Abstract
This study was performed to evaluate the therapeutic effects of wedge resection on male paraurethral duct dilatation following gonococcal paraurethral duct infection. Twenty-six men with paraurethral duct dilatation following gonococcal paraurethral duct infection were enrolled. Their lesions underwent wedge resection after examination using an ACUSON X300 ultrasound system. The anesthetic method, surgical duration, intraoperative blood loss, wound healing time, complications, sequelae, and curative effect were assessed. All 26 men received local infiltration anesthesia. The mean surgical duration was 18.65 ± 2.50 min (range, 14–23 min), the mean intraoperative blood loss was 10.50 ± 1.68 ml (range, 8–14 ml), and the mean wound healing time was 14.73 ± 1.31 days (range, 13–17 days). The lesions were cured in all patients (100%). The postoperative wounds healed in all patients. No complications or glans defects were observed in any patients. The study identifies that high-frequency ultrasound imaging–guided wedge resection is an effective and safe therapy for paraurethral duct dilatation following gonococcal paraurethral duct infection in men.
The paraurethral ducts in men are small, blind channels lined with columnar epithelium (Harkness, 1948). These ducts run parallel to the terminal part of the urethra for varying distances and open near or within the lips of the external meatus (King & Nicol, 1969). The paraurethral ducts appear to be embryological remnants and are not visible to the naked eye (Gilhooly & Hensle, 1984). Neisseria gonorrhoeae can invade the paraurethral duct via its orifice, resulting in gonococcal inflammation of the paraurethral duct (Fan, 2010; Fan, & Zhang, 2012; Fan, Zhang, & Fan, 2012; Fan, Zhang, & Jiang, 2014; Sherrard, 2014). The inflammation clinically manifests as local erythematous swelling at the external urethral orifice, with a pinhead-like ostium at the center indicating the orifice of the paraurethral duct. Pressure can result in purulent excretion from the ostium. Although N. gonorrhoeae infection can be cured by ceftriaxone followed by resolution of the local swelling and redness of the external urethral orifice and cessation of the purulent secretions, the ostium remains open and pressure can still cause the expression of a transparent pathogen-free liquid. Such a lesion can be pathologically confirmed as paraurethral duct dilatation (Fan et al., 2015; Fan, Zhang, & Song, 2013), which is considered a sequela of gonococcal inflammation of the paraurethral ducts (Fan et al., 2015) and is a stressful condition for the patient. Furthermore, pathogens can enter the paraurethral duct via the enlarged orifice and cause inflammation of the paraurethral ducts and even urethritis. Konrad (1976) described a patient with recurrence of gonorrhea caused by gonorrheal paraurethritis. Paraurethral duct dilatation should thus be actively treated to prevent these complications.
Methods
This study was approved by the medical ethics committee of the First People’s Hospital of Changshu (study approval number: csyy1992-1a). All patients provided written informed consent for the study.
Patients
The patients in this study were men with paraurethral duct dilatation following paraurethral duct infection by N. gonorrhoeae. All of them were treated in the First People’s Hospital of Changshu from January 1993 to December 2016.
The inclusion criteria were as follows: The patient initially had local erythematous swelling at the external urethral orifice, with an ostium at its center; pressure caused expression of purulent excretion from the ostium, and N. gonorrhoeae was confirmed as the pathogen; the local erythematous swelling at the external urethral orifice resolved and the purulent excretion disappeared after ceftriaxone therapy, but the ostium remained open and pressure caused the expression of transparent pathogen-free fluid from the ostium; high-frequency ultrasound revealed tubular structures; the ostium did not close during the 3-month follow-up period, and paraurethral duct dilatation was diagnosed; and the patient consented to undergo wedge resection of the paraurethral duct.
Patients with two or more skin lesions, with pathogens in the transparent excretion from the ostium, or who failed to provide written informed consent were excluded from the study.
Clinical Data
The following clinical and demographic characteristics of the patients were recorded: age, marital status, nonmarital sexual contact, disease course (from the occurrence of the secondary dilatation of the paraurethral duct), and clinical manifestations.
