Abstract
The aim of the study was to analyze population-based prostate cancer (PCa) screening and the incidence of PCa among males ≥50 years of age residing in the Luqiao district of Taizhou, China. From October to December 2020, male residents ≥50 years of age were screened for serum total prostate-specific antigen (total-PSA). If t-PSA re-test levels persisted above 4 μg/L, subjects underwent further noninvasive examinations, including digital rectal examination or multiparameter magnetic resonance imaging (mpMRI) of the prostate. Subjects underwent prostate biopsy of pathological tissue based on t-PSA and mpMRI results. A total of 3524 (49.1%) residents participated in this PCa screening study. In total, 285 (8.1%) subjects exhibited t-PSA levels ≥4.0 μg/L and 112 (3.2%) underwent noninvasive examinations. Forty-two (1.2%) residents underwent prostate biopsy, of which 16 (0.45%) were diagnosed with PCa. Of those diagnosed with PCa, three (19%) had localized PCa (cT1-cT2N0M0), six (37%) had locally advanced PCa (cT3a- cT4N0-1M0), and seven (44%) had advanced metastatic PCa (M1). Unfortunately, 3477 (48.5%) residents did not participate in the study, mainly due to lack of awareness of PCa based on feedback from local health centers. Age and t-PSA were used as primary screening indicators and, when further combined with mpMRI and prostate biopsy, confirmed the diagnosis of PCa among participating residents. Although this was a relatively economical and convenient screening method, education and knowledge should be further enhanced to increase the participation rate in PCa screening programs.
Keywords
Introduction
Prostate cancer (PCa) is one of the most common malignant tumors among elderly males in China. In 2015, the national tumor incidence report revealed that the incidence of PCa among Chinese males reached 10.23/100,000 population and the mortality rate was 4.36/100,000 and, moreover, that its incidence was increasing (Fu et al., 2020). Some studies and surveys have reported that two thirds of PCa patients in China are in the advanced stages of disease at the time of consultation, and the incidence of PCa is increasing over time (Fu et al., 2020; Liu et al., 2019). The survival of patients with PCa is influenced by many factors. The 5-year relative survival rate of patients with limited PCa or with only regional spread of PCa was up to 100%; however, the 5-year survival rate among those with distant metastases was only 29.3% (Ma et al., 2008). There is no established model for PCa screening in China. A meta-analysis revealed that a cut-off value of >4 μg/L for prostate-specific antigen (PSA) for detecting PCa in the Chinese population has high sensitivity but low specificity, and that PCa screening should be further based on PSA levels (B. H. Wang et al., 2020). To this end, we used age and total-PSA (t-PSA) as the main indicators to conduct PCa screening among local male residents of Luqiao, Taizhou, China. The aim was to investigate population-based PCa screening and to analyze the incidence of PCa among male residents ≥50 years of age.
Method
Study Population
The present study included 7173 males ≥50 years of age who resided in the Zhejiang province, China (Figure 1). The protocol was supported by the government (Taizhou Luqiao District Health Committee) and approved by the Ethics Committee of Taizhou Hospital (NO. K20210402). Included subjects were asked to sign an informed consent before undergoing serum t-PSA testing. Considering that t-PSA screening may result in medical overdiagnosis and overtreatment, the risks and benefits of prostate screening were detailed in the informed consent form, in accordance with the requirements of Consensus of Chinese Experts on Prostate Cancer Screening (Prostate Cancer Working Group of Genitourinary Cancer Committee in Chinese Anti-Cancer Association, 2017) at the beginning of the study.

Presentation of PCa Screening Data in This Study
Study Protocol
Exclusion Criteria
Serum t-PSA levels are influenced by many factors, including enlarged prostate volume, lower urinary tract symptoms, acute urinary retention, medications, and prostate inflammation. Taking into account the consensus of experts and the experience of published studies (Bernal-Soriano et al., 2019; Merriel et al., 2018; Palsdottir et al., 2019; Prostate Cancer Working Group of Genitourinary Cancer Committee in Chinese Anti-Cancer Association, 2017), several exclusion criteria were defined, including poor health and life expectancy <10 years, use of 5-alpha-reductase inhibitors within the previous 6 months, recent urinary tract infections or urological procedures, acute urinary retention, and acute or chronic bacterial or nonbacterial prostatitis within the past 6 weeks. Ultimately, 172 subjects were excluded from the study.
