Abstract
Variations in the estrogen receptor genes, particularly the Xba1 (rs9340799) SNP in the ESR1 gene, may influence the effects of estrogen on male fertility. Results from previous studies on this SNP for male infertility have been inconclusive. This review aimed to determine the association of ESR1 Xba1 (rs9340799) with male infertility. Relevant case–control studies published in English were searched from Google Scholar, Embase, Scopus, Web of Science, Cochrane Library, and PubMed using keywords of ESR, polymorphism, and male infertility. Studies on animals, reviews, and abstracts were excluded. Pooled odds ratios (ORs) were calculated for four genetic models, with heterogeneity assessed by I2. A fixed or random effect model was applied based on I2, and trial sequential analysis (TSA) was conducted with 5% significance for type I error and 95% power. ESR1 expression levels were examined in testes, hypothalamus, prostate, and pituitary using GTEx Analysis. Nine studies (four Asian, four Caucasian, one African) met the criteria. The G allele was protective against infertility overall (OR: 0.80; 95% confidence interval [CI] = [0.70, 0.92]) and in Caucasian men (OR: 0.71; 95% CI = [0.54, 0.92]). Lower infertility risk was observed in Asian (AA vs. GG OR: 0.65; 95% CI = [0.43, 0.98]) and Caucasian men (OR: 0.49; 95% CI = [0.28, 0.83]). TSA indicated no further studies are likely to change these results. No significant change in expression of ESR1 was observed due to this SNP. The present meta-analysis suggests that the SNP Xba1 (rs9340799) in ESR1 is protective against male infertility, with current data sufficient to confirm these findings.
Introduction
Infertility is the failure to achieve pregnancy after 12 months or more of regular unprotected sexual intercourse with the same partner (Sharma, 2017). Male factor solely or partially contributes to 20% to 50% of infertility cases (Hamada et al., 2012). The origins of primary male infertility may start in childhood due to infections such as mumps and measles, testicular injuries, chemical, radiation, heat and medicinal exposure, pubertal disorders, personal habits, other physiological and metabolic conditions, and environmental hazards (Kolesnikova et al., 2015). These internal and external factors may affect male fertility in previously fertile men and cause secondary male infertility. Genetic abnormalities are one of the major contributing factors to male infertility (Ge et al., 2014). Genetic male infertility disorders include chromosomal alterations, Y-chromosome microdeletions, gene mutations, and epigenetic disorders (Neto et al., 2016).
The testes produce aromatase, which converts testosterone into estrogens. As a result, there are relatively high levels of estrogen at the site of sperm production even than serum levels in females (Dostalova et al., 2017; Rivera-Diaz et al., 2021). Role of the estrogen in sperm production and male fertility is evident and is under investigation.
Estrogen receptors (ERs) belong to the superfamily and are members of the nuclear receptor (NR) which mediates the pleiotropic effects of estrogen in a vast range of physiological and developmental measures playing a vital part in mediating estrogen action on the targeted sites of tissues. Two further subtypes of ERs are known: ER-a encoded by the ESR1 gene and ER-b encoded by the ESR2 gene (Zalata et al., 2014). ESR1 is located on chromosome 6q25 and consists of eight exons and seven introns, with an overall length of 140 kb (Ponglikitmongkol et al., 1988). It encodes for the estrogen receptor alpha (ERα), responsible for mediating physiological responses to estrogen and reported to be present in testes (Vu et al., 2023).
Among genetic mutations, single-nucleotide polymorphisms are important for associating with various diseases at the population level. Several reports are published on the association of a SNP 351 A/G (rs9340799) in ESR1 and male infertility. These studies have contrasting results within the same population, different populations, and types of infertility. The present meta-analysis is conducted to unravel the association of SNP 351 A/G in ESR1 with male infertility. The trial sequential analysis (TSA) was also carried out to determine the power of the analysis.
Materials and Methods
Sear Strategies
A comprehensive search was conducted on Google Scholar, Embase, Scopus, Web of Science, Cochrane Library, and PubMed for articles using Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) as presented in Figure 1. The keywords used during the search were ESR, polymorphism, and male infertility. In addition, the reference lists of articles were thoroughly checked to identify any relevant studies.
