Abstract
This study retrospectively examines the effectiveness of low-frequency electrical stimulation for addressing nocturia in patients experiencing urinary incontinence after undergoing radical prostatectomy. We reviewed the outcomes of 32 patients who had undergone radical prostatectomy and subsequently experienced urinary incontinence. These patients were divided into the control group (n = 16) who received pelvic floor muscle training and the treatment group (n = 16) who underwent electrophysiological appropriate technique treatment in conjunction with pelvic floor muscle training. We assessed changes in nocturnal voided volume, polyuria index, and nocturia-related quality of life at three different time points: before treatment, after 2 weeks, and at the 3-month follow-up. After 3-month follow-up, both groups exhibit reductions in nocturnal voided volume, and polyuria index compared with baseline and the 2-week mark (p < .05). Treatment group outperformed the control group (p < .05). The quality of life in the treatment group remained consistently high, while the control group did not show a statistically significant difference from baseline (p > .05). The total effective rate was significantly higher in the treatment group (93.75%) than in the control group (75.00%, p = .044). This retrospective analysis suggests that electrophysiological appropriate technique treatment effectively mitigates nocturnal, leading to improved quality of life in patients with urinary incontinence following radical prostatectomy in the near term.
Introduction
Prostate cancer stands as one of the most prevalent malignant tumors within the urinary and reproductive systems, and it ranks second in terms of cancer incidence among men (Bray et al., 2018). The primary treatment approach for localized prostate cancer is radical prostatectomy, a widely accepted and highly effective option in the realm of clinical oncology (H. Zhu & Deng, 2020). However, a notable drawback associated with radical prostatectomy is the occurrence of postprostatectomy incontinence (PPI), which can have a profound impact on the physical and psychological well-being of affected individuals. PPI patients frequently grapple with distressing symptoms, including nocturia, which involves waking up at night to urinate. Interestingly, research has established a strong link between nocturia and various adverse health outcomes, such as an increased risk of falls and cardiovascular events (Lightner et al., 2012). Consequently, addressing nocturia in PPI patients emerges as a matter of considerable importance.
In response to the challenges posed by PPI and its associated symptoms, current medical approaches have seen significant advancements. Notably, electrophysiological treatments have demonstrated promising outcomes by targeting various aspects of the condition. These treatments aim to stimulate nerve regeneration, enhance external sphincter contraction, inhibit detrusor muscle contractions, and facilitate the repair of pelvic floor collagen, resulting in improved urinary control (Canning et al., 2022). In an exciting development, our research team has integrated precise electrophysiological diagnosis, traditional Chinese meridian theory, and electrophysiological interventions to develop a tailored, electro-therapeutic technique. This innovative approach has been applied in the treatment of nocturia in PPI patients (“Chinese Expert Consensus Compilation Group on the Application of Electrophysiological Appropriate Technology in the Diagnosis and Treatment of Andrology Diseases Andrology Branch of Chinese Medical Association, 2022”). Furthermore, we have systematically assessed its impact on the quality of life for these individuals.
This report provides comprehensive insights into our findings, detailing the methodology and outcomes of our electrophysiological treatment for nocturia in PPI patients, along with an analysis of its effects on their overall quality of life. By combining cutting-edge medical technology and traditional wisdom, our approach seeks to address a significant health challenge and improve the well-being of those affected by PPI-related symptoms.
Methods
Clinical Data
Diagnostic Criteria
The Western medical diagnostic criteria for PPI were defined according to the principles of urology (Wu, 2005) and Prabhu et al.’s (2013) study:
Involuntary loss of urine.
A history of radical prostatectomy with a corresponding pathological diagnosis.
Positive pad test: A 24-hr pad test showing an increase in pad weight of 4 g or more, or a daily pad usage of more than 1 pad.
The Traditional Chinese Medicine diagnostic criteria were formulated according to the principles of “Chinese Medical Diagnosis and Differential Diagnosis” (W. Zhu, 1999):
Urinary incontinence: In the awake state, urine flows out involuntarily.
