Abstract
The aim of this study is to systematically evaluate the association between metabolic syndrome (MetS) and key clinical parameters of benign prostatic hyperplasia (BPH), including International Prostate Symptom Score (IPSS), prostate volume (PV), and the prevalence of MetS among men with BPH. A systematic search of PubMed, Embase, Web of Science, Scopus, and the Cochrane Library was conducted from inception to March 31, 2025, with the search implementation completed on April 5, 2025 for all databases. Observational studies comparing BPH patients with and without MetS were included. Random-effects models were used to pool standardized mean differences (SMDs), mean differences (MDs), and prevalence estimates. Heterogeneity, publication bias, subgroup analyses, sensitivity analyses, and meta-regression were performed according to prespecified criteria. Ten studies involving 3,947 BPH patients met the inclusion criteria. Pooled analyses demonstrated that MetS was associated with larger prostate volume (PV) and a tendency toward higher IPSS, although the latter showed substantial variability across studies. The prevalence of MetS among BPH patients was high, with considerable geographic and temporal variation. Subgroup analyses indicated that sample size influenced effect estimates, and meta-regression identified age, sample size, and publication year as contributors to heterogeneity. MetS is closely associated with prostate enlargement and symptom burden in men with BPH. These findings highlight the metabolic–urological interplay and support the integration of metabolic assessment into BPH management strategies. Large-scale prospective studies are needed to clarify causality and evaluate whether metabolic interventions can alter BPH progression.
Introduction
Benign prostatic hyperplasia (BPH) is one of the most common urological conditions affecting aging men, frequently leading to lower urinary tract symptoms (LUTS) and reduced quality of life (Cai et al., 2025; Feng et al., 2025). Its prevalence increases sharply after the age of 50, reflecting complex interactions among hormonal, inflammatory, and metabolic factors (Chiarot et al., 2025; Ren et al., 2024). In parallel, metabolic syndrome (MetS)—a cluster of cardiometabolic abnormalities characterized by central obesity, insulin resistance, dyslipidemia, and hypertension—has emerged as a global health burden (He et al., 2025; O’Mahony et al., 2025; Yang et al., 2025). The growing coexistence of BPH and MetS in aging populations has prompted increasing interest in their potential pathophysiological links.
Accumulating evidence suggests that MetS may contribute to prostatic overgrowth and LUTS through multiple mechanisms, including hyperinsulinemia-driven stromal proliferation, chronic low-grade inflammation, endothelial dysfunction, and alterations in sex hormone metabolism (He et al., 2025; Jan et al., 2024; Ren et al., 2024). Several epidemiological studies have reported that men with MetS have larger PVs, higher International Prostate Symptom Score (IPSS), and a greater likelihood of BPH progression (Bertocchi et al., 2020; Bratchikov et al., 2018; Urgesa et al., 2025). However, findings across studies remain inconsistent, partly due to variations in study design, diagnostic criteria, population characteristics, and sample size.
Given these uncertainties, a comprehensive synthesis of available evidence is needed to clarify the relationship between MetS and the clinical manifestations of BPH. Understanding this association is clinically relevant, as it may inform risk stratification, early identification of high-risk individuals, and development of metabolic-targeted therapeutic strategies for BPH. Therefore, we conducted a systematic review and meta-analysis to evaluate the impact of MetS on PV, LUTS severity, and the prevalence of MetS among patients with BPH, and to explore potential sources of heterogeneity across studies.
Method
Literature Search Strategy and Registration
This systematic review and meta-analysis was conducted in accordance with the PRISMA 2020 guidelines (Parums, 2021). A comprehensive and database-specific search strategy was developed to identify observational studies examining the association between BPH and MetS. Five electronic databases—PubMed, Embase, Web of Science Core Collection, Scopus, and the Cochrane Library—were systematically searched from their inception to March 31, 2025, with the search implementation completed on April 5, 2025 for all databases.
PubMed: PubMed searches combined Medical Subject Headings (MeSH) with free-text terms using Boolean operators: (“Prostatic Hyperplasia”[Mesh] OR “benign prostatic hyperplasia” OR BPH OR “lower urinary tract symptoms” OR LUTS OR IPSS) AND (“Metabolic Syndrome”[Mesh] OR “metabolic syndrome” OR “insulin resistance” OR obesity OR dyslipidemia) AND (“prostate volume” OR “prostatic enlargement” OR inflammation).
Embase: In Embase, Emtree terms were combined with title/abstract keywords:(“benign prostatic hyperplasia”/exp OR “benign prostatic hyperplasia” OR BPH OR LUTS OR IPSS) AND (“metabolic syndrome”/exp OR “metabolic syndrome” OR “insulin resistance” OR obesity OR dyslipidemia) AND (“prostate volume” OR “prostatic growth” OR inflammation).
Web of Science Core Collection: Topic searches (title, abstract, keywords) were applied: TS = ((“benign prostatic hyperplasia” OR BPH OR LUTS OR IPSS) AND (“metabolic syndrome” OR insulin resistance OR obesity OR dyslipidemia) AND (“prostate volume” OR “prostatic enlargement” OR inflammation)).
Scopus: Searches were conducted in titles, abstracts, and keywords: TITLE-ABS-KEY (“benign prostatic hyperplasia” OR BPH OR LUTS OR IPSS) AND TITLE-ABS-KEY (“metabolic syndrome” OR insulin resistance OR obesity OR dyslipidemia) AND TITLE-ABS-KEY (“prostate volume” OR “prostatic growth” OR inflammation).
