Abstract
Male infertility affects millions worldwide, yet its underlying causes remain incompletely understood. Total round cell concentration (TRCC) in semen, particularly leukocytospermia, has been suggested as a biomarker of impaired sperm function. However, its relationship with sperm DNA fragmentation index (DFI) remains unclear, particularly in sub-Saharan Africa, where research is scarce. This study examines the prevalence of elevated TRCC and its associations with semen parameters and sperm DNA fragmentation among men attending a fertility clinic in Kumasi, Ghana. A cross-sectional study was conducted among 227 men, with semen samples analyzed following WHO guidelines. Sperm concentration, motility, and morphology were assessed, while TRCC was quantified using a Neubauer hemocytometer and light microscopy. Sperm DNA fragmentation was determined using the Sperm Chromatin Structure Assay, and multivariate logistic regression models were employed to evaluate associations between TRCC and semen quality parameters. Elevated TRCC was detected in 19.4% of participants. Higher TRCC levels were significantly associated with lower odds of oligozoospermia (c
Keywords
Introduction
Infertility is a growing global health concern with profound physical, psychological, and socioeconomic consequences, particularly for reproductive-age couples. An estimated 15% of couples worldwide struggle with infertility, affecting approximately 50 to 80 million individuals (Inhorn & Patrizio, 2014). Male-related factors contribute to 20% to 30% of these cases, yet male infertility remains an underrecognized and often misunderstood condition (Agarwal et al., 2021). Defined as the inability to achieve conception with a fertile female after at least 1 year of regular, unprotected intercourse, male infertility is a complex disorder influenced by genetic, environmental, infectious, and idiopathic factors, many of which remain poorly understood (Eisenberg et al., 2023; Tvrdá et al., 2022).
Infections of the male reproductive tract (testes, epididymis, and prostate) are significant disruptors of spermatogenesis and sperm function (Alzaidi & Kareem, 2024). A key diagnostic marker of testicular dysfunction and reproductive tract infections is an elevated total round cell concentration (TRCC) in semen, which includes both leukocytes (white blood cells) and immature germ cells (IGCs) (Palermo et al., 2016; Patil et al., 2013). The World Health Organization (WHO) recommends further evaluation when TRCC exceeds 1 million cells/mL, as elevated levels may indicate inflammation, oxidative stress, or defects in spermatogenesis (Long & Kenworthy, 2022). While leukocytes play an essential role in immune defense, excessive leukocytospermia generates reactive oxygen species (ROS), leading to sperm membrane damage, lipid peroxidation, and DNA fragmentation—key mechanisms implicated in male infertility (Aitken, 2017). Similarly, the presence of IGCs in semen suggests abnormal spermatogenesis, potentially compromising sperm quality and fertilization potential (Dissanayake, 2022).
Elevated TRCC has been associated with reduced sperm motility, abnormal morphology, increased sperm DNA fragmentation, and higher rates of apoptosis and necrosis—factors that collectively impair male fertility (Andradc-Rocha & Maya, 2022). However, studies yield conflicting results on whether TRCC is a direct cause of male infertility or merely a marker of underlying testicular dysfunction. Some evidence suggests that TRCC, particularly leukocytes, contributes to oxidative sperm damage, whereas other studies argue that its presence reflects an altered spermatogenic process without directly inducing DNA fragmentation (Balasubramaniam & Pawar, 2018; Hussain et al., 2024; Iommiello et al., 2015; Palermo et al., 2016).
Despite increasing global attention on male infertility, research in sub-Saharan Africa—particularly Ghana—remains scarce. As a resource-limited setting, Ghana faces significant challenges in diagnosing and managing male infertility due to limited access to specialized tests and advanced semen analysis techniques. This has contributed to a critical gap in understanding various factors affecting male fertility, including TRCC, a key marker of male reproductive health, and its impact on semen quality and sperm DNA integrity. While TRCC has been linked to testicular dysfunction, as well as inflammatory and infectious conditions affecting fertility, its clinical significance remains underexplored, particularly in African populations where unique genetic, environmental, and lifestyle factors may influence reproductive health outcomes. Investigating the prevalence of TRCC and its relationship with sperm DNA fragmentation index (DFI) is therefore crucial, not only to bridge this knowledge gap but also to inform fertility management in low-resource settings globally.
