Abstract
Age-related decline in dehydroepiandrosterone (DHEA) and its sulfate metabolite, dehydroepiandrosterone-sulfate (DHEAS), affects steroid synthesis in men. DHEA(S) acts as a direct neurosteroid and weak androgen and is the unique steroid unaffected by dutasteride’s inhibitory effect on 5α reductases. This study examined the relationship between dutasteride’s side effects, specifically erectile dysfunction (ED) and mood disorders, and the age-related decline in DHEAS. The study included 250 patients with benign prostatic hyperplasia (BPH), divided into two age groups (<60 and ≥60 years), and treated with tamsulosin or tamsulosin plus dutasteride. DHEAS levels were measured, and patients were assessed for ED and mood disorders using standardized questionnaires. Some patients experienced worsening ED and mood disorders during the 6-month follow-up period. The study found a correlation between DHEAS levels and erectile function and mood status in both age groups receiving dutasteride combination therapy (p < .05). Regression analyses showed that DHEAS was a positive predictor for ED in older patients treated with tamsulosin (p < .001) and in both age groups receiving dutasteride combination therapy (p < .001). In addition, DHEAS was a significant negative predictor for mood changes in both age groups receiving combination therapy (p < .001). This study suggests that baseline DHEAS levels can predict changes in erectile function and mood status in BPH patients treated with dutasteride therapy. It is, therefore, recommended to perform DHEAS assessment before starting dutasteride treatment in order to reduce the risk of ED and mood disorders.
Keywords
Introduction
The pharmacological treatment of benign prostatic hyperplasia (BPH) relies on two medication groups: alpha-adrenergic blockers (tamsulosin) and 5α-reductase inhibitors (dutasteride) (Gravas et al., 2023). Combining these medications has been shown to reduce the risk of BPH progression compared to monotherapy with tamsulosin. (Roehrborn et al., 2010). Dutasteride acts by blocking the conversion of testosterone (TT) to dihydrotestosterone (DHT), which is essential for prostate function. By inhibiting both type 1 and type 2 5α-reductase enzymes (5ARIs), dutasteride effectively lowers DHT levels in the body (Roehrborn et al., 2002), which decreases the prostatic volume (Miernik & Gratzke, 2020). Dutasteride is also the cause of the decrease in NO synthesis in the vascular endothelium of the corpora cavernosa, resulting in inadequate relaxation of smooth muscle cells in the blood vessels of the corpora cavernosa (Dean & Lue, 2005). Eventually, dutasteride can cause erectile dysfunction (ED) and decreased libido (Canguven & Burnett, 2008; Traish, 2020; Traish et al., 2011). Since neurosteroid biosynthesis also depends on 5α-reductases, medical treatment with dutasteride impacts the production of neurosteroids in the brain, thus affecting mood status (Mellon et al., 2001; Saengmearnuparp et al., 2021). However, not all patients receiving 5ARIs experience these side effects. Tamsulosin, as a selective alpha1-blocker, leads to smooth muscle relaxation of the prostate and the lower urinary tract and does not affect sexual function when used as monotherapy (van Dijk et al., 2006).
Dehydroepiandrosterone (DHEA) is a steroid prohormone produced in the adrenal cortex, gonads, adipose tissue, brain, and skin. After production, as a sulfated metabolite, dehydroepiandrosterone-sulfate (DHEAS) is transferred into the serum, the levels of which are approximately 250 times higher than free DHEA. In target tissues such as the brain, bone, breast, and adipose tissue, DHEAS is converted to DHEA by the sulfatase enzyme. DHEA can then be metabolized into various hormones such as androstenediol, androstenedione, estrone, TT, DHT, and 17β-estradiol (Labrie et al., 1997). This prohormone has potentially positive effects on age-related disorders, as it can be transformed based on local and overall hormone requirements (Orentreich et al., 1984). In addition, it is the only steroid in the human body independent of 5α-reductases action. When dutasteride inhibits the 5α-reductases, the “de novo” biosynthesis of neurosteroids in the brain remains impaired by DHEA levels (Mellon et al., 2001). As a potential androgen, DHEA may contribute to the maintenance of erectile function in addition to the effects of TT and/or DHT (El-Sakka, 2018). Since DHEA declines with aging, its functions as a neurosteroid and weak androgen also decrease, leading to symptoms that may not be present at normal DHEA levels (Voznesensky et al., 2009).
