Abstract
Gonococcal paraurethral duct infection in males has previously been regarded as a local complication of urethral gonorrhea. To verify this, pathogens were investigated in urethral secretions from 81 male patients with gonococcal paraurethral duct infection. In patients with gonococcal infections of both the urethra and the paraurethral duct, the times of onset of the first symptoms associated with urethral gonorrheal and gonococcal paraurethral duct infection were compared. Among 81 male patients with gonococcal paraurethral duct infection, gonococci were detected in the urethras of 76 patients and no pathogens were detected in the urethras of the remaining 5 patients. The first symptom associated with urethral gonorrhea and gonococcal paraurethral duct infection occurred simultaneously in 10 cases. In 7 cases, the first symptom of gonococcal paraurethral duct infection occurred 2–4 days (2.29 ± 0.76 days) earlier than that of urethral gonorrhea and in 59 cases, the first symptom of urethral gonorrhea occurred 1–6 days (3.07 ± 1.19 days) earlier than that of gonococcal paraurethral duct infection. This study shows that gonococcal paraurethral duct infection in males can be caused by primary infection of Neisseria gonorrhoeae.
The paraurethral ducts in men are small, blind channels lined with columnar epithelium (Harkness, 1948). These ducts run parallel to the terminal part of the urethra for varying distances and open near or within the lips of the external meatus (King & Nicol, 1969). The paraurethral ducts appear to be embryological remnants and are not visible to the naked eye (Gilhooly & Hensle, 1984). Neisseria gonorrhoeae can invade the paraurethral duct via its orifice, resulting in gonococcal inflammation of the paraurethral duct (Fan, 2010; Fan, & Zhang, 2012; Fan, Zhang, & Fan, 2012; Fan, Zhang, & Jiang, 2014; Fan, Zhang, & Ye, 2016; Sherrard, 2014). The inflammation clinically manifests as local erythematous swelling at the external urethral orifice, with a pinhead-like ostium at the center indicating the orifice of the paraurethral duct. Pressure can result in purulent excretion from the ostium. Gonococcal paraurethral duct infection in males has been regarded as a local complication of urethral gonorrhea in the literature (Fan, & Zhang, 2012; Harkness, 1948; King & Nicol, 1969; Sherrard, 2014). To investigate whether this is the case, the present study investigated pathogens in urethral secretions collected from male patients with gonococcal paraurethral duct infection and compared the time of onset of the first symptoms of urethral gonorrhea and gonococcal paraurethral duct infection in male patients with gonococcal infections of both the urethra and the paraurethral duct .
Methods
This study was approved by the Medical Ethics Committee of the First People’s Hospital of Changshu, Changshu Hospital Affiliated to Soochow University (Study approval number: csyy1999-1a). All patients provided written informed consent to participate in the study.
Patients
The patients in this study were men diagnosed with gonococcal paraurethral duct infection. All patients were treated at the First People’s Hospital of Changshu from January 2000 to October 2017. Inclusion criteria were (a) initial local erythematous swelling at the external urethral orifice, with a central ostium; (b) pressure-induced release of purulent excretions from the ostium; (c) N. gonorrhoeae as the confirmed pathogen; (d) no previous history of gonococcal paraurethral duct infection or gonorrhea; and (e) a maximum of one extramarital sexual encounter within 1 month prior to disease onset. Exclusion criteria were (a) pathogens other than N. gonorrhoeae detected in paraurethral duct discharge; (b) the use of antibiotics within 1 month before presentation; and (c) refusal to sign informed consent.
Data Collection
Demographic data including age, occupation, marital status, sexual orientation, sexual behavior pattern, condom use, and prepuce condition were recorded. The following information was also collected: (a) the interval from extramarital coitus to onset of the first symptom of urethral gonorrhea (painful urination, increased urinary frequency or urinary urgency, or urethral pus overflow); and (b) the interval from extramarital coitus to onset of the first symptom of gonococcal paraurethral duct infection (local erythematous swelling at the external urethral orifice, an ostium at the center of local erythematous swelling, or pressure-triggered release of purulent excretions from the ostium). The time of onset of the first symptoms of urethral gonorrhea and gonococcal paraurethral duct infection were compared in male patients with gonococcal infections of both the urethra and the paraurethral duct.
Laboratory Tests
To avoid cross-contamination of discharges from the orifices of the paraurethral duct and the urethras, the penis glans was repeatedly rinsed with normal saline for 3 min. Thereafter, pressure was applied to the external orifice of the urethra and discharge was collected from the orifice of the paraurethral duct using a sterile cotton swab. The glans penis was again repeatedly rinsed with normal saline for 3 min, then a thin cotton urethral swab was inserted 4 cm into the urethra and gently rotated to collect samples of urethral secretions. After Gram staining, microscopy was performed to detect intracellular Gram-negative diplococci within phagocytes. Specimens were also cultured to detect the presence of N. gonorrhoeae, Ureaplasma urealyticum, or other bacteria. The genetic materials of gonococci, Chlamydia trachomatis, U. urealyticum, and herpes simplex virus (HSV) type 1 or 2 were detected using polymerase chain reaction (PCR). Venous blood samples were collected and analyzed using a rapid plasma reagin (RPR) test, Treponema pallidum hemagglutination assay (TPHA), and human immunodeficiency virus antibody assay.
