Abstract
Ejaculatory duct obstruction (EDO) is a rare but treatable cause of male infertility. This case report describes a 28-year-old male with obstructive azoospermia. The patient came to our hospital after a fertility check-up revealed azoospermia. A subsequent semen analysis confirmed azoospermia. Transrectal ultrasonography (TRUS) and magnetic resonance imaging (MRI) revealed bilaterally enlarged seminal vesicles and thickened, calcified ejaculatory duct walls. The patient underwent transurethral seminal vesiculoscopy and transurethral resection of the ejaculatory ducts (TURED) for presumed partial EDO. Despite two transurethral seminal vesiculoscopy and TURED procedures, postoperative semen analysis still showed azoospermia. TRUS indicated non-contractile seminal vesicles and an unobstructed ejaculatory duct. The patient ultimately underwent percutaneous epididymal sperm aspiration for assisted reproductive technology and his spouse got pregnant. We identified a case of azoospermia caused by a rare combination of partial and functional ejaculatory duct obstruction. There are currently no reports of similar cases. This case report aims to provide valuable insights for diagnosing and treating EDO.
Introduction
Ejaculatory duct obstruction (EDO) is a rare condition, either congenital or acquired, accounting for 1% to 5% of male infertility cases. Azoospermia (absence of sperm in ejaculate) occurs in 15% of infertile men, with obstructive azoospermia accounting for 40% of these cases. (Modgil et al., 2016). Azoospermia due to EDO is uncommon, but advancements in seminal plasma biochemical techniques and imaging have increased its diagnosis. Unlike vas deferens and epididymal obstruction, EDO involves abnormal pathology in the distal ejaculatory duct, obstructing sperm excretion. It is one of the obstructive factors causing male infertility and can be treated surgically (Meacham et al., 1993). Most EDO cases are treated with transurethral resection of the ejaculatory ducts (TURED).
However, we identified a patient with azoospermia due to functional and partial EDO who underwent seminal vesiculoscopy followed by two TURED procedures with a holmium laser. Postoperative semen analysis still showed azoospermia. After unsuccessful surgeries, the patient underwent percutaneous epididymal sperm aspiration for assisted reproductive technology. This study reports a rare case of azoospermia due to functional and partial EDO and reviews the current literature.
Case Presentation
A 28-year-old unmarried male with his girlfriend presented to our hospital in August 2017 after a semen analysis revealed azoospermia. The patient reported that azoospermia was detected during a premarital examination a year ago, and subsequent semen examinations over the past year consistently showed an absence of sperm after centrifugation. A subsequent testicular biopsy revealed sperm in the testes. The patient had normal urination and did not report any associated diseases, such as orchitis, epididymitis, testicular trauma, gonorrheal urethritis, or polycystic kidneys. Physical examination revealed normal size and texture of both testicles, no varicocele, and palpable bilateral epididymides and vas deferens. Semen analysis showed a volume of 3.5 ml, pH of 6.0, and a fructose concentration in seminal plasma of 0.5 mmol/L. Sex hormone levels were within normal reference ranges. A scrotal ultrasound examination showed normal-sized testes. Transrectal ultrasonography (TRUS) revealed bilateral enlargement of seminal vesicles with fluid retention and thickened, calcified ejaculatory duct walls. Magnetic resonance imaging (MRI) of the seminal vesicles showed bilateral enlargement (left: 3.98×2.28 cm; right: 4.79×2.78 cm) with intraluminal mucinous material retention (high-protein substance) and multiple stones (Figure 1).

