Abstract
Prostate cancer is the second most common cancer in U.S. men, with higher incidence and mortality among African Americans and those with genetic or familial risk. Despite guidelines promoting early detection, prostate-specific antigen (PSA) screening in primary care remains inconsistent. We assessed PSA screening patterns and the association between risk factors and testing.
A retrospective analysis was conducted on men aged ≥40 seen at primary care clinics from 2020 to 2022. Risk scores were created based on the number of prostate cancer risk factors. Univariate and multivariate analyses assessed associations between the risk scores, PSA screening, and cancer diagnosis. Of 176,326 patients, 31.4% underwent PSA testing, rising to 78.1% with a risk score of 4. The predictive model was significant (χ2 = 19,527.021, df = 11, p < .001). PSA testing odds were higher with Medicaid or commercial insurance (vs. Medicare) and lower for self-pay patients. Former and never smokers were more likely to be tested than current smokers. Age, African American race, and family history increased the odds of receiving PSA testing. Prostate cancer diagnosis reached 25% at a risk score of 5, with significant predictors including older age, African American race, family history, and mention of “prostate” in clinical notes. PSA screening remains underused in primary care, even among high-risk patients. More risk factors were linked to higher testing rates, but African American and low-income groups were under-tested. Provider education and decision-support tools may improve guideline adherence and equity.
Introduction
Prostate cancer is the second most common cancer in men, with an incidence of 114.7 per 100,000 men in the United States (USCS Data Visualizations, n.d.). Men are typically diagnosed with prostate cancer starting in their 40s, and the risk increases to their 70s, from 5.2 per 100,000 men to 688.9 per 100,000 men (USCS Data Visualizations, n.d.). In addition to age, prostate cancer has many risk factors that guide the necessity of screening procedures. African American men have a 64% higher risk of prostate cancer than Caucasian males, and they have an increased risk of prostate cancer-specific mortality (hazard ratio [HR] = 1.30; 95% confidence interval [CI] = 1.23–1.37; p < .001) (Cancer Statistics Review, 1975–2012—Previous Version—SEER Cancer Statistics Review, n.d.; Dess et al., 2019; Hinata & Fujisawa, 2022). Additional risk factors associated with prostate cancer pertain to family history, the presence of germline mutations, urinary problems, smoking, and obesity [Early Detection of Prostate Cancer: AUA/SUO Guideline (2023)—American Urological Association, n.d.; Harmatz et al., 2024; US Preventive Services Task Force et al., 2018]. Screening for prostate cancer is typically initiated by conducting a prostate-specific antigen (PSA) test (Denijs et al., 2024; Tidd-Johnson et al., 2022). PSA-based screening was introduced in the 1980s and initially demonstrated success in the 1990s, significantly reducing prostate cancer-related deaths (Denijs et al., 2024; Tidd-Johnson et al., 2022). This success was short-lived, as increased testing rates resulted in a high number of false positives (Denijs et al., 2024; Tidd-Johnson et al., 2022). Many patients with slow-growing cancers underwent numerous tests with minimal impact on overall survival (Denijs et al., 2024; Tidd-Johnson et al., 2022). This has led to lower rates of PSA testing and increasing mortality from end-stage prostate cancer (Denijs et al., 2024). This issue can be addressed by using PSA-based screening at the primary care level. Primary Care Physicians (PCPs), as the first point of contact, play a crucial role in ensuring the judicious use of PSA-based screening (Denijs et al., 2024).