Kidney qi deficiency type: Urinary incontinence, clear and thin urine, aversion to cold, fatigue, weakness in the lower back and knees, pale tongue, slow or weak pulse.
Inclusion Criteria
Patients who had undergone radical prostatectomy with a pathological diagnosis of prostate cancer, met the PPI diagnostic standards and having undergone surgery within the past 2 weeks, were at least 2 weeks post-surgery, experienced waking up at night due to the urge to urinate at least two times, were between 50 and 80 years of age, voluntarily participated in this clinical study, and signed informed consent forms were included.
Exclusion Criteria
Patients underwent open conversion of laparoscopic radical prostatectomy.
Patients who experience total incontinence with empty bladder.
Patients who experience discomfort during the use of electrophysiological techniques. This includes symptoms such as skin itching or lesions, dizziness, limb numbness, and so on.
Patients with cognitive impairments.
Patients with urinary tract infections, and those participating in other clinical trials were excluded.
General Information
A total of 32 PPI patients who visited our hospital between June 2022 and December 2022 were selected. They were divided into two groups, with 16 patients in each group. The sample size was estimated using the formula N = 2([tα+ tβ] S/δ)2, where α = 0.05, β = 0.1, tα = 1.96, tβ = 1.282. Based on the preliminary results of this study, S = 0.98, δ = 1.20. After substituting these values into the formula, N1 = N2 = 14 cases were obtained. Considering a 10% dropout rate, 16 cases per group were included, resulting in a total of 32 cases.
Treatment Methods
Control group: Pelvic floor muscle training in addition to routine PPI treatment.
Patients received pelvic floor muscle training according to the 2019 Chinese Urology Disease Diagnosis Guidelines. Patients were instructed to contract their pelvic floor muscles for 2 to 6 s, relax for 2 to 6 s, and repeat this 10 to 15 times per set, for a total of three to eight sets per day, continuously for 3 months. Training could be done in three positions: standing, sitting, and lying down.
Treatment group: Low-frequency electrical stimulation in addition to the control group’s treatment.
Electrophysiological Diagnosis to Determine Precise Electrical Stimulation Patterns
Electrophysiological diagnosis was conducted using a medical infrared thermography camera (Foshan Shanshan Datang Medical Technology Co., Ltd., Model PRISM 640A) and a low-frequency neuromuscular therapy device (Foshan Shanshan Datang Medical Technology Co., Ltd., Model Biostim ble). This helped determine the precise electrical stimulation patterns and develop individualized treatment plans.
First, patients were instructed to remove all clothing and face the infrared camera while performing specific movements as directed. The medical infrared thermography camera displayed thermal images of the patients’ movements and automatically measured the lowest temperature, highest temperature, and average temperature, as shown in Figure 1. Next, a low-frequency neuromuscular therapy device was used to provide different modes of electrical stimulation to acupuncture points such as Qihai, Guanyuan, and Baliao. After stimulation, temperature measurements were taken again, and an average temperature difference of >0.5°C indicated the optimal electrical stimulation pattern, as shown in Figure 2. In this study, the electrical stimulation patterns included combinations of DD+ and CC+ frequencies, such as the DD297+DD603+CC1 pattern. This pattern represents electrical stimulation at 297 Hz for striated muscle, 603 Hz for striated muscle again, and 1 Hz for the parasympathetic nervous system. DD stands for electrical stimulation of striated muscles, which increases muscle fiber strength. The frequencies 297 and 603 correspond to Type I and Type II muscle fibers, respectively. Type I fibers are slow-twitch muscles that stabilize internal organs, while Type II fibers are fast-twitch muscles that control functions such as urinary incontinence, urinary frequency, and sexual function.

