Abstract
The purpose of this study is to assess the influence of sex hormones and other indicators on risk factors for hypercoagulable states in male patients with type 2 diabetes mellitus. Ninety-two diabetic patients were divided into two groups based on testosterone levels: T1 group (testosterone <12 mmol/L) and T2 group (testosterone >12 mmol/L). Fifty-four non-diabetic patients were used as the control group. Sex hormone index, glucose, insulin, C-peptide, 24-h urinary free cortisol, thromboelastography, and insulin resistance index were measured by radioimmunoassay. Testosterone was lower in the diabetic men than in the control group (12.02 vs 14.77, p < .05), and was inversely related to blood coagulation status, blood glucose level, and cortisol level. Body mass index was positively correlated with estradiol and insulin resistance index. Testosterone was independently associated with the clotting process after controlling for age. Low testosterone is a risk factor for hypercoagulable state in diabetic men. Elevated estradiol and insulin resistance are influential factors for increased body mass index.
Keywords
Introduction
Uncontrolled glucose can impair male reproductive function in men with type 2 diabetes (Mahbouli et al., 2021), and a previous study reported that about 39.4% of male diabetic patients had low serum total testosterone levels (Sepu et al., 2021). The severity of glucose metabolism influenced the decrease in androgen levels (Cheng et al., 2017). Hypercoagulation can lead to thrombosis, which further causes ischemia, hypoxia, and necrosis in the corresponding tissues and organs (Jiang & Luo, 2021). Reduced testosterone levels can predispose patients to hypercoagulation and the degree of free testosterone reduction correlates with the degree of changes in coagulation and fibrinolytic activity (Si & Li, 2005). However, some studies detected that testosterone therapy induced venous thromboembolism (Glueck & Wang, 2014) and it remained dispute how testosterone influences coagulation.
Chronic hyperglycemia in patients with type 2 diabetes can exacerbate the dysfunction of the hypothalamic–pituitary–adrenal axis, leading to increased cortisol secretion (Joseph & Golden, 2017). Diabetic patients experience oxidative stress due to the accumulation of non-enzymatic glycation end products in the body, which in turn leads to the development and occurrence of peripheral vascular lesions (Höbaus et al., 2020). Thromboelastography (TEG), a new technology, assesses coagulation throughout all phases of clot formation and is the most commonly used viscoelastic assay (Subramanian et al., 2019).
Our study focused on the analysis of sex hormones, 24-h urinary free cortisol, insulin dosage, insulin resistance index, and TEG correlations in men with type 2 diabetes to assess the impact of sex hormones and other risk factors for hypercoagulable states.
Data and Methods
Grouping of Study Subjects
A total of 92 male patients with type 2 diabetes mellitus (T2DM) admitted to the Department of Endocrinology, Chongming Branch, Shanghai Tenth People’s Hospital, Shanghai, China from April 2021 to March 2023, who met the admission criteria and completed hormone-level examination, were selected as the observation group. The recommended therapeutic split point of 12 mmol/L by the International Society of Gynecology (Nieschlag et al., 2005) was used as the basis for grouping. Blood testosterone concentration <12 mmol/L was considered as the T1 group and >12 mmol/L as the T2 group; 54 cases of non-diabetic patients with outpatient physical examination were used as the control group. The admission criteria included: (1) completing hormone-level examination by immunoassay method (the golden standard) using the Roche Cobas e601 analyzer (Roche; Geneva, Switzerland); (2) meeting one of the following diagnostic criteria for diabetes according to the American Diabetes Association (2021): (a) fasting blood glucose >7.1 mmol/L; (b) 2-h postprandial blood glucose >11.1 mmol/L by oral glucose tolerance test (the most commonly used diagnostic criteria); (c) patients with three more and one less symptoms accompanied by random blood glucose >11.1 mmol/L; (d) repeated multiple blood glucose tests greater than the diagnostic criteria under non-stressful conditions. Exclusion criteria: (1) history of abnormal adrenocortical function (hypoadrenocorticism, Cushing’s syndrome, adrenal tumors); (2) history of pituitary and hypothalamic disorders; (3) history of thyroid disorders; (4) patients taking oral hormonal drugs; (5) hepatic insufficiency (glutathione aminotransferase >120 U/L); (6) renal insufficiency; (7) coagulation abnormalities, or in anticoagulant therapy patients; (8) severe acute complications of diabetes mellitus (diabetic ketoacidosis, diabetic hyperosmolar coma); (9) infectious febrile diseases; (10) previous history of diagnosed hypogonadism or oral 5α-reductase inhibitors; (11) patients on oral aromatase inhibitors; (12) history of psychiatric disorders with oral medication. All subjects signed informed consent and this study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of our hospital (No. 2020-YF-02).
