Abstract
Sexual dysfunction, which is increasingly recognized as a cause of male infertility, may be influenced by environmental factors such as phthalic acid diesters (phthalates) and prenatal androgen exposure. This study aimed to investigate the association between male sexual function and recent exposure to phthalates and prenatal testosterone. The cross-sectional study included 177 males seeking fertility treatment. The index-to-ring-finger length ratio (2D:4D) was calculated as a measure of prenatal androgen exposure. To evaluate recent phthalate exposure, urinary concentrations of seven phthalate metabolites were measured. Their associations with International Index of Erectile Function (IIEF) scores were analyzed using generalized linear models. One standard deviation increase of mono-methyl phthalate was negatively associated with IIEF domains A (erectile function; exponentiated coefficient, 0.95; exponentiated 95% confidence interval, CI, [0.91, 0.98]) and D (intercourse satisfaction; exponentiated coefficient, 0.91; exponentiated [0.85, 0.97]). While mono-butyl phthalate was positively associated with domain A, mono-ethyl phthalate, monobenzyl phthalate, and the molar sum of di (2-ethylhexyl) phthalate metabolites showed no significant association with IIEF domain scores. When the serum testosterone concentration was included as an exposure variable, the overall association between urinary phthalates and IIEF domain scores remained similar. The left- and right-hand 2D:4D ratios did not show significant associations with any IIEF domain scores. Recent phthalate exposure may affect male sexual function by decreasing testosterone levels and other potential mechanisms.
Keywords
Introduction
The global total fertility rate (TFR) has continuously declined for over five decades and reached 2.3 in 2021 (The World Bank, 2025). Numerous countries and regions, including the European Union, the United States, Japan, Hong Kong, Singapore, and China, exhibit TFR levels significantly below the population replacement level. Extensive research has explored the impact of social and economic factors, along with the widespread availability of effective contraceptive methods and induced abortions, on the decline in the TFR. However, it has been posited that alterations in human fecundity due to biological factors may also play a role.
According to the World Health Organization (2023), infertility is diagnosed when a couple engaging in normal sexual intercourse fails to conceive within 12 months after birth. The estimated lifetime prevalence of infertility was 17.5% in 2022. Notably, approximately 20% of infertility cases are attributed solely to male factors, whereas in 30% to 40% of cases, both male and female factors contribute (Thonneau et al., 1991). In other words, men were responsible for infertility in approximately half of all infertile couples. A 2015 nationwide survey in Japan revealed that testicular factors account for 82.6% of male infertility, with sexual dysfunction and seminal tract obstruction contributing 13.5% and 3.9%, respectively (Yumura et al., 2018).
Although the exact causes of testicular factors are not fully understood (Punab et al., 2017; Yumura et al., 2018), the testicular dysgenesis syndrome (TDS) hypothesis by Skakkebaek et al. (2001) suggests a potential connection between issues in embryonal programming and gonadal development in the womb, which may contribute to conditions such as testicular cancer, hypospadias, cryptorchidism, decreased testosterone production, and impaired spermatogenesis (Skakkebaek et al., 2001, 2016). Lifestyle and environmental factors are anticipated to influence fetal gonad development during early pregnancy, resulting in decreased Leydig cell function and disturbed Sertoli cell function, ultimately causing TDS.
The primary mechanism by which TDS affects fertility is through impaired spermatogenesis. However, it may also affect fertility through the onset of sexual dysfunction. TDS reduces testosterone production, which is crucial for maintaining normal male sexual function. Existing literature lacks reports from this perspective. The significance of sexual dysfunction in male infertility is underscored by the fact that this percentage has increased from 3.3% in 1998 to 13.5% in 2015 in Japan (Shirai & Miura, 1998; Yumura et al., 2018). Male sexual dysfunction included erectile dysfunction, diminished libido, and ejaculatory disorders. A comprehensive study of a representative population sample of 1,410 adult males in the United States revealed various risk factors for male sexual dysfunction, including advanced age, health conditions such as diabetes, heart disease, and obesity, and psychological factors that include stress, anxiety, and depression (Laumann et al., 1999). This report implies that both physiological and psychological statuses are independent factors that affect sexual functioning.