Medical Infrared Thermal Imaging Inspection Interface

Medical Infrared Thermal Imaging Comparison Interface
Electrophysiological Appropriate Technology Treatment
Utilize a low-frequency neuromuscular treatment device for electrophysiological appropriate technology treatment that begins 2 weeks after radical prostatectomy surgery (J. Zhu et al., 2023). Instruct the patient to assume a supine or sitting position, apply electrode pads to the corresponding acupoints according to the prescribed protocol, and select the treatment mode. Adjust the electrical stimulation intensity based on the patient’s condition and tolerance, administering treatment for 60 min each session, once daily, for a continuous period of 2 weeks.
Observation Parameters and Methods
Urine Volume and Frequency of Urination
Both groups recorded a voiding diary before treatment, 2 weeks after treatment, and 3 months after treatment, obtaining the following data: Nocturnal urine volume: The total volume of urine passed from bedtime to waking up, including the first urination upon waking in the morning. Nocturnal urination frequency: The number of times a person urinates from bedtime to waking up, excluding the first morning urination. Recorded 24-hr total urine volume and calculated the nocturnal polyuria index, which is the nocturnal urine volume divided by the 24-hr total urine volume.
Quality of Life
Assessment of quality of life was conducted on both groups of the patients using the Chinese version of the nocturia quality of life questionnaire (Bu et al., 2018) before treatment, 2 weeks after treatment, and 3 months after treatment.
Efficacy Assessment Criteria
Assessments were conducted on both groups of relevant treatment indicators before treatment, 2 weeks after treatment, and 3 months after treatment, using a reduction in the frequency of nocturia as the efficacy assessment criterion.
Cure: Nocturia frequency reduced to 0 or 1 times after treatment.
Significant improvement: Nocturia frequency reduced by 50% to 75% after treatment but still more than once per night.
Effective: Nocturia frequency reduced by 25% to 50% after treatment.
Ineffective: Nocturia frequency reduced by <25% after treatment, or no change, or an increase in frequency.
Overall effective: Cure + significant improvement + effective.
Statistical Methods
Statistical analysis of data was performed using SPSS 23.0 software. For continuous data that followed a normal distribution and showed homogeneity of variances, the mean ± standard deviation (x±s) was used for representation. Between-group comparisons were conducted using independent samples t-tests, and within-group comparisons before and after treatment were done using paired samples t-tests. For continuous data that did not follow a normal distribution or exhibit homogeneity of variances, the median and quartiles (M [P25, P75]) were used for representation. Between-group comparisons were performed using the Mann–Whitney U test, and within-group comparisons were conducted using the Wilcoxon signed-rank test. Categorical data were presented as proportions or percentages, and between-group comparisons were performed using the Mann–Whitney U test. All statistical tests were two-tailed, and a p value < .05 was considered statistically significant.
Results
Comparison of Pad Test Results, Urine Volume, and Frequency of Urination Before Treatment, 2 Weeks After Treatment, and 3 Months After Treatment in Both Groups of Patients
As shown in Table 1, there was no statistically significant difference in the pad test results between the treatment groups after 2 weeks (χ2 = 2.133, p = .144). After 3 months of treatment, the treatment group showed an obvious lack of urine in the pad test, and the difference was statistically significant (χ2 = 3.865, p = .049).
Comparison of Urine Pad Test Results Between Two Groups of Patients Before and After Treatment

Note. There was no statistically significant difference in the pad test results between the treatment groups after 2 weeks (χ2 = 2.133, p = .144). After 3 months of treatment, the treatment group showed an obvious lack of urine in the pad test, and the difference was statistically significant (χ2 = 3.865, p = .049).
As shown in Table 2, there were no statistically significant differences in nocturnal urine volume, nocturnal urination frequency, and nocturnal polyuria index between the two groups of patients before treatment (p > .05). Two weeks after treatment, both groups of patients exhibited a significant reduction in nocturnal urine volume, nocturnal urination frequency, and nocturnal polyuria index compared with before treatment (p < .05), with the treatment group showing a significantly greater reduction than the control group (p < .05). Three months after treatment, both groups of patients showed a significant reduction in nocturnal urine volume, nocturnal urination frequency, and nocturnal polyuria index compared with before treatment and 2 weeks after treatment (p < .05), and the treatment group had significantly lower values than the control group (p < .05).