This study provides the first comprehensive analysis of TRCC and DFI in Ghanaian males, generating context-specific evidence despite the limitations of a resource-constrained environment. While it may not employ the most advanced diagnostic tools available in high-resource countries, the findings contribute valuable epidemiological and clinical data that can inform targeted interventions, healthcare policy, and the development of cost-effective fertility diagnostics in similar settings. By providing region-specific evidence, this study enhances understanding of TRCC’s role in male infertility, particularly in low-resource settings and lays the groundwork for future research and clinical interventions to improve male fertility management in sub-Saharan Africa.
Methodology
Study Design
This hospital-based, cross-sectional study was conducted in the Ashanti Region of Ghana, specifically within the Kumasi Metropolis, from February to July 2024. Participants provided semen samples after 2 to 7 days of abstinence. Exclusion criteria included lack of informed consent, systemic or localized illnesses affecting semen quality (e.g., diabetes, varicocele, orchitis, cryptorchidism, testicular surgery/trauma), recent febrile illnesses, sexually transmitted infections (STIs), and azoospermia confirmed by microscopic analysis (Kumurya et al., 2018; Nam & Mehta, 2023). To reduce confounding, individuals using medications that could alter semen parameters—such as hormonal therapies (e.g., testosterone, anabolic steroids), and chemotherapeutic agents—were excluded (Samplaski & Nangia, 2015). Semen analysis followed WHO guidelines, assessing volume, concentration, motility, and morphology. Demographic data, including age, marital status, and occupational exposure duration, were recorded.
Occupations were classified into high-, moderate-, and low-risk groups based on exposure to reproductive hazards. High-risk occupations involved significant exposure to heat, toxins, electromagnetic fields, or radiation (e.g., miners, electricians, mechanics, sonographers, spare parts dealers). Moderate-risk occupations included those with mixed exposures, stress, or sedentary work (e.g., engineers, contractors, drivers, farmers). Low-risk occupations were primarily office-based with minimal hazardous exposure (e.g., bankers, police officers, teachers, lecturers, journalists, health workers; Meyer et al., 2022; Ranasinghe et al., 2022). While this classification provides a structured risk assessment, individual variations exist based on job roles, exposure duration, and use of protective measures. The study adhered to the STROBE guidelines in carrying out the work and reporting the findings.
Study Population
The sample size was calculated using Cochran’s formula for unlimited population.

Where
Substituting the variables into the formula yields a minimum sample size of 205. The study, however, recruited a total of 227 participants.
Laboratory Procedures and Analysis
Semen Collection and Analysis
Semen samples were collected in sterile containers through masturbation after an abstinence period of 2 to 7 days. Various semen parameters, including appearance, volume, pH, liquefaction, count, motility, and morphology, were analyzed according to WHO guidelines.
Appearance
After liquefaction, semen appearance was assessed for color, transparency, and presence of mucus streaks at room temperature. Any alterations in visual appearance, such as changes in color, clarity, or the presence of mucous streaks, were noted.
Volume
The volume of each semen sample was measured using a graduated measuring cylinder, with the volume recorded in milliliters.
pH
Semen pH was determined using a narrow-range pH paper (pH 6.4–8.0). A drop of the sample was evenly spread onto the pH paper (Sperm Processer, India), and the color change was compared to a calibrated strip after 30 s.
Concentration
Sperm concentration was assessed using a Makler counting chamber (Sefi-Medical Instruments, Italy). The semen sample was diluted, and loaded onto the chamber, and sperm cells were counted under a microscope, with the concentration calculated using the chamber’s specific formula.
Motility
Total motility was assessed by applying a drop (10 μl) of well-mixed, liquefied semen onto the counting chamber. The sample was analyzed within 3 to 5 min under a microscope with a X10 or X20 objective lens. Sperm were categorized into four grades based on their progression: rapid (+4), moderate (+3), slow (+2), and non-progressive (+1).
