Abstract
The present meta-analysis is performed to determine the association of C1236T and C3435T polymorphisms in the MDR1 gene. Google Scholar, PubMed, and Science Direct were searched. A total of 47 studies were retrieved, of which only three case–control studies, consisting of 490 cases and 423 controls, met the selection criteria. Odds ratios (ORs) for MDR1 C1236T were as follows: Allelic model (T vs. C): OR = 1.06 [0.83, 1.35]; Additive model (TT vs. CC): OR = 0.91 [0.53, 1.56]; Dominant model (TT+CT vs. CC): OR = 0.83 [0.55, 1.24]; and Recessive model (TT vs. CT+CC): OR = 1.43 [0.95, 2.17]. However, for MDR1 C3435T: Allelic model (T vs. C): OR = 1.06 [0.83, 1.35]; Additive model (TT vs. CC): OR = 1.18 [0.75, 1.88]; Dominant model (TT+CT vs. CC): OR = 1.42 [0.99, 2.04]; and Recessive model (TT vs. CT+CC): OR = 0.90 [0.61, 1.33]. None of the four models presented a significant association of either polymorphism with the risk of infertility in men (p >.05). The present study indicates that MDR1 gene polymorphisms might not be a risk factor for male infertility. Further studies with a larger sample size are needed to be conducted to confirm the findings of the present study.
Introduction
In addition to other social problems, the developing world is also facing the threat of a declining birth rate (Miyamoto et al., 2012). Infertility refers to the failure of conception within 1 year of unprotected sex resulting from some kind of reproductive health issue of either of the partner (Practice Committee of the American Society for Reproductive Medicine., 2020). It is a major health concern affecting around 190 million couples worldwide. The condition may emerge due to different pathological conditions related to female or male factors, with the male factor observed to play the role in up to 50% of the cases (Arafa et al., 2020).
Infertility is undoubtedly a complicated pathological condition affecting 4% to 7% of the male population across the globe and is demonstrated as a broad range of heterogeneous phenotypes, from congenital or acquired urogenital abnormalities, endocrine disturbances, and immunological factors to spermatogenic quantitative and qualitative defects. It has been observed that 15% of infertile males have a genetic defect (Aydos et al., 2015; Xavier et al., 2021).
The gene MDR1 encodes for a protein that resides in the plasma membrane functioning as an ATP-driven efflux pump (Miller et al., 2008). P-glycoprotein was discovered almost three decades ago as a gene which overexpressed in multidrug-resistant tumor cells in humans (Juliano & Ling, 1976). The product of the MDR1 gene is P-glycoprotein, which is a probable link between the environmental and genetic factors that lead to the development of male infertility (Droździk al., 2009). It has a shielding role and removes toxic substances such as xenobiotics from cells and prevents their buildup within cells, which serves as a protective measure for the organism (Aydos et al., 2015). Xenobiotics, including pesticides, act as a substrate for P-glycoprotein. P-glycoprotein is found in the blood–testis barrier and protects the testis by preventing the penetration of xenobiotics (Droździk et al., 2009; Su et al., 2009). In testes, P-glycoprotein is expressed at the luminal surface of capillary endothelial cells and in Leydig cells, testicular macrophages, and Sertoli cells, which defend the somatic cells against detrimental toxic substances. P-glycoprotein influences the microenvironment of the seminiferous tubules by transporting testicular steroids. P-glycoprotein transfers a large number of xenobiotics including pesticides, which are well-known factors responsible for reducing male fertility (Aydos et al., 2015).
The nucleotide variation in MDR1 dictates the function and expression of P-glycoprotein. The level of expression of this gene is highly fluctuating between subjects. This fluctuation exhibits the genetic polymorphism of the MDR1 gene associated with a variation in expression level (Rüstemoglu et al., 2011). About 28 exons make up the human MDR1 gene. In the MDR1 gene, 29 single-nucleotide polymorphisms (SNPs) have been reported from which 19 SNPs are in the exonic regions and 11 are nonsynonymous. SNPs at exon 21 in position 2677 may eventually result in two distinct amino acid changes, namely, Ala893Ser (G2677T) and Ala893Thr (G2677A). However, a synonymous SNP in exon 26 (C3435T) was the first variant to be associated with altered protein expression, although the SNP does not change the encoded amino acid (Marzolini et al., 2004).
