Abstract
The AZFc partial deletions of Y chromosome and lifestyle/epidemiological factors such as the use of smokeless chewing tobacco (SCT) exhibit intriguing variations in their association with male infertility across the population, ethnicity, and genetic background. Here, a pioneering attempt has been made to elucidate the interactions of such deletions with the habits of SCT consumption among the participating individuals, using their large epidemiological data. This screening program was conducted among Bengali-speaking men in West Bengal, India. We screened the prevalence and association of distinct partial deletions (gr/gr, b1/b3, and b2/b3) of the AZFc region using locus-specific sequence-tagged site (STS) markers among 728 case subjects and compared them with 264 ethnicity- and age-matched proven-fertile control men. The recorded epidemiological data of the study group and the outcome of partial deletion analysis were compiled to frame the plausible Gene × Epidemiological factor (G × E) interactions. The gr/gr deletion was reported to be significantly associated with azoospermic (p = .0015, odds ratio [OR] = 3.413) and oligozoospermic (p = .0382, OR = 3.012) case subgroups, and b1/b3 deletions were also detected among the infertile persons only. The G × E model revealed that men who carried microdeletions as well as were SCT users had an elevated risk of infertility (p = .002, OR = 6.38). The study highlights the fact that AZFc partial deletions and SCT, when co-occurred, synergistically increase the risk of infertility among men. This work helps to get more insight into the etiology of male infertility in the light of gene–environmental interaction.
Keywords
Introduction
Infertility is one of the major health challenges across the globe, and approximately 15% of couples suffer from the issue of infertility. The underpinning cause of infertility includes both male and female factors, and in nearly 50% of cases, the cause of male infertility is of genetic origin (Agarwal et al., 2015; Choy & Eisenberg, 2018). Among all the genetic factors, Y chromosomal microdeletions (YCMDs) are the most prevalent structural anomaly (O’Flynn O’Brien et al., 2010; Rao et al., 2004). Azoospermia factor (AZF) region, located on the distal arm of the Y chromosome (Yq), acts as a hotspot for microdeletion (Tiepolo & Zuffardi, 1976). It carries functionally active genes that control testicular maturation and differentiation related to spermatogenesis (Pieri et al., 2002; Vogt et al., 1992). Among all the Y chromosomal AZF microdeletions, the AZFc deletion was reported to be the most prevalent (about 80%) among azoospermic and severe oligozoospermic men, followed by AZFb (1%–5%) and AZFa (0.5%–4%) deletions (Krausz et al., 2014; Pieri et al., 2002; Shamsi et al., 2011).
The AZFc region, spans approximately 4.5 Mb of distal part of the Yq, is one of the most comprehensively studied AZF loci due to its high deletion frequency and it harbors multicopy genes associated with male infertility, leads to varied spermatogenic dysfunction (Cram et al., 2000; Foresta et al., 2001; Krausz et al., 2000; McElreavey & Krausz, 1999). Alongside classical AZFc complete (b2/b4; ~3.5 Mb) deletions, three major partial deletions within the AZFc sub-region have been identified using sequence-tagged site (STS) genotyping. These are gr/gr, b1/b3, and b2/b3 deletions, which eliminate about 1.6 Mb, 1.6 Mb, and 1.8 Mb segments, respectively, from the AZFc locus (Figure 1; Kuroda-Kawaguchi et al., 2001; Repping et al., 2002; Skaletsky et al., 2003). Among the AZFc partial deletions, the most prevalent is gr/gr deletion (Stouffs et al., 2011), results in the removal of two of the four copies of DAZ (Deleted in azoospermia) gene, one of the two CDY1 (Chromodomain protein on Y, 1) and BPY2 (Basic protein Y, 2) genes. Thus, gr/gr deletions lower the number of copies of gene families on the Y chromosome but do not fully eliminate any genes exclusive to the testis. In comparison to gr/gr deletion, the b2/b3 is a slightly larger deletion (1.6 Mb vs. 1.8 Mb) and removes almost the same genetic segment as the gr/gr deletion. The b1/b3 deletion, which differs from the gr/gr and b2/b3 deletions by its overlap with the part of the AZFb segment, results in the loss of the six copies of RBMY1 (Ribonucleic acid binding motif, Y, 1) and both functional copies of PRY (PTPN13 like Y-linked) gene (Colaco & Modi, 2018; Repping et al., 2003).

Y chromosome with AZFc Sub-deletions defined by Sequence-Tagged Site (STS) markers. A schematic presentation shows the AZFc region with three partial deletions, gr/gr (1.6 Mb), b1/b3 (1.6 Mb), and b2/b3 (1.8 Mb) on the long arm (Yq) and SRY on the short arm (Yp) of the Y chromosome. The partial deletions are identified by specific STS markers used in screening. Ampliconic sequences are annotated as green (g), red (r), and blue (b) here
Besides genetic risk, epidemiological risk factors such as an incorrect lifestyle and addiction have been reported as risk factors for infertility among men (Barratt et al., 2017; Rai & Rai, 2016). Different chemical and physical agents (Leisegang & Dutta, 2021), tobacco (both in smoke and chewing forms), have been identified as potential epidemiological risk factors that impair spermatogenesis. According to Global Adult Tobacco Survey GATS 2 India 2016–2017, India possesses world’s largest tobacco chewers’ population and there are about 28.6% of people who are addicted to tobacco, of whom 10.7% smoke cigarettes and 21.4% use smokeless chewing tobacco (SCT; Global Adult Tobacco Survey GATS 2 India 2016–2017). Tobacco use is known to affect sperm concentration, motility, and morphology, as well as cause DNA damage (Borkhataria et al., 2020; Dikshit et al., 1987; Said et al., 2005; R. Sharma et al., 2016; Sofikitis et al., 1995; Sunanda et al., 2014). However, studies on the combined effects of genetic risk factors like partial Y microdeletion and tobacco exposure on infertility among males are almost lacking. But it is of the utmost interest to know how genetic and habitual risk factors act synergistically to increase the risk of spermatogenic impairment.