Screening Process for PCa
First, the local government counted the number of eligible male residents ≥50 years of age and sent the statistics to the local health centers. Second, subjects included in this study were scheduled to be screened at the local health center. Third, each subject underwent a serum t-PSA test, and name, age, contact information, urinary history, and relevant family history were recorded. Primary care physicians would be required to administer counseling and answer questions. Fourth, information from each resident was kept confidential and the result of serum testing was sent individually by mail to the subject or family members. Finally, the team maintained contact with the directors of health centers through “WeChat” or telephone. For subjects with t-PSA levels ≥4 μg/L, the director informed them of further examination within 1 week (Figure 2).

Flowchart of Prostate Cancer Screening
Serum t-PSA Protocol
Blood samples were tested according to protocol in the clinical laboratory of the authors’ medical center. The immunoassay instrument and assay kit used for testing were acquired from Beckman Coulter (Brea, CA, USA), and operation of the instrument was performed by trained personnel in strict accordance with manufacturer’s instructions.
Noninvasive Examination
Digital rectal examination is a simple and effective method and has been widely used in clinical practice and, moreover, in combination with PSA, can improve the detection rate of early PCa (Ilic et al., 2013). Because PSA levels can be affected by digital rectal examination, only residents with t-PSA levels ≥4 μg/L underwent palpation of the prostate in this screening at the beginning of the study. The specificity of ultrasound or computed tomography (CT) examination for the early diagnosis of PCa is not high (De Visschere et al., 2010; Klotz et al., 2021). Ultrasound and CT were mainly used to rule out other comorbid diseases such as bladder stones, urethral stones, and bladder tumors. It has been reported that multiparameter magnetic resonance imaging (mpMRI) and the prostate imaging reporting and data system (PI-RADS) can be used to localize, diagnose, and assess the risk for PCa, and has an important role in clinical practice (Ahmed et al., 2017; Hötker & Donati, 2021; Weinreb et al., 2016). PI-RADS (Version 2) score mainly referred to the quantitative evaluation of prostate magnetic resonance imaging (MRI) after comparison of different parameters and comprehensive analysis. According to PI-RADS, each patient was scored with a total score of 5 points. When the score was 1 or 2, the patient was less likely to have prostate cancer, whereas when the score ≥ 3, the patient might have prostate cancer. A PI-RADS score ≥3 was used as the cut-off value. Enrolled residents were required to undergo 3.0 Tesla mpMRI of the prostate before prostate biopsy.
Transperineal Biopsy of the Prostate
Although transperineal prostate biopsy did not require intestinal cleaning and had relatively few complications, it required linear rectal probe or biplanar probe, and had high requirements for operator experience. At present, transrectal ultrasound-guided (TRUS) transperineal biopsy has become the main development trend of prostate biopsy. Subjects who agreed to undergo invasive examination underwent systematic perineal puncture (12 needles) performed by a senior urologist. Prostate biopsies of subjects were performed under local anesthesia and the same puncture needle was used intraoperatively.
TNM Staging and Pathological Grading
According to TNM staging, cT1 ~ cT2N0M0 was limited PCa, cT3a ~ cT4N0 ~1M0 was locally advanced PCa, and M1 was advanced metastatic PCa. Pathology was graded and grouped according to the World Health Organization/International Society of Urological Pathology system. Limited PCa was classified as low, intermediate, and high risk according to D’Amico risk stratification.
Treatment and Follow-Up
Treatment options for PCa included close observation, surgery, drugs, and radiation. Patients diagnosed with PCa were followed up for 1 year.
Statistical Analysis
Measurement data are expressed as median (range) and count data are expressed as percentage.
Results
Subject Characteristics
From October to December 2020, 3,524 (49.1%) male subjects participated in the present screening study. The median age of the 3,524 subjects screened was 65 (range = 50–83) years; Figure 3), with subjects distributed across age groups as follows: 50 to 59 (n = 759 [21.5%]), 60 to 69 (n = 2,029 [57.5%]), 70 to 79 (n = 637 [18.1%]), and ≥80 years (n = 63 [1.9%]). Median (range) t-PSA levels among these age groups were as follows: 1.47 μg/L (0.11–57.6 μg/L), 2.01 μg/L (0.16–121.3 μg/L), 1.94 μg/L (0.23–115.8 μg/L), and 2.3 μg/L (0.7–216.6 μg/L), respectively (Table 1). In total, 3,477 (48.5%) residents did not provide informed consent and, as such, did not participate in this study. The directors of each health center communicated with these 3,477 residents by telephone and summarized the major reasons for nonparticipation (Table 2).

