Abstract
The purpose of this study was to develop a nomogram using hemogram inflammatory markers to predict the risk of infertility in patients with varicocele (VC). Patients with VC from March 2022 to June 2024 were retrospectively investigated. We divided the patients into two groups based on their fertility status. A total of 162 patients were enrolled: 81 in the infertile group and 81 in the fertile group. Statistical differences were observed between the two groups in lymphocyte, monocyte, erythrocyte, red cell distribution width (RDW), mean erythrocyte volume (MCV), mean platelet volume (MPV), platelet distribution width (PDW), neutrophil/lymphocyte ratio (NLR), platelet/lymphocyte ratio (PLR), MPV/platelet ratio (MPVPR), and systemic inflammation response index (SIRI) (p < .05). The 162 patients were divided into a modeling cohort and a validation cohort in a 7:3 ratio. A predictive nomogram was constructed based on independent influencing factors identified through univariate and multivariate logistic regression analyses. Receiver operating characteristic curve analysis, calibration curve, and decision curve analysis were used to assess the model’s performance. Multivariate logistic regression analysis indicated that erythrocyte count, PDW, NLR, and SIRI were independent influencing factors. The area under the curve for the nomogram predicting the risk of infertility in patients with VC was 0.869 in the validation cohort. The nomogram demonstrated good predictive performance. In this study, we developed an effective predictive nomogram for assessing the risk of infertility in VC patients using inflammatory markers. However, further external validation is crucial.
Background
Varicocele (VC) occurs when blood flows retrograde into the internal spermatic vein, leading to the dilation of veins in the pampiniform plexus (Jensen et al., 2017). It is a common urological vascular condition caused by deficiency or absence of valves in the spermatic veins, which commonly develops in young individuals, predominantly affecting the left side, VC can cause pain, discomfort, testicular dysfunction, and infertility in men (Clavijo et al., 2017). Most individuals with this condition are asymptomatic (Paick & Choi, 2019), but those who are symptomatic may exhibit visibly dilated veins on the scrotum’s surface resembling earthworms. VC is classified into two types: primary (idiopathic, with no identified cause) and secondary. Primary VC occurs due to insufficient valves in the internal spermatic veins. Retrograde blood reflux occurs in the pampiniform plexus. Secondary VC arises when intra-abdominal and retroperitoneal conditions increase pressure on the spermatic veins (Beddy et al., 2005; Zucchi et al., 2006). Approximately 15% of adult males in the general population have VC. It is observed in 35% to 44% of men with primary infertility and 45% to 81% of men with secondary infertility, although some patients can conceive naturally. Given that VC is likely a progressive condition, timely management is essential (Zavattaro et al., 2018).
VC is linked to valve dysfunction, which increases local pressure and causes blood to flow backward into the internal cremasteric and spermatic veins (Agarwal et al., 2009; Gat et al., 2006; Lomboy & Coward, 2016). This condition leads to ischemia, increased testicular pressure and temperature, and disrupted testicular microcirculation (Gat et al., 2006; Samanta et al., 2018). Consequently, VC increases reactive oxidative species (ROS), which damage cells and cause persistent inflammation (Agarwal et al., 2009; Moretti et al., 2009).
Various hematological and biochemical indicators can identify systemic inflammation. Recent research has identified key indicators of systemic inflammation, including the neutrophil/lymphocyte ratio (NLR), platelet/lymphocyte ratio (PLR), MPV/PLT ratio (MPVPR), and red cell distribution width (RDW) (Ates et al., 2019; Noori Alavije et al., 2022; Şahin et al., 2019; Semba et al., 2010). Specific alterations in peripheral blood cell components indicate reactions to various inflammatory illnesses (Macrez et al., 2011). In addition, researchers have studied more sophisticated inflammatory markers such as the systemic immune inflammation index (SII, neutrophil × platelet/lymphocyte) and systemic inflammation response index (SIRI, neutrophil × monocyte/lymphocyte) in inflammatory disorders and various malignancies (Tanacan et al., 2021; Yun et al., 2021).
By comparing blood samples and biochemical parameters in infertile and fertile patients diagnosed with VC, our study aims to formulate a nomogram model to predict infertility risk in VC patients using inflammatory markers.