The guidelines for screening and diagnosis of prostate cancer have been updated periodically to address the current issue of a lack of proper screening and utilization of PSA-based screening, as it is recommended in the United States. The National Comprehensive Cancer Network (NCCN) 2024 guidelines advocate screening for men aged 40 to 75 years at high risk for prostate cancer (Guidelines Detail, n.d.). The high-risk category in this age group includes individuals with any one of the following: African American descent, a family or personal history of cancer (prostate, ovarian, breast, colorectal, endometrial, or pancreatic cancer), or the presence of germline mutations in BRCA2, BRCA1, ATM, CHEK2, PALB2, HOXB13, MLH1, MSH2, MSH6, PMS2, EPCAM, or TP53 (Guidelines Detail, n.d.). Similarly, the American Urological Association (AUA) recommends offering a baseline PSA test to men aged 45 to 50 years and initiating screening as early as 40 to 45 years for individuals who are identified as an increased risk [Early Detection of Prostate Cancer: AUA/SUO Guideline (2023)—American Urological Association, n.d.]. The United States Preventive Services Task Force (USPSTF) recommends that men aged 55 to 69 be engaged in individualized decision-making and informed discussions (grade C recommendation) (US Preventive Services Task Force et al., 2018). Most shared decision-making happens at the primary care level (Estevan-Ortega et al., 2024).
Prostate cancer screening at the primary care level has inconsistencies in the approach to PSA testing, and adherence to updated guidelines is evident (Merrill & Gibbons, 2024). In the United States, only 50% of the primary care providers engaged in shared decision-making with their patients, and less than half of these providers (24%) felt comfortable doing so (Estevan-Ortega et al., 2024). In addition, only 29% of primary care providers engaged in shared decision-making with their African American patients (Merrill & Gibbons, 2024). This could be because the primary care providers perceive PSA testing as less beneficial and time-consuming or are unaware of the latest recommendations. The major medical societies emphasize the importance of PSA-based screening and shared decision-making for patients aged 40 and above. However, a lack of clear and accessible tools to streamline administrative tasks and educate patients on the risks and benefits of PSA testing effectively remains a significant barrier (Morgan et al., 2024).
We aimed to perform a retrospective chart review of male patients aged 40 and older seen in the primary care clinics at a community-based health care system between 2017 and 2022. Our objective was to assess the proportion of patients who underwent PSA screening and determine whether risk factors influenced the likelihood of screening.
Methods
Study Design and Setting
A retrospective chart review was conducted of all male patients aged 40 and older who were seen in any of the primary care clinics at a medium-sized health center in northeast Georgia between January 2020 and December 31, 2022. All patients included in the study had at least one risk factor for prostate cancer and were eligible for PSA testing at the time of their visit. The study was reviewed and received ethical approval by the Institutional Review Board (IRB) at Brenau University.
Data Collection
All data related to the study were collected via chart abstraction utilizing the health system’s EMR EPIC database from January 2020 to December 2022. The data were initially retrieved by the department research analyst, who acts as the department’s IRB honest broker. Once the data were retrieved, the investigators in the study conducted a manual validation of the data to ensure it correctly represented the study’s criteria and that all variables were appropriately flagged before the statistical analysis was conducted. In addition to demographic information, all risk factors including obesity (body mass index [BMI] >30 kg/m2), smoking status at time of visit (never, former, and current), prior cancer history, age, family history of prostate cancer or other cancers, signs and symptoms, history of benign prostate hyperplasia (BPH), prior prostate surgery biopsy, and prior elevated PSA levels were all obtained for the patients included in the study. All demographic information was collected through the patients’ electronic medical records at the time of the encounter collected for this study, and all signs, symptoms, and history of detected risk factors were collected with the use of the International Classification of Disease–Tenth Edition codes, and all recorded PSA screenings were retrieved with the use of Current Procedural Terminology (CPT) codes.
Outcomes
The primary outcome of this study was to identify the relation between the cumulative number of risk factors, identified as risk score, and the probability of receiving PSA screening and having a prostate cancer diagnosis to help provide a level of necessity for evaluating PSA testing based on the individual patient’s cumulative risk factors. In addition, we evaluated individual outcomes as predictors for prostate cancer diagnosis to spread awareness of significant predictors to increase awareness for primary care clinicians.