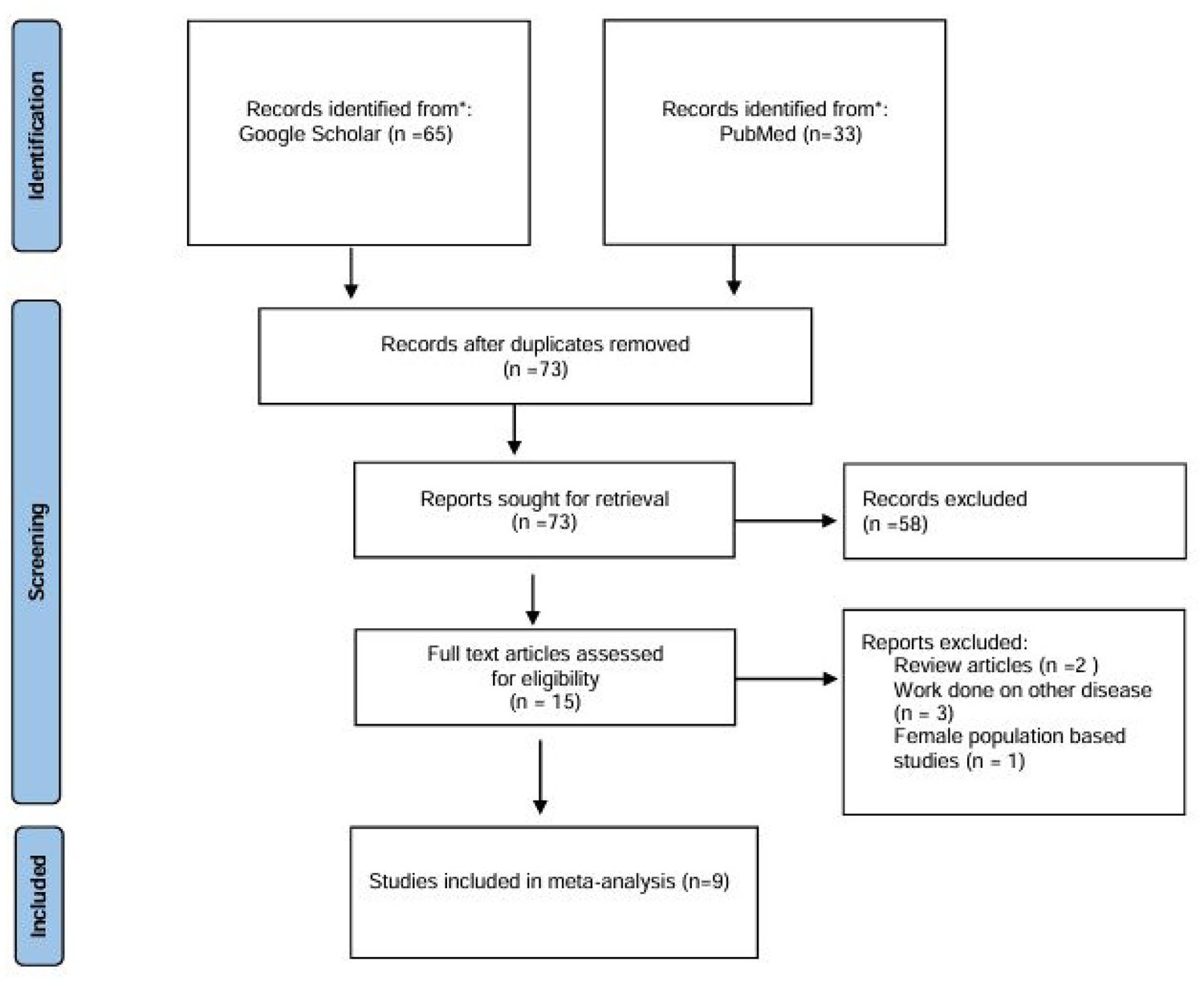
Flowchart Showing the Selected Studies
Inclusion and Exclusion Criteria
The studies included full-text articles published in English with genotypic and allelic data for both cases (infertile men) and control (fertile) groups sufficient to calculate odds ratios (ORs) and confidence intervals (CIs). Studies conducted on animal models, reviews, and abstracts were excluded. In addition, the studies were considered for inclusion if the men in the control group had at least one child and the case group had unknown causes of infertility. Two authors (HQ and MI) independently screened studies for inclusion and any discrepancy was resolved by discussion.
Data Extraction Method
The data extracted from included studies were the name of the first author, country name, ethnicity, age, and inclusion and exclusion criteria of subjects, genotyping method, and genotypic and allelic frequencies.
Quality Assessment
Assessment of the quality of studies was done based on the quality of genetic association studies (Q-Genie) tool (Version 1.1) (Sohani et al., 2015). It evaluates studies based on 11 items and with a maximum score of 1 to 7 (poor to excellent) each. The questions are on adequacy of the presented hypothesis and rationale, classification of outcomes, description of comparison groups, classification of genetic variants, disclosure of potential sources of bias, adequacy of study power, description of planned analyses, and statistical methods employed. The aggregate score of each study if ≤35 indicate poor quality, >35 and ≤45 indicate moderate quality, and >45 indicate good quality studies.