Examination Index
Observation Index. (1) Sex hormone indexes: Blood was collected from the subjects in the morning on an empty stomach, and serum specimens were measured by the immunoassay method using the Roche Cobas e601 analyzer produced in 2012. These include follicle-stimulating hormone (FSH), serum luteinizing hormone (LH), prolactin (PRL), and testosterone (T). Total testosterone (TT) levels are widely considered to be the most useful diagnostic indicator of androgen status (Anupam et al., 2020). (2) Glucose and lipid metabolism indexes: Subjects were fed a light diet in the evening before the examination, and did not eat after 20:00, fasted for 8 to 12 h. Blood was collected in the morning on an empty stomach, and serum specimens were determined by chemiluminescence using the Beckman Coulter UniCel DxI800 analyzer (Beckman Coulter, Fullerton, CA, USA) produced in 2016. Fasting blood glucose (FBG), fasting insulin (FINS), and fasting C-peptide were included; insulin sensitivity was assessed by the steady-state model index; and the steady-state model insulin resistance (IR) index (HO-MA Insulin Resistance, HOMA) was calculated: (IR, FBG × FINS / 22.5) (Yang et al., 2005). (3) Insulin dosage, the total daily insulin application at the time of discharge when the patient achieved glycemic control, was used as an index for analysis. (4) Body mass index = weight/height2 (in kg/m2). (5) Thromboelastogram display information including R time, K time, Angle, maximum amplitude (MA) value, and coagulation (CL) value.
Statistical Methods
SPSS 22.0 statistical software was used for statistical analysis of the study data. The study data were measured, and data conforming to normal distribution were expressed as mean ± standard deviation (
Results
Differential Analysis of Age and Testosterone in the Observation Control Group
There was no statistical significance in the comparison of age between the control group and the observation group (67.49 vs 68.26, p = .730). The testosterone in the observation group was significantly lower than that in the control group (12.02 vs 14.77, p = .026; Table 1).
Differential Analysis of Age and Testosterone in the Observation Control Group (

p < .05 was statistically significant.
Difference Test for Each Index Between T1 and T2 Groups
All diabetic subjects were divided into two groups according the testosterone level. The K time in group T1 was significantly smaller than that in group T2 (p = .006). Angle, MA value, CL value, fasting glucose, fasting blood sugar, and urinary free cortisol in group T1 were significantly larger than those in group T2 (p < .05; Table 2). There was no difference in the insulin dosage, R time, BMI, glycated hemoglobin (HbA1c), HOMA-IR, girth, hip measurement, age, fasting peptide, FSH, LH, estradiol, and lactogen between the two groups (all p > .05).
Difference Test for Each Index Between T1 and T2 Groups (

Note. MA = maximum amplitude; CL = coagulation.
Correlation of Body Mass Index With Insulin Dosage, Thromboelastography, Glycated Hemoglobin, 24-h Urinary Free Cortisol, Estradiol, and Insulin Resistance Index
BMI was positively correlated with estradiol and IR index (r = .225, .447, p = .04, .000; Table 3).