Age Distribution of the Residents Who Participated in the Screening
Age-Related Distribution of the Serum t-PSA Level in the 3,524 Male Residents

Note. t-PSA = total prostate-specific antigen.
Major Reasons of 3477 Residents Who Did Not Participate in Screening

PCa Screening Results
Of the 3,524 subjects included in the study, 285 (8.1%) had t-PSA levels ≥4.0 μg/L and 112 (3.2%) underwent noninvasive examination. Sixty-two (1.8%) subjects with t-PSA levels between 4 and 10 μg/L and PI-RADS scores <3 were referred to follow-up. Forty-two (1.2%) subjects underwent prostate biopsy and 16 (0.45%) had a confirmed diagnosis of PCa. The 42 subjects had no postoperative fever, urinary frequency, or urinary urgency after prostate biopsy; however, 35 experienced postoperative hematuria, which improved after observation or administration of finasteride. Eight (0.2%) subjects refused to undergo prostate biopsy, among whom five (0.01%) feared complications associated with puncture such as bleeding, pain, infection, long hospitalization, and/or financial constraints. The remaining three (0.08%) subjects had doubted the results of the noninvasive examination and visited a higher level hospital for further consultation at that time. Nevertheless, contact has been maintained with these patients.
Characteristics of Subjects With Confirmed PCa
Pathological data were available for all 16 patients diagnosed with PCa (Table 3). The incidence of PCa in the 50 to 59, 60 to 69, 70 to 79, and ≥80 years’ age groups was 0.1% (1/795), 0.2% (4/2,029), 1.1% (7/637), and 6.3% (4/63), respectively. All pathological types were adenocarcinoma of the prostate. There were three (19%) cases of limited PCa (cT1 to cT2 N0 M0), six (37%) of locally advanced PCa (cT3aÿ§cT4 N0ÿ§1 M0), and seven (44%) of advanced metastatic PCa (M1) among patients with confirmed diagnosis. There was one (78%) case of low-risk and two (22%) cases of intermediate-risk in limited PCa.
Clinical Stages and Pathological Grades of PCa Confirmed in Different Age Groups of the 16 Males
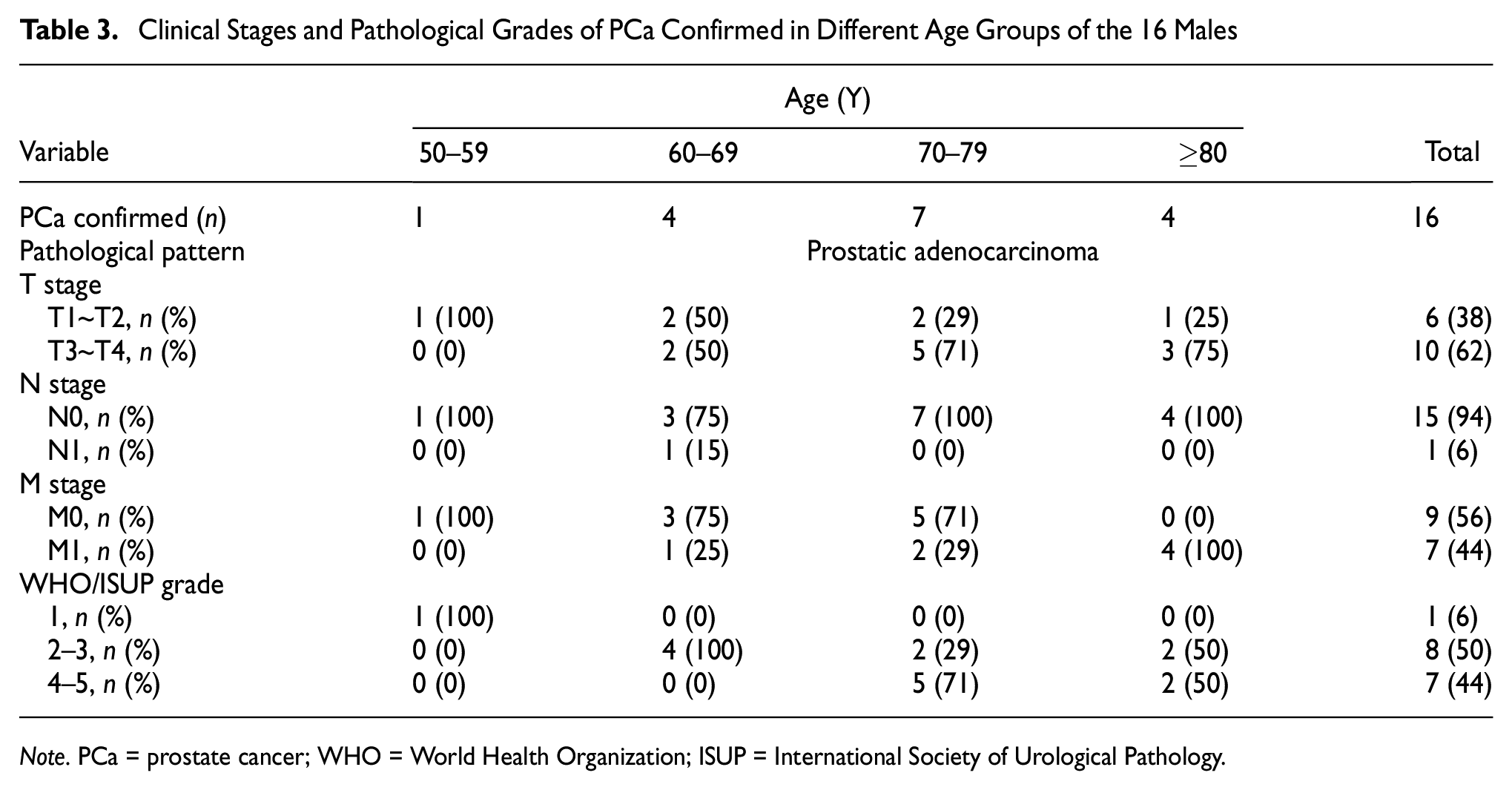
Note. PCa = prostate cancer; WHO = World Health Organization; ISUP = International Society of Urological Pathology.