Statistical Analysis
The statistical analysis for the meta-analysis was conducted using Review Manager (version 5.0). The strength of the association of ESR1SNP Xba1 was determined by calculating the OR and 95% CI. Pooled ORs were calculated for four genetic models: 1. Allelic model: A allele versus G allele; 2. Genotypic model: AA versus GG; AG + GG versus AA; and AA + AG versus GG for XbaI. The heterogeneity of the studies was assessed using I2, and the fixed effect or random effect model was chosen based on whether I2 was <50% or >50%, respectively (Supplementary Figure 1 to Supplementary Figure 26). Sensitivity analysis was conducted to assess the robustness of the pooled effect size and heterogeneity (I2). A leave-one-out approach was applied, where each study was sequentially excluded, and the meta-analysis was recalculated to evaluate the influence of individual studies. Publication bias was assessed using both graphical and statistical methods. Funnel plots were visually inspected for symmetry, with asymmetry suggesting potential small-study effects or selective reporting. Egger’s regression asymmetry test was performed to detect potential publication bias in studies evaluating the ESR1 Xba1 polymorphism and male infertility. A p-value <.05 was considered indicative of significant publication bias.
Trial Sequential Analysis
TSA was conducted by using Trial Sequential Analysis Viewer software, version 0.9.5.10 Beta, Copenhagen Trial Unit, Centre for Clinical Intervention Research, Rigshospitalet, 2016 (Gordon Lan & DeMets, 1983) by using the method of Meng et al. (2018). A significance of 5% was selected for type I error, whereas 95% power was used to calculate the required sample size and set TSA boundaries.
In Silico Analysis of ESR1 Expression
The influence of polymorphisms on ESR1 expression levels was analyzed by using GTEx Analysis Release V8 (dbGaP Accession phs000424.v8.p2) data (Gibson, 2015).
Results
Characteristics of Eligible Studies
The initial search yielded 98 studies, of which nine met the selection criteria for inclusion in the meta-analysis (Figure 1). These included four studies from Asian populations, four from Caucasian populations, and one from an African population. A total of seven studies reported low sperm count (azoospermia and oligospermia). Two genotyping methods, polymerase chain reaction (PCR)-restriction fragment length polymorphism (RFLP) and TaqMan, were utilized to identify the SNP (Table 1).
Characteristics of the Studies Included

Note. AZ/ISO = azoospermia or idiopathic severe oligospermia; AZ/OS = azoospermia or oligospermia; HWE = Hardy–Weinberg equilibrium; IMO = idiopathic moderate oligospermia; NA = not available; NOA = non-obstructive azoospermia; SD = standard deviation; SO = severe oligospermia; QA = quality assessment.
Qualitative Analysis of Studies
The qualitative characteristics of studies are presented in Table 1.
Sample Size
The sample size varied from 29 to 204 infertile men (Lazaros et al., 2010; Meng et al., 2013). The control group comprised 28 to 252 men (Meng et al., 2013; Zalata et al., 2014) (Table 1).
Definition of Infertility and Its Types
All the studies included well-defined infertility types, such as idiopathic azoospermia and/or oligospermia (Bianco et al., 2011; Kukuvitis et al. 2002; Lazaros et al., 2010; Meng et al., 2013; Vladoiu et al., 2016; Vu et al., 2023) and oligoasthenoteratozoospermia was also considered (Safarinejad et al., 2010; Zalata et al., 2014) (Table 1).
Infertility was defined as the inability of a couple to conceive after 1 to more than 2 years of attempting conception (Bianco et al., 2011; Safarinejad et al., 2010). Oligozoospermia was defined as a sperm count of fewer than 20×106/mL, while azoospermia referred to the complete absence of sperm in the ejaculate (Meng et al., 2013). In a study by Vladoiu et al. (2016), the observed semen characteristics for infertile group included a sperm concentration of 4.51 ± 1.17×106/mL (range: 0–46), motility (grade A + B) of 13.05% ± 1.94% (range: 0–50), and normal morphology of 5.39% ± 1.63% (range: 0–70). Severe oligozoospermia was classified as a sperm concentration of fewer than 5 million/mL (Bianco et al., 2011). Kukuvitis et al. (2002) categorized patients into idiopathic moderate oligospermia (sperm concentration = 19.2 ± 3.0 million/mL), azoospermia (1.0 ± 0.2 million/mL) or idiopathic severe oligospermia, and azoospermia or oligospermia (6.4 ± 1.3 million/mL) caused by known etiologies (Kukuvitis et al., 2002). Normal semen parameters included a sperm density of more than 20×106/mL, progressive motility (grade A+B) greater than 50%, normal morphology above 30%, and a semen volume exceeding 2 mL (Safarinejad et al., 2010).
Definition of Controls
Most of the studies included a well-defined control group of normospermic fertile men who had fathered at least one child without the use of assisted reproductive technology (Bianco et al., 2011; Liaqat et al., 2015; Meng et al., 2013; Safarinejad et al., 2010; Vu et al., 2023; Zalata et al., 2014). Both Zalata et al. (2014) and Meng et al. (2013) defined fertile men as those who had fathered a child within 1 to 2 years, respectively. One study also considered normal serum hormones and inhibin B levels for the control group (Safarinejad et al., 2010).
