, body weight, disease incubation period, duration of gonococcal paraurethral duct infection, and duration of urethral gonorrhea (all p > .05). All 48 patients were heterosexual, with a history of non-marital sexual activity; none had used condoms during these encounters. All patients presented with local erythematous swelling at the external urethral orifice, characterized by a pinhole-like opening at the center, from which purulent discharge was expressed upon pressure (Figure 1). The lesions were tender on palpation. Lower urinary tract symptoms, including dysuria, frequent urination, urinary urgency, and urethral pus discharge, were reported by all patients.
Demographic and Clinical Characteristics of Patients in the Three Groups.

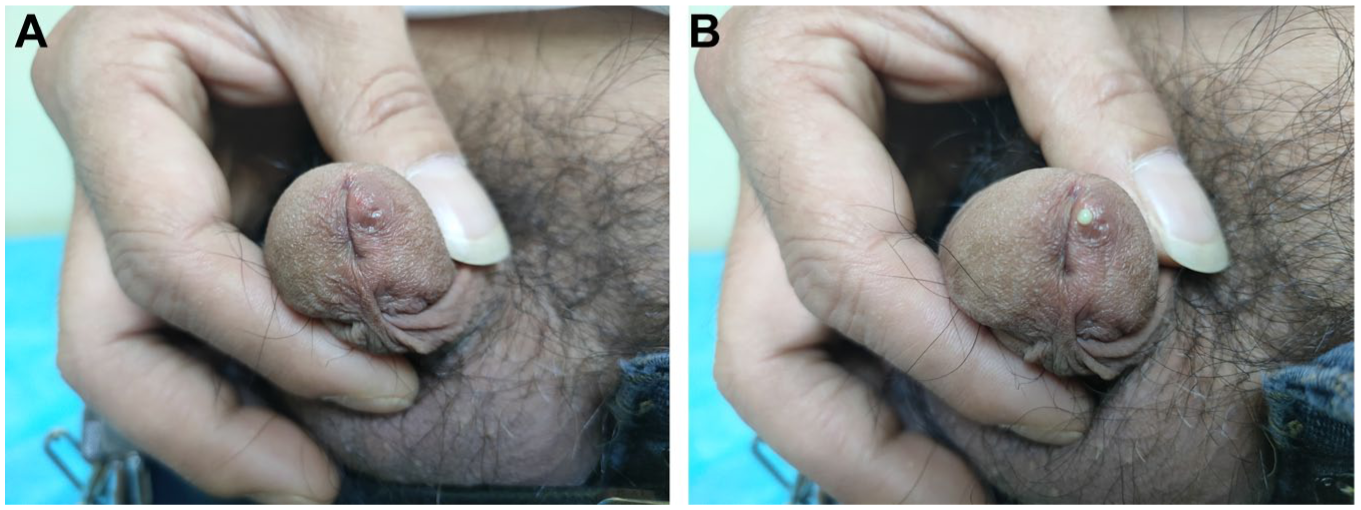
Paraurethral Duct Infection With Neisseria gonorrhoeae. (A) Local Swelling Was Seen on the Left Side of the External Urethral Orifice, With a Pinhead-Like Ostium at the 3 o’clock Position. (B) Pressure on the Glans Caused Expression of a Purulent Discharge From the Ostium
Results of Laboratory Tests
Intracellular Gram-negative diplococci within phagocytes were identified in the paraurethral and urethral secretions of all 48 patients. General bacterial and fungal cultures were negative in all cases. PCR test results were positive for N. gonorrhoeae DNA; C. trachomatis, U. urealyticum, and herpes simplex virus DNA were not detected in any specimens. Rapid plasma reagin, T. pallidum hemagglutination, and human immunodeficiency virus antibody test results were also negative in all cases.
Efficacy and Adverse Reactions
The gonococcal paraurethral duct infection was cured in all 48 cases, along with resolution of gonococcal urethritis symptoms. After treatment, intracellular Gram-negative diplococci within phagocytes were no longer detected in urethral secretions. PCR tests for gonococcal DNA in urethral secretions returned negative results in all cases. Table 2 shows a comparison of the efficacies of different single-dose ceftriaxone regimens for treating paraurethral duct gonococcal infection in men across the three groups.
Comparison of Ceftriaxone Efficacies in Treating Gonococcal Paraurethral Duct Infection in Men.

p < .05, compared among three groups; #p < .05, compared between two groups.
In terms of adverse reactions, as shown in Table 3, one patient (6.25%) in Group A experienced mild dizziness, one patient (6.25%) in Group B reported nausea, and two patients (12.5%) in Group C experienced dizziness (p > .05). These symptoms spontaneously resolved without any specific treatment.
Adverse Events Reported by Group in the Study.