Materials and Methods
Study Design
This study was approved by the Medical Ethics Committee. A retrospective analysis was conducted on VC patients from March 2022 to June 2024 in the Department of Andrology and Sexual Medicine. Physical examinations and scrotal color ultrasounds identified 162 VC patients. Patients were divided into two groups based on infertility status. Blood samples were taken before breakfast between 8 and 10 a.m., with 5 mL of elbow venous blood collected. Data on age, neutrophil, lymphocyte, monocyte, eosinophil, erythrocyte, red cell distribution width (RDW), MCV, platelet (PLT), MPV, platelet distribution width (PDW), NLR, PLR, monocyte/lymphocyte ratio (MLR), monocyte/eosinophil ratio (MER), MPVPR, SII, and SIRI were collected retrospectively. Differences in hemogram between the two groups were compared. Patient data were split into modeling and validation cohorts in a 7:3 ratio to develop a nomogram model and verify its predictive performance.
Inclusion and Exclusion Criteria
We divided the VC patients into two groups: infertile and fertile. The inclusion criteria for the infertile group were: (1) regular sexual activity without contraception for over a year without conception; (2) physical examination and color Doppler ultrasound evidence of VC; (3) aged 18 to 40 years; (4) normal fertility examination of the spouse; and (5) complete hemogram results available. The inclusion criteria for the fertile group were: (1) spouse has been pregnant after diagnosing VC; (2) physical examination and color Doppler ultrasound evidence of VC which was initially suspected due to the symptom of scrotal pain; (3) aged 18 to 40 years; and (4) complete hemogram results available.
Exclusion criteria included known chronic diseases (e.g., leukemia and lymphoma), gonadotoxic medication, current medications, smoking, diabetes, peripheral vascular disease, sleep apnea, diseases causing sperm excretion disorders or sperm gene abnormalities, azoospermia, acute infections affecting hemogram parameters, and those outside the defined age group.
Statistical Analysis
The Kolmogorov–Smirnov test was used to assess the normality of continuous variables. Mean and standard deviation described normally distributed continuous data, while median (interquartile range) represented non-normally distributed continuous data. The independent-sample t-test compared means of two normally distributed variables, and the Mann–Whitney U-test compared two non-normally distributed variables. Categorical variables were presented as numbers (percentages), and compared using Fisher’s exact test and chi-square test. Independent risk factors were identified using univariate and multivariate logistic regression analyses. The final model was determined by the forward LR stepwise method, including all significant variables from the multivariate analysis. A nomogram was constructed using independent risk factors from the logistic regression analysis. The model’s effectiveness was evaluated with a separate validation cohort. Receiver operating characteristic (ROC) curve analysis, calibration curves, and decision curve analysis (DCA) validated the model’s discriminatory power, calibration, clinical utility, and advantages. Data were analyzed using IBM SPSS Statistics 26 (IBM Corp, Armonk, NY, USA) and R software (version 4.3.2; R Foundation for Statistical Analysis, Vienna, Austria). Statistical significance was defined as p < .05 in two-sided analyses.
Results
A retrospective analysis was conducted on 162 VC patients meeting the research criteria, included from March 2022 to June 2024. This included 81 patients with infertility and 81 patients with fertility. The two groups did not differ significantly in the frequency of sexual activity.
Table 1 compares the two groups. The groups were comparable by age (p > .05). Significant differences were found in lymphocyte, monocyte, erythrocyte, RDW, MCV, MPV, PDW, NLR, PLR, MPVPR, and SIRI between the groups (p < .05). The infertile group had lower lymphocyte, monocyte, MCV, and SIRI, but higher erythrocyte, MPV, PDW, NLR, PLR, and MPVPR compared to the fertile group. No significant differences were found in neutrophil, eosinophil, PLT, MLR, MER, and SII.