Criteria for Semen Analysis
Four studies categorized semen parameters according to the guidelines of WHO (1999) (Bianco et al., 2011; Kukuvitis et al., 2002; Lazaros et al., 2010; Safarinejad et al., 2010) and two other used guidelines by WHO (2010) (Vladoiu et al., 2016; Zalata et al., 2014). Liaqat et al. (2015) and Meng et al. (2013) used guidelines of WHO (1991) and WHO (1992), respectively. One study did not describe the criteria of semen analysis (Vu et al., 2023).
Most of the studies provided information on the abstinence of men from sexual activity for 3 to 5 days before semen analysis (Lazaros et al., 2010; Meng et al. 2013; Safarinejad et al., 2010; Vladoiu et al., 2016; Zalata et al., 2014).
Most studies conducted two semen analyses, 10 days to 1 month apart, and calculated the means of these analyses to determine semen parameters (Bianco et al., 2011; Kukuvitis et al., 2002; Lazaros et al., 2010; Safarinejad et al., 2010; Zalata et al., 2014). In contrast, a single semen sample was analyzed in other studies (Liaqat et al., 2015; Meng et al., 2013; Vladoiu et al., 2016).
Exclusion and Inclusion Criteria Set by Individual Studies
Most of the studies implemented rigorous inclusion and exclusion criteria to ensure the selection of appropriate participants and control groups. Infertile men were excluded if they exhibited known causes of infertility, including karyotypic abnormalities and/or Y chromosome microdeletions (Bianco et al., 2011; Lazaros et al., 2010; Meng et al., 2013; Safarinejad et al., 2010; Vladoiu et al., 2016; Vu et al., 2023; Zalata et al., 2014), varicocele (Bianco et al., 2011; Lazaros et al., 2010; Meng et al., 2013; Safarinejad et al., 2010; Vu et al., 2023; Zalata et al., 2014), cryptorchidism (Bianco et al., 2011; Kukuvitis et al., 2002; Meng et al., 2013; Safarinejad et al., 2010), obstructive azoospermia (Bianco et al., 2011; Lazaros et al., 2010; Meng et al., 2013; Safarinejad et al., 2010), endocrinological defects (Lazaros et al., 2010; Meng et al., 2013; Safarinejad et al., 2010), testicular trauma (Safarinejad et al., 2010; Vu et al., 2023), congenital absence of the testes (Safarinejad et al., 2010; Vu et al., 2023), cystic fibrosis (Liaqat et al., 2015), or hypogonadotropic hypogonadism (Bianco et al., 2011; Kukuvitis et al., 2002; Lazaros et al., 2010).
Lifestyle factors such as smoking and alcoholism (Bianco et al., 2011; Vladoiu et al., 2016; Zalata et al., 2014), exposure to occupational toxins (Bianco et al., 2011; Vladoiu et al., 2016), use of cytotoxic drugs (Bianco et al., 2011; Safarinejad et al., 2010; Vladoiu et al., 2016), immunosuppressants (Bianco et al., 2011; Safarinejad et al., 2010; Vladoiu et al., 2016), anticonvulsives (Bianco et al., 2011; Safarinejad et al., 2010; Vladoiu et al., 2016), androgens or antiandrogens (Safarinejad et al., 2010), and substance abuse were also grounds for exclusion (Bianco et al., 2011; Vladoiu et al., 2016; Zalata et al., 2014). In addition, men under spermatogenesis-impairing medications (Bianco et al., 2011; Lazaros et al., 2010; Vladoiu et al., 2016), or with a history of chemotherapy and radiotherapy (Bianco et al., 2011; Safarinejad et al., 2010; Vladoiu et al., 2016), systemic illnesses (Bianco et al., 2011; Liaqat et al., 2015; Safarinejad et al., 2010), mumps infection (Vu et al., 2023), leukocytospermia (Safarinejad et al., 2010), hepatobiliary disease (Safarinejad et al., 2010), significant renal insufficiency (Safarinejad et al., 2010), AIDS (Liaqat et al., 2015), hepatitis (Liaqat et al., 2015), or other sexually transmitted infections (Bianco et al., 2011; Safarinejad et al., 2010; Vu et al., 2023), were excluded (Table 1). Men who have normal female partners according to the extensive investigation were selected (Bianco et al., 2011; Safarinejad et al., 2010).
All the studies, except three (Liaqat et al., 2015; Vu et al., 2023; Zalata et al., 2014), reported the average age or minimum and maximum age for patients and controls. The minimum age was 22 years, and the maximum average age was 59.9 ± 5.2 years for fertile control men. The minimum age for infertile men was 23 years and the maximum average age was 40 years (Table 1).
Hardy–Weinberg Equilibrium
All studies determined the deviation of Xbal polymorphism frequencies from Hardy–Weinberg equilibrium (HWE) except a Caucasian study (Zalata et al., 2014). Another Caucasian study did not mention the results of HWE (Kukuvitis et al., 2002). Safarinejad et al. (2010) and Liaqat et al. (2015) reported deviation from HWE (Table 1).