Statistical Analyses
All baseline demographics and characteristics of the study sample were reported by descriptive statistics. The baseline demographics table was broken down into patients that received PSA testing and those who did not receive PSA testing. All variables were reported as categorical variables and were summarized by frequencies with percentages. A chi-squared analysis was conducted to determine the association between patients receiving PSA testing and a prostate cancer diagnosis by their cumulative risk score, and a multinominal logistic regression was utilized to determine which individual risk factors were predictors for PSA testing and subsequent prostate cancer diagnosis. All statistical tests were performed on IBM SPSS version 29.0 software, and an alpha level of 0.05 was used for all statistical tests.
Results
Study Demographics and Characteristics
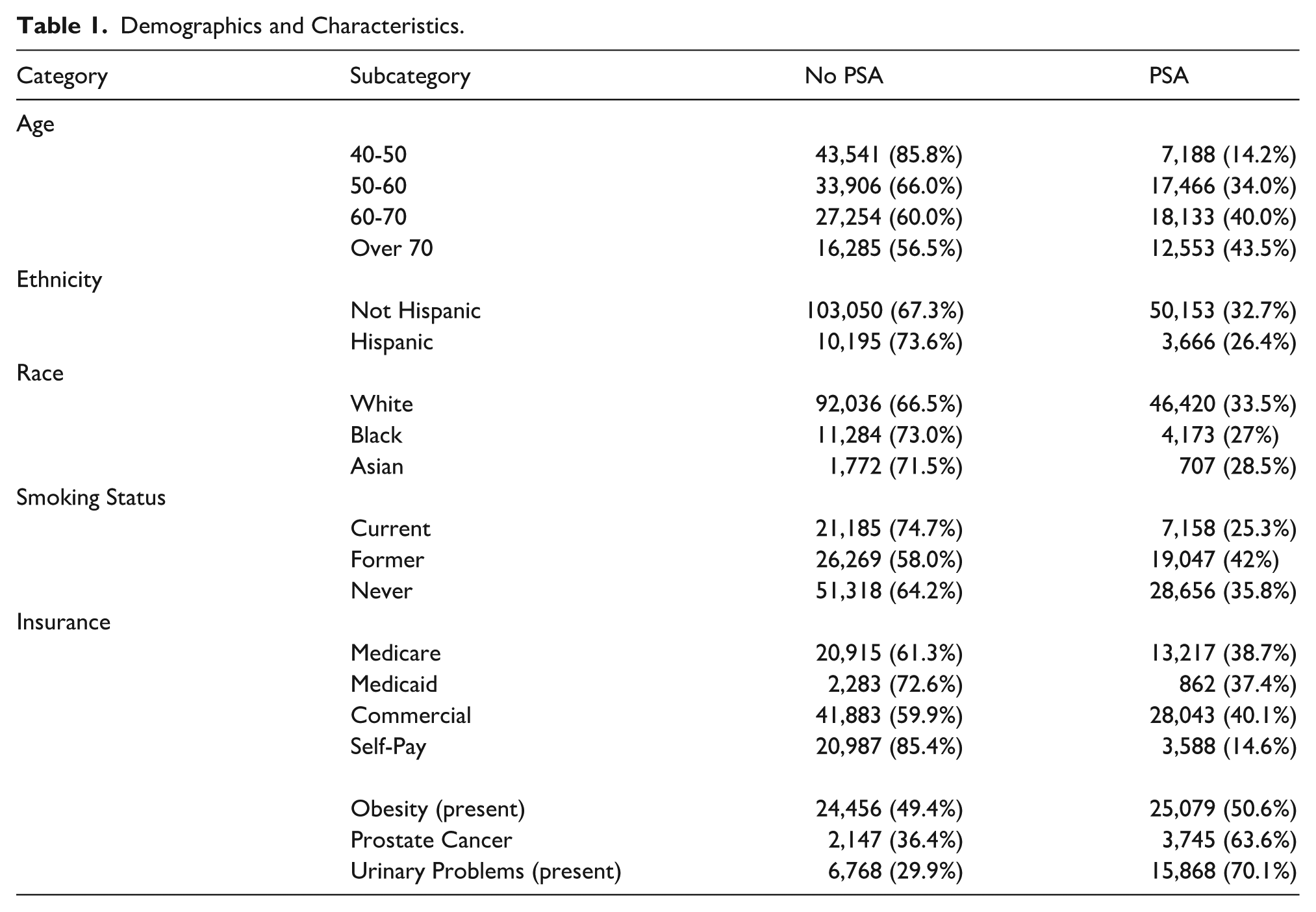
A total of 176,326 male patients with encounters in a rural Georgia health system were identified for the study. Of these patients, 55,340 (31.4%) received PSA testing. Most patients in the study were between 50 and 60 years of age, White, and non-Hispanic who never smoked. All baseline and demographic information of these patients are summarized in Table 1.