As the previous studies reported contrasting results, this meta-analysis aimed to determine the association of MDR1 polymorphisms, C1236T (rs1128503) and C3435T (rs10456452), with male infertility.
Method
Search Strategy
Google Scholar, PubMed, and ScienceDirect were used to retrieve the relevant studies, using the keywords “MDR1,” “C1236T,” “C3435T,” and “male infertility.”
Inclusion Exclusion Criteria
The case–control studies with full text available, investigating the association of MDR1 polymorphisms and male infertility, with complete genotypic or allelic frequencies for cases and controls, were included in the meta-analysis. Whereas cohort studies, animal studies, reviews, conference abstracts, and editorials were excluded from the present study.
Data Extraction
Data such as author name, year of publication, country, inclusion and exclusion criteria, the total number of cases and controls, age of the participants, genotyping method, and genotypic and allelic frequencies were extracted from the included studies.
Statistical Analysis
The relationship between MDR1gene polymorphisms (C1236T and C3435T) and male infertility was analyzed by four models, that is, allelic model (T vs. C), additive model (TT vs. CC), dominant model (TT+CT vs. CC), and recessive model (TT vs. CT+CC). The odd ratios (ORs) with 95% confidence intervals (CIs) were calculated to determine the association between the polymorphism and the risk of male infertility. Heterogeneity among the studies was assessed by using the Q test and I 2 statistics. p≤.05 was considered statistically significant. Review Manager 5.4 was used for all these analyses.
Trial Sequential Analysis
Trial sequential analysis (TSA) was conducted by using Trial Sequential Analysis Viewer software, version 0.9.5.10 Beta, Copenhagen Trial Unit, Centre for Clinical Intervention Research, Rigshospitalet, 2016 (Gordon Lan & DeMets, 1983) by using the method of Meng et al. (2018a). A significance of 5% was selected for type I error, whereas 60% power was used to calculate the required sample size and set TSA boundaries.
In Silico Analysis of MDR1 Expression
The influence of polymorphisms on MDR1 expression levels was analyzed by using GTEx Analysis Release V8 (dbGaP Accession phs000424.v8.p2) data (Gibson, 2015). The differential expression of MDR1 for normal and cancer tissues was also determined by using GEPIA (http://gepia.cancer-pku.cn/) (Fan et al., 2019; Meng et al., 2018b, 2018c).
Results
Study Selection
A total of 47 studies were retrieved by searching the three electronic databases. After duplicate removal, the titles and abstracts of 43 records were screened. The full texts of eight studies were then assessed, and finally, three studies were included in the current meta-analysis (Figure 1).

Flowchart of Included Studies Selection
Characteristics of Included Studies
The studies (n = 3) consisted of 490 cases and 423 controls. The characteristics of included studies are summarized in Table 1.
Characteristics of the Included Studies

Note. SNP, single-nucleotide polymorphism; PCR, polymerase chain reaction; RFLP, restriction fragment length polymorphism.
MDR1 C1236T Polymorphism and Male Infertility
A total of 328 cases and 232 controls were included in this assessment. Figure 2 presents the odd ratios of all comparison models, as the Allelic model (T vs. C): OR = 1.06 [0.83, 1.35], (p =.65); Additive model (TT vs. CC): OR = 0.91 [0.53, 1.56], (p = .73); Dominant model (TT+CT vs. CC): OR = 0.83 [0.55, 1.24], (p = .35); and Recessive model (TT vs. CT+CC): OR = 1.43 [0.95, 2.17], (p = .09). None of the four models showed a significant association of MDR1 C1236T with the risk of male infertility.