This study aims to explore how the age-related declines in DHEA(S) levels impact dutasteride-related side effects, ED, and mood disorders in men with BPH.
Material and Methods
The study examined the baseline characteristics of 250 participants aged 40 to 70 and assessed BPH treatment outcomes at the 6-month follow-up. The institution’s Ethics Committee approved the study protocol. Patients provided voluntary written consent after the informed consent form was reviewed by the Institutional Review Board at each site. Data entry into the registry required a signed informed consent form. The study included only patients presenting with symptoms and signs of BPH and seeking urological evaluation for the first time. Before administering the first dose of medication(s), a physical examination was conducted, including measuring the prostate volume, recording concomitant medications, and assessing comorbidities. Serum analyses at baseline and follow-up appointments included prostate-specific antigen (PSA) and DHEAS. Laboratory measurements were obtained at the same laboratory between 7 a.m. and 9 a.m. The primary outcomes focused on dutasteride side effects, specifically ED and mood disorders. Erectile function and mood regulation were assessed using the International Index of Erectile Function (IIEF-5) questionnaire and the Patient Health Questionnaire-9 (PHQ-9), respectively.
The inclusion criteria were moderate to severe lower urinary tract symptoms (LUTS; International Prostate Symptom Score [IPSS] between 12 and 26), 40 to 65 ml prostate volume, and a total serum PSA concentration of less than 4 ng/ml. The studied groups included patients <60 and ≥60 years old, divided into two subgroups according to the treatment, either with tamsulosin as monotherapy or tamsulosin and dutasteride as combination therapy. The assessment of LUTS was performed using the IPSS questionnaire, prostate volume was measured using transabdominal ultrasound, and serum levels of total PSA were measured in the same laboratory to confirm BPH diagnosis.
Patients were excluded from the study if they had dementia, deafness, vascular illness, myocardial infarction, diabetes mellitus, known/suspected carcinoma of the prostate, HIV/AIDS, metabolic syndrome, obesity, or were taking opioids.
Results
Baseline demographic and clinical characteristics were compared between the two age groups, as shown in Table 1. Older patients had larger prostate volumes (p = .001), higher IPSS scores (p = .003), lower IIEF-5 scores (p = .002), and higher PHQ-9 (p = .004).
Baseline Demographic Characteristics
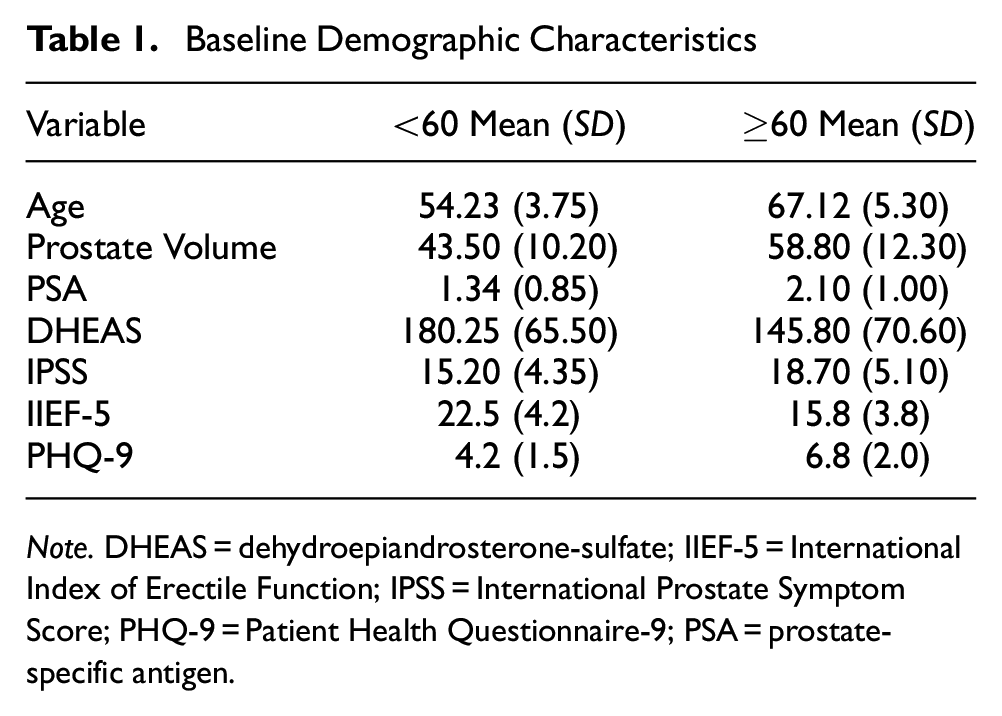
Note. DHEAS = dehydroepiandrosterone-sulfate; IIEF-5 = International Index of Erectile Function; IPSS = International Prostate Symptom Score; PHQ-9 = Patient Health Questionnaire-9; PSA = prostate-specific antigen.