Association of Infertility With ESR Xbal Genotypes
A total of four studies found no significant difference in genotypic and allelic frequencies and no significant association of Xbal polymorphism with male infertility and sperm disorders (Bianco et al., 2011; Lazaros et al. 2010; Vladoiu et al. 2016; Vu et al., 2023). One of these studies is Asian (Vu et al., 2023) and another is the African population (Bianco et al., 2011). Two studies reported a protective role of G allelic frequency compared with the A allelic frequency and the heterozygous form of the G allele (AG) compared with AA genotype frequency (Meng et al., 2013; Safarinejad et al., 2010). Two studies reported that the GG genotype increased the odds of infertility compared with the AA (Kukuvitis et al. 2002; Zalata et al. 2014) and another study reported that AG and AG+GG frequencies increased the odds of infertility compared with AA (Liaqat et al. 2015).
Association of Hormonal Parameters With ESR Xbal Genotypes
The AG genotype was associated with significantly higher levels of sex hormone-binding globulin (SHBG), total testosterone (T), and total estradiol (E2) compared with the AA genotype. In addition, serum E2 levels were significantly higher in individuals with GG genotypes compared with those with AA genotypes (Safarinejad et al., 2010; Zalata et al., 2014). Contradictory findings were reported by Safarinejad et al. (2010) and Meng et al. (2013) for follicle-stimulating hormone (FSH) and luteinizing hormone (LH) levels in individuals with AG or GG genotypes versus those with the AA genotype. Men with the AG genotype exhibited higher LH and lower FSH levels compared with those with the GG genotype (Safarinejad et al., 2010). Men with the AG genotype had significantly lower free testosterone, FSH, and free E2 levels compared with those with the AA and GG genotypes (Safarinejad et al., 2010).
Association of Semen Parameters With ESR Xbal Genotypes
Safarinejad et al. (2010) observed a positive correlation between sperm concentration and the presence of the AG genotype. Lazaros et al. (2010) found that men with the AG and GG genotypes had lower sperm concentrations compared with those with the AA genotype. Both Safarinejad et al. (2010) and Lazaros et al. (2010), along with Vladoiu et al. (2016), reported that sperm motility was higher in individuals with the AG genotype compared with the AA genotype. The GG genotype also resulted in higher sperm motility compared with the AA genotype (Lazaros et al., 2010; Vladoiu et al., 2016). A trend suggesting a positive correlation between the percentage of sperm with normal morphology and the presence of the AG genotype was also observed (Safarinejad et al., 2010). Zalata et al. (2014) reported that the median values for sperm count, sperm linear velocity, linearity index, normal sperm morphology, and acrosin activity index were significantly higher in individuals with the GG genotype than in those with the AA genotype.
Quantitative Analysis
The combined sample size from the nine studies included 1,036 cases and 1,154 controls.
Male Infertility
According to the results in Table 2, the G allele was protective against infertility in overall analysis (OR: 0.80; 95% CI = [0.70, 0.92]) and in Caucasian men (OR: 0.71; 95% CI = [0.54, 0.92]). Lower infertility risk was observed in Asian (AA vs. GG OR: 0.65; 95% CI = [0.43, 0.98]) and Caucasian men (OR: 0.49; 95% CI = [0.28, 0.83]).
Stratification Analyses of Genetic Susceptibility of rs rs9340799 Polymorphism to Male Infertility

Note. OR = odds ratio; CI = confidence interval.
Low Sperm Count (Azoospermia and Oligospermia)
According to Table 3, in cases of low sperm count, the Xba1 polymorphism was associated with a reduced risk of male infertility in the overall analysis of the recessive model, with the AA+AG genotypes showing a lower risk compared with the GG genotype (OR: 0.70; 95% CI = [0.53, 0.94]).
Stratification Analyses of Genetic Susceptibility of rs rs9340799 Polymorphism to Low Sperm Count in Males

Note. OR = odds ratio; CI = confidence interval.
Publication Bias
The Egger’s test of asymmetry performed for overall included studies (allelic and genotypic comparison) and subgroup analysis (Asian and Caucasian). The intercept p-values varied from .365 to .099, indicating no significant evidence of publication bias. The funnel plot also did not show any publication bias (Figure 2).

Funnel Plot Assessing Publication Bias for the Association Between ESR1 Xbal Polymorphism and Male Infertility
Sensitivity Analysis
A leave-one-out sensitivity analysis revealed that heterogeneity (I2) fluctuated between 52% and 30% depending on the excluded study. Removing Vu et al. (2023) reduced I2 from 49% to 30%, suggesting that this study contributed significantly to between-study variability. However, the pooled effect size remained stable, ranging from 0.76 (95% CI = [0.66, 0.87]) to 0.85 (95% CI = [0.71, 0.98]), indicating that the overall findings are robust.
Quality of the Studies
The quality assessment depicted that all included studies were of good quality (score > 45) (Table 1; Supplementary Table 1).