Sperm Morphology
A smear was prepared to assess sperm morphology. The slide was well cleaned and then washed in 70% ethanol and air-dried. 5 µl of semen was applied to the slides. These slides were fixed with 95% v/V ethanol for 5 to 10 min and allowed to air-dry. The smear was washed with sodium bicarbonate-formalin solution to remove any mucus that may be present and rinsed several times under running water. The smears were then logged with diluted (1 in 20) carbol fuchsin, crystal violet solution, and allowed to stain for 2 min, and then the stain was washed off with water. Lugol’s solution was added to the smear for 1 min as a mordant and washed with distilled water. The smear was then counterstained with dilute (1 in 20) Loeffler’s methylene blue safranin (0.1%) solution for 2 min and washed with distilled water, drained, and air-dried. The samples were stained and examined under a microscope using the ×40 and ×100 objective lenses. Abnormalities in sperm head, middle piece, and tail were recorded, with 100 spermatozoa assessed per slide (Agyepong & Bedu-Addo, 2019).
Determination of TRCC
The TRCC in semen was determined manually using a sperm counter and a light microscope (Olympus Corporation, Japan; ×200 magnification), following the WHO Laboratory Manual for the Examination and Processing of Human Semen. A well-mixed semen sample was loaded into the counting chamber and allowed to settle under a coverslip. Non-sperm round cells were identified and counted within the designated grid areas of the hemocytometer at ×200 magnification to ensure optimal visualization and accuracy. The final concentration was calculated using the formula:

No further differentiation was performed to distinguish leukocytes from IGCs. To enhance accuracy and minimize variability, all counts were conducted in duplicate, and the mean value was recorded as the TRCC.
Sperm DFI Test
Semen samples with elevated TRCC were processed within 1 h of collection and allowed to liquefy at 37°C for 20 to 30 min. Samples were then centrifuged at 3,000 rpm for 10 min to pellet the sperm cells. Sperm DNA fragmentation of semen samples received was assessed following the manufacturer’s instructions for the Sperm Chromatin Structure Assay kit (Sperm Processor Pvt., India). The slides were stained with DAPI (4’6–diamino-2-phenylindole). Spermaotozoa with non-dispersed and dispersed chromatin loops were identified with bright-field microscopy. DFI was then calculated as the percentage of sperm with fragmented DNA, with a DFI > 30%. The DFI was recorded in the patient’s file and interpreted based on the reference ranges (Normal/Excellent: <15%, Good/Fair: 15–24%, Fair to Poor: 25–29%, Poor: 30–50%, Severely Elevated: >50%).
Ethical Consideration
The study was approved by the Committee on Human Research, Publications, and Ethics (CHRPE) of Kwame Nkrumah University of Science and Technology (reference number CHRPE/AP/107/24).
Statistical Analysis