Cochrane Library: Given the simpler indexing structure, a broad search strategy was applied: (“benign prostatic hyperplasia” OR BPH) AND (“metabolic syndrome”) AND (“prostate volume” OR LUTS OR IPSS).
No restrictions were applied regarding language, region, or publication year. To ensure completeness, the reference lists of all eligible articles and related reviews were manually screened, and forward citation tracking was conducted through Web of Science and Google Scholar. All search procedures and protocol amendments were documented in compliance with PROSPERO (International Prospective Register of Systematic Reviews) and PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) standards.
Eligibility Criteria
Studies were considered eligible if they met the following criteria: (1) Population: Adult men diagnosed with BPH based on clinical and/or imaging criteria. (2) Exposure: Diagnosis of MetS using standardized criteria. (3) Comparator: BPH patients without MetS. (4) Outcomes: Reported at least one of the following—IPSS, PV, maximum urinary flow rate (Qmax), or the prevalence of MetS among BPH patients. (5) Design: Observational studies (cross-sectional, case–control, or cohort). (6) Data: Provided sufficient quantitative information to derive effect estimates.
Exclusion criteria included reviews, case reports, conference abstracts, animal or laboratory research, studies lacking extractable outcome data, and duplicate or overlapping cohorts.
Study Selection
All retrieved records were imported into EndNote for de-duplication. Two reviewers independently screened titles/abstracts and subsequently assessed full texts using prespecified inclusion and exclusion criteria. Any discrepancies were resolved through consensus or adjudication by a third reviewer. The study selection workflow followed the PRISMA 2020 guidelines.
Data Extraction
Data extraction was performed independently by two investigators using a piloted and standardized extraction template. The following variables were collected from each study: general study information (author, year, country/region, study design), sample size of MetS and non-MetS groups, participant characteristics (mean age), diagnostic criteria for MetS, outcome data: IPSS, PV, Qmax, and prevalence of MetS. Continuous outcomes were extracted as means with standard deviations; proportion data were extracted as event counts. When required, corresponding authors were contacted to clarify missing or ambiguous information.
Quality Assessment
The methodological quality of all included observational studies was evaluated using the Newcastle–Ottawa Scale (NOS), adapted for cross-sectional and cohort designs (Stang, 2010). The NOS assesses three methodological domains: (1) selection of participants, (2) comparability of exposure groups, and (3) outcome or exposure ascertainment. Each study could receive up to 10 points, and, consistent with commonly accepted thresholds in high-impact epidemiologic literature, studies were categorized as follows: high quality: NOS ≥7 points; moderate quality: NOS 4–6 points; low quality: NOS 0–3 points (none met this criterion).