Comparison of Urine Volume and Urination Frequency Before Treatment, 2 Weeks After Treatment, and 3 Months After Treatment in Both Groups of Patients

Compared with before treatment in this group, p < .05. bCompared with 2 weeks after treatment in this group, p < .05. cCompared with the control group, p < .05.
Comparison of Quality of Life Before Treatment, 2 Weeks After Treatment, and 3 Months After Treatment in Both Groups of Patients
As shown in Table 3, there were no statistically significant differences in the quality of life between both groups of patients before treatment (p > .05). Two weeks after treatment, patients in the treatment group had significantly better quality of life compared with the control group (p < .05), but the difference compared with before treatment was not statistically significant (p > .05). In contrast, the quality of life for patients in the control group was significantly lower than before treatment (p < .05).
Comparison of Quality of Life Before and After Treatment in Both Groups of Patients
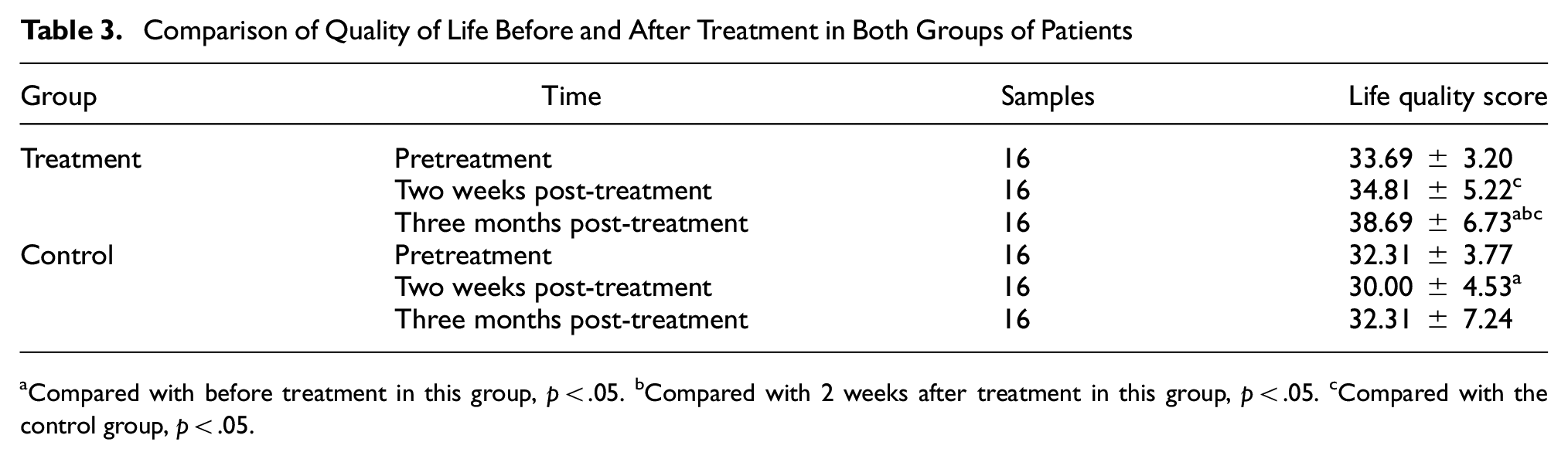
Compared with before treatment in this group, p < .05. bCompared with 2 weeks after treatment in this group, p < .05. cCompared with the control group, p < .05.
Three months after treatment, patients in the treatment group had significantly higher quality of life compared with the control group (p < .05), and it was also higher than before treatment and 2 weeks after treatment (p < .05). However, the quality of life for patients in the control group did not show statistically significant differences compared with before treatment and 2 weeks after treatment (p > .05).