Combined with estimation of life expectancy, four (25%) residents with locally advanced PCa underwent laparoscopic radical prostatectomy and lymph node dissection. One (6%) subject with low-risk limited PCa underwent laparoscopic radical prostatectomy. Two (13%) subjects with intermediate-risk limited PCa underwent implantation of I-125 particles and endocrine therapy. Seven (44%) subjects with bone metastatic PCa were receiving zoledronic acid as an anti-bone metastatic therapy. Nine (56%) subjects underwent endocrine therapy, including the seven previously mentioned subjects who were currently undergoing treatment for bone metastases.
Outpatient Follow-Up in Patients Diagnosed With PCa
Patients who underwent surgery all had low PSA levels and did not experience severe urinary incontinence after 1 year of follow-up. Patients who underwent I-125 particle implantation and endocrine therapy also had low PSA levels after 1 year of follow-up. However, of patients with bone metastases, four developed metastatic castration-resistant PCa. These four patients had agreed to undergo new endocrine therapy or chemotherapy, with the latest PSA level <2 μg/L at follow-up. The remaining patients who underwent endocrine therapy alone or combination therapy all had low current PSA levels and had not developed metastatic castration-resistant PCa or non-metastatic castration-resistant PCa.
Discussion
Clinical Implications
PSA is a serine protease produced by prostate epithelial cells and is secreted directly into the prostatic ductal system. Because prostate tissue has a natural barrier to high levels of PSA, serum levels of PSA are normally very low (Rao et al., 2008). However, if prostate cells rupture, large amounts of PSA can enter the serum. Serum PSA exists in the form of free PSA (f-PSA) and t-PSA. Through a review of the literature addressing PCa screening, we found that the incidence of PCa varied over time, age, race, and screening methods (Table 4). A systematic review and meta-analysis of the accuracy of mpMRI for the diagnosis of PCa reported that the sensitivity and specificity of mpMRI were 0.87 (95% confidence interval [CI] = [0.81, 0.91]) and 0.68 (95% CI = [0.56, 0.79]; Zhen et al., 2019). In the present study, we initially used t-PSA and age as the primary screening indicators and mpMRI and prostate biopsy for subsequent screening, which reduced the need for blind prostate biopsy and enabled targeted observation of selected residents. Therefore, mpMRI represented an important auxiliary tool in PCa screening.