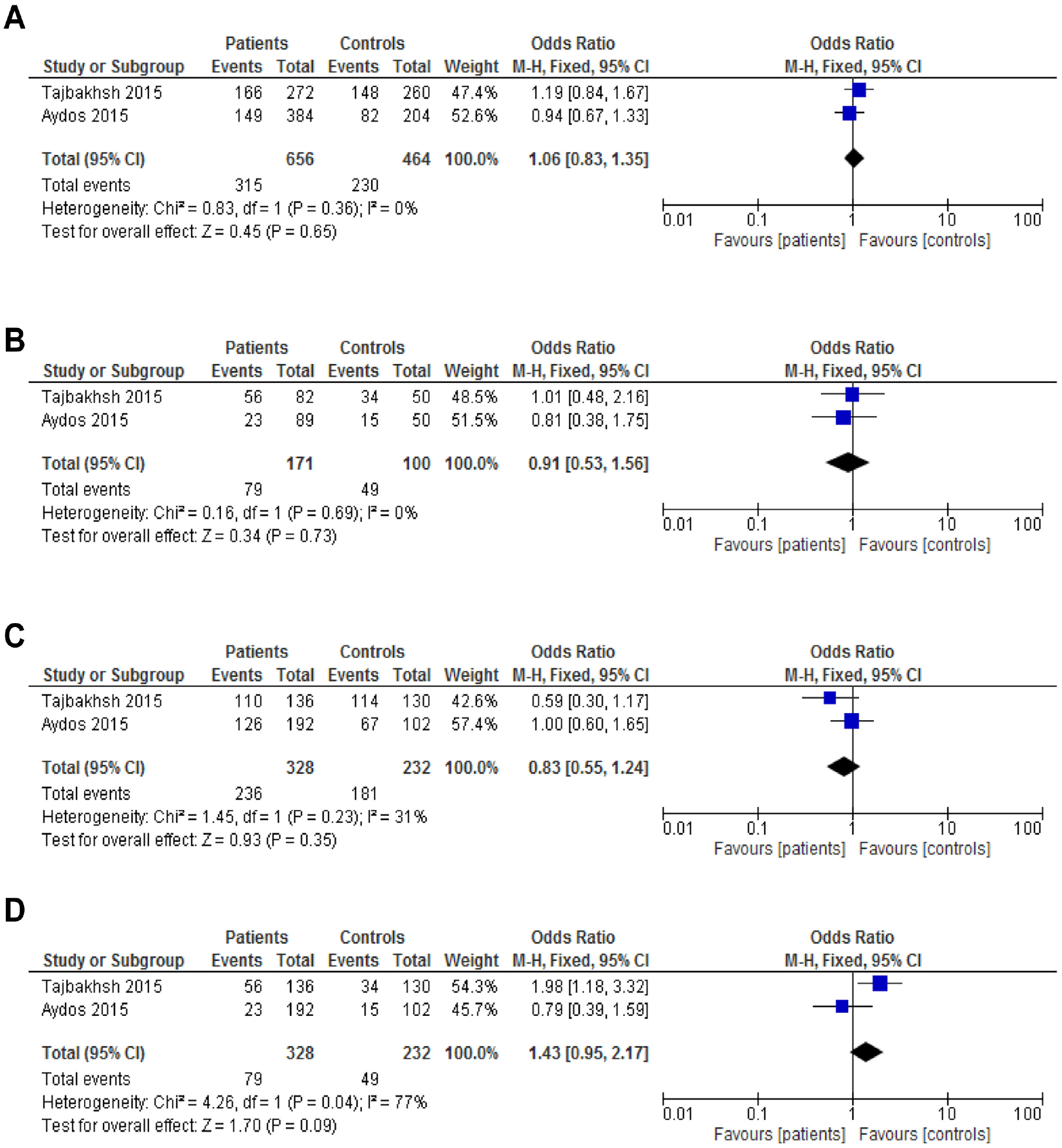
Forest Plot of the Studies Assessing the Association Between MDR1 C1236T Polymorphism and Male Infertility. (A) Allelic Model: T Versus C; (B) Additive Model: TT Versus CC; (C) Dominant Model: TT+CT Versus CC; and (D) Recessive Model: TT Versus CT+TT
MDR1 C3435T Polymorphism and Male Infertility
A total of 354 cases and 293 controls were included in this analysis. Figure 3 presents the odd ratios of all comparison models, as the Allelic model (T vs. C): OR = 1.06 [0.83, 1.35], (p = .65); Additive model (TT vs. CC): OR = 1.18 [0.75, 1.88], (p =.48); Dominant model (TT+CT vs. CC): OR = 1.42 [0.99, 2.04], (p =.06); and Recessive model (TT vs. CT+CC): OR = 0.90 [0.61, 1.33], (p = .61). Similarly, none of the four models presented a significant association of MDR1 C3435T with the risk of infertility in men.