Trial Sequential Analysis
For the allelic model of SNP 351 A/G (rs9340799) in ESR1, the cumulative sample size (n = 4,216) exceeded the required sample size (n = 4,153) at 95% power, with the cumulative z-curve crossing the upper trial sequential monitoring boundary (Figure 3). This indicates that further studies are unlikely to alter the finding that the G allele is protective against male infertility.
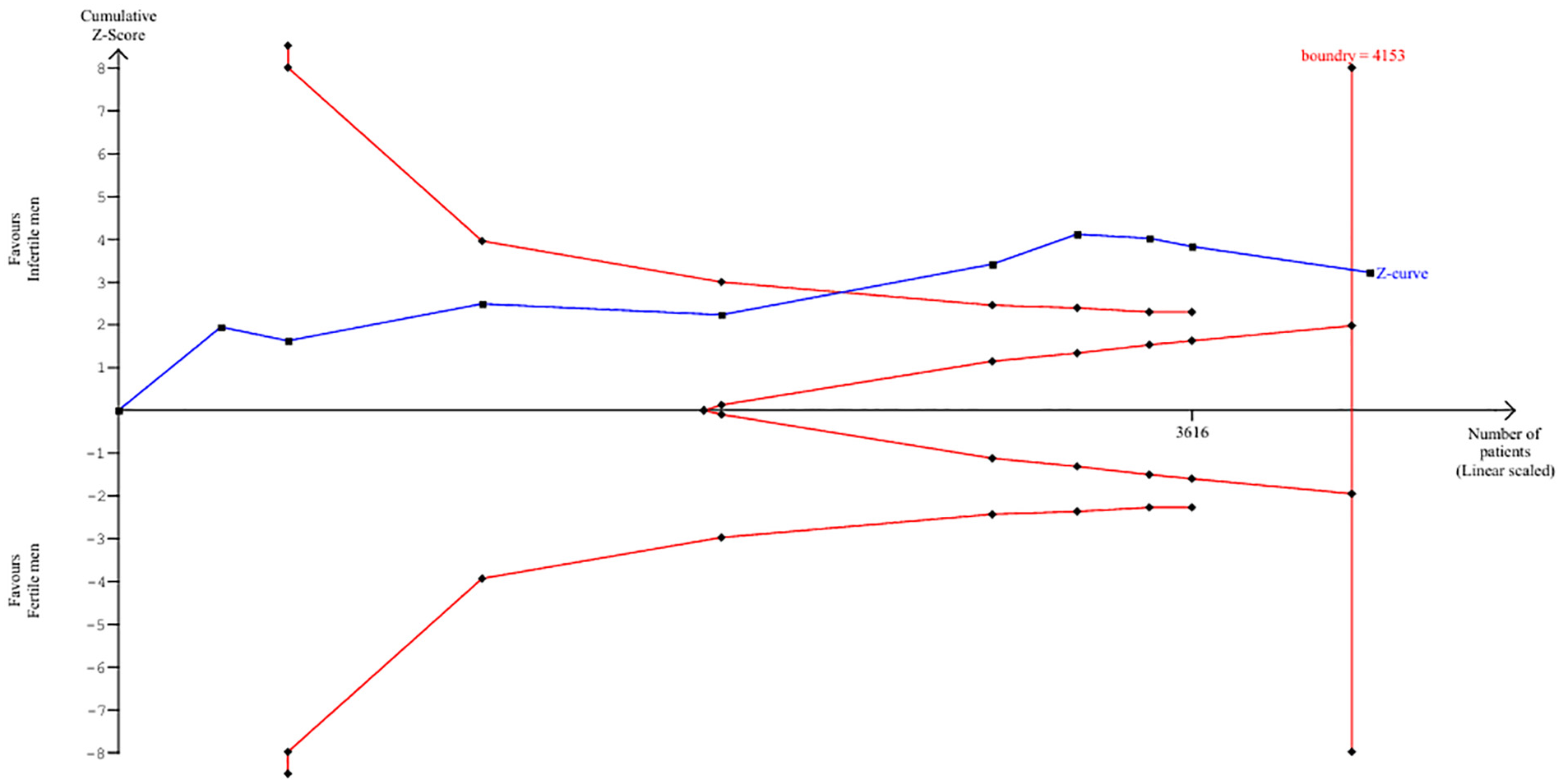
Trial Sequential Analysis for A to G
In Silico Analysis of ESR1 Expression
GTExportal data showed a non-significant increase in ESR1 expression for the rs9340799 AG and GG genotypes compared with AA in the testis, hypothalamus, and prostate, with a marginal increase in the pituitary (Figure 4).