Laboratory Tests
Paraurethral duct discharge and urethral discharge specimens were collected separately and examined under a microscope to detect intracellular Gram-negative diplococci within phagocytes. Specimens were also cultured to detect N. gonorrhoeae, Ureaplasma urealyticum, or other bacteria. DNA of gonococci, Chlamydia trachomatis, U. urealyticum, and herpes simplex virus type 1 or 2 were detected by polymerase chain reaction. Venous blood samples were taken and analyzed using a rapid plasma reagin test, Treponema pallidum hemagglutination assay, and human immunodeficiency virus antibody assay. Platelets and the blood coagulation status (including the prothrombin time, international normalized ratio, activated partial thromboplastin time, thrombin time, fibrinogen level, and D-dimer level) were tested.
High-Frequency Ultrasound
High-frequency ultrasound examination was carried out using an ACUSON X300 ultrasound system (Siemens, Germany) with a probe frequency of 7.5 to 10 MHz. The patient lay in the supine position. After disinfection of the glans penis, the probe was covered with a sterile sheath and placed on the lesion at the glans and its surrounding tissues for continuous multi-sectional scanning. The shape, size, orientation, borders, and internal echogenicity of the lesions were determined by two-dimensional ultrasound.
Wedge Resection of the Paraurethral Duct
All patients underwent wedge resection of the paraurethral duct. With the patient in the supine position, the perineum was sterilized with 5% povidone iodine solution over a 15-cm-diameter area centered on the urethral meatus. The glans penis was exposed after spreading a sterilized towel over the area. Wedge resection was carried out following infiltration of 1% lidocaine solution applied 0.5 cm from the lesion borders, and the lesion was excised for a distance of about 1 mm from the left and right sides of the ostium and to a depth slightly greater than the length of the paraurethral duct. Bleeding was stopped by applying pressure, and patients were observed for 2 hr after surgery.
Nursing and Follow-Up
After wedge resection, the patients were requested to wear sterile pants, which were changed on a daily basis. If the wound became contaminated by urine, it was washed with 1/5,000 potassium permanganate solution. Patients visited once a day until the wound healed.
Safety Assessment
The anesthetic method, operation difficulty, surgical duration, intraoperative blood loss, wound healing time, complications, and sequelae were evaluated.
Determination of Intraoperative Blood Loss
Intraoperative blood loss was calculated as the weight of the blood-saturated gauze minus the preoperative weight of the dry gauze. One gram of blood was approximately equivalent to 1 ml.
Evaluation Criteria for Curative Effect
The patient was considered to be cured if the ostium at the external urethral orifice had disappeared, pressure did not cause fluid expression from the ostium site, and no tubular structures were detected by high-frequency ultrasound at the 4-week follow-up visit. The patient’s condition was considered to be unresponsive if the ostium was still present at the external urethral orifice, pressure still caused transparent fluid to be expressed, and tubular structures were still evident on high-frequency ultrasound.
Results
In total, 273 men with paraurethral duct infection by N. gonorrhoeae were diagnosed and treated in the First People’s Hospital of Changshu City from January 1993 to December 2016. All patients were treated with intramuscular ceftriaxone sodium (1 g once daily for 1–5 days). After treatment, paraurethral duct dilatation was reported in 50 patients (18.32%), and 26 patients (age, 19–71 years; mean, 40.73 ± 12.39 years) were enrolled in this study. Twenty-one patients were married and five were unmarried. The mean disease course was 43.69 ± 11.28 days (range, 28–66 days). All 26 men demonstrated a pinhead-like ostium at the external urethral orifice, through which transparent liquid could be expressed with pressure (Figures 1a, 1b). Specimens of discharge from the paraurethral ducts and urethra were collected separately for Gram staining. No Gram-negative intracellular diplococci within phagocytes or trichomonads were identified in any specimens, and all cultures tested negative for N. gonorrhoeae, U. urealyticum, and other bacteria. Polymerase chain reaction analysis of both paraurethral and urethral specimens was negative for N. gonorrhoeae, C. trachomatis, U. urealyticum, and herpes simplex virus types 1 and 2 DNA. The serum rapid plasma reagin, T. pallidum hemagglutination assay, and human immunodeficiency virus antibody test results were also negative in all patients. Platelet and blood coagulation tests were normal.