This study aimed to examine the Y-chromosomal AZFc partial deletion as a genetic risk factor and the habits of using SCT as an epidemiological/habitual risk factor. We capitalized on a large sample of Indian Bengali males of West Bengal, distinguished by their SCT consumption patterns. Our study aimed to address two primary research questions: first, whether AZFc partial deletions and SCT use are independently associated with male infertility incidence; second, in cases where these two factors co-occur, how they interact to elevate the risk of male infertility. For the first time ever, we have attempted to elucidate a comprehensive exploration of the multifactorial risks of spermatogenic failure, offering insights into the Gene × Environment model for male infertility.
Method
Ethics Approval
This study was approved by the Institutional Ethics Committee constituted by the University (Institutional Review Board [Approval No. CU/BIOETHICS/HUMAN/2306/3044/2018]).
Inclusion and Exclusion Criteria of the Study Subjects
We recruited case subjects who were clinically diagnosed with impaired fertility based on semen analysis results. Individuals with chromosomal aberrations, abnormal karyotypes, endocrinological abnormalities, immunological defects, and infections were excluded. Infertile cases with obstructive azoospermia, congenital/acquired anatomical defects, or any history of surgical intervention of the urogenital system were all excluded. Men with classical AZF microdeletions were not considered in the present study.
The control group consisted of proven age-matched fertile men. They shared the same ethnicity and demographic attributes as the case subjects. They reported to the clinic for health issues other than infertility and were proven fertile as they all had their own biological offspring.
Study Subjects
Based on the above-mentioned criteria, a cohort of 728 infertile males aged between 20 and 50 years (mean age 34.1 ± 3.98 years) were recruited from the clinic between December 2018 and January 2023. Simultaneously, a group of 264 fertile men of same age range (mean age 33.6 ± 4.21 years) with proven biological fatherhood were included as controls. All participants in this study were Bengali-speaking, demographically homogeneous, and residents of the state of West Bengal, India, over generations. These criteria negate the possibility of population substructure within the sample cohort. Participants were personally interviewed, providing epidemiological information on personal, conjugal, and lifestyle attributes using a pre-printed questionnaire. To ensure utmost confidentiality, all participant records were kept anonymously in the laboratory, identified only by a specific code. Each individual who agreed to participate signed a written informed consent for the use of their donated tissue (blood and semen) samples in the study while maintaining confidentiality.
Stratification of Study Groups
The study population was categorized as non-obstructive azoospermic [NOA] (n = 332; absence of sperm in the ejaculate), oligozoospermic (n = 93; sperm concentration: <15 million/mL of ejaculate), severe oligozoospermic (n = 67; sperm concentration: ≤5 million/mL of ejaculate), normozoospermic (n = 174; sperm concentration: >15 million/mL of ejaculate) with idiopathic infertility, and asthenozoospermic (n = 62; sperm motility: <32% and sperm count: ≥15 million/mL of ejaculate) individuals following guidelines laid by the World Health Organization (WHO, 2010) with the help of clinicians. As “normozoospermia” and “fertility” are not synonymous, normozoospermic men who were reported to be infertile were classified as “idiopathic” cases. All the “control” men had fathered at least one child and identified normozoospermic upon seminogram. In addition, all participants (both case and control men) were stratified as “SCT users” or “SCT non-users” based on their declared SCT consumption habits in their epidemiological records.
Tissue Sample Collection
Participants’ blood samples were taken with the help of laboratory technicians through the venepuncture method and collected in EDTA vacutainer tubes.
DNA Extraction
Genomic DNA (gDNA) was extracted from whole blood samples with the help of DNA extraction and purification kit (QIAamp Blood Mini Kit, Qiagen), based on the manufacturer’s protocol. The isolated DNA’s quality (purity) and quantity were assessed using 0.8% agarose (SRL) gel electrophoresis and NanoPhotometer (Implen), respectively. The gDNA was then stored at −20°C for future analysis.
Screening for AZFc Partial Deletions
A total of six sequence-tagged site (STS) markers were used, with five being specific to Y chromosome AZFc partial deletions (sY1191, sY1291, sY1161, sY1206, and sY1201; Krausz et al., 2014; Repping et al., 2003). The marker sY14 served as an internal control and is specific to the sex-determining region on Y-chromosome (SRY). Detailed information on each of the STS markers is presented in supplementary Table S1. Failure to amplify a certain STS marker or group of markers indicates partial Y-chromosome sub-deletions within AZFc region. The gr/gr partial deletion is characterized by the absence of sY1291, while the lack of sY1191 detects the b2/b3 sub-deletion. The b1/b3 sub-deletion was validated by the absences of sY1161, sY1191, and sY1291. Thus, a conventional plus (+) or minus (−) sign was used to denote the presence or absence of the sub-deletions as presented in Table 1.
Deletion-Specific Coverage of STS Makers in AZFc Regions

Note. For each sequence-tagged site (STS) marker, plus (+) and minus (−) signs, respectively, denote the presence or absence of the amplicon.