Special attention was given to critical indicators that influence internal validity, including explicit diagnostic criteria for BPH and MetS, representativeness of cohorts, adjustment for confounders (e.g., age, body mass index [BMI], comorbidities), and validated outcome measurement (such as TRUS for PV and standardized IPSS scoring).
Two reviewers independently conducted the quality assessment. Discrepancies were resolved through discussion or adjudication by a third senior reviewer. Quality assessments informed interpretation of findings but were not used to exclude studies.
Statistical Analysis
Meta-analyses were conducted using random-effects models (DerSimonian–Laird estimator) to accommodate expected clinical and methodological heterogeneity across study settings, populations, and measurement techniques. For continuous outcomes (IPSS and PV), standardized mean differences (SMDs) were calculated when measurement scales varied, and mean differences (MDs) were used for comparable units. For prevalence outcomes, pooled estimates were derived using the inverse variance method, applying appropriate variance-stabilizing transformations when required.
Assessment of heterogeneity: Between-study heterogeneity was quantified using Cochran’s
Sensitivity and publication bias analysis: To assess the robustness of summary estimates, leave-one-out sensitivity analyses were performed for each outcome. Potential publication bias was evaluated visually using funnel plots, interpreted with attention to asymmetry patterns commonly associated with small-study effects or selective reporting.
Software and significance thresholds: All analyses were performed using R (version 4.1.2) with the meta and metafor packages. Statistical significance was defined as a two-sided
Result
Study Selection
The initial database search yielded a total of 1,383 records, including PubMed (
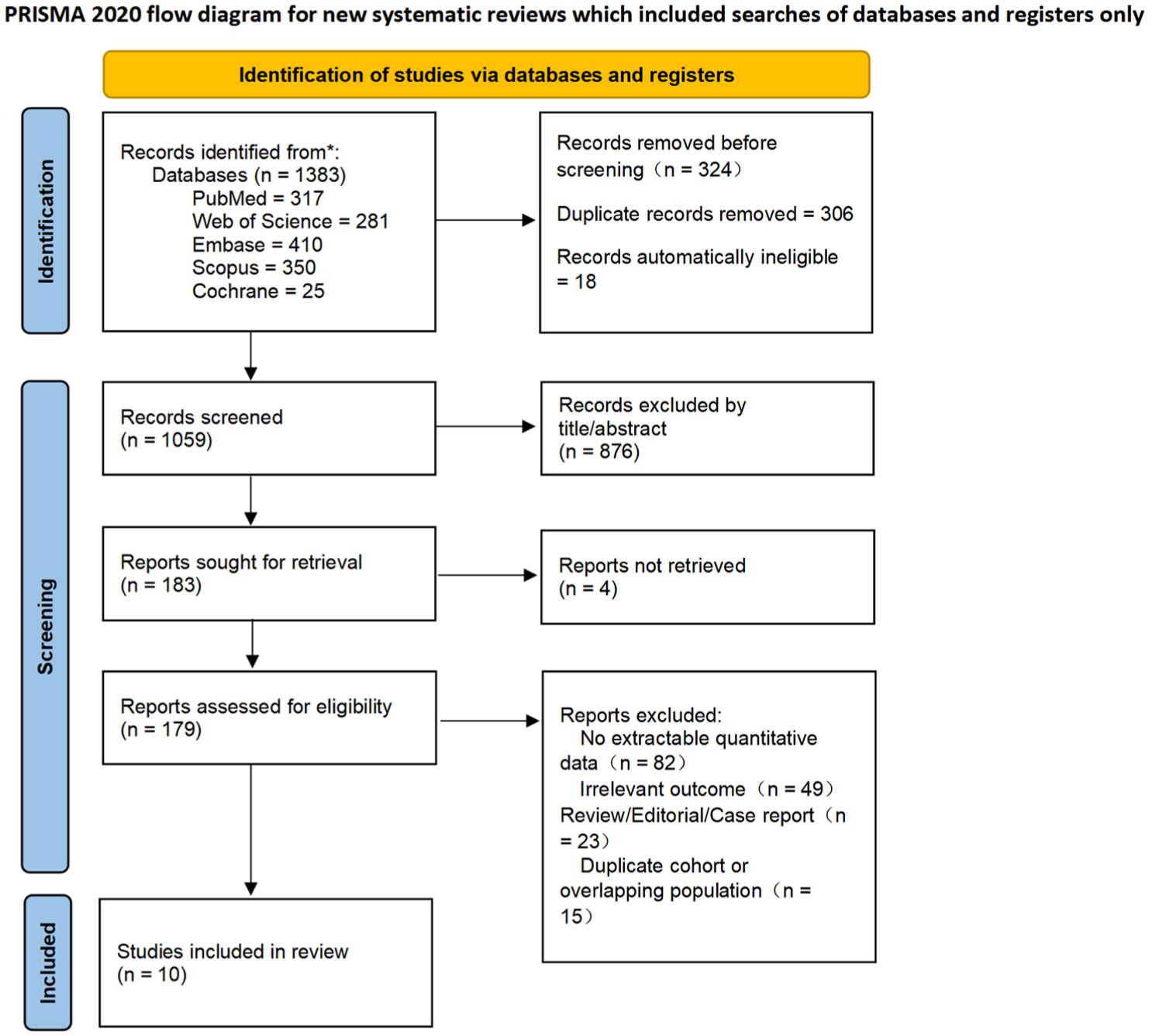
PRISMA Flow Diagram of Study Selection.
Study Characteristics
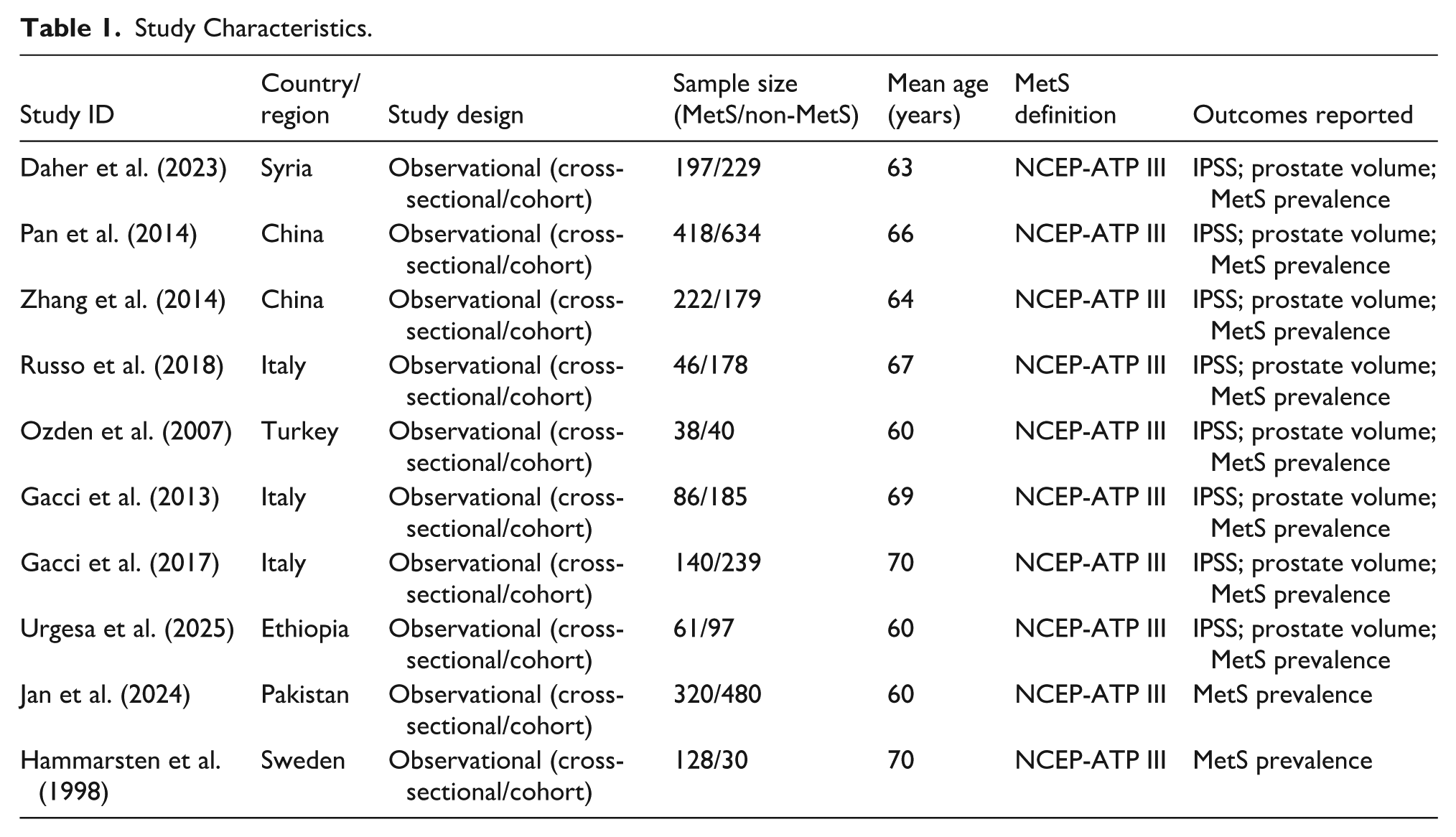
Characteristics of the 10 included studies are summarized in Table 1. Studies were published between 1998 and 2025, and the core characteristics were as follows: the total sample size was 3,947 patients with BPH; all studies adopted the NCEP-ATP III to diagnose MetS, reporting of outcome indicators: eight studies reported PV, eight studies reported the IPSS, and 10 studies reported the prevalence of MetS; the study types were all cross-sectional studies or cohort studies; the age range of the study subjects was 50 to 80 years; geographical distribution: five studies in Asia, four studies in Europe, and one study in the Middle East.