(a) A pinhead-like ostium was present at the 8 o’clock position on the right side of the external urethral orifice and (b) pressure on the ostium caused expression of clear liquid.
High-frequency ultrasound showed tubular hypoechoic structures with well-defined borders and smooth margins in all patients. One end of the tubules was blind and the other was open to the atmosphere, and the ducts ran parallel to the urethra (Figure 2). The mean diameter and length of the paraurethral ducts before treatment were1.08 ± 0.24 mm (range, 0.7–1.7 mm) and 9.00 ± 1.22 mm (range, 6.6–11.9 mm), respectively.
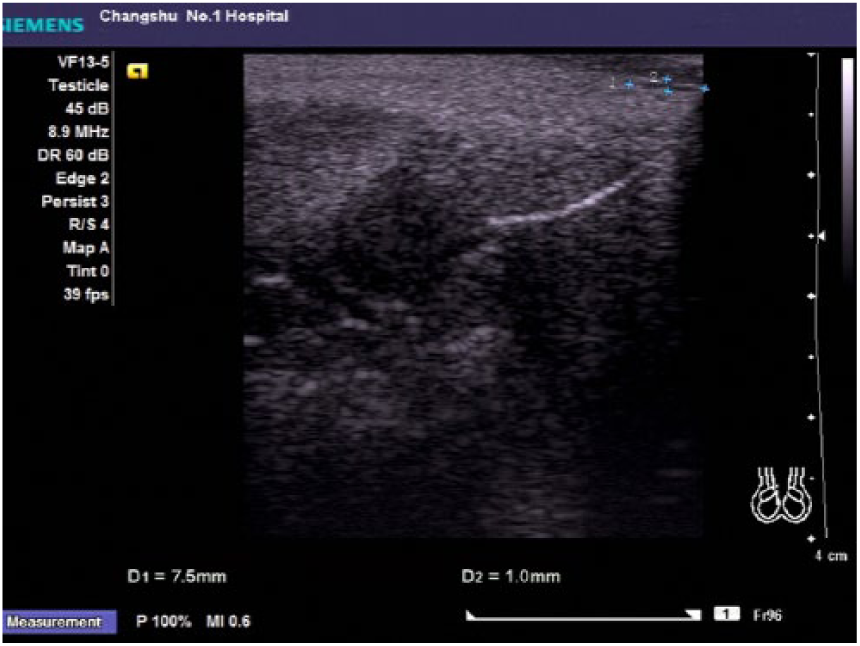
High-frequency ultrasound showed a tubular hypoechoic area with well-defined borders and smooth margins. One end was blind and the other was open to the environment. The duct lay parallel to the urethra. The lumen diameter was 1.0 mm, and the duct length was 7.5 mm.
All 26 men received local infiltration anesthesia. The mean surgical duration (including local disinfection, anesthesia, wedge resection, and hemostasis by compression; Figures 3–5) was 18.65 ± 2.50 min (range, 14–23 min), the mean intraoperative blood loss was 10.50 ± 1.68 ml (range, 8–14 ml), and the mean wound healing time was 14.73 ± 1.31 days (range, 13–17 days). The lesions were cured in 25 of the 26 men (96.15%; Figure 6), but the treatment was ineffective in 1 (3.85%). This patient was cured after a second wedge resection. The postoperative pathological examination revealed a tubular dermal structure that was connected to the epidermis and lined with stratified squamous epithelium, and the peripheral stroma was fibrous and infiltrated by inflammatory cells (Figure 7).

Local infiltration anesthesia.

Wedge resection of the paraurethral duct.

Hemostasis by compression.

The dilated paraurethral duct disappeared after wedge resection.

Epidermal acanthosis, which was connected to a tubular structure in the dermis, was evident. The inner wall of this structure was lined with stratified squamous epithelium, and the peripheral stroma was fibrous and infiltrated by inflammatory cells (hematoxylin–eosin stain, ×100).
The postoperative wounds healed in all patients. No complications or glans defects were observed in any patients.