Comparison of Age and Blood Parameters Between the Infertile and Fertile Groups
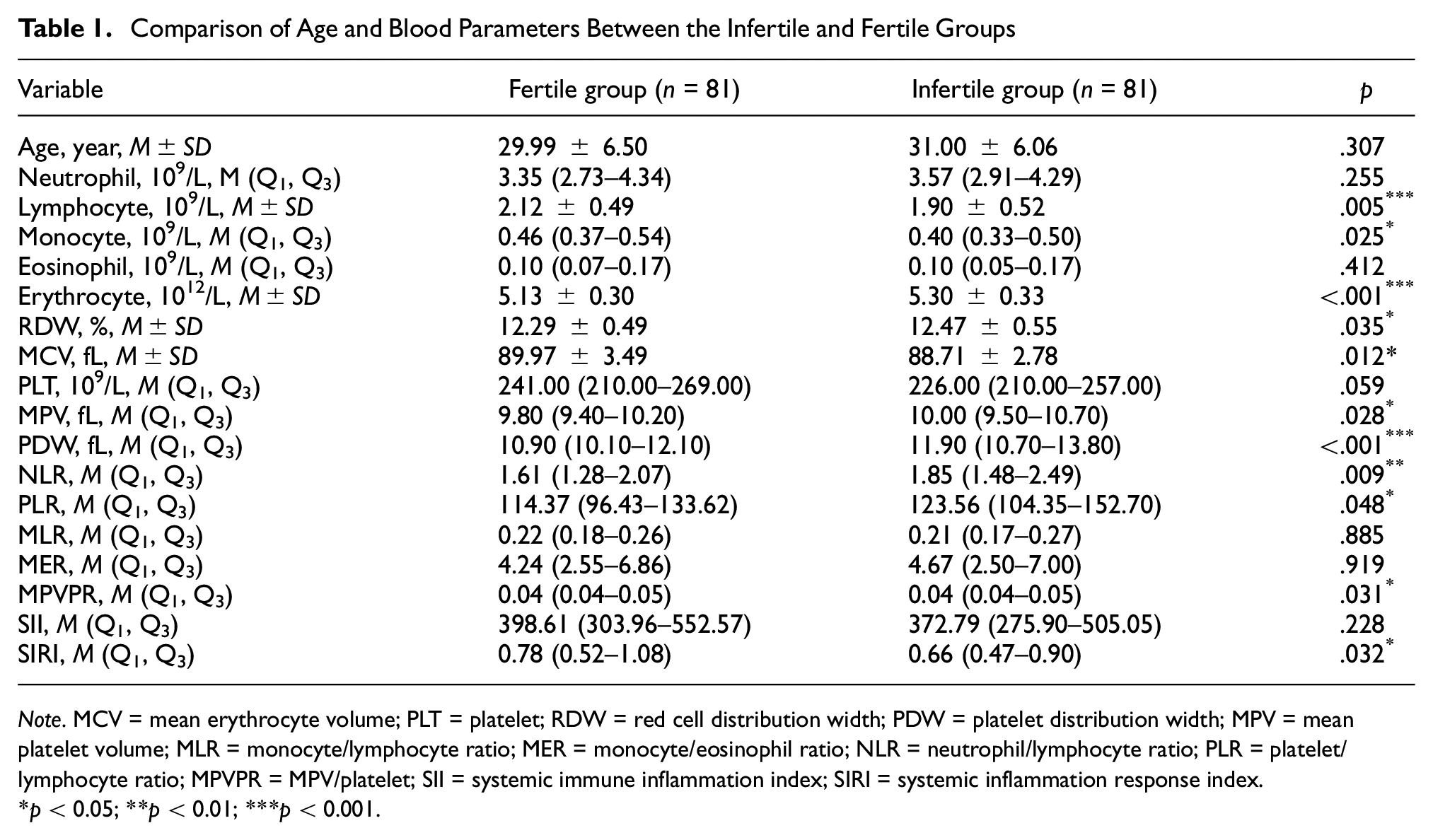
Note. MCV = mean erythrocyte volume; PLT = platelet; RDW = red cell distribution width; PDW = platelet distribution width; MPV = mean platelet volume; MLR = monocyte/lymphocyte ratio; MER = monocyte/eosinophil ratio; NLR = neutrophil/lymphocyte ratio; PLR = platelet/lymphocyte ratio; MPVPR = MPV/platelet; SII = systemic immune inflammation index; SIRI = systemic inflammation response index.
p < 0.05; **p < 0.01; ***p < 0.001.
Table 2 shows the baseline characteristics of the study patients. There were no statistically significant differences between the two cohorts in baseline characteristics.