Study Characteristics.
Quality Assessment
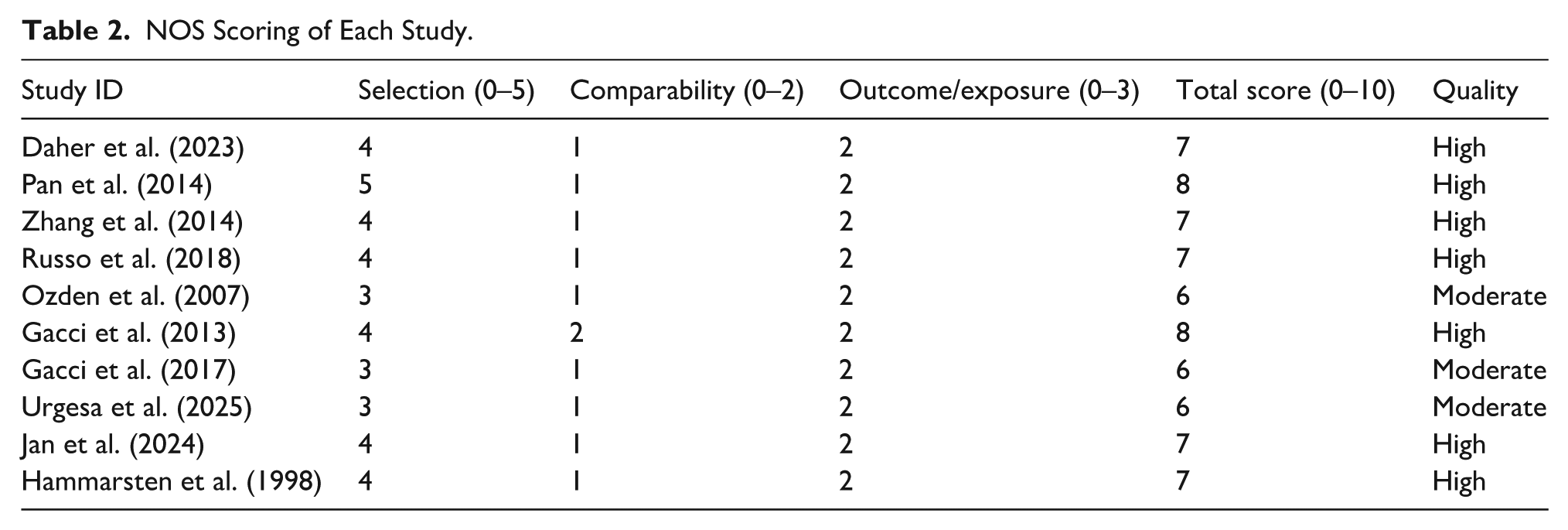
All included observational studies were evaluated using the NOS for cross-sectional and cohort studies. The NOS assesses three major domains: selection of participants, comparability between exposure groups, and outcome/exposure assessment. Across the 10 included studies, seven were rated as high quality (NOS ≥ 7 points) and three were rated as moderate quality (NOS 5–6 points). No study was classified as low quality (<5 points). Most studies clearly defined BPH and MetS using standardized diagnostic criteria (NCEP ATP III), employed validated outcome measures such as IPSS and transrectal ultrasound for PV, and used appropriate statistical methods. However, common methodological limitations included inadequate reporting of nonrespondents, limited adjustment for confounding factors (such as age, BMI, or comorbidities), and lack of blinding in outcome ascertainment. The detailed NOS scoring of each study is presented in Table 2.
NOS Scoring of Each Study.