As shown in Figure 1, the older age group had lower levels of DHEAS (p = .002).

Graphic Representation of Baseline Serum Levels of DHEAS by Age Group.
The IIEF-5 score in both age groups, as shown in Figure 2, was within normal ranges (22–25) for satisfactory erectile function, although the mean score was higher in the younger group. The baseline PHQ-9 score in both age groups was also within normal ranges (0–4; Figure 2).
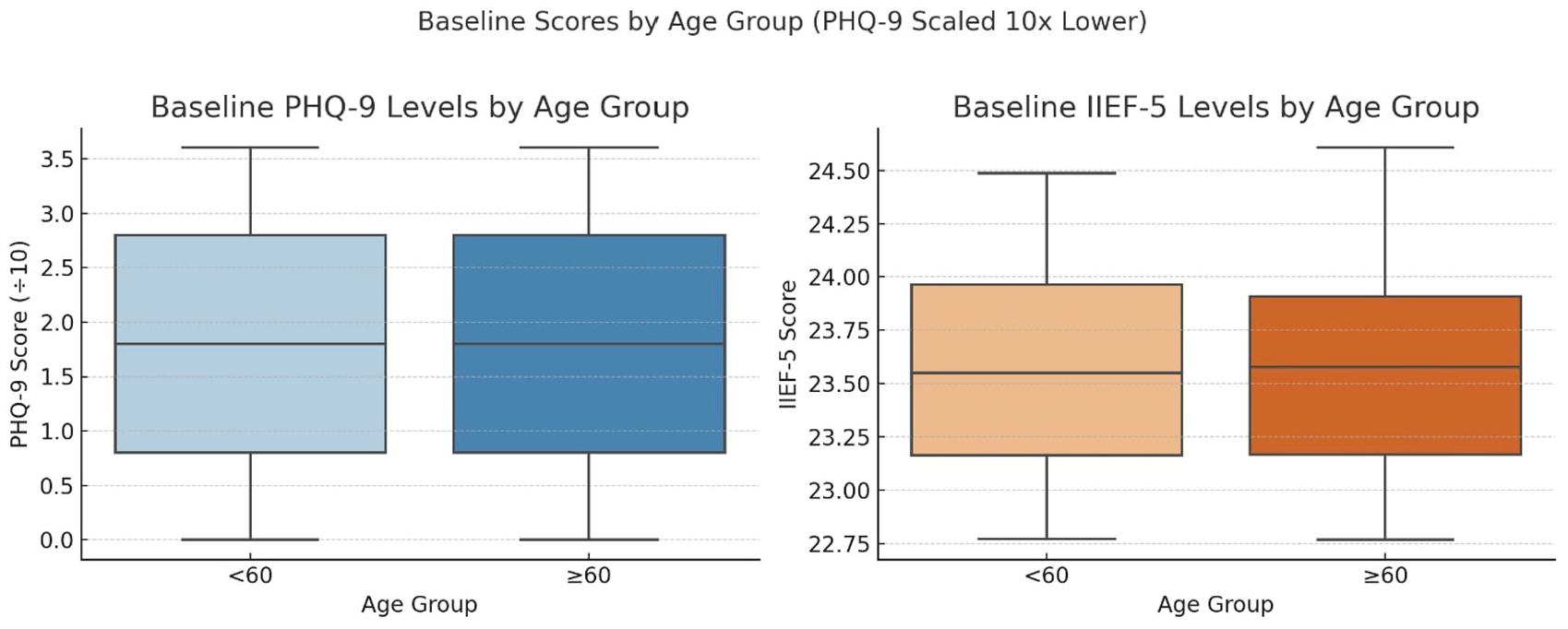
Graphic Representation of Baseline PHQ-9 Score and IIEF-5 Score by Age Group.