Demographics and Characteristics.
Factors Affecting PSA Testing
The model predicting PSA testing is significant (chi-square = 19,527.021, df = 11, p < .001). Medicaid and commercial insurance patients have higher adjusted odds of being tested than Medicare. On the contrary, self-pay customers have lower odds of being tested than Medicare customers. Former smokers and never smokers have higher adjusted odds of being tested than current smokers. Ethnicity was not a significant predictor. However, being Black and having a family history of cancer increased the odds of testing. Finally, age slightly increased the odds of receiving PSA testing (Table 2).
Factors Predicting PSA Testing.

Risk Score and PSA Testing
There is an association between risk score and PSA test (Pearson chi-square = 17,879.128a, df = 5, p < .001). Patients with a risk score less than 2 have a lower prevalence of PSA test. Prevalence of PSA test in patients with risk scores of 2 and over increases, reaching 78.1% for patients with a risk score of 4 (Table 3).
Association between Risk Score and PSA Testing.

Risk Score and Prostate Cancer Diagnosis
There is an association between risk score and prostate cancer (Pearson chi-square = 1,851.069, df = 5, p < .001). Prostate cancer in patients with risk scores below 2 is less prevalent. Only 1.8% of patients with a score of 0 are diagnosed with cancer. The diagnosis becomes more prevalent as the risk increases, reaching 75% for risk score 5 (Table 4).