International Prostate Symptom Score
Eight studies reported continuous IPSS values, involving 1,208 patients with MetS and 1,781 without MetS. The pooled SMD was 0.36 (95% confidence interval [CI] = [−0.24, 0.95]) (Figure 2), suggesting a trend toward more severe LUTS in the MetS group, although the CI crossed zero. Heterogeneity was extremely high (
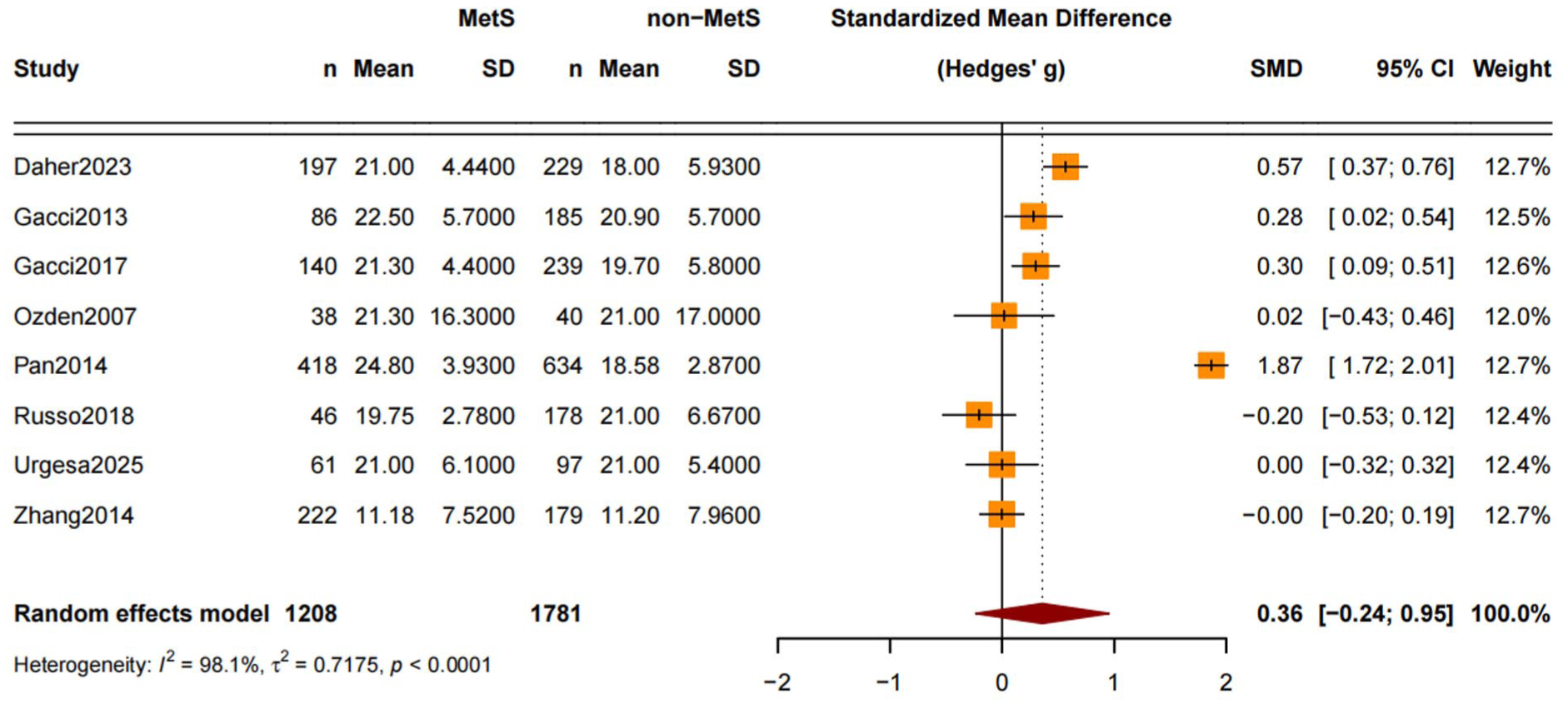
Forest Plot for IPSS SMD Between MetS and Non-MetS Groups.
Prevalence of MetS Among BPH Patients
A total of 10 studies (
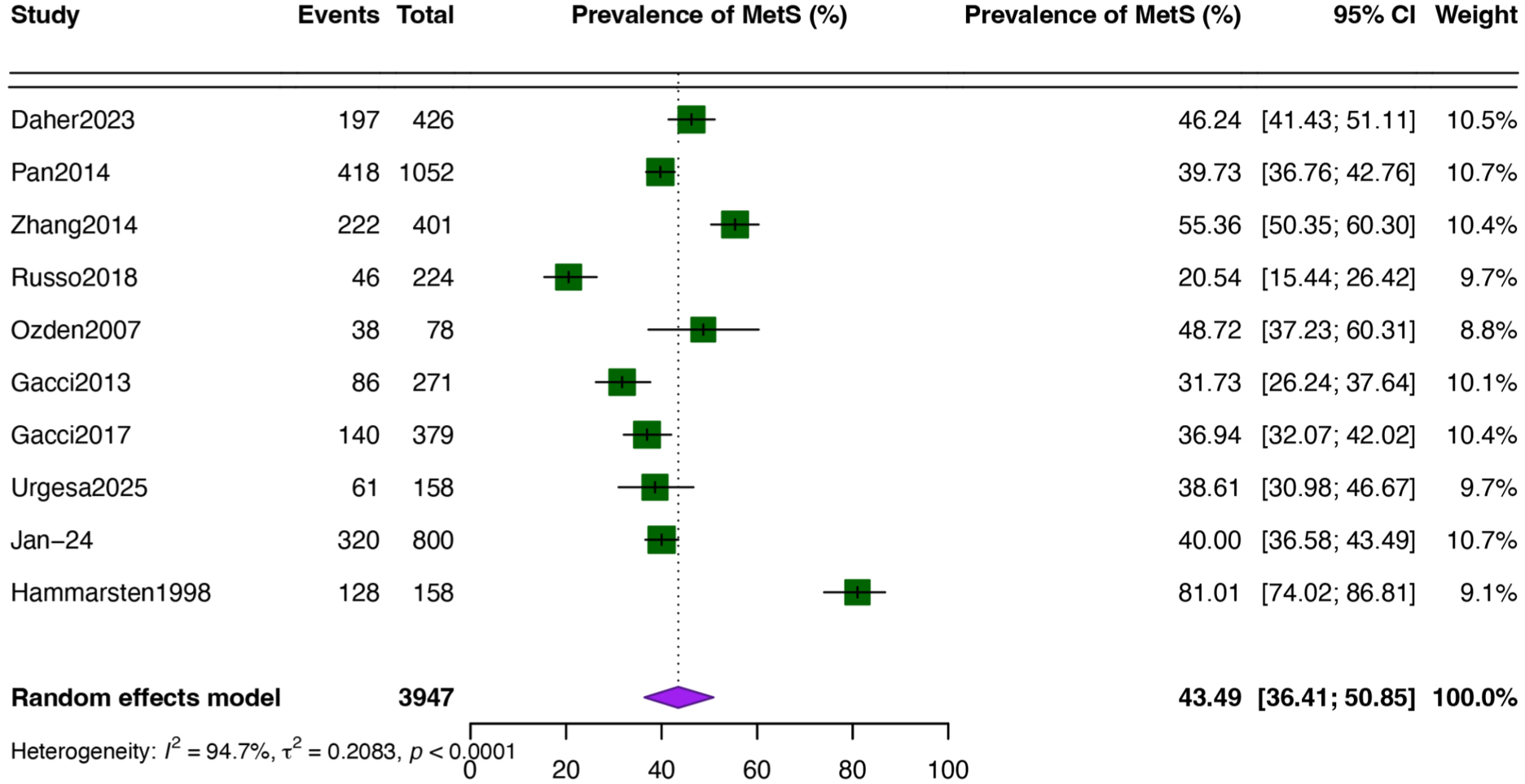
Pooled Prevalence of Metabolic Syndrome Among BPH Patients.