Comparison of Clinical Efficacy Between Both Groups of Patients 2 Weeks and 3 Months After Treatment
As shown in Table 4, 2 weeks after treatment, the total effective rate in the treatment group (81.25%) was significantly higher than in the control group (56.25%), with a statistically significant difference (Z = −2.480, p = .013). Three months after treatment, the total effective rate in the treatment group (93.75%) was significantly higher than in the control group (75.00%), with a statistically significant difference (Z = −2.013, p = .044).
Comparison of Clinical Efficacy Between Both Groups of Patients

Discussion
Urinary incontinence is a common adverse reaction after radical prostatectomy surgery, with an incidence rate ranging from 6% to 20% (Mayekar et al., 2017), which significantly affects various aspects of patients’ physical, mental, and social activities. Urinary incontinence after radical prostatectomy is mainly stress urinary incontinence, with some cases being urge, mixed, or overflow incontinence, and a very small proportion being total incontinence. This study excludes total incontinence because these patients experience continuous and involuntary urine leakage, regularly wearing diapers, making it difficult to assess the frequency of urination. Currently, the specific causes of PPI are not yet clear, but most studies suggest it may be related to injury to the urethral sphincter and abnormalities in the detrusor muscle (Song et al., 2022). In addition, PPI patients often experience increased nocturia symptoms, and it has been reported that long-term nocturia not only reduces patients’ quality of life but also greatly increases the risk of falls (Vaughan et al., 2010). Therefore, addressing the issue of increased nocturia in PPI patients is of paramount importance.
Urinary incontinence occurring within 6 months after radical prostatectomy surgery is classified as early PPI (Wang et al, 2022). Currently, early-stage PPI patients are often treated with pelvic floor muscle training, behavioral therapy, and medication (Bauer et al., 2009). Among these, pelvic floor muscle training is preferred due to its simplicity and high safety. However, clinical observation has revealed that some patients do not accurately target the correct muscle groups during training, limiting its effectiveness (Shao et al., 2022).
Electrophysiological appropriate technology is a diagnostic and therapeutic technique that combines traditional Chinese medicine theory with modern electrophysiological principles. It involves determining precise electrical stimulation patterns under infrared visualization, selecting acupuncture points based on traditional Chinese medicine theory, and using electrical stimulation to promote blood circulation, stimulate nerve-muscle tissues, and clear meridians. Current research has indicated that electrophysiological treatment has good clinical efficacy. According to the results of a study by Su (2021), the cure rate after receiving electrophysiological treatment twice a week for 30 min each session was 24%, with an overall effective rate of 68%. It is believed that a certain frequency of electrical stimulation can increase cyclic adenosine monophosphate (cAMP) levels within damaged nerve cells, promote the expression of neurotrophic factors and receptors, facilitate nerve repair and regeneration, cause contractions of the urethral sphincter and pelvic floor muscle groups through a series of complex reflexes, and promote collagen repair in the pelvic floor, among other effects. Combining electrophysiological diagnosis and traditional Chinese medicine meridian theory can enhance the overall therapeutic effect.
Currently, there is no unified definition for urinary incontinence after radical prostatectomy. The diagnostic criteria most scholars accept include “urination that is not under subjective control; a history of radical prostatectomy with postoperative pathology confirming prostate cancer; and a positive 24-hr pad test.” Research results show that the incidence of nocturia in patients after radical prostatectomy surgery is as high as 76.1% and is correlated with urinary incontinence (Pan, 2023). Patients with an International Consultation on Incontinence Questionnaire–Short Form (ICIQ-SF) score of 6 to 10 experience a significant increase in the frequency of nocturia postoperatively (p = .023; Zhang et al, 2024). Since nocturia is closely related to falls and cardiovascular incidents, this study focuses on the nocturia condition in patients after radical prostatectomy surgery. The study subjects included are those who experienced urinary incontinence and had a nocturia frequency of two or more times after radical prostatectomy surgery.