Forest Plot of the Studies Assessing the Association Between MDR1 C3435T Polymorphism and Male Infertility. (A) Allelic Model: T Versus C; (B) Additive Model: TT Versus CC; (C) Dominant Model: TT+CT Versus CC; (D) Recessive Model: TT Versus CT+TT
Results of Trial Sequential Analysis
For the allelic model of C1236T, the required sample size was 20,379 at 60% power, whereas the cumulative z-curve did not cross the trial sequential monitoring boundaries (Figure 4). However, for C3435T allelic model, the z-curve did not reach the trial sequential monitoring boundaries even at 20% power when the required sample size was 22,709 (Figure 5). It shows that more studies, with larger sample sizes, are required to provide sufficient evidence for confirming the results of the current meta-analysis.

Trial Sequential Analysis (5% Type I Error and 60% Power) for C1236T Polymorphism for the Allelic Model

Trial Sequential Analysis (5% Type I Error and 20% Power) for C3435T Polymorphism for the Allelic Model
In Silico Expression Analysis
The GTExpotal data indicated a significant decrease in the expression of MDR1 for rs1128503 (p = 1.17 × 10−13) and rs1045642 (p = 8.71 × 10−31) in CC genotype compared with the TT genotype in testis only (Figure 6). The differential expression of MDR1 in normal and different types of cancer tissues is also indicated in Figure 7.

Graphs Showing Significant Influence of the Polymorphisms (A. rs1128503: C1236T; B. rs1045642: C3435T) on the Expression of MDR1 Gene in the Testicular Tissue

The Gene Expression Profile Across Various Tumour Samples (Red Peaks) and Paired Normal Tissues (Green Peaks)
Discussion
The P-glycoprotein is a member of the ATP-binding cassette family. It is the product of the MDR1 gene, also known as the ABCB1 gene (Marzolini et al., 2004), which acts as ATP-driven pump (Zhang et al., 2021). Human P-glycoprotein is a phosphorylated protein that is composed of 1280 amino acids having two symmetrical and homologous sequences, and each sequence contains a binding motif for ATP and the transmembrane domain. The energy needed for the activation of the drug transporter comes from ATP hydrolysis. P-glycoprotein prevents the accumulation of toxic substances in different body organs such as gonads, bone marrow, brain, and the fetus (Marzolini et al., 2004). In humans, MDR1 is normally found in a tissue-specific manner (Fung & Gottesman, 2009). The P-glycoprotein’s functions and expressions are influenced by MDR1 gene SNPs (Hoffmeyer et al., 2000). C1236T and C3435T are synonymous mutations. The functioning of P-glycoprotein may be affected by changing protein folding (Zhang et al., 2021). C3435T exhibits strong linkage disequilibrium with the C1236T polymorphism (Aydos et al., 2015; Kim et al., 2001). C3435T polymorphism affects the stability of mRNA (Wang et al., 2005).
Subjects with MDR1 3435 TT genotype had significantly lower mRNA expression levels compared with those with 3435 CC genotypes (Fromm, 2002; Hoffmeyer et al., 2000; Li et al., 2006; Wang et al., 2012). The mRNA derived from the 3435T allele has the potential to decay more rapidly, after its synthesis (Wang et al., 2005). Significantly higher plasma levels of P-gp substrate (digoxin) in individuals with the T allele indicate the weaker expression and functioning of MDR1 (Hoffmeyer et al., 2000). Our in silico analysis also reported a significant decrease in MDR1 mRNA expression in the testis, due to the presence of mutant allele T in both of the MDR1 polymorphisms studied.
Previous epidemiological studies reported an association between male infertility and cancer. This association could be explained by the direct effect of cancer on fertility such as due to poor health conditions, lesions in testicular tissues, or shared pathophysiology. Infertile men with poor semen quality and their first-degree relatives are reported to have increased odds of cancers compared with control (Nagirnaja et al., 2018). Nagirnaja et al. (2018) identified 25 common candidate genes for male infertility and cancer. About half (n = 13) of these 25 genes also underlie a specific hereditary cancer predisposition syndrome.