Prostate Volume
Eight studies reported PV, totaling the same patient groups as IPSS analysis. The pooled MD indicated that BPH patients with MetS had significantly larger prostates than those without MetS, with an MD of 10.17 mL (95% CI = [5.46, 14.87]) (Figure 4). Substantial heterogeneity was observed (
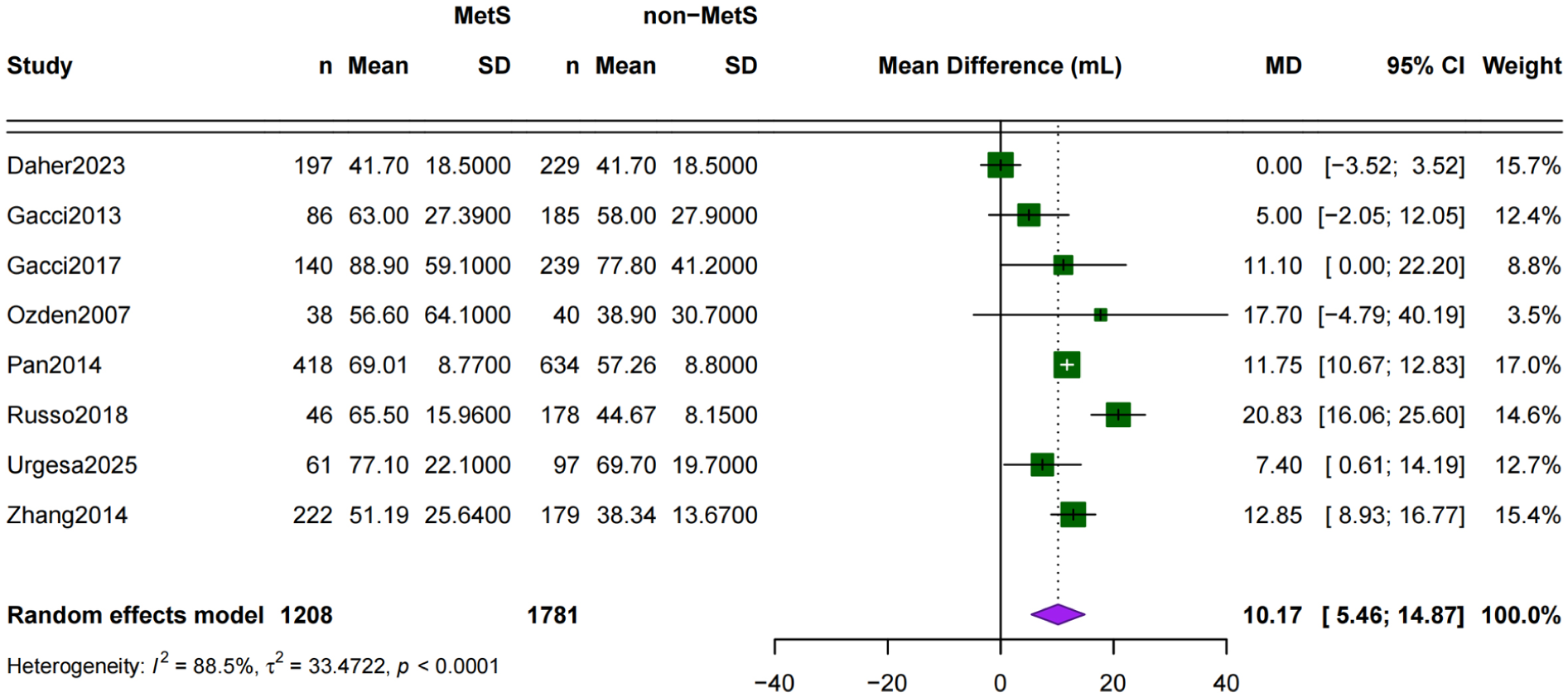
Forest Plot for Prostate Volume MD Between MetS and Non-MetS Groups.