The length ratio of the index finger to the ring finger (2D:4D) may indicate prenatal androgen exposure (Berenbaum et al., 2009; Manning et al., 1998). Lower ratios were linked to higher exposure, with boys showing lower ratios than girls by the age of 2 years. Notably, considerable within-group variability and between-group overlap were observed, indicating that the 2D:4D ratio is not a precise marker of individual differences in prenatal androgen exposure (Berenbaum et al., 2009). It serves as a retrospective marker of the average differences in prenatal androgen action between groups and correlates with specific behaviors and characteristics within a group (Breedlove, 2010). Researchers leveraging 2D:4D as a correlate have found evidence that prenatal androgen exposure influences various human diseases, including schizophrenia (Collinson et al., 2010; Y. Han et al., 2020), cancer (Fonseca et al., 2022), and ischemic heart disease (Manning, Bundred, et al., 2019), as well as behaviors such as autism (Manning et al., 2001), aggression, and risk-taking (Breedlove, 2010). Despite several reports on the relationship between 2D:4D ratios and semen parameters, the results have been mixed and inconclusive, showing a positive (Bang et al., 2005), negative (Auger & Eustache, 2011; Manning et al., 1998), or no relationship (Firman et al., 2003). Our recent examination of infertile Japanese men revealed no clear relationship between 2D:4D ratios and semen parameters (Uchida et al., 2023).
Phthalic acid diesters (phthalates), a class of synthetic and multifunctional chemicals used in a wide array of consumer and industrial products, exhibit well-documented antiandrogenic activities in rodent studies (Johnson et al., 2012). Human exposure to phthalates is widespread throughout life, with multiple reports indicating their impact on semen parameters and anogenital distance, another marker of prenatal androgen exposure (Radke et al., 2018). However, few studies have investigated the association between phthalate exposure and male sexual function (Lopez et al., 2017).
To assess the impact of prenatal testosterone and current phthalate exposure on male sexual function, we investigated the relationship between 2D:4D and urinary phthalate concentrations and the International Index of Erectile Function (IIEF) domain scores in Japanese male patients seeking fertility treatment.
Materials and Methods
This study used samples and information collected from the Interdisciplinary Investigation on Technology, Environment, and Fertility (IITEF) project (Uchida et al., 2023). The IITEF project was a cross-sectional survey. Participants were recruited from two hospitals in Tsukuba and Tokyo, Japan, between September 2020 and January 2021. The eligibility criteria were male patients aged 20 to 55 years who could provide urine and semen specimens on the same day and who had never undergone vasectomy. As previously described (Uchida et al., 2023), 198 participants responded to a questionnaire on age, height, and weight, and 15 questions on the IIEF (Rosen et al., 1997). The minimum sample size was set as 150, considering previously reported correlations between sperm concentration and phthalate exposure (Pant et al., 2008). The palms of both hands were scanned at both clinics using an EP-882AW scanner (SEIKO EPSON Co., Ltd., Suwa, Japan) at 300 dots per inch and saved as JPG files. The participants provided urine and semen specimens on the same day as the hand scans. Venous blood was collected on the same day (N = 23), 15 days later (N = 1), or 3 to 1,757 days prior to recruitment (N = 154). Serum testosterone concentration was quantified using an electrochemiluminescence immunoassay (ECLIA; SRL Co., Ltd., Tokyo, Japan) using an ECLIA. Blood collection times were recorded and ranged from 8:30 to 15:00. However, the diurnal variation in testosterone levels is a recognized limitation, as discussed below.
The scanned images of the participants’ palms were sent to the University of Tokyo. The lengths of the second and fourth digits were measured in pixels to two decimal places by an investigator using MATLAB Image Viewer. Preliminary to the present study, the researchers conducted an exploratory analysis to assess the variability of the 2D:4D ratio measurements between the two researchers. The intraclass correlation coefficient was .9732 when two researchers independently scanned the palms of the same seven subjects.
Urine specimens were stored at −80°C at each hospital and transferred to the University of Tokyo, where specific gravity (SG) was measured using a pocket refractometer (Atago Co., Ltd., Tokyo, Japan). The urine specimens were sent to IDEA Consultations, Inc. (Yaizu, Japan), where the urinary concentrations of phthalate metabolites were quantified using liquid chromatography-tandem mass spectrometry. The seven metabolites measured were mono-methyl phthalate (MMP), mono-ethyl phthalate (MEP), mono (2-ethylhexyl) phthalate (MEHP), mono (2-ethyl-5-oxohexyl) phthalate (MEOHP), mono (2-ethyl-5-hydroxyhexyl) phthalate (MEHHP), mono-butyl phthalate (MBP), and monobenzyl phthalate (MBzP). The limits of detection (LODs) for MMP, MEP, MBzP, MEHP, MEHHP, and MEOHP were 0.52, 0.51, 0.12, 0.34, 0.11, and 0.074 ng/mL, respectively. Urinary concentrations below the LOD were substituted with the LOD value divided by the square root of the two values. The urinary concentrations of these phthalate metabolites were adjusted for SG using a previously published equation (Moore et al., 2018). The urinary concentrations of phthalate metabolites mentioned in this study were SG-corrected values.