Statistical Analysis
Statistical analysis was performed using R software (R Core Team, Vienna, Austria). In male patients with gonococcal infections of both the urethra and the paraurethral duct, the intervals from extramarital coitus to the onset of the first symptom of urethral gonorrhea and from extramarital coitus to the onset of the first symptom of gonococcal paraurethral duct infection were compared using nonparametric pairwise Mann–Whitney U tests. The correlation of redundant prepuce and the time sequence of the first symptom associated with urethral gonorrhea and gonococcal paraurethral duct infection in male patients with gonococcal infections of both the urethra and the paraurethral duct was compared with the chi-square test. Statistical significance was assumed at p < .05.
Results
Demographic Data
A total of 92 male patients with gonococcal paraurethral duct infection were diagnosed and treated in the First People’s Hospital of Changzhou from January 2000 to October 2017; of these, 81 met the inclusion criteria. The patients enrolled in this study were between 18 and 63 years of age. Their professions included student (4 patients, 4.94%), unemployed (2 patients, 2.47%), short-term employee (25 patients, 30.86%), long-term employee (43 patients, 53.09%), and employer (7 patients, 8.64%). Fifteen patients (18.52%) were married or had been previously married and 66 patients (81.48%) were unmarried. All 81 patients were heterosexual and all admitted to having had extramarital coitus without a condom. The mode of extramarital coitus was genital-to-genital contact for 78 patients (96.30%) and oral-to-genital contact for 3 patients (3.70%). The prepuces of 48 patients (59.26%) were normal; however, redundant prepuce and phimosis were observed in 32 patients (39.51%) and 1 patient (1.23%), respectively.
Clinical Manifestations
In all 81 patients, local swelling and redness was observed at the external urethral orifice, with a pinhead-like ostium at the center, and increased pressure caused purulent excretions from the ostium. Seventy-six patients (93.83%) suffered from urodynia, frequent micturition and urinary urgency, and urethral pus overflow. Five patients (6.17%) had no symptoms of urethritis or urethral pus overflow (Figure 1a and b). In all 81 patients, the interval from extramarital coitus to the onset of the first symptom of gonococcal paraurethral duct infection was 1–10 days (mean: 5.21 ± 1.85 days), and the duration of gonococcal paraurethral duct infection symptoms was 1–9 days (mean: 3.15 ± 1.61 days). In 76 patients (93.83%) with gonococcal infections of both the urethra and the paraurethral duct (Figure 2a and b), the first symptom of urethral gonorrhea and gonococcal paraurethral duct infection occurred simultaneously in 10 patients (13.16%), of whom 4 had redundant prepuce (40%). In 7 patients (9.21%), the first symptom of gonococcal paraurethral duct infection occurred 2–4 days (2.29 ± 0.76 days) earlier than the first symptom of urethral gonorrhea, of whom 3 had redundant prepuce (42.86%), while in 59 patients (77.63%) the first symptom of gonorrhea occurred 1–6 days (3.07 ± 1.19 days) earlier than the first symptom of gonococcal paraurethral duct infection, of whom 26 had redundant prepuce or phimosis (44.07%). In the 76 patients with gonococcal infections of both the urethra and the paraurethral duct, the interval from extramarital coitus to the first symptom of gonococcal paraurethral duct infection was 2.11 days longer than the interval from extramarital coitus to the first symptom of urethral gonorrhea (95% confidence interval: 1.64–2.57 days), and this difference was statistically significant (W = 1997, p < .001). There was no significant difference between redundant prepuce and the time sequence of the first symptom associated with urethral gonorrhea and gonococcal paraurethral duct infection in male patients with gonococcal infections of both the urethra and the paraurethral duct (χ2 = 0.058, p = .97).