Baseline Characteristics of the Study Patients

Note. MCV = mean erythrocyte volume; PLT = platelet; RDW = red cell distribution width; PDW = platelet distribution width; MPV = mean platelet volume; MLR = monocyte/lymphocyte ratio; MER = monocyte/eosinophil ratio; NLR = neutrophil/lymphocyte ratio; PLR = platelet/lymphocyte ratio; MPVPR = MPV/platelet; SII = systemic immune inflammation index; SIRI = systemic inflammation response index.
Univariate logistic analysis showed that erythrocyte, PDW, and MPVPR were positively correlated with the risk of infertility, while lymphocyte, monocyte, MCV, and PLT were negatively correlated (Table 3). Multivariate logistic regression analysis indicated that erythrocyte (odds ratio [OR]: 4.60, 95% confidence interval [CI]: [1.14, 18.63]; p = .032), PDW (OR: 1.51, 95% CI: [1.18, 1.95]; p = .001), NLR (OR: 5.17, 95% CI: [1.63, 16.44]; p = .005), and SIRI (OR: 0.19, 95% CI: [0.04, 0.84]; p = .029) were independent factors for infertility risk. SIRI was a protective factor, while erythrocyte, PDW, and NLR were risk factors for infertility.
The Univariate and Multivariate Logistic Analysis of Influence Factors

Note. MCV = mean erythrocyte volume; PLT = platelet; RDW = red cell distribution width; PDW = platelet distribution width; MPV = mean platelet volume; MLR = monocyte/lymphocyte ratio; MER = monocyte/eosinophil ratio; NLR = neutrophil/lymphocyte ratio; PLR = platelet/lymphocyte ratio; MPVPR = MPV/platelet; SII = systemic immune inflammation index; SIRI = systemic inflammation response index.
The ROC analysis results are summarized in Table 4. The best cut-off value of erythrocyte was 5.31 × 1012/L, with an area under the curve (AUC) of 0.684 (95% CI: [0.520, 0.848]). The highest sensitivity and specificity were 0.529 and 0.781 (p < .05). The optimal PDW cut-off for predicting infertility was 13.00 fL, with an AUC of 0.740 (95% CI: [0.591, 0.889]), and the highest sensitivity and specificity of 0.412 and 0.969 (p < .05). The best NLR cut-off was 1.88, with an AUC of 0.759 (95% CI: [0.617, 0.902]), and the highest sensitivity and specificity of 0.594 and 0.824 (p < .05). The optimal SIRI cut-off was 0.78, with an AUC of 0.676 (95% CI: [0.516, 0.836]), and the highest sensitivity and specificity of 0.594 and 0.765 (p < .05) (Figure 1).
ROC Analysis of Factors Affecting the Risk of Infertility

Note. AUC = area under the curve; CI = confidence interval; PDW = platelet distribution width; NLR = neutrophil/lymphocyte ratio; SIRI = systemic inflammation response.
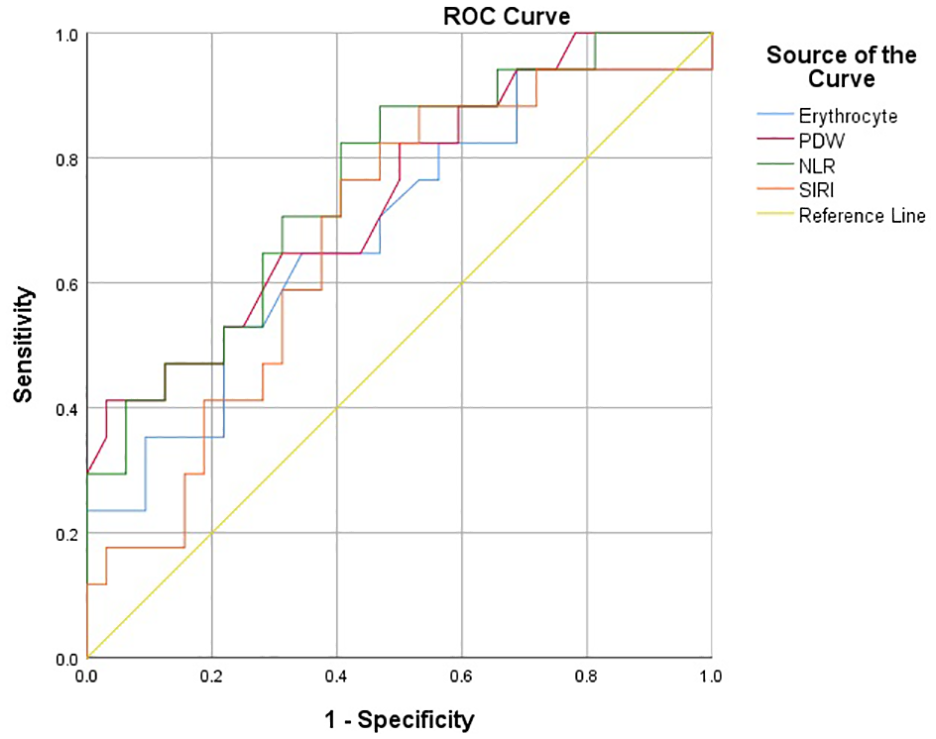
Receiver Operating Characteristic (ROC) Analysis of Erythrocyte, Platelet Distribution Width (PDW), Neutrophil/Lymphocyte Ratio (NLR), and Systemic Inflammation Response Index (SIRI)
The nomogram model for predicting infertility risk in VC patients and its performance evaluation in the validation cohort are presented (Figure 2). In the validation cohort, the AUC was 0.869, indicating high accuracy. Calibration plots of the nomogram in the modeling and validation cohorts showed good calibration for predicting infertility in VC patients. DCA demonstrated the nomogram’s clinical applicability, with good net benefits for VC patients.