The Incidence of Prostate Cancer Varied With Different Time, Age, Race, and Screening Methods

Note. t-PSA = total prostate-specific antigen; PCa = prostate cancer; DRE = digital rectal examination; TRUS = transrectal ultrasound-guided; mpMRI = multiparameter magnetic resonance imaging.
Although the Taizhou and Nanjing areas are on the east coast of China, the PCa detection rate (0.45% [16/3,524]) in this study was different from previously reported results (1.14% [79/6,903]) of PCa screening by Liang and other Chinese investigators in Nanjing, China (Liang et al., 2019). Unfortunately, of the 16 residents who have been diagnosed, we also found one patient with pathologically confirmed intermediate-risk limited PCa between 50 and 59 years of age and one with advanced metastatic PCa between 60 and 69 years of age. This may also provide us with clues about the increasing incidence of PCa in younger individuals and the importance of early screening. Data from the European Randomized Study of Screening for Prostate Cancer (ERSPC) revealed that PSA screening resulted in a significant 20% reduction in mortality from PCa (Schröder et al., 2014). However, the Prostate, Lung, Colorectal and Ovarian Cancer (PLCO) Screening trial in the United States revealed an increased incidence of cancer in the screening group, but no cancer-specific mortality benefit from PSA screening after 13 years of follow-up (Andriole et al., 2012). However, it is important to consider differences in economy, culture, and medical health care level across different regions. Presently, China’s medical health care level remains lagging behind that of Europe and the United States. Only three cases of early localized PCa were detected in our study, and the proportion of early localized PCa still needs to be improved.
For surgical treatment, Bill-Axelson et al. (2018) reported that the tumor-specific survival rate of limited, medium-low risk patients after radical prostatectomy reached 80.4% at 23 years. Radical prostatectomy may be the first choice of treatment for these patients. Wallis et al. (2016) reported that among patients with localized PCa, there was no significant difference in 10-year overall survival rate between radical prostatectomy and radical radiotherapy. However, we found four patients with localized PCa, who received internal radiotherapy and endocrine therapy for fear of urinary incontinence and other complications after radical prostatectomy. Many studies have reported that comprehensive treatment with radical prostatectomy as the main treatment for locally advanced PCa could yield good survival benefits to patients (Costello, 2020; Marra et al., 2020; Pansadoro & Brassetti, 2019). In our study, four patients with locally advanced PCa, who underwent radical prostatectomy and lymphadenectomy, were currently undergoing outpatient follow-up for PSA. Adjuvant therapy is recommended if the indicator of PSA rebounds or metastases occurs.
Clinical Practice
We found that 3,477 (48.5%) age-eligible residents in the Luqiao district of Taizhou, China, did not participate in this PCa screening study. This means that there remains a large number of residents with PCa who were not diagnosed in this study. We received some feedback from the local health center directors and found that more than one half of the residents did not participate in screening because they were not aware of PCa. Mobilizing the local population to actively participate in PCa screening was an issue in this study that warranted attention. Durkin et al. (2020) reported that mass media campaigns increased participation in bowel cancer screening. Hoffmeister et al. (2017) reported that targeted invitations significantly increased participation in cancer screening. A systematic review and meta-analysis of rectal cancer screening suggested that the decision to participate in colorectal cancer screening depends on individual perceptions of colorectal cancer screening (Honein-AbouHaidar et al., 2016). Factors mediating awareness included public education to address misconceptions, primary care physician efforts to recommend screening, and the influence of friends and family (Honein-AbouHaidar et al., 2016; Wilkes et al., 2013). Perhaps compliance and actual participation in PCa screening can be improved when residents’ awareness shifts from passive to active screening.
Clinical Suggestions
Establishment of a Professional Team
There were 356 (10%) male residents who did not participate in the screening and reported that they did not receive easy-to-understand answers to their questions on site. This may be because primary care physicians do not have specialized knowledge of PCa and lack awareness of early screening. A randomized controlled study reported that primary care physicians have the ability to screen for PCa. A randomized controlled study reported that primary care physicians’ education and patient activation had a significant impact on their attitude toward screening (Wilkes et al., 2013). We need to make some changes to this current screening model, such as establishing a professional team to regularly train primary care physicians to improve their knowledge and sending urologists to conduct on-site consultations.