All PCR were conducted in a simplex reaction. Each PCR contained a total of 10 μL of reaction mixture consisting of PCR master mix (GoTaq Green Master Mix, Promega), nuclease-free water (Promega), STS marker-specific forward and reverse primers (Integrated DNA Technologies), and gDNA template (25 ng/µL concentration). PCR amplification was performed using a thermal cycler (Applied Biosystems, Thermo-Fischer Scientific) with the reaction conditions of initial denaturation at 94°C for 5 min, followed by 35 cycles of denaturation at 94°C for 30 s with respective annealing temperatures (supplementary Table S1), and extension at 72°C for 30 s. The final extension lasted for 5 min at 72°C before being kept at 4°C. The amplified PCR products were separated on a 2% agarose gel for 1 hr at 90 V and 400 mA current. The Gel Doc EZ imager (Bio-Rad) was utilized to visualize the amplicons and document the results. All samples were tested for the presence of SRY gene (sY14). The deletion-positive reactions were carried out three times to confirm their presence in each individual. All the deletion screening experiments were performed blindly without knowing the SCT use status of the subjects.
Statistical Analysis
The differences among frequencies of AZFc partial sub-deletions were calculated and compared between several case and control groups using Fisher’s exact test through GraphPad InStat (GraphPad Software, Inc., San Diego, USA, version 3.06). The total sample size underwent verification with G* power software to ensure its adequacy in meeting the 80% threshold of statistical power for all conducted tests. t-tests assessed the differences of clinical attributes between case and control groups, while Fisher’s exact tests compared other epidemiological parameters in the study cohort using GraphPad InStat. There are two types of variables. The presence of mDel/genetic variations, as well as the SCT use status of the individuals, is taken as “predictor” variables (independent variable), while the fertility status of the men is considered an “outcome” (dependent variable). The study questions were answered with either Yes or No. In this case, we considered partial deletion as a “genetic predictor” and SCT as an “epidemiological/environmental predictor.” A binary logistic regression analysis was conducted to study the Gene × Epidemiological (G × E) interaction in the respective study cohort. This analysis was carried out using the software package STATA 13 (StataCorp LP, College Station, Texas). p values <.05 were considered statistically significant. Odds ratios (ORs) with their respective 95% confidence intervals (CIs) were computed to determine the risk of developing the condition. Statistical tests were repeated at least twice to confirm the findings.
Results
Epidemiological and Clinical Attributes of the Study Participants
Various clinical parameters and epidemiological factors were examined in both case and control individuals. The infertile men (including all subgroups) exhibited significantly (p < .0001) elevated serum follicle-stimulating hormone (FSH) levels (11.24 ± 2.16 mIU/mL) in comparison to the control group (7.18 ± 1.07 mIU/mL), while the testosterone levels in both infertile (13.82 ± 2.74 nmol/L) and control groups (14.11 ± 1.9 nmol/L) fell within the normal reference range, and no significant difference was recorded. Upon comparison, the total sperm count was considerably lower in the case (47.4 ± 2.10 million/mL) group than the fertile (54.2 ± 1.83 million/mL) group (p < .0001; Table 2). Sociodemographic variables, including income status of the individuals, location, and religion, did not significantly differ across the groups. However, the AZFc partial deletion status was significant (p = .0019) in the case groups (freq. 0.09; 62/728) when compared with the fertile controls (freq. 0.03; 8/264). The details are presented in Table 2.
Demographic and Epidemiological Details of the Participating Individuals Involved in This Study

Note. SD: standard deviation; t-tests were used to test for differences between clinical parameters, and Fisher’s exact tests were used to compare other epidemiological parameters between the case and control groups of the study cohort.
p value <.05 was considered statistically significant. b Indicates a value significantly different from fertile controls.
Prevalence of AZFc Partial Deletion
Upon screening, an incidence of 7.06% (70/992) of AZFc partial deletions was observed in the study cohort, including cases (6.25%; 62/992) and controls (0.81; 8/992). Frequency was recorded much higher in the infertile group (including all subgroups) than in the fertile control group; we identified that 8.52% (62/728) and 3.03% (8/264) of participants carried partial deletions from the case and control groups, respectively. The frequency distribution of each type of partial deletion and its combination among the case subgroups and fertile controls is presented in Table 3.