This study was performed with formal approval from the Institutional Research Ethics Committees of the Graduate School of Medicine and Faculty of Medicine, the University of Tokyo (2020057NI-(2)), Tsukuba Gakuen Hospital (20-07), and the International University of Health and Welfare (20-S-8).
Statistical Analyses
Responses to the 15-item IIEF questions were used to calculate five domain scores: domain A for erectile function (sum of items 1, 2, 3, 4, 5, and 15), domain B for orgasmic function (items 9 and 10), domain C for sexual desire (items 11 and 12), domain D for intercourse satisfaction (items 6, 7, and 8), and domain E for overall satisfaction (items 13 and 14). The maximum scores for domains A to E are 30, 10, 10, 15, and 10, respectively (Rosen et al., 1997).
The molar sum of di (2-ethylhexyl) phthalate metabolites (DEHP sum) was calculated by summing the moles of MEHP, MEHHP, and MEOHP. Spearman’s correlation coefficients between the 2D:4D ratio of the right and left hands, IIEF domain scores, testosterone, SG-adjusted urinary concentrations of phthalate metabolites (MMP, MEP, MEHP, MEOHP, MEHHP, MBP, and MBzP), and the DEHP sum were calculated (Supplemental Table 1). Generalized linear models (GLMs) with Poisson distribution were constructed using each IIEF domain score as an outcome, resulting in five different GLMs. For each GLM, all exposure variables, that is, 2D:4D of the left and right hands and urinary MMP, MEP, MBP, MBzP, and DEHP sum, and all covariates of age, body mass index (BMI), smoking, drinking, and sleep duration, were included (see Supplemental Tables 2–4). Serum testosterone levels were not included in the covariates of the GLM model shown in Supplemental Tables 2 and 4. However, they were included in Supplemental Table 3 for the reasons discussed below. All the exposure variables were simultaneously included in each GLM. The Variance Inflation Factor (VIF) was calculated to confirm the absence of multicollinearity in the GLMs. In the five GLMs, the VIFs of all the variables were <2 (data not shown), suggesting that there were no multicollinearity issues. Covariates were selected because they have been associated with erectile function in previous studies (McVary et al., 2001; Nicolosi et al., 2003; Pizzol et al., 2020; F. Zhang et al., 2022). As the urinary concentrations of phthalates were skewed (Supplemental Figure 1), they were log-transformed for inclusion in the GLMs. Coefficients and 95% confidence intervals (CIs) per standard deviation (SD) increase for each exposure variable were calculated and exponentiated for data presentation. Adjustments for multiple testing were not performed in this study because it was an exploratory analysis of any potential association between 2D:4D or phthalate exposure and male sexual function.
We conducted two sets of sensitivity analyses. The first set was the same as the GLMs of the primary analyses, except that serum testosterone levels were adjusted (Supplemental Table 3). The second set used creatinine-adjusted instead of SG-adjusted phthalate concentrations as explanatory variables (Supplemental Table 4). All statistical analyses were performed using R ver. 4.4.2 (R Core Team, 2025).
Results
In total, 198 participants were enrolled in the IITEF project. The analytical sample comprised 177 men who provided complete data. The basic characteristics of the participants are presented in Table 1. The mean (SD) 2D:4D ratios were 0.937 (0.031) and 0.950 (0.032) for the left and right hands, respectively (Table 1 and Supplemental Figure 2). Summary statistics and distributions of the IIEF domain scores are shown in Table 2 and Figure 1.