Data were entered into Microsoft Excel 2019 (Microsoft Inc., USA) and analyzed using the Statistical Package for the Social Sciences (SPSS; SPSS Inc., Chicago, Illinois, USA) software version 26. Descriptive analyses were performed to determine data distribution. The Shapiro-Wilks test was done to assess the normality of continuous variables before selecting appropriate parametric or non-parametric tests. Spearman’s rank correlation was employed to evaluate the strength and direction of associations between quantitative and qualitative variables. Group comparisons were conducted using Student’s
Results
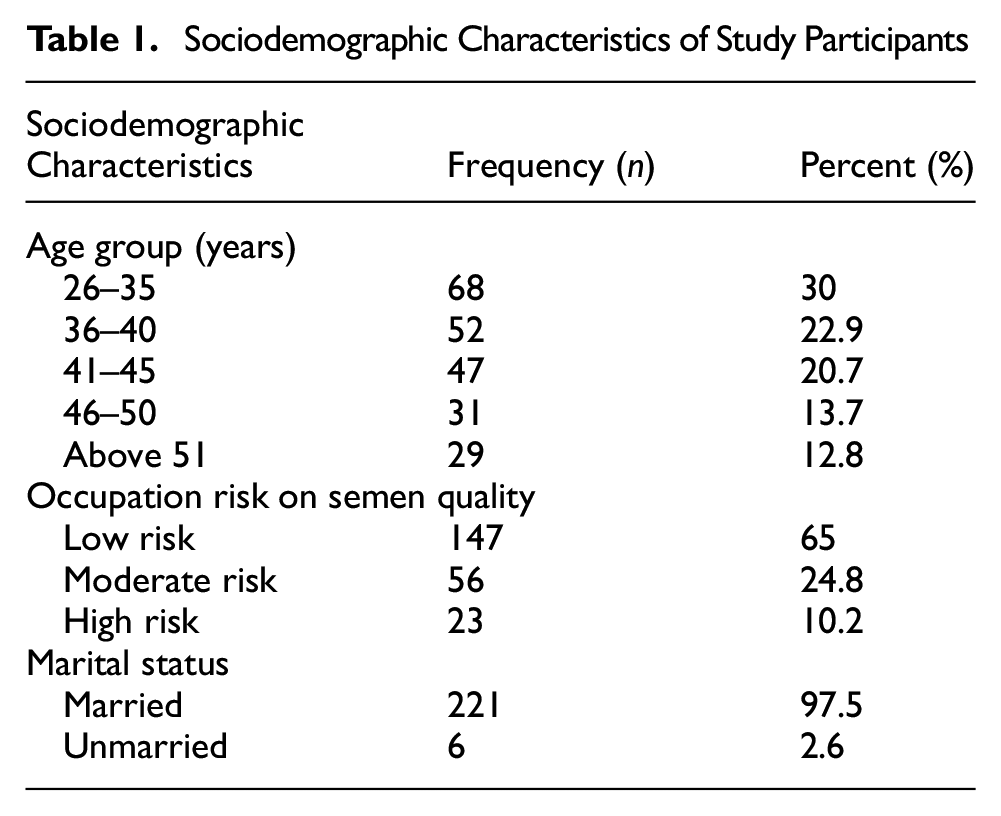
The study recruited 227 participants with a median age of 40 years (IQR: 34–46 years) and ages ranging from 26 to 64 years. The majority of the participants were found in the cohort 26 to 35 years (68, 30.0%), followed by–36 to 40 years (52, 22.9%) and 41 to 45 years (47, 20.7%). Additionally, 24.8% (56) of the participants had occupations classified as moderate risk, while 10.2% (23) had high-risk occupations. Most participants were married (221, 97.4%), as shown in Table 1.
Sociodemographic Characteristics of Study Participants
Prevalence of Elevated Round Cells Among Study Participants
This study found that 19.4% of the participants had elevated round cells, whereas the majority (80.6%) did not (Figure 1).

Prevalence of Elevated Round Cells Among Study Participants Showing the Frequency and Percentage.
Association of Elevated Round Cells with Semen Parameters
TRCC was significantly associated with reduced odds of oligozoospermia (c
Association of TRCC With Semen Motility and Morphology

Association of TRCC With Semen Motility and Morphology


Correlation Between TRCC and semen DFI among study participants. Non-adjusted model for age, waist to hip ratio, alcohol, and smoking (A) and Adjusted model for age, waist to hip ratio, alcohol, and smoking (B).
Discussion
In this study, 19.4% of participants exhibited elevated TRCC, suggesting that one in five men show elevated TRCC, making it a potential biomarker for male infertility.
Though Krishnamurthy and Gowda (2019) reported a 57% prevalence of elevated round cells in semen samples with reduced sperm counts; our study’s lower prevalence suggests a relatively lower burden of spermatogenic failure or genital tract inflammation. Differences in population characteristics, laboratory techniques, and diagnostic criteria may explain this discrepancy. Notably, Krishnamurthy and Gowda (2019) distinguished IGCs (75–80%) from leukocytes (20–25%), emphasizing their distinct clinical implications—spermatogenic failure and inflammation, respectively. In contrast, our study assessed TRCC as a single entity, a common limitation in routine semen analysis. This approach may have underestimated TRCC’s clinical significance, as differentiating leukocytes from IGCs offers deeper insights into underlying pathologies (Dissanayake, 2022). This distinction is critical, as elevated leukocytes may indicate genital tract infections, while high IGC levels may reflect defective spermatogenesis (Long & Kenworthy, 2022; Sharma et al., 2021). The variation in prevalence estimates brings to bear how differences in diagnostic thresholds and cell differentiation methods impact reported TRCC values, limiting direct comparisons across studies.