Incidence of AZFc Partial Deletions in Different Case Subgroups and Fertile Controls
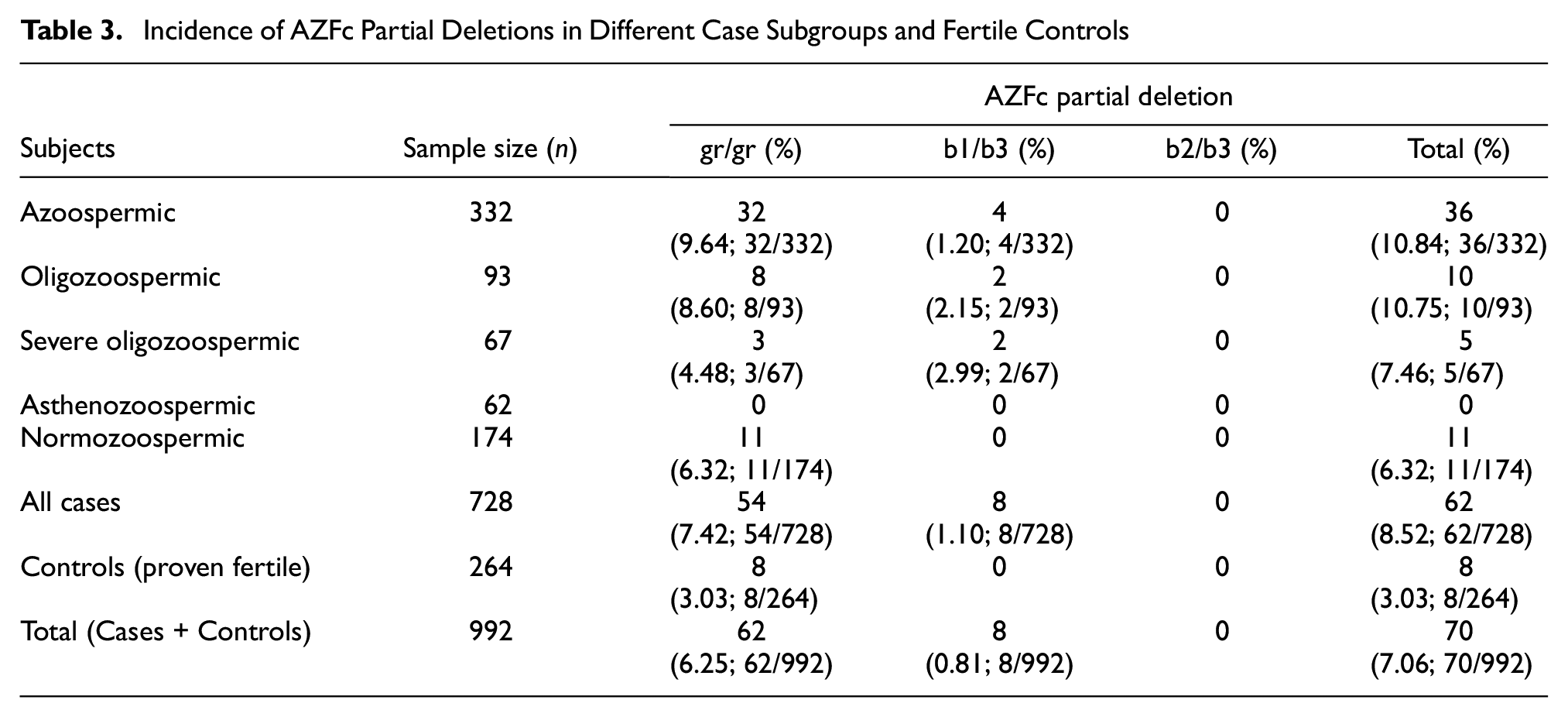
gr/gr Deletion
Among the 728 case samples, 54 (7.42%; 54/728) men exhibited gr/gr deletions. Of them, 32 (9.64%, 32/332) were detected in azoospermic (n = 332), eight (8.60%; 8/93) in oligozoospermic (n = 93), three (4.48%; 3/67) in severe oligozoospermic (n = 67), and 11 (6.32%; 11/174) in infertile normozoospermic (n = 174) subgroups, respectively. Out of 264 fertile control males, eight (3.03%, 8/264) exhibited the presence of this deletion. No such deletion was detected in the asthenozoospermic subgroup. The frequency of gr/gr deletion was significantly different in pair-wise comparison among the total cases (including all subgroups) versus fertile controls (54 vs. 8; p = .011), azoospermic versus fertile controls (32 vs. 8; p = .0015), and oligozoospermic versus fertile controls (8 vs. 8; p = .0382). We did not observe a significant association for severe oligozoospermic (3 vs. 8; p = .4703) and normozoospermic (11 vs. 8; p = .1483) groups when compared to that of the control group.
b1/b3 Deletion
The b1/b3 sub-deletion was observed in eight (1.10%; 8/728) individuals from infertile groups (n = 728), while no such deletion was detected among the fertile controls. In the infertile group, four (1.20%; 4/332) subjects were azoospermic (n = 332), two (2.15%; 2/93) were oligozoospermic (n = 93), and two (2.99%; 2/67) were severe oligozoospermic (n = 67) cases. However, this type of sub-deletion was unidentified, neither in normozoospermic nor in asthenozoospermic individuals. The case-control comparison was not carried out because this deletion was not detected among control subjects.
Combined Incidence of AZFc Sub-Deletions
All the sub-deletions (gr/gr and b1/b3, no b2/b3 sub-deletions were observed in this study) were combined, and it was identified that 62 men, that is, 8.51% (62/728) of the infertile group (n = 728), carried partial deletions in contrast to eight, that is, 3.03% (8/264) of the fertile controls (n = 264). Of the 62 partial deletion-bearing case subjects, 36 (10.84%; 36/332) were from the azoospermic (n = 332) group, 10 (10.75%; 10/93) from the oligozoospermic (n = 93) group, five (7.46%; 5/67) from the severe oligozoospermic group (n = 67), and 11 (6.32%; 11/174) from the infertile normozoospermic (n = 174) group. The frequency of sub-deletions (combining all partial deletions) was much higher among the case subjects than in fertile controls (62 vs. 8), and the difference was identified substantially significant (p = .0019).
Table 4 summarizes the results of association study for all partial deletions, including their respective odds ratio (OR), 95% confidence interval (CI), and p values at a significance level of <.05.