MRI of the Seminal Vesicles Before the First Surgery Showed Bilateral Enlargement With Intraluminal Mucinous Material Retention and Multiple Stones
Based on the patient’s medical history and examination results, we diagnosed him with obstructive azoospermia caused by EDO. The patient underwent his first surgery, which consisted of seminal vesiculoscopy and TURED using a holmium laser. During the operation, the right ejaculatory duct opening was identified at the prostatic utricle. A guide wire was used to access the right seminal vesicle, thoroughly flushing out calcifications and old blood clots. The normal opening of the left ejaculatory duct could not be found. Therefore, a weak spot in the left wall of the prostatic utricle was breached to enter the left seminal vesicle, revealing significant dilation and numerous gray-white secretions, which were thoroughly flushed out using irrigation. Before irrigation, a sample of seminal vesicle contents was extracted for pathological examination, revealing red blood cells in all fields under the microscope, a white blood cell count of 6 to 8 per high-power field, and a large number of sperm. Subsequently, a holmium laser fiber was inserted to dilate the ejaculatory duct opening with a power setting of 1.0 J/12 Hz. A urethral catheter was left in place for 2 days. During the first 2 weeks post-operation, the patient was encouraged to ejaculate every 3 days. Despite multiple semen analyses over the year post-operation, no sperm was detected. The patient was readmitted in November 2018 due to persistent azoospermia. Semen analysis at our hospital showed a volume of 2 ml, a pH of 6.7, and a fructose concentration in seminal plasma of 1.1 mmol/L. MRI of the seminal vesicles revealed bilateral enlargement with signs of mucinous or proteinaceous fluid retention (Figure 2). Compared with the MRI from August 2017, there was a slight improvement. The patient underwent a second surgical procedure due to EDO. Intraoperatively, we found that the bilateral ejaculatory duct openings, previously dilated during surgery, were normal. The conditions observed inside the bilateral seminal vesicles were similar to those during the first surgery, characterized by wall calcifications, cavity dilatation, and numerous old blood clots and gray-white secretions. We performed thorough irrigation again and used a holmium laser to further dilate the ejaculatory duct openings to the verumontanum. Postoperative TRUS monitoring of the patient’s ejaculation process in real-time showed non-contractile seminal vesicles, unobstructed ejaculatory ducts (Figure 3), and retrograde semen flow within the vesicles (Supplemental Video 1). After the urethral catheter was left in place for 2 days, the patient was advised to ejaculate every 3 days. Despite two surgical procedures, multiple postoperative semen analyses still showed azoospermia. Four months postoperatively, semen analysis showed a volume of 2.5 ml, a pH of 7.1, and a fructose concentration in seminal plasma of 0.1 mmol/L.

MRI of the Seminal Vesicles Before the Second Surgery Showed Bilateral Enlargement With Signs of Mucinous or Proteinaceous Fluid Retention
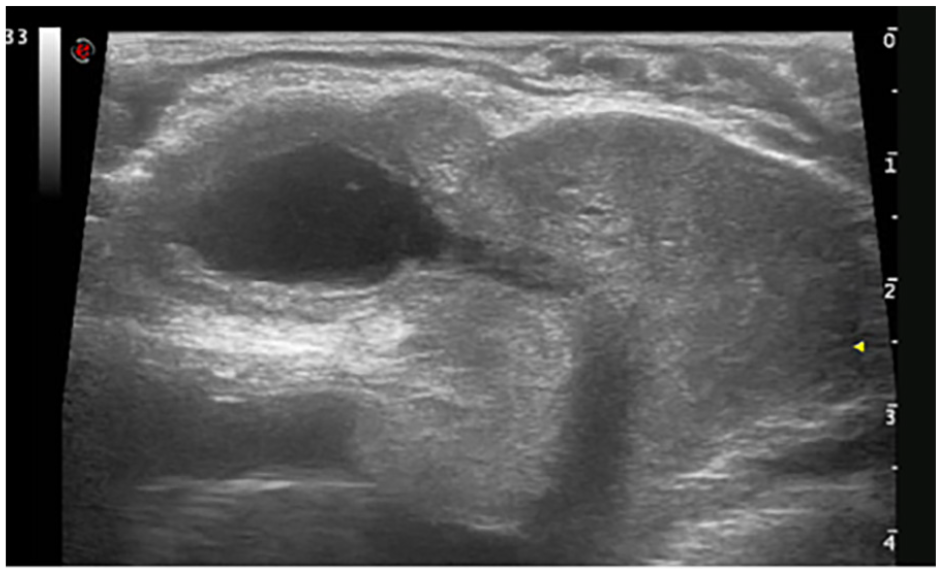
TRUS Showed Non-Contractile Seminal Vesicles and Unobstructed Ejaculatory Ducts When Monitoring the Patient’s Ejaculation Process in Real-Time
Literature Review
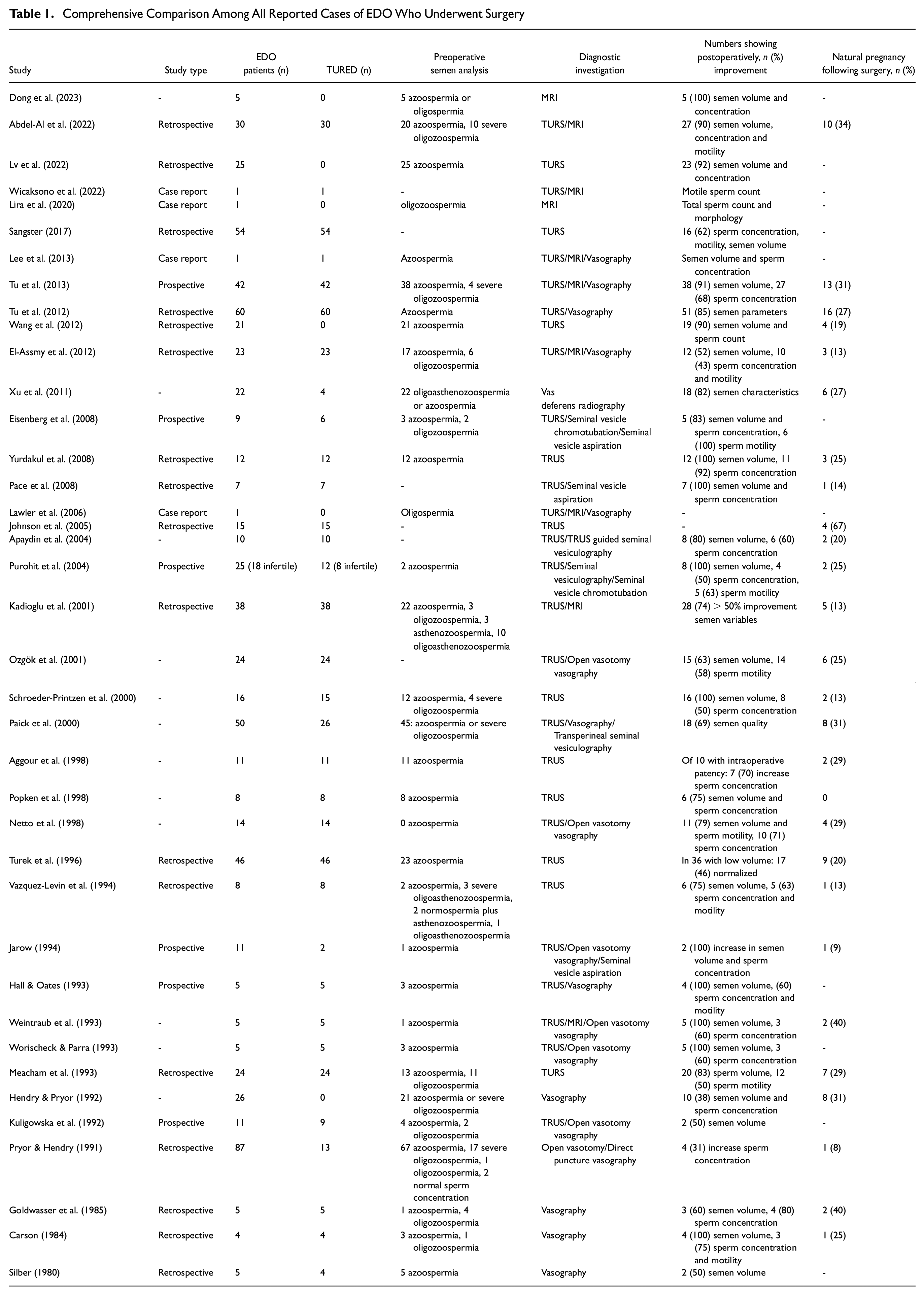
We searched the PubMed and Embase databases for similar cases and compared the findings across them. Thirty-nine articles were finally included. All included patients had EDO and underwent surgery due to poor semen quality (Table 1).