Figure 3 presents IIEF-5 and PhQ-9 scores at the follow-up in both age and therapy subgroups. In the subgroup receiving combination therapy, there was a sudden drop in IIEF-5 score (5–13) (Figure 3A, 3B-encircled). Also, as shown in Figures 3C and 3D, the PHQ-9 score decreased over time for both age groups within normal ranges (0–4) in the tamsulosin group, but there were individuals receiving combination therapy with a sudden increase in the score (7–12) in both age groups (Figures 3C and 3D, encircled).

IIEF-5 (A, B) and PhQ-9 (C, D) scores at the follow-up in both age and therapy subgroups.
As shown in Table 2, DHEAS positively correlated with IIEF-5 in younger patients treated with combination therapy (p = .015) and in older patients in both therapeutic modalities (p = .049, p = .01, respectively). The correlation between DHEAS and PHQ-9 was inverse and significant in both age groups and all therapeutic subgroups (p < .05).
The Correlation Between DHEAS and IIEF-5 and PHQ-9

Note. DHEAS = Dehydroepiandrosterone Sulfate; IIEF-5 = International Index of Erectile Function-5; PHQ-9 = Patient Health Questionnaire-9.
As shown in Table 3, regression analyses were performed to identify the predictors of change in IIEF-5 and PHQ-9.
Regression Analyses to identify DHEAS as a Predictor of Changes in IIEF-5and PHQ-9

Note. DHEAS=Dehydroepiandrosterone Sulfate; IIEF-5=International Index of Erectile Function-5; PHQ-9=Patient Health Questionnaire-9.
Table 3. Regression analyses to identify DHEAS as a predictor of changes in IIEF-5 and PHQ-9.
DHEAS was found to be a positive predictor of IIEF-5 changes in the older patient group treated with tamsulosin (p < .001) and in both age groups treated with combination therapy (p < .001) at the 6-month follow-up. Also, DHEAS was a significant negative predictor of the change in PHQ-9 in both age groups receiving combination therapy at the 6-month follow-up (p < .001).
Discussion
DHEA and its sulfated metabolite, DHEAS, decline with aging, reaching levels as low as 10 to 20% in individuals aged 70 to 80 compared to young individuals (Orentreich et al., 1984). Multiple studies have found lower levels of DHEA in men with ED, indicating that reduced DHEA(S) secretion is a significant risk factor for ED in older men (Basar et al., 2005; Feldman et al., 1994; Morales et al., 2000). The Massachusetts Male Aging Study findings revealed a correlation only between DHEA and ED among 17 investigated hormones (Feldman et al., 1994). Our study revealed lower baseline serum levels of DHEAS in older patients, reflecting the hormonal decline associated with aging. Even though the older patients had lower IIEF-5 scores at the baseline, indicating worse erectile function, this was expected due to a decrease in male sexual activity in aging males (Chung, 2019). In the younger patient group, the baseline IIEF-5 score was along the reference scale for normal erectile function (22–25; Rosen et al., 1999). In our study, there was a positive correlation between DHEAS and IIEF-5, confirming that DHEA(S) decline correlates with ED.
There is a proven connection between low DHEAS levels and mood disorders, particularly depression symptoms (Michael et al., 2000). DHEA(S) is a direct neurosteroid with antidepressant and cognitive properties known to increase feelings of well-being (Stárka et al., 2015). Age-related decline in serum DHEAS is evident, but in some individuals, this decline is greater than expected and could precipitate disorders related to its deficiency (Voznesensky et al., 2009), which is confirmed based on our study results. In this study, the older patients had higher PHQ-9 scores at baseline but within normal ranges, indicating none or minimal mood disturbances (Kroenke et al., 2001), which is consistent with the findings of more common mood impairment in the aging population (Valiengo et al., 2016). The study results showed a significant correlation between DHEAS decline and impaired mood status in both age groups and both therapeutic subgroups.