The Association of AZFc Partial Deletions Between the Cases and Controls
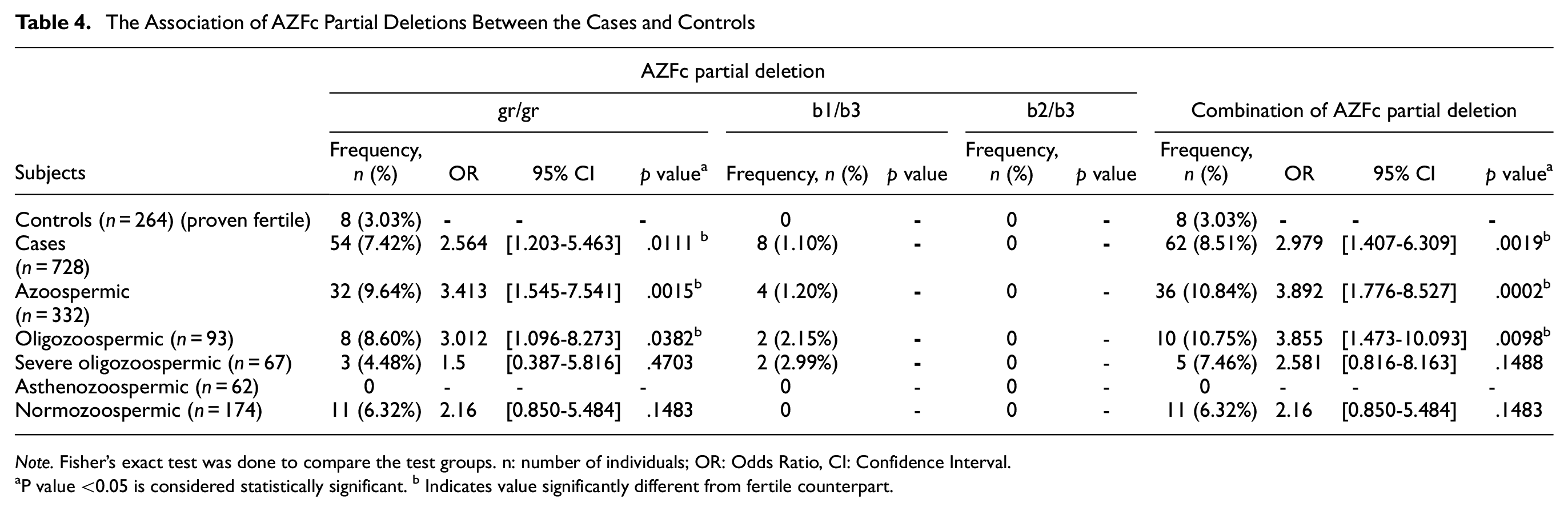
Note. Fisher’s exact test was done to compare the test groups. n: number of individuals; OR: Odds Ratio, CI: Confidence Interval.
P value <0.05 is considered statistically significant. b Indicates value significantly different from fertile counterpart.
Frequency of SCT Use in Control and Case Groups
A total of 728 case individuals and 264 control men were recruited in this study and each of them was categorized as “SCT non-users” and “SCT users” according to the declared SCT use status in their epidemiological record. We observed 102 (14.01%; 102/728) SCT users from the case subjects (n = 728) in comparison to 15 (5.68%; 15/264) from the control groups (n = 264). Fisher’s exact test was performed for comparing the frequencies of SCT use status in case and control groups, and a significant difference was reported in SCT use with odds in favor of the case subjects (OR = 2.705, 95% CI = [1.542-4.743], p = .0002; Table 5). Among the AZFc partial deletion-bearing case subjects (n = 62), 48 (77.42%; 48/62) were recorded as SCT users, whereas 14 (22.58%; 14/62) were non-users. In contrast, persons without microdeletions exhibited 54 (8.10%; 54/666) SCT users and 612 (91.89%; 612/666) SCT non-users. Among the fertile controls, eight individuals were identified with partial deletions, of whom three (37.5%; 3/8) were SCT users and five (62.5%; 5/8) were SCT non-users. These details are summarized in supplementary Table S2.
Status of Smokeless Chewing-Tobacco (SCT) Use Among Case (All Infertile Subgroups) and Fertile Controls Involved in This Study Cohort

Note. n = number of individuals; OR = odds ratio; CI = confidence interval.
p value <.05 was considered statistically significant. b Indicates value significantly different in the analysis.
Genetic × Environmental Interaction: Synergistic Effect of AZFc Partial Deletion and SCT Use
Frequent occurrences of SCT use (i.e., gutka, khaini, gudaku, pan masala) were observed among the participants through the scouting of information collected in their epidemiological records. Taking advantage of this unique population sample, an initiative was taken to investigate the potential synergistic effects, if any, between SCT usage and partial microdeletions on male infertility. In other words, an approach was made to design a Gene × Environment (G × E) model for male infertility, considering partial microdeletions are genetic risk factors and SCT uses an environmental/epidemiological risk factor.
We designed a logistic regression model, considering microdeletions and SCT use as “predictors” and fertility status of the individuals as an “outcome.” In binary logistic regression, we tested three interactions among the predictors, where SCT non-user × mDel (partial microdeletion) absent group was considered as the reference group. We observed a statistically significant (p = .002) interaction between SCT use and microdeletion (SCT user [n = 102] × mDel [n = 48]) in the infertile group, with significantly elevated odds (OR = 6.38) compared to the other two interactions, that is, SCT non-user (n = 626) × mDel (n = 14) [OR = 1.11; p = .834] and SCT user (n = 102) × mDel absent (n = 54) [OR = 1.79; p = .075] (presented in Table 6). This finding suggests SCT has a strong interaction with partial microdeletion among infertile Bengali-speaking men, and it may be a significant epidemiological risk factor for developing infertility among men.