In traditional Chinese medicine, while there may not be a specific condition called “nocturia” in the field, based on its etiology, pathogenesis, and clinical presentation, the increased nocturia symptoms in PPI patients can be classified into categories such as “urinary incontinence” and “enuresis.”Zhang Jiebin (2011) proposed the concept of “qi unable to transform water” in his work “Jing Yue Quan Shu,” suggesting that qi deficiency leads to the inability of the bladder to transform and transport urine, with the pathological site being the bladder. This condition is closely related to the kidneys, lungs, heart, liver, spleen, and other organs. Factors such as the invasion of pathogenic toxins into the body, intraoperative damage to qi and blood, and aging lead to a gradual decrease in the body’s vital energy (yuan qi). Yuan qi primarily originates from kidney essence and is the main component of kidney qi. The kidneys are the foundation of congenital essence, govern water, control the two excretory functions (urination and defecation), and are closely related to the bladder. The bladder controls the opening and closing of urination, and kidney qi provides the motive force. If kidney qi is not consolidated, and the bladder’s opening and closing are uncontrolled, urinary incontinence may occur. Therefore, by stimulating relevant acupuncture points such as those along the Bladder Meridian of Foot-Taiyang, we can invigorate yang qi in the body, activate the body’s qi circulation, enhance the organs’ ability to consolidate, and thus achieve the effect of nourishing yuan qi, strengthening the kidneys, and consolidating the foundation. Baliao, also known as Shangliao, Ciliao, Zhongliao, and Xiaoliao, includes eight acupuncture points in total, four on each side, located in the first to fourth sacral foramina, collectively referred to as the “Eight Erroneous Points” and belonging to the Bladder Meridian of Foot-Taiyang. Electrical stimulation of Baliao can warm the kidneys and strengthen yang, unblock the qi of the bladder meridian, and restore the bladder’s ability to transform and transport qi, ensuring controlled opening and closing. Furthermore, in traditional Chinese medicine, the roots of yin and yang are interdependent, and supplementing qi and consolidating the root must start with yin leading to yang. When treating PPI patients, supine acupuncture points are often selected to enhance the body’s yang qi, with Qihai playing a role in nourishing qi and warming yang.
The results of this study show that, in terms of urine volume, urination frequency, and clinical efficacy, electrophysiological appropriate technology has favorable short-term and long-term effects and can effectively improve patients’ symptoms of increased nocturia. In terms of quality of life, the treatment group’s quality of life was significantly higher than that of the control group at 2 weeks after treatment, but there was no significant difference compared with before treatment. In contrast, the control group’s quality of life was significantly lower than before treatment. Three months after treatment, the treatment group’s quality of life was significantly higher than that of the control group, and both were higher than before treatment and 2 weeks after treatment. The control group’s quality of life did not show significant differences compared with before treatment and 2 weeks after treatment. The possible reasons for these findings are as follows: (1) While electrophysiological appropriate technology can significantly alleviate patients’ symptoms of increased nocturia, it is still challenging to achieve a cure within a short period, resulting in limited improvement in patients’ quality of life. (2) Pure pelvic floor muscle training may not significantly relieve symptoms of increased nocturia in the short term and may further exacerbate patients’ anxiety and fear, leading to a lower quality of life than before treatment. In summary, electrophysiological appropriate technology has shown clear advantages and has had a positive therapeutic effect on the nocturia symptoms of PPI patients. However, this study also has certain limitations, such as a relatively small sample size, which limit its persuasiveness. Therefore, further multicenter, large-sample randomized controlled trials are needed to provide more evidence for clinical nursing decision-makers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the ethics committee of Hangzhou Hospital of Traditional Chinese Medicine (approval no. [2020]478). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to Participate
Written informed consent was obtained from individual participants.
Data Availability Statement
The datasets generated and analyzed during the present study are available from the corresponding author on reasonable request.