A significant inverse association between TRCC and oligozoospermia was observed, challenging the conventional assumption that elevated TRCC universally impairs semen quality (Andradc-Rocha & Maya, 2022; Patil et al., 2013). TRCC is frequently implicated in oxidative stress-mediated sperm damage, primarily due to the release of ROS by leukocytes (Agarwal et al., 2014; Ayad et al., 2022). However, emerging evidence highlights the importance of distinguishing leukocytes from IGCs, which constitute 80% to 90% of round cells in semen (Long & Kenworthy, 2022; Patil et al., 2013). Unlike leukocytes, IGCs may not necessarily indicate inflammatory pathology but rather reflect transient disruptions in spermatogenesis. Such disruptions may arise due to testicular stress, subclinical infections, hormonal fluctuations, or delayed germ cell maturation, all of which may be reversible under favorable conditions (Palermo et al., 2016; Patil et al., 2013).
The inverse association between TRCC and oligozoospermia may represent a compensatory response to impaired spermatogenesis (Hasan et al., 2022). Increased IGC levels might reflect an upregulated spermatogenic process attempting to recover sperm production despite underlying dysfunction (La & Hobbs, 2019). This interpretation aligns with Patil et al. (2013), who suggested that elevated IGC levels may serve as a marker of transient spermatogenic disruption rather than irreversible infertility.
The study found no significant correlation between TRCC and sperm DFI. This finding contrasts with previous research suggesting that round cells, particularly leukocytes and IGCs, contribute to oxidative stress-mediated sperm DNA damage (Aitken & Lewis, 2023; Henkel, 2024; Muratori et al., 2019; Sharma et al., 2021). The lack of association in our study suggests that the relationship between TRCC and DNA fragmentation may be complex and influenced by multiple biological and methodological factors.
Leukocytes, especially neutrophils and macrophages, are known to produce significant amounts of ROS through the activation of Nicotinamide Adenine Dinucleotide Phosphate (NADPH) oxidase and myeloperoxidase (Winterbourn et al., 2016). Excessive ROS levels can overwhelm the seminal antioxidant defense system, leading to lipid peroxidation, mitochondrial dysfunction, and direct oxidative modifications to sperm DNA. Studies have demonstrated that leukocytospermia (≥1 × 106 leukocytes/mL) is often associated with increased sperm DNA fragmentation, likely due to excessive ROS generation within the seminal plasma (Alahmar, 2019; Bisht et al., 2017, 2022; Dutta et al., 2019). However, in our study, the TRCC did not correlate with DFI, suggesting that either leukocyte counts in our cohort were below the pathological threshold required to induce oxidative stress, or that seminal antioxidant defenses effectively neutralized ROS effects. This highlights the importance of quantifying leukocyte subpopulations in future studies, as not all leukocytes exert the same oxidative burden on spermatozoa (Micillo et al., 2016; Wang et al., 2025). Beyond leukocytes, accumulating evidence suggests that IGCs, which account for the majority of non-leukocytic round cells in semen, may also contribute to oxidative stress and sperm DNA fragmentation (Muratori et al., 2019; Patil et al., 2013; Takeshima et al., 2018). IGCs are prematurely released from the seminiferous tubules due to impaired spermatogenesis, and their presence in semen is often linked to testicular dysfunction. While they lack phagocytic activity, IGCs have been implicated in pro-oxidant and pro-inflammatory processes that may indirectly contribute to sperm DNA damage. One proposed mechanism involves mitochondrial dysfunction in IGCs, leading to ROS leakage and oxidative stress propagation within the seminiferous tubules. Additionally, apoptotic IGCs can release oxidizing enzymes and inflammatory mediators, further amplifying oxidative damage to neighboring sperm cells (Alfano et al., 2021; Mannucci et al., 2022; Song et al., 2024). Another pathway through which IGCs may influence sperm DNA integrity is through cytokine-mediated inflammation. IGCs have been shown to secrete tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and interleukin-8 (IL-8), which promote a pro-inflammatory testicular microenvironment. This inflammation may impair Sertoli cell function, reducing their ability to provide antioxidant protection to developing spermatozoa. Moreover, the presence of residual histones and incomplete chromatin condensation in IGCs renders sperm DNA more susceptible to oxidative damage. Studies have reported that sperm with defective protamination exhibit increased vulnerability to ROS-induced strand breaks, a key mechanism of DNA fragmentation (Duque & Descoteaux, 2014; Mannucci et al., 2022).