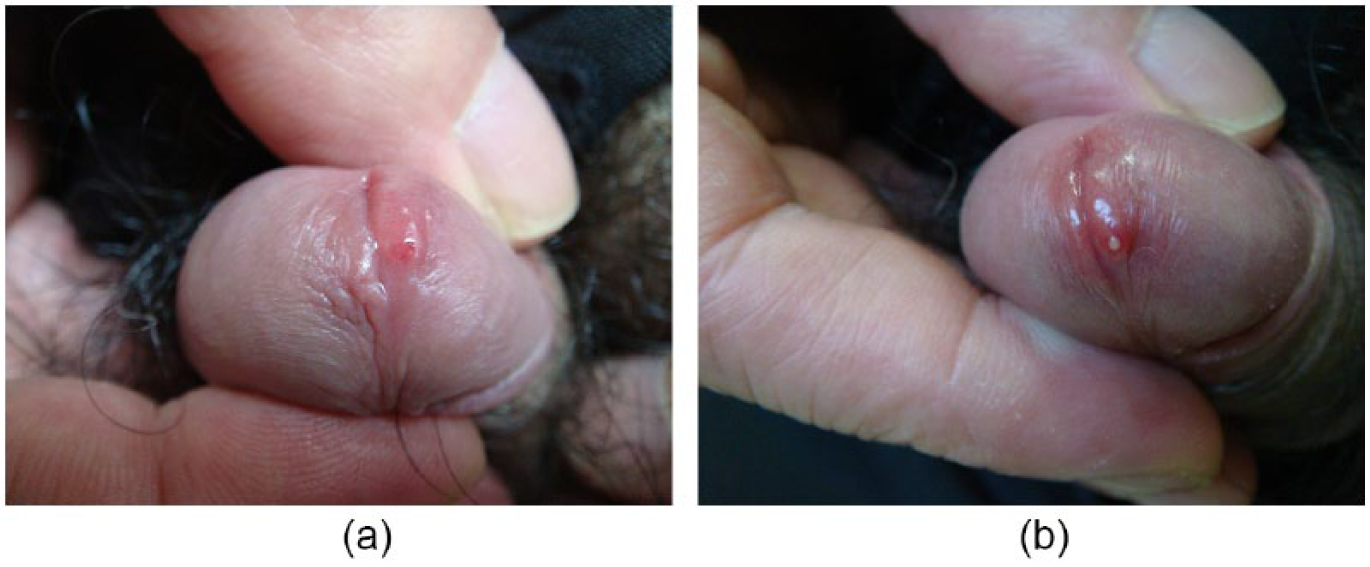
Gonococcal paraurethral duct infection without comorbid urethral gonorrhea.

Gonococcal paraurethral duct infection with comorbid urethral gonorrhea.
Laboratory Findings
Specimens of paraurethral duct discharge from 81 patients and urethral discharge from 76 patients with accompanying symptoms of urethritis were collected separately for Gram staining. Gram-negative intracellular diplococci within phagocytes were observed in these specimens, and cultures and PCR were both positive for N. gonorrhoeae. Urethral discharge specimens of the five patients without symptoms of urethritis showed no Gram-negative intracellular diplococci within phagocytes, and cultures and PCR were both negative for N. gonorrhoeae. All specimens were negative for other bacterial cultures including U. urealyticum, and PCR for DNA of C. trachomatis, U. urealyticum, and HSV types 1 and 2 were also negative. Testing for blood RPR, TPHA, and HIV also showed negative results.
Discussion
Gonorrhea, caused by N. gonorrhoeae, primarily manifests as a purulent infection of the urogenital system but may also cause infections of the eyes, pharynx, and rectum as well as disseminated gonococcal infection. Local complications of male urethral gonorrhea include tysonitis, paraurethral duct infection, periurethral abscess, epididymitis, penile edema, and penile lymphangitis (Sherrard, 2014).
Gonococcal paraurethral duct infection in males was previously known as gonococcal inflammation of the paraurethral glands (Fan, 2010; Fan, & Zhang, 2012; Fan et al., 2014; Fan et al., 2016); however, high-frequency ultrasound of affected patients revealed a lumenlike structure of the lesion (Fan, Zhang, Fan, Ye, & Jiang, 2018), and histopathologic examination of such lesions showed no gland but instead a tubular structure in the dermis connected to the epidermis (Fan et al., 2015). Gonococcal infection of the paraurethral ducts is therefore a more appropriate description than gonococcal inflammation of the paraurethral glands (Fan et al., 2018).
The pathogenic mechanisms of gonococcal paraurethral duct infection in males may be as follows: N. gonorrhoeae enters the paraurethral duct via its orifice and adheres to the surface of the columnar epithelial cells using adherent factors present on their surface pili; subsequently, gonococci enter columnar epithelial cells phagocytosis and reproduce, leading to cell lysis and rupture. The endotoxin and outer membrane lipopolysaccharide of N. gonorrhoeae combined with complement mediators produce chemical toxins and inflammatory infiltrates, inducing neutrophil aggregation and phagocytosis, which cause inflammation of the paraurethral duct. Together, these effectors contribute to ductal edema, stenosis, and poor drainage and cause the ducts to become dilated. Pus accumulates within the ducts, resulting in abscess formation, and the pus then leaks out of the abscess under pressure (Fan, 2010).