Age, Anthropometry, Lifestyle Characteristics, Serum Testosterone, 2D:4D, and Urinary Phthalate Concentrations of 177 Men in the IITEF Study, 2020 to 2021

Note. DEHP = di (2-ethylhexyl) phthalate; IITEF = Interdisciplinary Investigation on Technology, Environment, and Fertility; IQR = interquartile range; LOD = limit of detection; MBP = mono-butyl phthalate; MBzP = monobenzyl phthalate; MEHHP = mono (2-ethyl-5-hydroxyhexyl) phthalate; MEHP = mono (2-ethylhexyl) phthalate; MEOHP = mono (2-ethyl-5-oxohexyl) phthalate; MEP = mono-ethyl phthalate; MMP = mono-methyl phthalate; SD = standard deviation; BMI = body mass index.
Adjusted for specific gravity of urine.
The LODs were 0.52 ng/mL for MMP, 0.51 ng/mL for MEP, 0.12 ng/mL for MBzP, 0.34 ng/mL for MEHP, 0.11 ng/mL for MEHHP, and 0.074 ng/mL for MEOHP. A urinary concentration below LOD was substituted with the LOD values divided by the square root of two.
IIEF Domain Scores of 177 Men in the IITEF Study, 2020 to 2021

Note. IIEF = International Index of Erectile Function; IITEF = Interdisciplinary Investigation on Technology, Environment, and Fertility; IQR = interquartile range; SD = standard deviation.
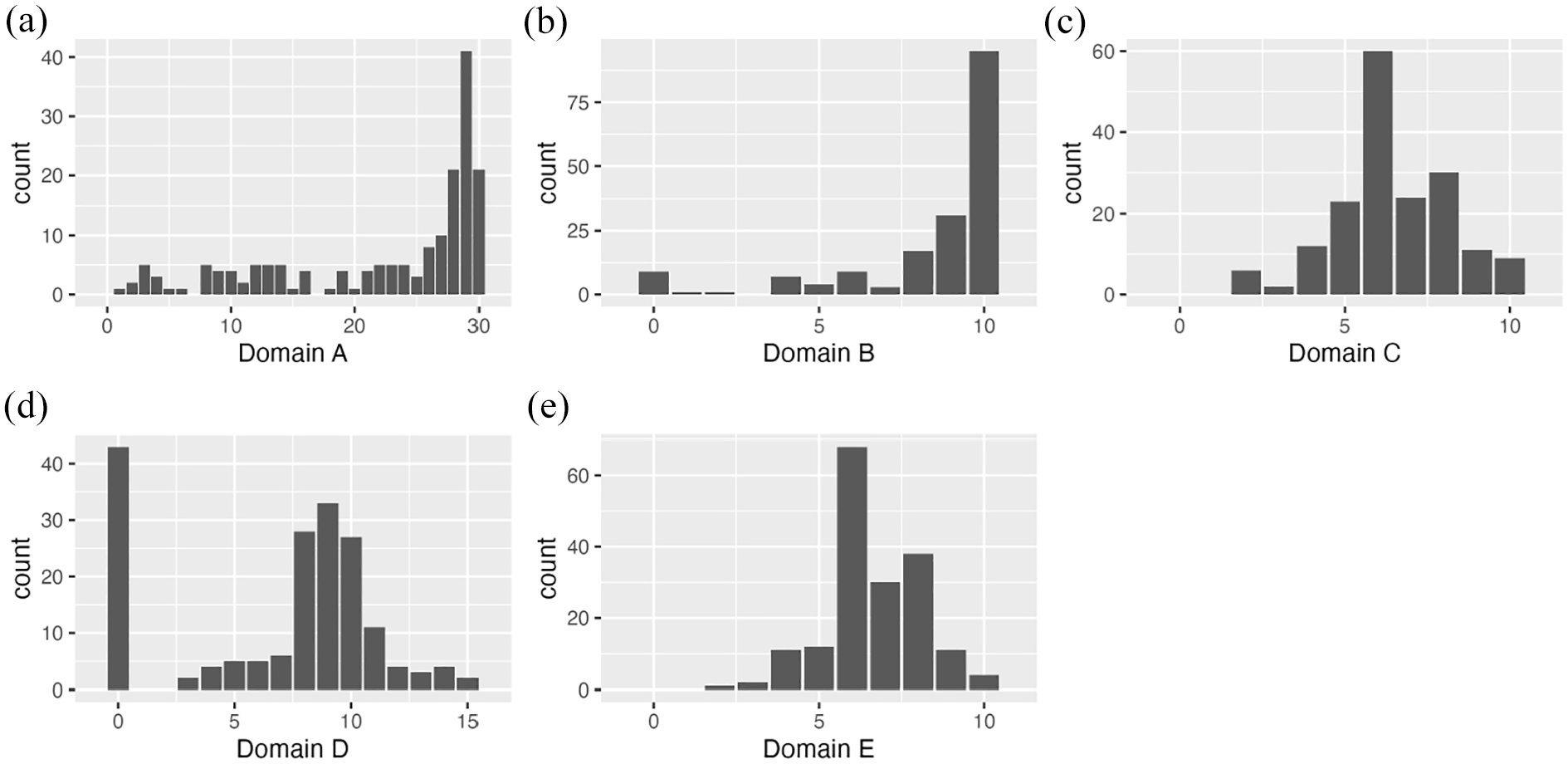
Histograms Showing the Distribution of IIEF Domain Scores of (a) A (Erectile Function), (b) B (Orgasmic Function), (c) C (Sexual Desire), (d) D (Intercourse Satisfaction), and (e) E (Overall Satisfaction) of 177 Men in the IITEF Study, 2020 to 2021
Spearman’s correlation analysis revealed a negative correlation between urinary MMP and the IIEF domains A, D, and E (Supplemental Table 1). Urinary MBzP was negatively correlated with IIEF domains D and E. Urinary MEP, MEHP, MEOHP, MEHHP, MBP, and DEHP sum levels were not significantly correlated with any of the IIEF domains. Left-hand 2D:4D showed weak positive correlations with IIEF domains B and C, whereas right-hand 2D:4D showed no correlation with any domain scores. Serum testosterone positively correlated with IIEF domains A and D, and negatively correlated with urinary MMP, MEOHP, MEHHP, MBzP, DEHP sum, and both 2D:4D of the left and right hands.
In the GLM analysis, in which age, BMI, smoking, alcohol consumption, and sleep duration were controlled, the urinary MMP concentration showed a significant negative association with domain A (exponentiated coefficient 0.95, exponentiated 95% CI [0.91, 0.98]) and domain D (0.91, [0.85, 0.97]) scores (Figure 2 and Supplemental Table 2). No significant associations were observed with