Publication Bias
IPSS
The funnel plot (Figure 5A) demonstrated mild asymmetry, with small studies scattering widely and large studies clustering near the right upper quadrant, suggesting potential small-study effects.
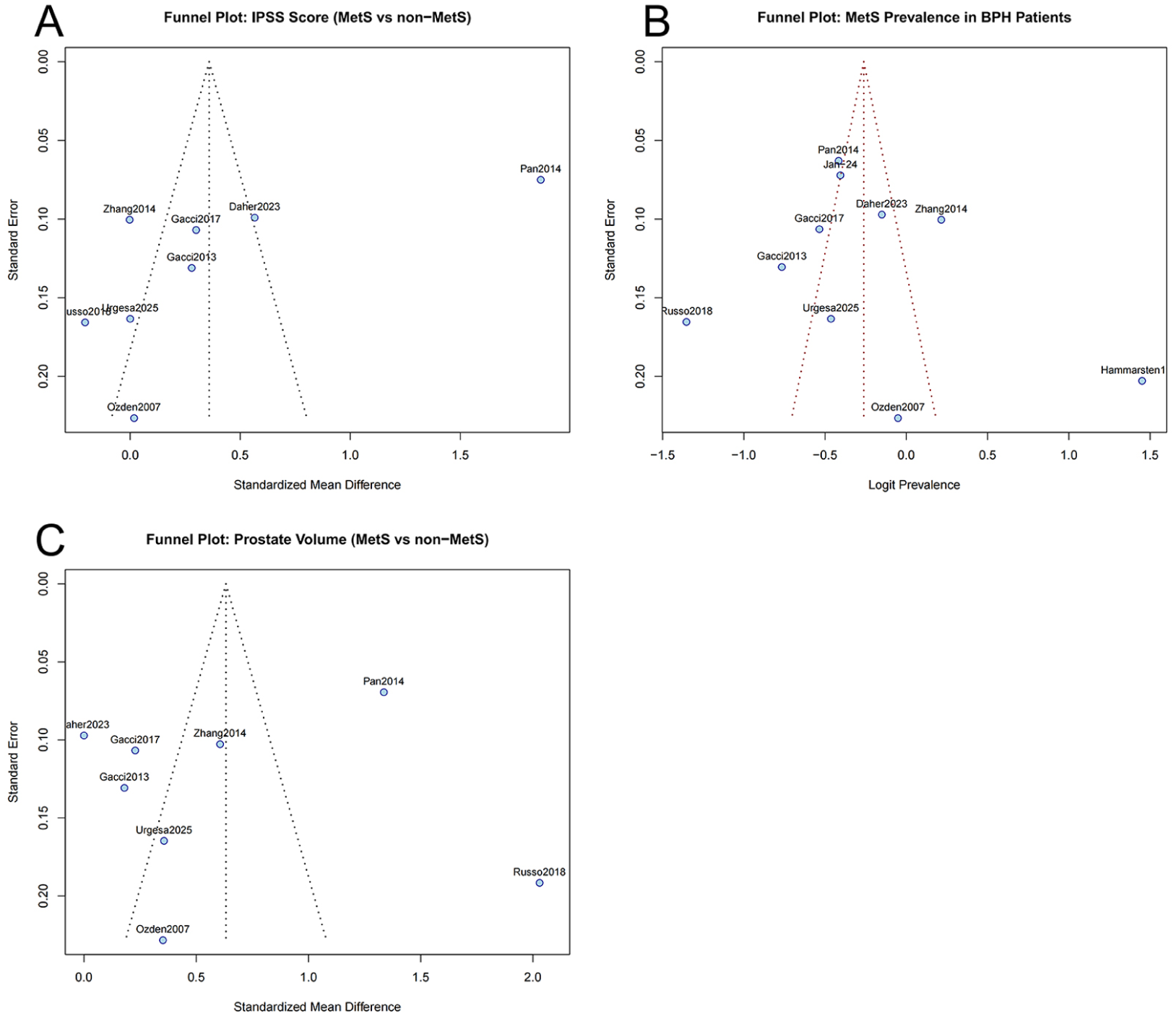
Funnel Plots (A: IPSS; B: MetS Prevalence; C: Prostate Volume).
MetS Prevalence
The funnel plot for prevalence (Figure 5B) revealed marked asymmetry, with high-prevalence studies clustering to the right, whereas small-sample, low-prevalence studies appeared in the lower left. This pattern indicates possible publication bias and strong between-study heterogeneity.
Prostate Volume
The PV funnel plot (Figure 5C) showed moderate asymmetry, indicating potential influence from sample size, region, or measurement variation.
Subgroup Analysis
Subgroup analysis stratified by sample size revealed a significant between-group difference (χ2 = 219.89,
Across sample size categories, no statistically significant subgroup differences were detected (χ2 = 1.61,
A significant subgroup effect was observed for PV (χ2 = 46.46,
Sensitivity Analysis
To evaluate the stability of the association between MetS and IPSS, a leave-one-out sensitivity analysis was performed. Sequential omission of each study generated SMD estimates ranging from 0.33 to 0.44, and no single study changed the direction or magnitude of the overall effect (Supplemental Figure S2A). The narrow fluctuation range suggests that the pooled estimate was not disproportionately influenced by any individual dataset, confirming the high robustness of the primary findings.
Sensitivity analysis for the pooled prevalence of MetS among BPH patients showed similarly stable results. Excluding studies one at a time produced prevalence estimates ranging from 39.38% to 46.27%, which closely aligned with the main pooled rate (Supplemental Figure S2B). The consistency across iterations indicates that the observed prevalence was not driven by outliers or low-quality studies, further strengthening the reliability of the prevalence estimate.