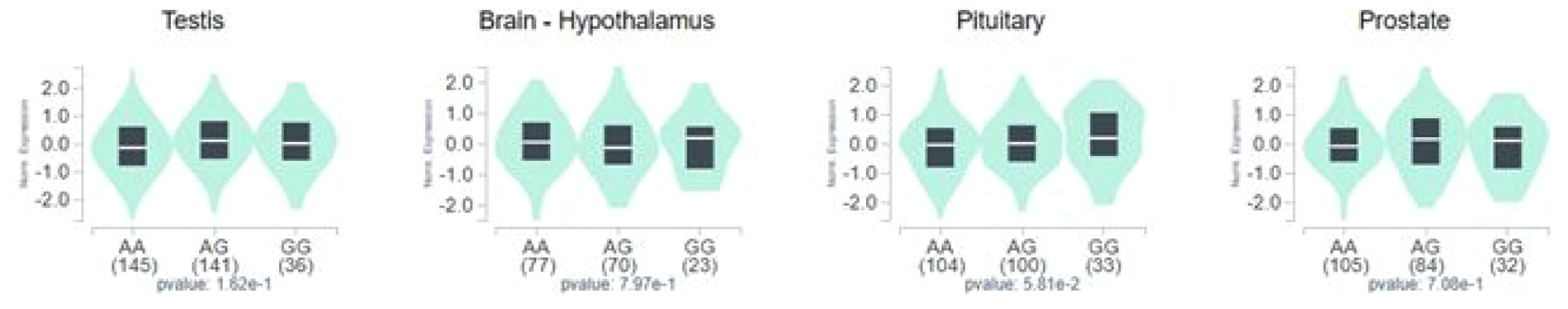
Graphs Showing Influence of the Polymorphisms (rs9340799) on the Expression of ESR1 Gene in the Testicular, Hypothalamus, Pituitary, and Prostate Tissue
Discussion
Estrogens and their receptor gene polymorphisms have been studied to understand their impact on male infertility (Ghadirkhomi et al., 2022). Estrogen action is mediated mainly by two specific receptors: estrogen receptor a (ERa), encoded by the ESR1 gene located on chromosome 6q25, and estrogen receptor b (ERb), encoded by the ESR2 gene located on chromosome 14q23–24. Both receptors are present in human testicular germ cells at different stages of spermatogenesis, from spermatogonia to elongated spermatids (Bianco et al., 2011). ERα comprises 595 amino acids and has a molecular weight of 66 kDa. It consists of six domains, A–F, from the N- to the C terminal regions A/B domains: The N-terminal domain contains an activation functional domain (AF-1) which interacts with co-regulators and is involved in transcriptional activation. This domain also contains several phosphorylation sites, which are important for transcriptional activity. C domain: The DNA-binding domain has two zinc finger structures and is critical for recognizing a specific DNA sequence, the estrogen response element motif (Habara & Shimada, 2022).
Estrogens are implicated in the development or progression of numerous diseases, including various types of cancer (breast, ovarian, colorectal, prostate, and endometrial), osteoporosis, neurodegenerative and cardiovascular diseases, insulin resistance, lupus erythematosus, endometriosis, and obesity (Ascenzi et al., 2006).
Although the role of estrogen in the developing male was established, studies could not identify the effects in the adult male reproductive organs for a long time. It was thought that an estrogen-binding ability was left over from developmental processes and that estrogen played only a small role in the adult male. Most interesting was the discovery that cytochrome P450 aromatase, which is capable of converting androgens into estrogens, is present in the testis (Hess, 2003).
Testosterone has long been known to be the dominant sex hormone in men. However, estrogen is found at detectable levels at multiple points of development and contributes to spermatogenesis. In addition, an abnormal T/E ratio (<10) has been associated with decreased semen parameters, and administration of an aromatase inhibitor normalized the ratio and improved sperm concentration, motility, and morphology targeting estrogen levels has clinical value when optimizing sperm retrieval rates in men with non-obstructive azoospermia (Hess & Cooke, 2018).
ERα has been reported to be absent in the testes of some species, including humans. However, estrogen receptors are abundant in the epithelium of the efferent ducts to regulate the expression of proteins involved in fluid reabsorption. Their expression in the efferent ducts is even higher than in the female reproductive tract (Hutson et al., 2019). Disruption of the α-receptor, either through knockout models or treatment with a pure antiestrogen, led to diluted cauda epididymal sperm, altered sperm morphology, inhibited sodium transport, reduced water reabsorption, increased secretion of chloride ions, and ultimately decreased fertility (Hess, 2003). In addition, ESR1 is present in round spermatids and plays a key role in regulating spermiogenesis (Bois et al., 2010; Talarczyk-Desole et al., 2021). Activation of estrogen signaling can affect spermatogenesis, potentially resulting in low sperm output. ESR1 gene is activated in male rats by treatment with an agonist, the expression of nucleoproteins such as transitional proteins 1 (Tnp1), transitional proteins 2 (Tnp2), and protamine 1 (Prm1) decreases. This downregulation can lead to an arrest in spermiogenesis, ultimately resulting in decreased sperm production and low sperm counts (Dumasia et al., 2016; Mobasseri et al., 2019).