Recently, there has been an increased interest in the side effects of medical treatment in BPH patients. The majority of research data indicates that patients receiving 5ARIs (finasteride and dutasteride) have a greater incidence of ED than those receiving a placebo (Skolarus & Wei, 2009) or alpha-blockers (Haque et al., 2018). In this study, even though all patients had normal sexual function and unimpaired mood status at the beginning, after the BPH medical treatment, some individuals in both age groups receiving a combination of tamsulosin and dutasteride experienced ED and mood impairment. These findings confirmed that dutasteride side effects, such as ED and mood disorders, are common in combination treatment for BPH (Roehrborn et al., 2008, 2010). The mechanism behind these side effects is still debatable, and there is concern that they may persist even after stopping the therapy (Traish et al., 2011). One of the proposed mechanisms is that dutasteride inhibits the conversion of testosterone into DHT, thus impairing steroidogenesis, altering neural networks, and reducing blood vessels’ smooth muscle cell elasticity in erectile tissue, leading to persistent sexual dysfunction (Traish, 2020). Reducing the conversion of testosterone decreases the amount of DHT that stimulates NO synthesis in the vascular endothelium of the corpora cavernosa (Clark et al., 2004; Dean & Lue, 2005), resulting in insufficient relaxation of the smooth muscle cells of the blood vessels in the corpora cavernosa and consequently leading to an insufficient erection. (Dean & Lue, 2005). In our study, patients with declined DHEAS levels treated with tamsulosin alone did not experience ED or mood disturbances. DHEAS was found to be a predictor of the changes in erectile function and mood status only in patients receiving combination therapy with tamsulosin and dutasteride. One possible explanation is that DHEAS, a steroid unaffected by 5ARIs (Mellon et al., 2001), may help maintain erectile function since has been shown that DHEA(S) activates potassium channels, stimulates guanylate cyclase, enhances nitric oxide synthesis, and promotes arterial dilation while inhibiting vasoconstriction (El-Sakka, 2018).
Current evidence indicates that users of 5ARIs, including dutasteride, have a 1.1 to 1.9 times higher rate of depressive symptoms compared to non-users (Saengmearnuparp et al., 2021). Inhibition of brain 5α-reductases by 5ARIs, including dutasteride, leads to impaired neurosteroid biosynthesis (Mellon et al., 2001) but does not affect DHEA(S) action in the brain since they are direct neuroactive steroids (Stárka et al., 2015). These findings make DHEA(S) unique in maintaining normal mood regulation when 5α-reductase is inhibited by dutasteride.
The study was limited by its single-center design and narrow patient sample due to strict inclusion criteria.
Conclusion
In conclusion, baseline DHEAS should be a reliable predictor of changes in erectile function and mood status in patients undergoing BPH combination therapy with dutasteride. It is advisable to consider assessing DHEAS levels prior to designing treatment plans for BPH patients to prevent potential side effects like ED and mood disorders. Further research is encouraged to enhance understanding of this issue and inform clinical practice.
Footnotes
Appendix A. Variable Names and Definitions
PSA- Prostate-specific antigen is a protein produced by normal, as well as malignant, cells of the prostate gland.
DHEAS- dehydroepiandrosterone sulfate is an endogenous steroid hormone precursor.
IPSS-International Prostate Symptom Score is an eight-question written questionnaire used to screen for, rapidly diagnose, track the symptoms of, and suggest management of the lower urinary tract symptoms related to benign prostatic hyperplasia.
IIEF-5-International Index of Erectile Function is a diagnostic tool to assess erectile dysfunction.
PHQ-9-Patient Health Questionnaire-9 is a multipurpose instrument for screening, diagnosing, monitoring, and measuring the severity of depression.
Acknowledgements
none
Authors’ contribution
MSG designed the work and article draft, collect the data and wrote the article; ST revised it critically for important intellectual content; SA analyzed and interpreted the data. All authors approved the publication of the version.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