Comprehensive Comparison Among All Reported Cases of EDO Who Underwent Surgery
Discussion
EDO is a rare cause of male infertility, often presenting with symptoms such as low ejaculate volume, post-ejaculatory pain, hematospermia, or infertility. This condition, although uncommon, can arise from congenital or acquired disorders. Acquired EDO often results from seminal vesicle calculi, post-inflammatory scar formation at the prostate, a history of indwelling urethral catheter use, urethral trauma, or prior transurethral surgeries such as bladder-neck incision or transurethral resection of the prostate. Congenital EDO may be caused by atresia or stenosis of the ejaculatory ducts as well as by Müllerian duct (utricular) or Wolffian duct (diverticular) cysts. Rare Wolffian duct developmental abnormalities that lead to seminal vesicle cysts, ipsilateral renal agenesis, ejaculatory pain, and EDO are known as Zinner syndrome.
Diagnosing EDO requires a high index of suspicion. Early endoscopic treatment can reverse symptoms and prevent partial obstruction from progressing to complete bilateral obstruction (Johnson et al., 2005). In EDO patients, semen analysis frequently reveals the only abnormal test results, with pathognomonic parameters such as low-volume azoospermia or severe oligozoospermia, low pH, and low or absent fructose, despite normal secondary sex characteristics, testicular findings, and hormonal profiles (Smith et al., 2008). Diagnosis relies on semen analysis, typically showing azoospermia with low ejaculate volume, followed by imaging for confirmation. Vasography was once the preferred diagnostic test, but due to the risks of vassal injury and stricture (Paick, 2000), TRUS and MRI are now the preferred imaging modalities (Belker & Steinbock, 1990). Once confirmed, EDO can be managed surgically, underscoring its significance in the differential diagnosis of male factor infertility (Yurdakul et al., 2008).
TRUS is the preferred method for evaluating patients suspected of EDO, with more than 85% of published studies supporting its use (Mekhaimar et al., 2020). In cases of ambiguous TRUS results, pelvic MRI may be utilized. TRUS plays a pivotal role in diagnosing EDO. Specific findings such as seminal vesicle width >1.5 cm or ejaculatory duct >2.3 cm, particularly when accompanied by ductal cysts or calcifications, help confirm the diagnosis (Jarow, 1993). In recent years, TRUS has replaced vasography as the primary diagnostic tool for suspected EDO, improving urologists’ diagnostic capabilities and treatment strategies. Treatment of EDO should be considered for patients with infertility due to obstructive azoospermia or severe oligozoospermia, recurrent hematospermia, and/or pre-ejaculatory pain. Before performing endoscopic resection, any medications that could potentially cause physiological EDO should be discontinued. TURED, first described by Farley and Barnes in 1973 for treating ejaculatory duct stenosis, remains the cornerstone of EDO treatment (Farley & Barnes, 1973). Most patients diagnosed with EDO through seminal vesiculography or chromotubation are candidates for TURED.
Regarding the success rate of TURED, Smith et al. (2008) demonstrated in a decade-long study that men treated with TURED for low-volume azoospermia or low-volume oligoasthenospermia had a similar likelihood (65%–70%) of semen quality improvement. Another study revealed that the underlying cause of EDO significantly influences the likelihood of successful symptom resolution following TURED. The study reported that patients with congenital EDO had excellent treatment success rates: 100% improvement in semen parameters (motility and volume), 83% improvement in sperm count, and a 66% conception success rate. In contrast, patients with acquired EDO showed only 37.5% improvement in semen parameters, with only 12.5% achieving conception (Netto et al., 1998). A smaller series involving 16 patients indicated that EDO caused by cysts responded better to TURED than obstruction caused by calcification. The same study noted that patients with partial EDO responded better to TURED, with 94% showing improvement, compared with 59% of men with complete EDO showing improvement in postoperative semen analysis (Kadioglu et al., 2001).