Painless Transperineal Prostate Biopsy
Regardless of the type of local prostate anesthesia used, it would—more or less—result in discomfort, including pain and anxiety, among patients (Hizli et al., 2015; Packiam et al., 2018). Five patients in our study declined the invasive procedure due to fear of complications associated with prostate biopsy. Therefore, it was important to provide effective and even painless control for prostate biopsy. For patients who did not want to undergo prostate biopsy with local anesthesia, transperineal prostate biopsy combined with general anesthesia may be a good option to reduce their concerns. Although TRUS prostate biopsy is a standard procedure for the diagnosis of PCa (Fabiani et al., 2016). In terms of complications, transperineal prostate biopsy—compared with transrectal prostate biopsy—had obvious advantages in reducing the incidence of infection and patients did not need to worry about intestinal bleeding (Borkowetz et al., 2018; Saito et al., 2017). In addition, Borkowetz et al. (2018) reported that the targeted puncture of MRI/ultrasonography fusion could significantly improve the detection rate of PCa and reduce the number of unnecessary needle punctures. In this study, our hospital did not have such dedicated equipment; as such, the 26 patients with abnormal PSA levels but negative biopsy results might have PCa.
Combination of Artificial Intelligence
Applying PSA or combining it with an advanced technology in PCa screening warrants serious consideration. Follow-up and observation are needed to determine whether screening models that include advanced technology and equipment, combined with new drugs or combination therapies, can reduce PCa-related mortality. Distler et al. (2017) reported that using PSA density combined with mpMRI improved the negative predictive value of PI-RADS scoring. In this study, a total of 82 residents did not undergo prostate biopsy but were recommended for follow-up with t-PSA and PI-RADS. Artificial intelligence is gradually changing the traditional medical model, which is the direction and trend of human medical development in the future. Reda et al. (2018) developed an artificial intelligence system based on MRI and PSA test results, which could avoid unnecessary prostate biopsies, reduce diagnostic costs, and avoid complications of diagnostic methods.
Limitations
A previous study reported that biopsy-detected PCa, including high-grade cancers, was not rare among men with PSA levels ≤4.0 ng/mL—levels generally believed to be in the normal range (Thompson et al., 2004). Therefore, there were some residents in whom PCa was undoubtedly missed in our screening model. Consensus among Chinese PCa experts suggested that serum PSA testing should be performed as early as possible for groups at high risk of PCa, including males >50 years of age, males >45 years of age with a family history of PCa, males >40 years of age with PSA levels > 1 μg/L, and males >40 years of age with BRCA2 (breast cancer 2) gene mutation (Prostate Cancer Group, Urogenital Tumor Professional Committee, Chinese Anti-cancer Association, 2017). However, a previous study (Fu et al., 2020) showed that the incidence of prostate cancer was not high in Chinese male residents below the age of 50, and the financial resources of this study were limited. The baseline (i.e., minimum) age of the subjects in this study was 50 years; as such, high-risk men <50 years of age were not screened for PCa. Finally, in this study, prostate biopsy was not recommended for subjects with t-PSA levels between 4 and 10 μg/L and PI-RADS < 3, in whom a diagnosis of PCa may have been missed. This study took place between October 2020 and December 2020. In analyzing why residents were not screened, we did not take into account that residents were unable or unwilling to be screened due to fear of Corona Virus Disease 2019 (COVID-19), lockdowns, social distancing, and other reasons. In addition, this study has limited generalizability outside of similar districts in China. In particular, it will have very little generalizability to countries with established screening guidelines and programs.
Conclusion
In this study, age and t-PSA were used as primary screening indicators, which were further combined with mpMRI and prostate biopsy to diagnose PCa. The detection rate of PCa in this study was 0.45% (16/3524); however, the actual incidence may be higher. Although this was a relatively effective and convenient screening mode, 3,477 (48.5%) age-eligible residents did not participate in this PCa screening study; as such, further enhancing public education and knowledge to increase resident awareness and participation in PCa screening programs should be promoted.
Footnotes
Acknowledgements
We thank the government and the hospital team for their support of this study.
Ethics Approval and Consent to Participate
Our hospital ethics committees (Taizhou Hospital of Zhejiang Province affiliated to Wenzhou Medical University, China) reviewed and approved this study (NO. K20210402). The principles of the Helsinki Declaration were followed in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in the scientific research fund of the Taizhou Enze Medical Center Group (No. 2021EZZDC04)