Despite these well-established pathways linking round cells to oxidative stress and sperm DNA fragmentation, our study did not find a significant association between TRCC and DFI. Several factors may account for this discrepancy. First, the relative proportions of leukocytes and IGCs within TRCC were not determined, and if our study population had a higher proportion of IGCs relative to leukocytes, the expected oxidative stress contribution from leukocytes may not have been pronounced. Since IGCs produce lower levels of ROS compared to leukocytes, their impact on sperm DNA integrity may be less immediate or indirect, thus explaining the lack of correlation observed (Nowicka-bauer, 2020; Ollero et al., 2001). Future studies should employ CD45 immunocytochemistry or flow cytometry-based methods to distinguish leukocytes from IGCs, providing more precise assessments of TRCC composition.
Second, seminal antioxidant defense mechanisms may have played a role in mitigating oxidative stress, thereby attenuating any potential impact of TRCC on sperm DNA fragmentation. The seminal plasma contains potent enzymatic antioxidants (superoxide dismutase, catalase, glutathione peroxidase) and non-enzymatic antioxidants (vitamin C, vitamin E, coenzyme Q10), which counteract ROS-induced sperm damage (Alahmar et al., 2021; Kowalczyk, 2022). If participants in our study had sufficient antioxidant capacity, this could have neutralized the oxidative effects of both leukocytes and IGCs, preventing a measurable increase in sperm DNA fragmentation. Future research should incorporate oxidative stress biomarkers (8-hydroxydeoxyguanosine, malondialdehyde, and total antioxidant capacity) to directly assess seminal oxidative status in relation to TRCC (Rocco, 2024).
Finally, individual variability in clinical and lifestyle factors may contribute to the observed findings. Factors such as age, smoking, obesity, diet, occupational toxin exposure, and reproductive tract infections can independently influence both TRCC levels and sperm DNA fragmentation, potentially masking any direct correlation between the two (Pacey, 2010; Szabó et al., 2023). Large-scale, multivariate-adjusted studies are required to disentangle these confounders and clarify the true biological relationship between TRCC and sperm DNA integrity.
Conclusion
This study identifies a notable prevalence of elevated TRCC (19.4%) among Ghanaian males and its inverse association with oligozoospermia, suggesting a potential role in sperm production. However, the lack of a significant correlation between TRCC and sperm DNA fragmentation challenges the assumption that increased round cells universally contribute to sperm DNA damage. These findings highlight the need for more precise diagnostic techniques, such as flow cytometry and the Peroxidase (Endtz) Test, to accurately differentiate leukocytes from IGCs and improve the clinical interpretation of TRCC in male infertility. Given the scarcity of research on male reproductive health in sub-Saharan Africa, this study provides crucial baseline data to guide future research, refine diagnostic approaches, and improve clinical decision-making in reproductive medicine.
Limitations and Recommendations
This study’s reliance on light microscopy for TRCC assessment limited the differentiation between leukocytes and IGCs, potentially affecting result accuracy. Incorporating more advanced techniques such as flow cytometry or the Peroxidase (Endtz) Test could improve precision in distinguishing these cell types and enhance clinical interpretation.
Semen concentration was assessed using the Makler counting chamber, which, while widely used in andrology laboratories, is prone to overestimation due to its shallow depth (10 µm), manual counting variability, and potential inter-observer discrepancies. The WHO 2010 guidelines recommend the Neubauer improved hemocytometer for more precise assessments; however, resource constraints necessitated the use of the Makler chamber in this study.
Footnotes
Acknowledgements
We acknowledge the technical staff of Oak Specialist Hospital who aided with sample collection.
Authors’ Contributions
All authors made a significant contribution to this study, whether that is in conception, data analysis or interpretation. All authors also took part in the drafting, and revising, and approved the publication of this manuscript.
Writing—review & editing: Evans Anokye Kumi, Victor Boachie Owusu, Peter Nyarko Coffie, Ebenezer Kojo Addae, Rex Kwadwo Mawuli Djokoto, Kweku Bedu-Addo, John Asiedu Larbi
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