domains B, C, and E. Urinary MBP concentration showed a significant positive association with domain A (1.06, [1.01, 1.10]) and no significant associations with other domains. The MEP, MBzP, and DEHP sums were not significantly associated with the IIEF domain scores (Supplemental Table 2). The predicted values of the GLMs are shown along with scatter plots of the domain scores and explanatory variables (Supplemental Figures 3–7). When the serum testosterone concentration was adjusted, the overall associations between urinary phthalates and IIEF domain scores remained similar (Supplemental Table 3). The use of creatinine-adjusted phthalate concentrations instead of SG-adjusted values did not alter the overall results (Supplemental Table 4). The left- and right-hand 2D:4D ratios were not significantly associated with the IIEF domain scores (Figure 2 and Supplemental Table 2).

Exponentiated Coefficients and Their 95% CIs of the Five Separate GLMs With Each of the IIEF Domain Score as an Outcome Variable (N = 177)
Discussion
This study explored the effects of prenatal androgen and phthalate exposure on male sexual function. We used 2D:4D in both hands as a marker of prenatal testosterone effects, and urinary phthalate concentrations as markers of recent phthalate exposure. We also examined the relationship between blood testosterone levels, which directly affect sexual function, 2D:4D ratios, and urinary phthalate concentrations.
Among the urinary phthalates measured, the urinary MMP concentration was negatively correlated with IIEF domains A, D, and E (Supplemental Table 1). The negative association between MMP and domains A and D was also confirmed by the GLM, suggesting that exposure to MMP or its parent compound, DMP, may negatively affect erectile function and sexual satisfaction (Supplemental Table 2). The positive association demonstrated in the GLM between urinary MBP and IIEF domain A may be attributable to the statistical effect of other phthalate metabolites because there was no significant correlation between MBP and domain A (rho = −.002, p > .05; Supplemental Table 1). Although there were no multicollinearity issues, as judged by the VIFs, it is likely that the inclusion of other phthalates in the GLM affected the MBP coefficient. When only age, BMI, smoking, drinking, sleep duration, and left- and right-hand 2D:4D were adjusted, urinary MBP was not associated with domain A (exponentiated coefficient 1.02, exponentiated 95% CI [0.99, 1.06]; data not shown).