Discussion
Gonococcal paraurethral duct infection in men was previously referred to as male gonococcal inflammation of the paraurethral glands. However, considering that biopsy of the lesions and surrounding tissues reveals only the paraurethral duct and its inflammation—without any associated glands (Fan et al., 2015; Fan, Zhang, Ye, et al., 2018)— the term “gonococcal paraurethral duct infection” may be more appropriate for this condition (Fan et al., 2018). Gonococcal paraurethral duct infection is a local complication of gonorrhea (Sherrard, 2014), with an incidence of approximately 1.5% (Fan, Zhang, & Ye, 2016). Risk factors include a prolonged course of gonorrhea, sexual intercourse during infection, repeated squeezing of the penis, and a redundant prepuce (Fan & Zhang, 2012).
Ceftriaxone is a broad-spectrum, semi-synthetic third-generation cephalosporin. Its long half-life and beta-lactamase stability make it effective in treating gonorrhea. Its pharmacokinetics include an albumin-bound fraction that serves as a reservoir, maintaining a prolonged effective antimicrobial concentration (Reiner, 1982). Guidelines across various countries recommend ceftriaxone as the preferred antibiotic for gonorrhea treatment, with increasing dosage recommendations over time (Bignell & Fitzgerald, 2011; Fifer et al., 2020; St. Cyr et al., 2020; Workowski & Bolan, 2015). Guidelines exist for treating local complications of gonorrhea, including gonococcal pelvic inflammatory disease, gonococcal epididymitis, and gonococcal conjunctivitis (Workowski & Bolan, 2015), as well as disseminated gonococcal infection (Workowski & Bolan, 2015). However, no specific guidelines are available for treating gonococcal paraurethral duct infection in men. Based on the guidelines for uncomplicated urethral gonorrhea treatment, we administered various ceftriaxone doses to men with gonococcal paraurethral duct infection. We found that single doses of ceftriaxone at 250 mg, 500 mg, and 1 g effectively treated the infection. This may be due to the persistent ceftriaxone susceptibility among nearly all gonococcal strains circulating in China (Li et al., 2014). In addition, the blood concentration achieved with a single 250-mg dose is sufficient to eradicate gonococci in the paraurethral duct. The times to resolution of purulent discharge from the paraurethral duct, erythematous swelling at skin lesions, tenderness, and closure of the paraurethral duct opening in Group A significantly differed from those times in Groups B and C, possibly because of the varying ceftriaxone concentrations in plasma and paraurethral tissue across the three groups; lower ceftriaxone concentrations may lead to reduced bactericidal efficacy against gonococci (Korting et al., 1987). However, there were no significant differences in efficacy between Groups B and C, suggesting that a single 500 mg dose of ceftriaxone achieved a saturating concentration sufficient to eliminate gonococci in the paraurethral duct.
The study also showed that during recovery, resolution of purulent discharge from the paraurethral duct was initially observed, followed by resolution of erythematous swelling and tenderness at skin lesions; closure of the paraurethral duct opening occurred last.
Increasing age, a prolonged course of gonococcal inflammation of the paraurethral duct, co-infection with C. trachomatis, and gonococcal reinfection are known risk factors for paraurethral dilatation secondary to gonococcal paraurethral duct infection (Fan, Zhang, Wang, et al., 2016). None of the patients in our study exhibited paraurethral dilation, likely due to the short disease duration, absence of co-existing C. trachomatis infection, and lack of a prior history of gonococcal paraurethral duct infection.
In an earlier study, Sequential therapy was examined for gonococcal inflammation of the paraurethral glands in men (Fan et al., 2012). The cure rate after intramuscular ceftriaxone injection was only 70.83%, possibly because some patients with gonococcal infection of the glans penis skin (Fan et al., 2023; manifesting as abscesses and nodules) were included in the treatment group for gonococcal paraurethral duct infection.
In China, high-dose ceftriaxone is often used to treat urethral gonorrhea without strict adherence to relevant guidelines (Han et al., 2018). As a complication of urethral gonorrhea, gonococcal paraurethral duct infection lacks specific treatment guidelines, which may lead to the inappropriate use of higher ceftriaxone doses in clinical practice. In our study, all three single-dose ceftriaxone regimens (250 mg, 500 mg, and 1 g) were effective in curing gonococcal paraurethral duct infection in men. A single 500 mg dose may represent a more suitable treatment option.
However, this study was limited by its small sample size; larger multicenter controlled studies are needed to validate our findings. In addition, we did not determine the minimum inhibitory concentration of ceftriaxone for gonococci in the paraurethral duct. According to the literature, the minimum inhibitory concentration of ceftriaxone for N. gonorrhoeae in the urethra ranges from 0.001 to 0.25 mg/L (Ito et al., 2016).
Footnotes
Acknowledgements
The authors would like to thank the clinic staff for their participation in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