A Nomogram Predicting the Risk of Infertility in Patients With VC and Its Performance Evaluation in the Validation Group. (A) Predict the Risk of Infertility by the Nomogram Model. (B) Receiver Operating Characteristic Curve Analysis. (C) Calibration Plots. (D) Decision Curve Analysis Was Used to Evaluate the Performance of the Risk of Infertility Predicted by the Nomogram Model in the Validation Group
Discussion
Current research links VC to various etiologies, including pathological hyperthermia, oxidative stress, apoptosis, inflammation, and hormonal effects on infertility (Hassanin et al., 2018). Inflammation, in particular, has received significant research attention regarding its detrimental effects on fertility. This study aimed to investigate the association between inflammatory markers and infertility in VC patients. Our findings showed that infertile VC patients had significantly higher erythrocyte, RDW, MPV, PDW, NLR, PLR, and MPVPR values than the fertile group. Infertile VC patients also had lower lymphocyte, monocyte, MCV, and SIRI values. We found that erythrocyte, PDW, NLR, and SIRI independently influenced the risk of infertility.
Inflammatory indicators are not only predictive in VC but also in various cancers and systemic disorders (Guthrie et al., 2013). High NLR, PLR, MPVPR, and RDW values forecast mortality and morbidity in cardiovascular disease and are linked to micro- and macro-vascular problems (Ates et al., 2019; Noori Alavije et al., 2022; Şahin et al., 2019; Tsuboi et al., 2013). Higher MPV is associated with VC (Bozkurt et al., 2012; Çoban et al., 2015), aligning with our findings. PDW measures platelet distribution and function in the blood. A high PDW indicates irregular platelet volume, with larger platelets being more metabolically and enzymatically active (Hong et al., 2014; Nena et al., 2012). Our study found significantly higher PDW in the infertile group. Previous studies on VC and PDW levels have had mixed results, and there are fewer studies on PDW and VC. Additional cohort and case–control studies are needed to support our findings.
It has been reported that oxidative stress triggers vascular inflammation, playing a key role in atherosclerotic disease progression (King & Loeken, 2004; May et al., 2019). Oxidative stress, directly and through cytokine release due to inflammation, damages erythrocytes, decreasing their survival and leading to anemia, which increases RDW (Friedman et al., 2004; Lippi et al., 2009). High oxidative stress can also shorten erythrocyte lifespan and cause the early release of large immature erythrocytes, possibly explaining the higher erythrocyte count in VC patients. Another important aspect of RDW is its strong correlation with inflammatory parameters (King & Loeken, 2004; May et al., 2019). RDW has been reported as a marker for vascular problems in adult diabetic patients (Malandrino et al., 2012). Our research found that RDW was significantly higher in the infertile group compared to the fertile group. These studies support our findings, indicating that the degree of inflammatory response is more severe in infertile patients and RDW may be used as a marker for venous disease, such as VC, which is associated with male infertility.