Total Risk Score and Probability of Prostate Cancer Diagnosis.

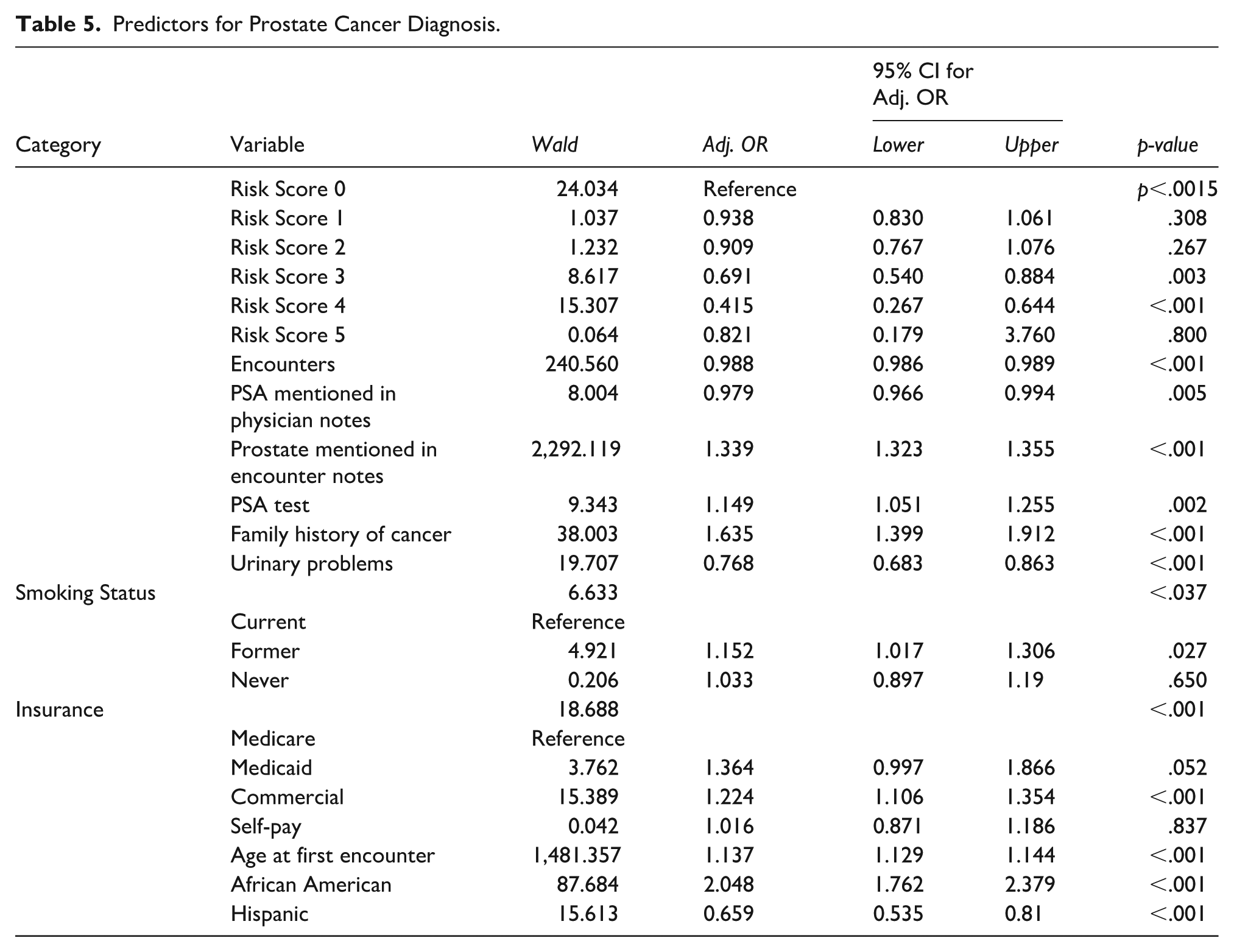
Predictors for Prostate Cancer Diagnosis
Prostate cancer diagnosis can be predicted by Risk Score, PSA mentioned in the notes, number of encounters, prostate mentioned in the notes, and whether or not the patient had a PSA test. In addition, smoking status, insurance, race, ethnicity, age, and family history of cancer diagnosis also predicted prostate cancer diagnosis (chi-square = 11,594, df = 9, p < .001). Specifically, with an increase in risk, adjusted odds ratios for diagnosis remained the same when compared to the reference (Risk Score 0). However, after reaching risks 3 and 4, the odds of being diagnosed with prostate cancer decreased. The diagnosis rates decreased with PSA mentioned in the notes and decreased with an increase in encounters, but when the prostate is mentioned in the notes, the odds of diagnosis increase. Family history of cancer increased the odds of being diagnosed, and so did the age at first encounter. Furthermore, when compared to the current smoker, a former smoker’s odds of being diagnosed increase but were not statistically different from the nonsmoker. African American patients had higher odds of being diagnosed than non-African American patients. Hispanic patients, however, had lower odds of being diagnosed than non-Hispanics. Finally, urinary tract problems decreased the odds of diagnosis (Table 5).
Predictors for Prostate Cancer Diagnosis.