Logistic Regression Analysis Revealed Significant Interaction Between the Predictors Smokeless Chewing-Tobacco (SCT) Use and Partial Microdeletion (mDel)
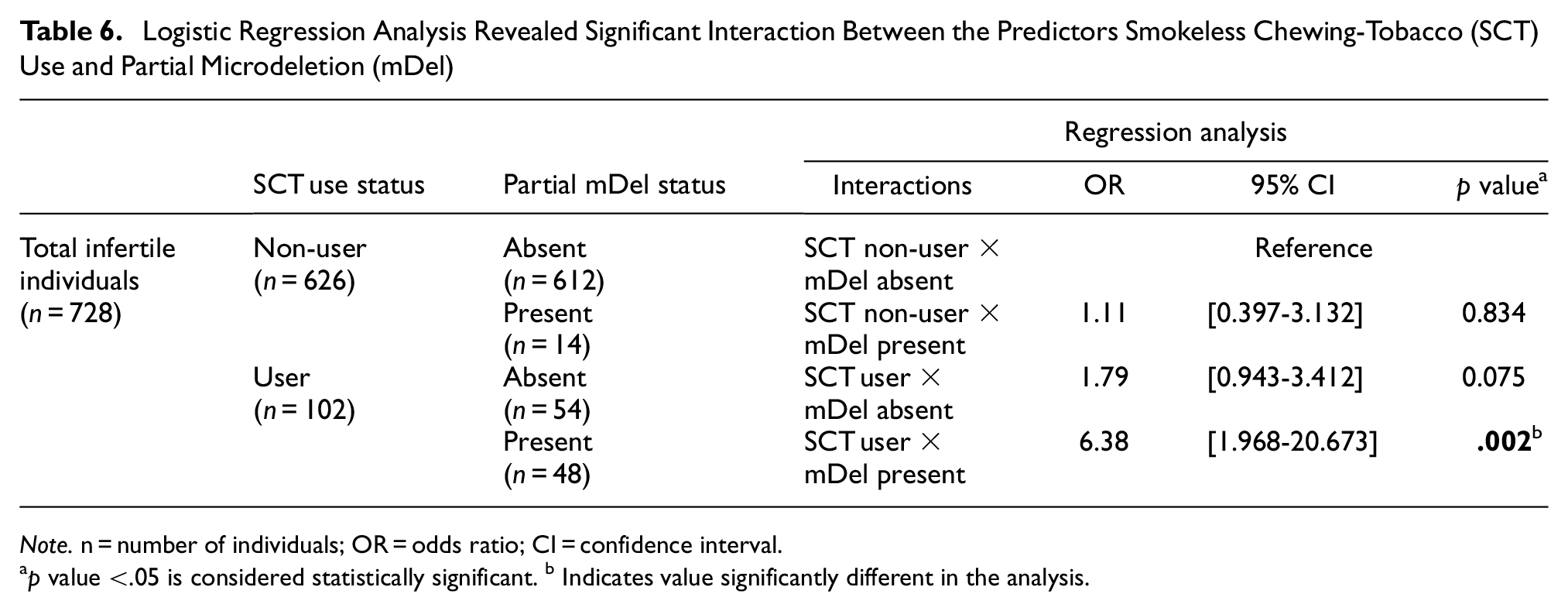
Note. n = number of individuals; OR = odds ratio; CI = confidence interval.
p value <.05 is considered statistically significant. b Indicates value significantly different in the analysis.
Discussion
In the current study, we demonstrated that two types of AZFc sub-deletions, gr/gr and b1/b3, are prevalent among infertile Bengali men. The gr/gr deletion was more frequent among the cases (7.42%; 54/728) than the b1/b3 deletion (1.10%; 8/728). Together, 8.52% (62/728) of the infertile males (including all subgroups) exhibited the presence of any of the partial deletion types, in contrast to 3.03% (8/264) in the controls. In addition, we did not find any incidence of the b2/b3 sub-deletion in our study cohort. The reason behind this is not clear. This could be a chance that the study sample might not have included individuals with the b2/b3 deletion; a larger sample size might reveal all sub-deletion categories. Alternatively, it may genuinely reflect the fact that Bengali men do not carry b2/b3 deletion.
These results receive support from several earlier studies that reported a more frequent occurrence of gr/gr deletion than other types. Since the initial finding of gr/gr sub-deletion (Repping et al., 2003) among men, several independent studies have reported incidence of gr/gr deletion, though results were inconsistent. Three meta-analyses (Bansal et al., 2016; Stouffs et al., 2011; Tüttelmann et al., 2007) suggested a significant association between the gr/gr deletion and infertility, and one meta-analysis (Visser et al., 2009) inferred that such deletion significantly reduced the sperm count. A population-based survey revealed gr/gr deletion causes severe spermatogenic failure (SSF; Rozen et al., 2012). Several independent case–control analyses conducted in different ethnic populations identified gr/gr deletion as a risk factor for male infertility (Choi et al., 2012; de Llanos et al., 2005; Ferlin et al., 2005; Giachini et al., 2005; Liu et al., 2019; Lynch et al., 2005; Navarro-Costa et al., 2007). Contrarily, several independent investigations from other populations failed to demonstrate such phenotypic impact in the context of gr/gr deletion, and they suggested this deletion might not contribute to spermatogenic failure causing male infertility (Alimardanian et al., 2016; Carvalho et al., 2006; de Carvalho et al., 2006; Fernando et al., 2006; Hucklenbroich et al., 2005; Imken et al., 2007; Lardone et al., 2007; Ravel et al., 2006; Stahl et al., 2011; Stouffs et al., 2008; Vijesh et al., 2015).