For PV, leave-one-out analysis revealed minor variations, with MD values fluctuating only between 0.44 and 0.73. None of the exclusions meaningfully altered the effect size or led to directional changes (Supplemental Figure S2C). This pattern demonstrates that the association between MetS and increased PV remained structurally stable, providing no indication of undue influence from any single study.
Meta-Regression
Sample size showed a strong positive linear relationship with IPSS effect estimates (
Publication year significantly predicted MetS prevalence (
Meta-regression demonstrated that mean age significantly contributed to heterogeneity in PV differences (β > 0;
Discussion
In this systematic review and meta-analysis, we synthesized current evidence regarding the association between MetS and clinical features of BPH. The findings demonstrate that MetS is associated with higher IPSS, increased PV, and a substantial prevalence of MetS among patients with BPH. These results align with an expanding body of literature, suggesting that metabolic dysregulation contributes to prostatic overgrowth and LUTS through multiple endocrine, inflammatory, and vascular mechanisms (Calmasini et al., 2020; Gacci et al., 2017; Sachdeva et al., 2022).
Several pathophysiological pathways may explain the observed associations. Hyperinsulinemia and insulin resistance may promote prostate stromal proliferation via activation of the Insulin-like Growth Factor-1 (IGF-1) axis (Gong et al., 2022; Sharkey et al., 2025), while visceral obesity-related inflammation increases local cytokine production, fostering tissue remodeling and enlargement (Colado-Velázquez et al., 2023; Urgesa et al., 2025; Xia & Li, 2017). Dyslipidemia has also been implicated in altering sex hormone metabolism, resulting in androgen–estrogen imbalances that stimulate prostatic hyperplasia (Wang et al., 2025). These mechanisms support the notion that MetS acts not only as a comorbidity but also as a potential driver of LUTS progression.
Our results are consistent with previous epidemiologic studies that reported larger prostate size and more severe LUTS among men with MetS (Bratchikov et al., 2020; Jan et al., 2024; Urgesa et al., 2025). However, the considerable heterogeneity observed across studies warrants careful interpretation. Differences in sample size, measurement techniques (e.g., TRUS vs. digital estimation), population age structure, and regional prevalence of obesity likely contributed to the variation. Meta-regression findings indicating publication year, age-, and sample-size effects further support these possibilities. In addition, variations in diagnostic rigor for MetS and lifestyle patterns across geographic regions may explain discrepancies among studies (Cai et al., 2025; Sharkey et al., 2025).
From a clinical perspective, these findings underscore the importance of metabolic health assessment in men presenting with BPH symptoms. Addressing modifiable metabolic disturbances may provide dual benefits in improving systemic risk profiles and mitigating LUTS severity. Emerging interventional studies suggest that weight reduction, glycemic control, and lipid optimization may help attenuate prostate growth and improve urinary symptoms, although evidence remains preliminary (O’Mahony et al., 2025; Wu et al., 2025).
This review has several strengths, including comprehensive database coverage, rigorous methodological assessment, and multiple sensitivity analyses confirming the robustness of findings. Nonetheless, limitations must be acknowledged. All included studies were observational, limiting causal inference. Residual confounding—particularly from lifestyle factors, medication use, or unmeasured metabolic markers—cannot be excluded. High heterogeneity, although explored through subgroup and meta-regression analyses, may still reflect unaccounted population-level differences. Finally, the reliance on cross-sectional designs in several studies restricts insights into temporal relationships between MetS and BPH progression.
Conclusion
In summary, MetS appears to be significantly associated with increased PV and more severe LUTS, highlighting an important metabolic–urological interface. Future large-scale prospective studies are needed to clarify causality and evaluate whether metabolic interventions can modify the clinical trajectory of BPH.
Supplemental Material
sj-jpg-1-jmh-10.1177_15579883261436335 – Supplemental material for The Association Between Metabolic Syndrome and Benign Prostatic Hyperplasia: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-1-jmh-10.1177_15579883261436335 for The Association Between Metabolic Syndrome and Benign Prostatic Hyperplasia: A Systematic Review and Meta-Analysis by Wenfeng Gao, Yingbo Guo, Xudong Yu, Tao Zhang, Huijie Gong and Yaosheng Zhang in American Journal of Men's Health
Supplemental Material
sj-jpg-2-jmh-10.1177_15579883261436335 – Supplemental material for The Association Between Metabolic Syndrome and Benign Prostatic Hyperplasia: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-2-jmh-10.1177_15579883261436335 for The Association Between Metabolic Syndrome and Benign Prostatic Hyperplasia: A Systematic Review and Meta-Analysis by Wenfeng Gao, Yingbo Guo, Xudong Yu, Tao Zhang, Huijie Gong and Yaosheng Zhang in American Journal of Men's Health
Supplemental Material
sj-jpg-3-jmh-10.1177_15579883261436335 – Supplemental material for The Association Between Metabolic Syndrome and Benign Prostatic Hyperplasia: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-3-jmh-10.1177_15579883261436335 for The Association Between Metabolic Syndrome and Benign Prostatic Hyperplasia: A Systematic Review and Meta-Analysis by Wenfeng Gao, Yingbo Guo, Xudong Yu, Tao Zhang, Huijie Gong and Yaosheng Zhang in American Journal of Men's Health
Footnotes
Funding
The authors disclosed receipt of the following financial sup port for the research, authorship, and/or publication of this article: This work was supported in part by the National High-level TCM Hospitals Clinical Research Fund (DZMG-ZLZX-25027), and the Beijing Traditional Chinese Medicine “Torch Inheritance 3+3 Project”.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