Discussion
The primary objective of our study was to evaluate PSA-based screening practices in the rural primary care setting in the United States and to determine the association between screening tests and the presence of risk factors. Our findings revealed PSA screening rates declined from 85.8% in the 40–50 age group to 60% in the 60–70 age group in primary care practices. PSA testing is essential to reduce the disease burden of prostate cancer, and the declining testing rates likely reflect a lack of awareness of updated guidelines in primary care practices (Garraway et al., 2024; Merrill & Gibbons, 2024; Nguyen et al., 2024). Monitoring PSA trends over time can facilitate the early and accurate diagnosis of prostate cancer (Sajadi Kaboudi et al., 2024). Patients with a PCP generally have a 71% increased likelihood of undergoing PSA screening compared to those without a PCP (Iyer et al., 2024). Despite having a PCP, the decreasing rates of PSA screening are alarming. The reduced prevalence of PSA screening could also be attributed to a lack of awareness among the rural population of Georgia regarding the importance of undergoing testing.
Our findings point out that prostate cancer increased from 11% with four risk factors to 75% with five risk factors. This shows that PSA screening should be done annually in patients with high-risk factors, as the incidence of high-grade tumors and de novo metastatic prostate cancer has been rising in recent years (Merrill & Gibbons, 2024). We identified that patients who had prostate cancer mentioned in their notes had an increased prostate cancer diagnosis, favoring the need for surveillance. African Americans face a heightened risk of prostate cancer with a younger age of diagnosis and more aggressive tumors compared to American White men, and annual health screening is particularly recommended in this population (Merrill & Gibbons, 2024). We also identified that African American ethnicity and a family history of cancer were important factors that have led to increased testing. The 2018 USPSTF update emphasized the importance of shared decision-making for PSA screening; however, screening rates have declined. Following this update, African Americans were found to have 32% lower odds of undergoing PSA screening compared to American Whites (Sajadi Kaboudi et al., 2024; Tidd-Johnson et al., 2022).
In 2020, Medicaid coverage was extended to childless adults earning <138% of the federal poverty level. However, no long-term effect on PSA screening prevalence was observed in the United States (Lucas et al., 2024). In our study, we found that Medicaid and commercial insurance patients had increased odds of undergoing PSA screening compared to Medicare patients and decreased likelihood of testing with self-pay patients. Cancer campaigns that raise awareness and provide support are essential for increasing testing (ZERO Prostate Cancer Connects People to Lifesaving Support, n.d.).
Our study also had some limitations. We were unable to determine whether PSA screening led to changes in the diagnosis of early-stage tumors or contributed to a reduction in the identification of late-stage tumors. Given that our study was conducted retrospectively between 2017 and 2022, larger studies are needed to explore these aspects and evaluate changes in prostate cancer-specific mortality. In addition, our research was conducted within primary care offices in rural Georgia, so the findings may not be generalizable to urban populations or applicable nationally. Furthermore, as our study relied on chart review, we could not account for potential coding errors or missing information in the charts. However, we implemented a validation procedure on 10% of the sample to minimize the impact of such errors and ensure consistency in our findings.
Future studies should adopt a robust multicentric approach to comprehensively evaluate the barriers to PSA testing from the perspectives of physicians. Research should also aim to assess the level of awareness among both patients and health care providers regarding the risk factors associated with prostate cancer. This will contribute to the development of improved strategies for prostate cancer prevention and management.
Conclusion
Prostate cancer is a prevalent form of cancer that affects many men. Shared decision-making remains the cornerstone in guiding whether to screen for prostate cancer. However, discussions regarding the importance of said need for a screen are paramount in forming shared decision-making. It remains essential that patients are appropriately screened for prostate cancer when they present with risk factors that are associated with prostate cancer development. Currently, many patients who would benefit from PSA screening are not being appropriately counseled and, therefore, are unable to make an informed decision on screening that could impact their health. Both patients and PCPs must be aware of the risk factors that contribute to prostate cancer, so that shared decision-making can be made appropriately regarding whether prostate cancer screening is warranted.
Footnotes
Acknowledgements
The authors want to extend our sincere gratitude to Dr. Louise Jones for her leadership of Graduate Medical Education research and support of this project through each of the phases.
Ethical Approval
This study was approved by the Institutional Review Board at Brenau University.
Informed Consent
Informed consent was not required, as this was a retrospective chart review using de-identified data.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon request.