Inflammatory pathologies play a role in the pathogenesis of VC, with increased inflammatory cytokine expression stimulating neutrophils in VC patients (Tetè et al., 2012). Studies have found significantly higher NLR in chronic venous insufficiency (Mosmiller et al., 2017). Given the involvement of systemic inflammation and venous insufficiency in VC, NLR becomes significant in these patients. The pathogenesis of VC-induced infertility has been linked to inflammation, with higher NLR in patients not showing improvement in sperm parameters after varicocelectomy (Ates et al., 2019). Our study indicates that NLR, PLR, and MPVPR can be markers of venous disease. We found significantly higher NLR, PLR, and MPVPR in infertile VC patients compared to the fertile group.
Recently, SII and SIRI have gained attention as new inflammatory indices that are straightforward, affordable, and simple to detect. They may represent the body’s systemic inflammatory response as they are derived from neutrophil, lymphocyte, monocyte, and platelet counts (Chen et al., 2017; Fu et al., 2018; Geng et al., 2018). SII and SIRI are linked to the development, course, and prognosis of various human diseases, including inflammatory diseases, connective tissue diseases, bleeding disorders, and malignant tumors (Aziz et al., 2019; Li et al., 2020). Different anti-inflammatory cytokines may be released, depressing the immune system and causing lymphocyte death (Liu et al., 2020). Previous studies found higher SII and SIRI levels associated with an enhanced inflammatory response and worse prognosis. Research on SIRI and SII values in various malignancies and illnesses exists, but there are no studies on VC patients with infertility based on SII and SIRI values. Our study found that infertile VC patients had significantly lower SIRI compared to the fertile group.
Testicular spermatogenic function is commonly evaluated using semen analysis. However, due to the private nature of the sperm collection process, some patients may feel tense and unable to adapt to the new environment. This tension, combined with improper collection methods and incorrect periods of abstinence, can result in insufficient or no semen samples. In addition, improper transport and contamination of semen samples can lead to inaccurate test results. Therefore, detecting inflammatory markers is more stable. In our study, high erythrocyte, PDW, and NLR values, along with low SIRI values, were found to be significant parameters predicting infertility. Specifically, when erythrocyte >5.31, PDW >13, NLR >1.88, and SIRI <0.78 were used as cut-off values, the risk of infertility was greatly increased. In terms of semen analysis, the fertile group exhibited higher sperm concentration and progressive motility compared to the infertile group.
If a man with VC is screened for abnormal blood parameters without other disease, he is considered to have a higher chance of infertility, prompting referral to an infertility specialist for VC repair. In addition, inflammatory markers can predict the success of varicocelectomy. Low NLR and MLR values before varicocelectomy were significant predictors of success (Duran et al., 2022). Erdogan et al. found that low preoperative SII (334.6 × 103/mm3) and SIRI (0.882 × 103/mm3) levels are beneficial in identifying patients who would benefit from surgery in infertile men with VC (Erdogan & Ok, 2024).
However, our study has limitations including its retrospective design, lack of sex hormone, testicular volume, color Doppler ultrasound data, and the small number of patients from a single center. Further prospective studies are needed to confirm the results.
Conclusion
Our study found statistically significant differences in lymphocyte, monocyte, erythrocyte, RDW, MCV, MPV, PDW, NLR, PLR, MPVPR, and SIRI between the infertile and fertile groups. Erythrocyte, PDW, NLR, and SIRI were identified as independent risk factors for infertility. Based on our results, we recommend using these inflammatory parameters as markers to screen for infertility in VC patients.
Footnotes
Acknowledgements
All authors have no acknowledgements to disclose.
Author Contributions
T.J. conceived of the study and drafted the manuscript. L.W. and L.Z. participated in the procedure. L.W. carried out data curation. L.Z. performed the statistical analysis. T.J. and H.J. participated in its design and coordination. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
This study was approved by the ethics committee of The Second Hospital of Dalian Medical University.