Gonococcal paraurethral duct infection in males has been regarded as a local complication of urethral gonorrhea in the literature. A prolonged disease course, sexual intercourse during gonorrhea, repeated squeezing of the penis, and a redundant prepuce are suggested risk factors for paraurethral duct infection by N. gonorrhoeae in male patients with urethral gonorrhea (Fan & Zhang, 2012). N. gonorrhoeae can infect both the urethra and the paraurethral duct when a healthy male has sexual contact with a partner infected with gonorrhea, so gonococcal paraurethral duct infection may be a primary gonococcal infection that occurs when N. gonorrhoeae directly colonizes the paraurethral duct. A gonococcal paraurethral duct infection should be regarded as a local complication of urethral gonorrhea only if subsequent to N. gonorrhoeae colonization of the male urethra (causing urethral gonorrhea), the bacteria in purulent secretions overflowing from the urethral orifice reinfect the paraurethral duct. In a male patient with gonococcal paraurethral duct infection, if N. gonorrhoeae in purulent secretions overflowing from the paraurethral duct reinfects the urethra and causes urethral gonorrhea, the urethral gonorrhea should be regarded as a local complication of gonococcal paraurethral duct infection.
Konrad (1976) has reported the recurrence of gonorrhea caused by gonorrheal paraurethritis in men. In the present study, among 81 male patients with gonococcal paraurethral duct infection, the pathogen was not detected in the urethras of 5 patients (6.17%). All five patients deliberately passed urine immediately after extramarital coitus in an attempt to prevent venereal disease. In these patients, gonococcal paraurethral duct infection should be the primary gonococcal infection. In the 76 patients with gonococcal infections of both the urethra and the paraurethral duct, the first symptom of gonococcal paraurethral duct infection occurred earlier than that of urethral gonorrhea in 7 patients (9.21%), and simultaneously in 10 patients (13.16%). It is highly probable that gonococcal paraurethral duct infection was the primary gonococcal infection in 17 patients (22.37%). The first symptom of urethral gonorrhea occurred earlier than the first symptom of gonococcal paraurethral duct infection in 59 patients (77.63%), and it is highly probable that urethral gonorrhea was the primary gonococcal infection in these 69 (10 + 59) patients (90.79%).
The risk of gonococcal infection after a single exposure is approximately 10% in men and 40% in women (Siracusano & Silvestri, 2014). A previous study inoculated male volunteers with N. gonorrhoeae and found that the incidence of gonorrhea depended on inoculum size (Cohen et al., 1994). Burgess (1971) reported a case of gonococcal tysonitis without urethritis after prophylactic postcoital urination. Based on a review of the literature, he concluded that deliberate postcoital urination might flush N. gonorrhoeae that had entered the urethra during sexual intercourse before they colonized the urethral mucosa and that postcoital urination might be a simple and harmless method to prevent urethritis. In the study, urination may also have prevented the occurrence of urethral gonorrhea in these five patients. By contrast, the paraurethral duct in males is a blind tube; without flushing with urine, N. gonorrhoeae is more likely to colonize and reproduce in the paraurethral duct.
The latency period of the five patients with primary gonococcal paraurethral duct infection in the study was 3–7 days, which is similar to that previously reported for urethral gonorrhea (2–8 days) in men (Annabelle, Vandana, & Stephen, 2008). The incubation periods of gonococcal paraurethral duct infection and urethral gonorrhea overlap, so the first symptoms of gonococcal paraurethral duct infection and urethral gonorrhea may appear on any day within their respective incubation periods. Thus, it was difficult to judge whether gonococcal paraurethral duct infections represented primary infections or a complication of urethral gonorrhea among the 59 patients in whom the first symptom of urethral gonorrhea occurred earlier than that of gonococcal paraurethral duct infection. Similarly, it was difficult to judge whether urethral gonorrhea represented primary infections or a complication of gonococcal paraurethral duct infection among the seven patients in whom the first symptom of gonococcal paraurethral duct infection occurred earlier than that of urethral gonorrhea. Experimental gonococcal infection in male volunteers (Hobbs et al., 2011) may further elucidate the relationships between gonococcal paraurethral duct infection and urethral gonorrhea.
This study showed that gonococcal paraurethral duct infection in males can be caused by primary infection of N. gonorrhoeae. Different doses of ceftriaxone are used to treat uncomplicated gonococcal infections (urethritis, cervicitis, and proctitis), gonorrhea complications, and disseminated gonococcal infections (Bignell & Fitzgerald, 2011; Bignell & Unemo, 2013). An accurate judgment of whether gonococcal paraurethral duct infection represents a primary infection or a complication of urethral gonorrhea can guide the dosing of ceftriaxone. Currently, the optimal dose of ceftriaxone for treating primary gonococcal paraurethral duct infection is being studied.
Footnotes
Acknowledgements
The authors would like to thank the clinic staff for their participation in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