Previous studies on India sample cohort have reported gr/gr deletions among infertile men from North India (Bansal et al., 2016; Shahid et al., 2011), Western India (Sen et al., 2015), and Central India (Rani et al., 2019). All these data are concordant with what we observed in present study on a sample cohort from the eastern part of India. Our findings suggest men diagnosed as azoo/oligozoospermic experienced frequent occurrences of gr/gr deletions, and the association was significant (32 vs. 8; p = .0015, OR = 3.413 for azoospermic; 8 vs. 8; p = .0382, OR = 3.012 for oligozoospermic) when compared with fertile controls (Table 4). We observed the presence of gr/gr deletions among normozoospermic infertile men (6.32%; 11/174) and proven fertile controls (3.03%; 8/264), though the association was insignificant. The presence of gr/gr deletions in normozoospermic/fertile men was also reported in other studies (Bansal et al., 2016; Machev et al., 2004; Sato et al., 2013; Shahid et al., 2011; Visser et al., 2009). From the above discussion, it can be inferred that the implication of gr/gr deletion in clinical manifestation of infertility remains contentious. Discrepancies in findings across studies may develop due to variations in study designs, association analysis techniques, geographical differences, and ethnic heterogeneity (Krausz et al., 2015). In addition, the Y chromosome background of the study group (allelic variations of other spermatogenic regulator genes) might influence the risk of developing spermatogenic impairment in combination with such sub-deletions in the AZFc region (Yang et al., 2008). When the outcome of the present study was compared with those obtained from studies on other Indian populations, the frequency of gr/gr deletion among Indian Bengali-speaking men (7.42%; 54/728) was higher than the findings from two separate studies on North Indian sample populations (7.17% [30/418]; 5.84% [48/822]; Bansal et al., 2016; Shahid et al., 2011) and also the findings of studies on South Indian (6.25%; 13/208; Vijesh et al., 2015) and Central Indian populations (5.1%; 50/973; Rani et al., 2019). In contrast, the above-mentioned frequency (7.42%; 54/728) was lower than that reported in the Western Indian population (9.90%; 51/515; Sen et al., 2015). The current study reveals a significant association of gr/gr sub-deletion with male infertility (54 vs. 8; p = .0111; OR = 2.564) in favor of the cases (Table 4), which is consistent with the findings from Asian and Caucasian men (Bansal et al., 2016; Rozen et al., 2012).
Our study revealed the presence of b1/b3 sub-deletion, in addition to gr/gr deletion, in the study cohort (1.09%, 8/728), and no such deletion was detected among control men. This deletion was reported to cause spermatogenic failure (Rozen et al., 2012) and maturation arrest (Li et al., 2016). In this study, b1/b3 deletion was observed among severe oligozoospermic (2.99%; 2/67) cases (Table 4), and it is consistent with the previous findings (Li et al., 2016; Rozen et al., 2012). In contrast, a study conducted on Northwest Chinese men in 2019 reported a higher prevalence of b1/b3 deletion among control men, suggesting that the deletion may not be a risk factor for male infertility (Liu et al., 2019). Earlier studies on Indian men revealed a significant association between b1/b3 sub-deletion and male infertility in the North (Shahid et al., 2011) and Western Indian (Sen et al., 2015) study cohorts. Support for this association also comes from a study conducted in 2019 in Central India that reported 2.7% (26/973) of the study cohort had b1/b3 deletion (Rani et al., 2019). Considerable population-specific variations in phenotypic manifestations of these deletions made the genotype–phenotype relationship enigmatic, and this needs more investigation in other populations as well.
Genetic risk factors for male infertility not only vary according to ethnicity or demography but also due to exposure to diverse epidemiological, lifestyle, and environmental factors. In complex or multifactorial disorders, the genetic make-up serves as “loaded gun,” while the environmental factors act as “trigger” (Halder et al., 2021). Several studies have confirmed association of tobacco and alcohol use with infertility or altered semen parameters (Barratt et al., 2017; Gunes et al., 2018; Marinelli et al., 2004; Rai & Rai, 2016; Sansone et al., 2018; A. Sharma, 2017; R. Sharma et al., 2016; Yao & Mills, 2016). In India, a number of studies have revealed the adverse impact of SCT on semen quality (Borkhataria et al., 2020; Dikshit et al., 1987; Said et al., 2005; Sunanda et al., 2014). After careful data mining from the epidemiological records, we observed the habit of SCT consumption was more frequent among infertile men compared with fertile controls. Then, we decided to consider SCT as representative of epidemiological/environmental risk factor in our subsequent analyses. In this study, we stratified the study participants as SCT users and SCT non-users and blindly screened the participants for AZFc partial deletions without looking into their SCT use status. Then, the two sets of data, that is, SCT use status and occurrence of partial deletions, were compiled (as presented in Supplementary Table S2) to check whether SCT use potentially interacts with partial deletion and increases the risk of male infertility. The synergistic or interactive effect of SCT use and partial deletions was checked using a binary logistic regression model, and it was observed that the SCT users with AZFc partial deletions (n = 48) were at significantly higher risk (p = .002; OR = 6.38) of being infertile than the other two groups, SCT non-users carrying those deletions (n = 14; p = .834; OR = 1.11) and SCT users not bearing those sub-deletions (n = 54; p = .075; OR = 1.79), while those who are SCT non-users and carry no such deletions as well are taken as the reference group in this study (Table 6). It is evident that the combined occurrence of AZFc partial deletion and SCT consumption significantly elevates the risk of infertility in the individuals through synergistic effects.