In the present study, one SNP of estrogen receptor alpha has been studied using the past nine studies that include different Asian and Caucasian populations, and the obtained results indicate that the rs9340799AA genotype was observed as a protective factor for infertility in both Asian and Caucasian populations. Our results are contrasting to a previous meta-analysis that indicated an increased risks for the comparison of AA versus GG (OR = 1.75, 95% CI [1.15, 2.68]) and AA versus GA+GG (OR = 1.38, 95% CI = [1.02, 1.88]) (Li et al., 2014).
We observed high heterogeneity in some analyses (I2 > 50%), indicating variability across studies. To explore potential sources of heterogeneity, we performed subgroup analyses based on study region and low sperm count and conducted sensitivity analyses by excluding individual studies. The funnel graph and Egger’s asymmetric test were not statistically significant, but the small number of studies in the subgroups limits the power of Egger’s test. Most of the studies examined idiopathic infertile men and adhered to strict criteria for fertile men. Certain unexplained variations in the results and high heterogeneity may be due to the differing semen analysis criteria set by WHO guidelines between 1991 and 2000, which were used in various studies. In addition, the sample sizes were variable and often small, leading to decreased statistical power in the results. Many studies excluded participants with karyotyping issues and Y chromosome microdeletions, and there could be several other underlying genetic polymorphisms and mutations that remain unknown. Moreover, ethnic differences may serve as a confounding factor.
TSA showed that the studies already done have a cumulative sample size larger than the required sample size at 95% power and the cumulative z-curve crossed the upper trial sequential monitoring boundaries. Therefore, conducting more studies may not change the results that the G allele is significantly protective for male infertility.
ESR1 Xbal gene may alter receptor function, disrupt hormonal balance, change gene expression, or affect downstream signaling pathways. These alterations could lead to impaired sperm production or motility. The findings suggest that ESR1 Xbal polymorphism could serve as biomarker for infertility risk, providing opportunities for early diagnosis and personalized treatment approaches. Genetic screening could identify individuals who might benefit from targeted interventions, such as hormonal therapies to compensate for ESR1 dysfunction. Counseling of couples undergoing assisted reproductive techniques could be done. Understanding the mechanisms behind these associations could also lead to the development of new therapeutic strategies, ultimately improving the management of male infertility.
Future research should focus on including more diverse populations and accounting for potential confounding factors such as age, smoking, and obesity to better understand the relationship between the rs9340799 and male infertility. The mechanism of action of ESR Xbal polymorphism need to be further investigated through functional studies.
The included studies predominantly represent Asian, Caucasian, and African populations. Differences in genetic backgrounds and environmental factors across these populations may contribute to variability in the observed association between the SNP and male infertility.
Some of the included studies did not adjust for potential confounding factors such as age, smoking, and obesity, which could influence the observed association. The absence of such adjustments may introduce bias.
Future research should focus on including more diverse populations and accounting for potential confounding factors such as age, smoking, and obesity to better understand the relationship between the SNP and male infertility.
Conclusion
In the conclusion, this review and meta-analysis suggests that SNP Xba1 (rs9340799) in ESR1 has a protective effect against male infertility and low sperm count. The cumulative sample size from previous studies is large enough that further may not change the results that the G allele is significantly protective against male infertility. Future research should focus on accounting for potential confounding factors such as age, smoking, and obesity to better understand the relationship between the rs9340799 and male infertility. The mechanism of action of ESR Xbal polymorphism needs to be further investigated.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883251319134 – Supplemental material for Association of ESR1 Xba1 (rs9340799) With Male Infertility: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-jmh-10.1177_15579883251319134 for Association of ESR1 Xba1 (rs9340799) With Male Infertility: A Systematic Review and Meta-Analysis by Hania Qamar, Sadia Bibi, Zeeshan Qadeer, Faiza Muzammil, Masooma Batool, Shaista Aslam, Ayesha Akram, Adina Arshad and Muhammad Irfan in American Journal of Men’s Health
Supplemental Material
sj-xlsx-2-jmh-10.1177_15579883251319134 – Supplemental material for Association of ESR1 Xba1 (rs9340799) With Male Infertility: A Systematic Review and Meta-Analysis
Supplemental material, sj-xlsx-2-jmh-10.1177_15579883251319134 for Association of ESR1 Xba1 (rs9340799) With Male Infertility: A Systematic Review and Meta-Analysis by Hania Qamar, Sadia Bibi, Zeeshan Qadeer, Faiza Muzammil, Masooma Batool, Shaista Aslam, Ayesha Akram, Adina Arshad and Muhammad Irfan in American Journal of Men’s Health
Footnotes
Acknowledgements
The authors would like to thank Naila Zuliqar (MPhil scholar) and Kainat Abbasi (PhD scholar), Department of Zoology, Wildlife and Fisheries, PMAS-Arid Agriculture University, Rawalpindi, Pakistan, for their assistance with database searches.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Access
All the relevant data are included in the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