A negative correlation was observed between urinary MMP and serum testosterone concentrations (Supplemental Table 1), suggesting that MMP may adversely affect erectile function and sexual satisfaction by reducing serum testosterone levels. The observed significant negative correlation between urinary MMP and serum testosterone concentrations aligns with previous studies that have demonstrated a negative relationship between urinary phthalate and serum testosterone concentrations. A study of 2,208 members of the general U.S. population indicated that elevated concentrations of multiple urinary phthalates were associated with lower total serum testosterone concentrations in men and women in various age groups (Meeker & Ferguson, 2014). In males, DEHP metabolites (MEHP, MEHHP, MEOHP, mono-2-ethyl-5-carboxypentyl phthalate, etc.) and MBP in the 40- to 60-year age group and DEHP metabolites in the 6- to 12-year age group were significantly associated with decreased serum testosterone concentrations. Notably, no association was observed between the MMP and testosterone levels in a U.S. study (Meeker & Ferguson, 2014). Another study involving 589 husbands of pregnant Greenlandic, Polish, and Ukrainian women investigated the association between serum phthalate concentrations and reproductive function. The results indicated that DEHP and DiNP metabolites (such as mono-(carboxyoctyl phthalate and mono-isononyl phthalate) were negatively correlated with serum testosterone concentrations (Specht et al., 2014). This study did not assess MMP. Conversely, a study examining the correlation between phthalate exposure and reproductive function in 232 Chinese men revealed a strong association between urinary MBP and sperm concentration. No significant correlation was found between urinary phthalate metabolites, including DEHP metabolites, and serum testosterone concentrations (X. Han et al., 2014). This study did not include MMP analysis. The specific phthalates that demonstrated significant correlations varied across the studies. One potential explanation for this discrepancy is that each phthalate metabolite may exert varying effects on testosterone concentrations and sexual function, which are contingent on the racial group under examination. A study of 3,746 U.S. men ≥20 years of age examined the association between urinary phthalate metabolites and erectile dysfunction in each of three racial/ethnic groups: non-Hispanic white, non-Hispanic black, and Mexican American (Lopez et al., 2017). In Mexican American men, MBP and MBzP concentrations were inversely correlated with the incidence of erectile dysfunction. In non-Hispanic Black men, MEHHP concentrations were inversely correlated with the incidence of erectile dysfunction. In contrast, no association was observed between phthalate metabolites and erectile dysfunction in non-Hispanic Caucasian men. Urinary MMP concentrations were not associated with erectile dysfunction in any racial/ethnic group, and testosterone levels were not measured. Therefore, given the potential for racial- and ethnic-group-specific effects of each phthalate metabolite on testosterone levels and sexual function, a larger study is needed to confirm these results in each racial and ethnic group, including the Japanese population, and to investigate the underlying causes of these differences. It is also essential to examine the potential factors contributing to these differences between racial and ethnic groups, such as variations in phthalate metabolic pathways and exposure routes.
Interestingly, in GLM adjusting for serum testosterone concentration, MMP was still significantly negatively correlated with both IIEF domains A and D (Supplemental Table 3), suggesting the potential of MMP to adversely affect male sexual function through an unidentified mechanism unrelated to a reduction in serum testosterone concentration. We speculate that other mechanisms, such as neurotoxicity and inhibition of angiogenesis mediated by increased oxidative stress, may be involved (Tseng et al., 2013; Y. Zhang et al., 2024). These possibilities merit further investigation in future studies.
In the GLMs with or without testosterone adjustment, the 2D:4D ratio was not associated with any IIEF domain score. Although both the left- and right-hand 2D:4D ratios demonstrated negative correlations with testosterone concentrations, it is unlikely that a higher 2D:4D ratio is linked to lower sexual function. Although several reports indicate that 2D:4D ratios do not correlate with testosterone concentration (Auger & Eustache, 2011; Muller et al., 2011), the existing literature on the relationship between 2D:4D ratios and male sexual function is notably scarce. In a study comparing the proportion of men in an age group receiving male hormone replacement therapy, a higher proportion of men with higher 2D:4D ratios received male hormone replacement therapy than those with lower 2D:4D ratios, suggesting that men with higher 2D:4D ratios may have higher rates of hypogonadism, including sexual dysfunction (Manning, Cook, & Crewther, 2019).