This observation underscores that lifestyle/habitual and epidemiological factors contribute significantly to the varied clinical manifestations of Y chromosome partial deletions in male infertility. The interaction of these factors with genetic makeup leads to diverse clinical sub-types of compromised semen parameters. Previously, SCT use has been identified as a risk factor for chromosome 21 nondisjunction in the oocyte that leads to Down syndrome birth (Ghosh et al., 2011; Halder et al., 2021) and for shorter telomere and accelerated molecular aging among women bearing Down syndrome children (Ray et al., 2016). At this point, it is unclear whether SCT use instigates aberrant intrachromosomal recombination and YCMD or if their association is just a stochastic event. This study is the first attempt to explore the interaction and synergistic effects of YCMD and SCT use, suggesting further comprehensive investigations in diverse populations for a deeper understanding of male infertility etiology and gene–environment interactions.
However, there are some limitations to consider. One of the limitations is its cross-sectional design, which may introduce a potential constraint in establishing a causal relationship between the AZFc partial deletion and chewing tobacco. A dose-response analysis based on the amount of SCT consumption per day could address this. Future research should prioritize prospective studies to better explore this relationship. Second, the study cohort included only Indian Bengali-speaking men aged 20 to 50 years from West Bengal, and this may restrict the generalization of proposed notion regarding G × E as risk of infertility among men from other parts of India or elsewhere. Further research is required to gain a more knowledge of the mechanisms underlying the association between tobacco consumption (smoking or chewing) and Y chromosome microdeletions and to assess the impact of tobacco cessation over the improvement of fertility challenges among men. More studies involving diverse populations or ethnic groups across the world would provide valuable insights into the long-term effects of smoking or chewing tobacco on the etiology of male infertility.
Conclusion
In summary, AZFc partial deletion screening reveals that such deletions (gr/gr and b1/b3) are more prevalent among infertile individuals, and when such deletions and SCT are present together, their interaction increases the risk of infertility among men. We succeeded in figuring out for the first time ever that genetic and environmental risk factors act synergistically to impair fertility among men. In addition, allelic variations of other genetic loci with which genes from AZFc interact to regulate spermatogenesis contribute to the clinical variability of semen quality that surrogates infertility phenotype. This study provides the foundation for the Gene × Environment model that can be successfully applied to elucidate the risk of female infertility or other lifestyle disorders in which both genetic make-up and environmental factors play pivotal roles.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883241279195 – Supplemental material for Screening of the Combined Risk of Genetics and Epidemiology on Infertility Among Indian Men: Synergistic Effect of AZFc Partial Deletions and Habits of Smokeless Chewing Tobacco
Supplemental material, sj-docx-1-jmh-10.1177_15579883241279195 for Screening of the Combined Risk of Genetics and Epidemiology on Infertility Among Indian Men: Synergistic Effect of AZFc Partial Deletions and Habits of Smokeless Chewing Tobacco by Saurav Dutta, Pranab Paladhi, Samudra Pal, Souvik Srimani, Gunja Bose, Papiya Ghosh, Ratna Chattopadhyay and Sujay Ghosh in American Journal of Men's Health
Supplemental Material
sj-docx-2-jmh-10.1177_15579883241279195 – Supplemental material for Screening of the Combined Risk of Genetics and Epidemiology on Infertility Among Indian Men: Synergistic Effect of AZFc Partial Deletions and Habits of Smokeless Chewing Tobacco
Supplemental material, sj-docx-2-jmh-10.1177_15579883241279195 for Screening of the Combined Risk of Genetics and Epidemiology on Infertility Among Indian Men: Synergistic Effect of AZFc Partial Deletions and Habits of Smokeless Chewing Tobacco by Saurav Dutta, Pranab Paladhi, Samudra Pal, Souvik Srimani, Gunja Bose, Papiya Ghosh, Ratna Chattopadhyay and Sujay Ghosh in American Journal of Men's Health
Footnotes
Acknowledgements
We express our sincere gratitude to the participating men and our physician friends who assisted in obtaining tissue samples for this study. We acknowledge the Department of Zoology, University of Calcutta, for helping with the necessary instruments, which were supported by UGC-UPE II, DST-FIST Programme, and DST-PURSE Programme.
Author Contributions
S.G. conceptualized and conceived the study; S.G., P.G., and S.D. designed the study; S.D., P.P., and S.P. helped in collection of samples, interviewed the couples, and recorded the epidemiological details. R.C. and G.B. cared for the participants and helped in sample collection and subsequent categorization. S.D., P.P., S.P., and S.S. performed experimentation and analyzed the results; S.G., P.G., and S.D. wrote the manuscript. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Indian Council of Medical Research (ICMR), Government of India, provided financial support for the research work (Grant number: 5/10/FR/10/2015-RCH) only. Saurav Dutta is thankful to the University Grants Commission (UGC), India, for the research fellowship. There is no financial support available for the publication of this article.
Ethics Approval
We followed the guidelines as specified in the “Declaration of Helsinki” and the Indian Council of Medical Research (ICMR) in working with human subjects and for the collection of biological samples. The study design was approved by the Institutional Ethics Committee constituted by the University of Calcutta, West Bengal, Kolkata, India (Approval No. CU/BIOETHICS/HUMAN/2306/3044/2018; Dated 04/05/2018).
Consent to Participate
All participating individuals involved in this study provided their written informed consent in the pre-printed questionnaire.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