MDR1 is highly expressed in multidrug-resistant cancer cells compared with the normal cells, in response to chemotherapy (Haque et al., 2020). Zaib et al. (2022) performed a meta-analysis as well as an experiment to evaluate MDR1 polymorphisms with breast cancer risk. Contradictory results were reported regarding MDR1 C3435T polymorphism, and two studies reported an association whereas four studies showed no association with breast cancer. The experimental analysis showed that this polymorphism increases the risk of breast cancer up to 3 times. MDR1 was significantly overexpressed in 44% of the cancerous tissue compared with the normal tissues.
Both, 1236 TT and 3435 TT mutations were associated with increased mRNA expression of MDR1 in resistant ovarian tumor patients (Haque et al., 2020). Human germ cell testicular tumors, particularly stage II–III, were detected to be correlated with MDR1 mRNA overexpression (Bak et al., 1995).
Among bladder cancer patients, MDR1 gene expression was reported to be highest in the 3435 CC genotype and lowest in the 3435 TT genotype (Duda, 2020), whereas no significant association was observed in a case–control study, conducted in Spain (Henríquez-Hernández et al., 2012). The presence of MDR1 C3435T-mutant allele “T” was related to the increased expression of P-gp protein among patients with hepatocellular carcinoma (Baldissera et al., 2012). In the Iranian population, the 3435 TT genotype was found to be associated with twice the lower expression of MDR1 in gastric cancer patients, compared with those with 3435 CC genotypes (Sabahi et al., 2010).
A study was performed on rats to check the function of the MDR1 gene blood–testis barrier (BTB) and its importance. Mruk and Cheng (2012) knock down rat Mdr1a and Mdr1b from Sertoli cells by using RNA interference (RNAi) which affects Sertoli cell barrier integrity. Silenced Mdr1 allowed the entry of harmful chemicals into the Sertoli cell adluminal compartment. The entry of these chemicals is stopped from entering or being pumped out by P-glycoprotein at the BTB (Mruk & Cheng, 2012).
The current meta-analysis showed that neither of the two polymorphisms in the MDR1 gene, that is, C3435T and C1236T, is significantly associated with infertility in men. However, all of the previous molecular epidemiological studies regarding MDR1 polymorphisms and male infertility reported contrasting results (Aydos et al., 2015; Droździk et al., 2009; Tajbakhsh et al., 2015). One study was conducted in the Asian population, that is, Iran (Droździk et al., 2009), while the other two were performed in the Caucasian population, that is, Turkey and Poland (Aydos et al., 2015; Tajbakhsh et al., 2015). The genotypic and allelic frequencies for both the polymorphisms between infertile men and controls were insignificant, but 3435 CT was reported to be significantly associated with high levels of sperm DNA damage (Aydos et al., 2015). Tajbaksh et al. (2015) reported a significant association between MDR1 1236 C>T polymorphism and male infertility. Droździk et al. (2009) worked on MDR1 3435C>T polymorphism and checked their association with male infertility. The result showed that there was no significant association between male infertility and MDR1 gene polymorphism. Infertility risk was significantly increased in subjects carrying the T allele, for example, 3435TT and 3435CT genotypes.
Inconsistency among the studies might be attributed to the factors like ethnicity, geographic variations, small sample size, as well as inclusion and exclusion criteria of the included studies (Irfan et al., 2016; Weiner et al., 2014).
Only three studies were available for the meta-analysis, which limited the sample size of this study. The data from the Asian and Caucasian populations were insufficient, whereas data from the African population were lacking. Therefore, subgroup analysis based on ethnicity, as well as sperm disorders, was not performed. The individual studies lacked unadjusted estimates for other covariates like age, smoking, and lifestyle factors, which might affect the precision of the results. An increased level of heterogeneity between the included studies might also influence the result. Moreover, genetic screenings such as Y-chromosome microdeletions were performed only in one of the included studies.
Conclusion
The present study performed to evaluate the association of MDR1 gene polymorphisms with male infertility indicates that this polymorphism might not be a risk factor for infertility in men. The availability of only three studies suggests that further studies are needed to be conducted, including large sample size, to confirm the findings of the current study.
Footnotes
Acknowledgements
The authors would like to thank Mr. Bilal Ishaq, PhD scholar, PMAS-Arid Agriculture University, Rawalpindi, Pakistan, for his assistance with database searches.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval does not apply to the present meta-analysis.