Complications following TURED occur in approximately 13% to 26% of patients (Eisenberg et al., 2008; Kadioglu et al., 2001; Pryor & Hendry, 1991). In the immediate postoperative period, patients may develop acute epididymoorchitis (Eisenberg et al., 2008) or gross hematuria (Meacham et al., 1993). The incidence of these immediate complications is typically low. In a study of 24 patients treated for EDO with TURED, two developed gross hematuria requiring repeat catheterization (Eisenberg et al., 2008). Another retrospective study involving 23 patients reported two cases of postoperative epididymoorchitis (El-Assmy et al., 2012). Early and late complications are less frequent. Clinicians should be aware of the potential for early postoperative complications. Patients should be counseled regarding the potential outcomes of TURED. Approximately 10% to 15% of patients undergoing TURED for low-volume azoospermia transition to normal-volume azoospermia (Xu et al., 2011), typically indicating more proximal obstruction at the epididymal level, which may be treatable with epididymovasostomy (Smith et al., 2008). Approximately 4% of patients treated with TURED for partial EDO become azoospermic, likely due to postoperative fibrosis of the ejaculatory ducts (Xu et al., 2011). Therefore, men with oligozoospermia due to partial EDO should be advised to undergo preoperative sperm cryopreservation for assisted reproduction in case they become azoospermic postoperatively.
EDO is categorized into three types: partial, complete, and functional. In this case, a normal ejaculate volume (3.5 ml preoperatively) suggested partial EDO. Based on the patient’s preoperative TRUS and MRI findings, we initially diagnosed partial obstruction and performed TURED. During the operation, we confirmed our diagnosis by accessing the right seminal vesicle via the right ejaculatory duct. However, the outcomes of both surgeries were unsatisfactory. During the second surgery, we observed normal dilation of the ejaculatory duct opening; however, the condition inside the seminal vesicles remained essentially unchanged from the first surgery. Therefore, we considered other potential factors, including the rare condition of functional EDO. There are currently no reported cases involving partial obstruction combined with functional obstruction. Approximately 50% to 80% of the male ejaculate is produced in the seminal vesicles (Tauber et al., 1975), and their compliance and contractility resemble those of the bladder. Functional or neurological dysfunction of the seminal vesicles can lead to functional EDO, analogous to neurogenic bladder-induced voiding dysfunction (Schroeder-Printzen et al., 2000; Turek et al., 1998). To confirm our hypothesis, we used TRUS to monitor the patient’s postoperative ejaculation process in real-time. We observed that the ejaculatory duct was unobstructed, but during ejaculation, the seminal vesicles did not contract, leading to semen reflux. Finally, we diagnosed the patient with partial obstruction combined with functional obstruction of the ejaculatory ducts. The patient underwent percutaneous epididymal sperm aspiration for assisted reproduction due to azoospermia.
From the diagnosis and treatment process of this patient, we conclude that for patients with obstructive azoospermia, if seminal vesicle MRI shows abnormal enlargement with mucus retention, wall thickening, and calcification, dynamic obstruction should be considered. These patients often have unsatisfactory postoperative outcomes, and alternative assisted reproductive technologies should be considered to address fertility issues.
Conclusion
This article presents a rare case of EDO. To our knowledge, this is the first documented case of partial obstruction combined with functional obstruction in EDO. The case’s novelty lies in the identification of dynamic factors through TRUS. We hope this report offers valuable insights into the diagnosis and treatment of EDO.
Footnotes
Author Contributions
TJ and HJ constructed this study. LZ and LHW performed figures plotted and writing. TJ and HJ were responsible for the critical reading of the manuscript. All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
This study was approved by the ethics committee of The First Affiliated Hospital of Dalian Medical University.
Patient Consent Statement
The authors bear full responsibility for all aspects of the work, ensuring that inquiries pertaining to the veracity or integrity of any fragment thereof are duly examined and rectified. The patient provided written informed consent for publication of the report and accompanying photos. He reviewed the submission version of the report and confirmed that its content was accurate. Informed consent was obtained from the patients. The authors certify that they have obtained informed consent from the patient to publish his details.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