The strengths of this study include its focus on the impacts of prenatal testosterone exposure and current phthalate exposure, and its detailed assessment of sexual function. Another strength was the relatively large number of participants (N = 177). However, this study was limited by its cross-sectional design. Another limitation is that the participants were male outpatients seeking fertility treatment, which may reflect the results from a population with lower testosterone levels and IIEF scores than the general healthy population. It is possible that phthalate exposure differs between populations. However, it is unknown how these sample characteristics may have biased the results. Further studies are required to determine whether similar results can be obtained in individuals with and without fertility issues. It is noteworthy that blood samples were not collected at a fixed time from all participants as men’s serum testosterone levels exhibit a circadian rhythm, peaking in the morning and decreasing in the evening (Brambilla et al., 2009). Collecting blood samples exclusively in the morning would have facilitated the assessment of the relationship between testosterone levels and other variables.
Conclusion
The results of our study indicate that urinary phthalate metabolites may be associated with sexual function in adult Japanese men, whereas no significant relationship was observed between 2D:4D ratios and sexual function. In adult males, serum testosterone concentration, which greatly influences sexual function, may be more strongly influenced by recent endocrine disruptors, including phthalates, than by environmental factors during the embryonic period, as proposed by the TDS hypothesis. Further research is required to examine the effects of each phthalate, as well as the effects of other endocrine disruptors, such as isoflavones, on a larger scale. Furthermore, it is essential to consider the influence of demographic variations such as race and fertility status on the effects of environmental exposures. If the above are clarified, it is hoped that measures to reduce exposure to certain phthalates and endocrine disruptors will reduce the incidence of male sexual dysfunction, which in turn could contribute to reducing infertility.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883251352972 – Supplemental material for Association of the Ratio of Second to Fourth Digit Length and Phthalate Exposure With Sexual Function in Japanese Men Seeking Fertility Treatment
Supplemental material, sj-docx-1-jmh-10.1177_15579883251352972 for Association of the Ratio of Second to Fourth Digit Length and Phthalate Exposure With Sexual Function in Japanese Men Seeking Fertility Treatment by Kazumitsu Yamasaki, Masahiro Uchida, Yuki Mizuno, Teruaki Iwamoto and Shoko Konishi in American Journal of Men's Health
Supplemental Material
sj-docx-2-jmh-10.1177_15579883251352972 – Supplemental material for Association of the Ratio of Second to Fourth Digit Length and Phthalate Exposure With Sexual Function in Japanese Men Seeking Fertility Treatment
Supplemental material, sj-docx-2-jmh-10.1177_15579883251352972 for Association of the Ratio of Second to Fourth Digit Length and Phthalate Exposure With Sexual Function in Japanese Men Seeking Fertility Treatment by Kazumitsu Yamasaki, Masahiro Uchida, Yuki Mizuno, Teruaki Iwamoto and Shoko Konishi in American Journal of Men's Health
Footnotes
Acknowledgements
The authors thank all participants and hospital staff members for their cooperation in this study. We thank Mr. Tatsuji Ihana and Ms. Fumiko Kariya for helping with data collection, and Prof. Jun Yoshinaga for his help in designing the study. We would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Ethical Considerations
This study was conducted after the formal approval was obtained from the Institutional Research Ethics Committees of the Graduate School of Medicine and Faculty of Medicine, the University of Tokyo (2020057NI-(1)) on September 16, 2020, Tsukuba Gakuen Hospital (20-07) on July 27, 2020, and the International University of Health and Welfare (20-S-8) on September 11, 2020. All participants provided written informed consent prior to enrolment in the study.
Consent to Participate
Written informed consent was obtained from all the participants upon enrollment.
Author Contributions
Kazumitsu Yamasaki, Yuki Mizuno, Teruaki Iwamoto, and Shoko Konishi contributed to the study’s conception and design. Data were acquired by Kazumitsu Yamasaki, Teruaki Iwamoto, Masahiro Uchida, and Shoko Konishi. Shoko Konishi and Yuki Mizuno analyzed and interpreted the data. The first draft of the manuscript was written by Kazumitsu Yamasaki and Shoko Konishi. All authors revised and agreed on the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) Topic-Setting Program to Advance Cutting-Edge Humanities and Social Sciences Research, Global Initiatives, Grant Number JSPS00119217822 (P.I.: Shoko Konishi) and KAKENHI 25H01377 (P.I.: Shoko Konishi).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