Discussion
Gonococcal infection of the paraurethral ducts in men was historically called gonococcal inflammation of the paraurethral glands (Fan, 2010; Fan & Zhang, 2012 ; Fan, Zhang, & Fan, 2012; Fan, Zhang, & Jiang, 2014); however, histopathologic examination of such lesions revealed no gland but instead a tubular structure in the dermis connected to the epidermis (Fan et al., 2015). Gonococcal infection of the paraurethral ducts is a more appropriate description than gonococcal inflammation of the paraurethral glands.
Infection of the paraurethral duct by N. gonorrhoeae is a localized complication in male patients with gonorrhea. In total, 14,652 male patients with gonorrhea confirmed both clinically and by laboratory test results were treated in the First People’s Hospital of Changshu from January 1993 to December 2016, and 273 (1.86%) of these patients had accompanying paraurethral duct infection by N. gonorrhoeae. After treatment, paraurethral duct dilatation was identified in 50 patients (18.32%). A prolonged disease course, sexual intercourse during gonorrheal infection, repeated squeezing of the penis, and a redundant prepuce may be risk factors for paraurethral duct infection by N. gonorrhoeae in male patients with gonorrhea (Fan & Zhang, 2012). Risk factors for paraurethral duct dilation secondary to paraurethral duct infection by N. gonorrhoeae may include delayed treatment, age, C. trachomatis infection, and a history of gonococcal infection in the paraurethral duct (Fan, Zhang, Wang, Ye, & Jiang, 2016).
The pathogenesis of paraurethral duct dilatation is likely to involve the entry of N. gonorrhoeae into the opening of the paraurethral ducts, leading to ductal edema, stenosis, and poor drainage and causing the ducts to become dilated. Pus accumulates within the ducts, resulting in abscess formation, and the pus then leaks out of the abscess under pressure. Although N. gonorrhoeae can be eradicated with ceftriaxone, the walls of the fibrotic paraurethral duct become less compliant or even nonelastic and the duct remains unable to close even after resolution of the infection (Fan, Zhang, Wang, Ye, & Jiang, 2016).
The paraurethral ducts are extremely thin and small, and the length of the ducts therefore cannot be measured accurately using a probe. Furthermore, although the biological stain methylene blue can be used to identify the sinus tract (Strevinas, Reid, & McGrouther, 2013), it cannot be used to provide guidance for surgical treatment of the paraurethral ducts because they are too narrow to allow methylene blue to enter. However, the present study has identified that the dilated paraurethral ducts can be visualized using high-frequency ultrasound. This method can be used to provide valuable information on the length, lumen diameter, and direction of the dilated paraurethral ducts before wedge resection (Fan, Zhang, & Ye, 2016).
Wedge resection involves the surgical removal of a triangle-shaped slice of tissue. The procedure may be used to remove a tumor or other abnormal tissue, typically together with a small amount of surrounding normal tissue. Based on high-frequency ultrasound images, wedge resection was performed to remove an inverted triangle of tissue containing the dilated paraurethral duct. This procedure is simple and easy to perform and enables the acquisition of complete pathological data. This site has a rich blood supply, and intraoperative bleeding is unavoidable; thus, the platelet and blood coagulation statuses should be carefully examined before surgery. Hemostasis by compression is usually effective in these patients. Fast-absorbing sutures may cause scars or ostia at the suture site and should therefore be avoided in the wedge resection. The first wedge resection in the present study was successful in 25 of the 26 patients; the one failure was pathologically confirmed to be associated with incomplete resection of the paraurethral duct. This patient was cured after a second wedge resection. The male paraurethral duct is lined with columnar epithelium (Harkness, 1948). Postoperative pathological examination in the current study identified that all paraurethral ducts were lined with squamous epithelium. Therefore, it is possible that the columnar epithelium had been destroyed by the gonococci and replaced by squamous epithelium (Harkness, 1948).
The results of the present study suggest that high-frequency ultrasound imaging–guided wedge resection is an effective and safe treatment for paraurethral duct dilation secondary to paraurethral duct infection by N. gonorrhoeae in men.
Limited by the relative small patients, we could not observe any complication of this surgery. Further study still should pay attention to the possible complications including long-term complications.
Footnotes
Acknowledgements
The authors would like to thank the clinic staff for their participation in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