Spearman Correlation Analysis of Insulin Dosage, Body Mass Index, Thromboelastography, Age, Glycated Hemoglobin, 24-h Urinary Free Cortisol, Estradiol, Testosterone, and Insulin Resistance Index
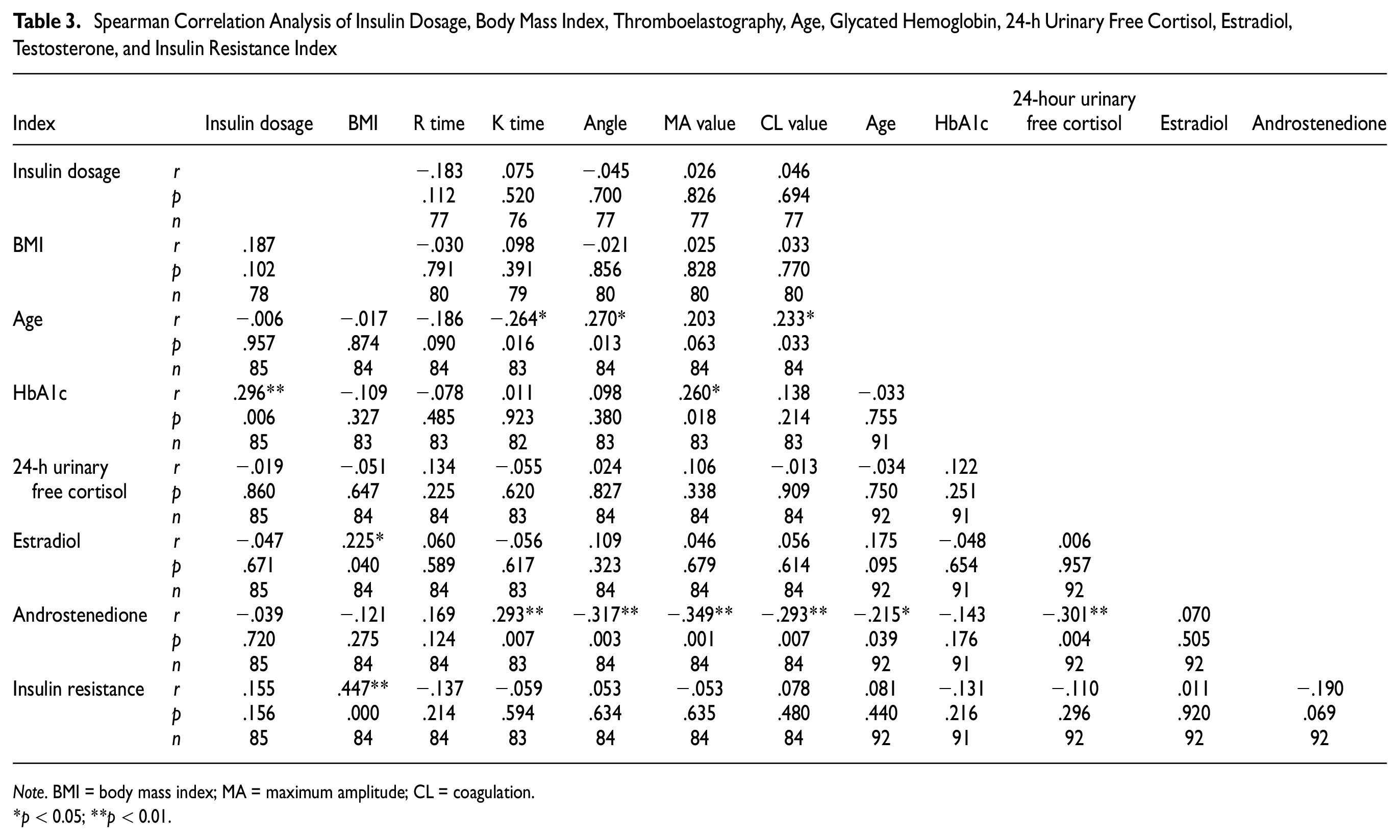
Note. BMI = body mass index; MA = maximum amplitude; CL = coagulation.
p < 0.05; **p < 0.01.
Glycosylated hemoglobin was positively correlated with insulin dosage and MA values (r = .296, .260, p = .006, .018).
The TEG parameters K time and testosterone reported a negative correlation with age (r = −.264, −.215, p = .016, .039); Angle and CL value reported a positive correlation with age (r = .270, .233, p = .013, .033; Table 3).
The TEG parameters Angle, MA value, CL value, and 24-h urinary free cortisol reported a negative correlation with testosterone (r = −.317, −.349, −.293, −.301, p = .003, .001, .007, .004); K time reported a positive correlation with testosterone (r = .293, p = .007); testosterone correlated with insulin dosage, BMI, R time, estradiol, IR index, and glycated hemoglobin were not correlated (Table 3).
Correlation of Age, Testosterone, and Thromboelastogram. Combined with the above analysis age was correlated with testosterone and thromboelastogram. Testosterone was correlated with TEG. Analysis of the bias correlation between thromboelastogram index and testosterone after controlling for age reported that: testosterone was negatively correlated with Angle, MA value, and CL value (r = −.313, −.345, −.320, p = .004, .001, .003); testosterone was positively correlated with K time (r = .228, p = .039). There was no significant correlation between testosterone and R time (Table 4).
Analysis of Testosterone and Thromboelastography Bias Correlation After Controlling for Age
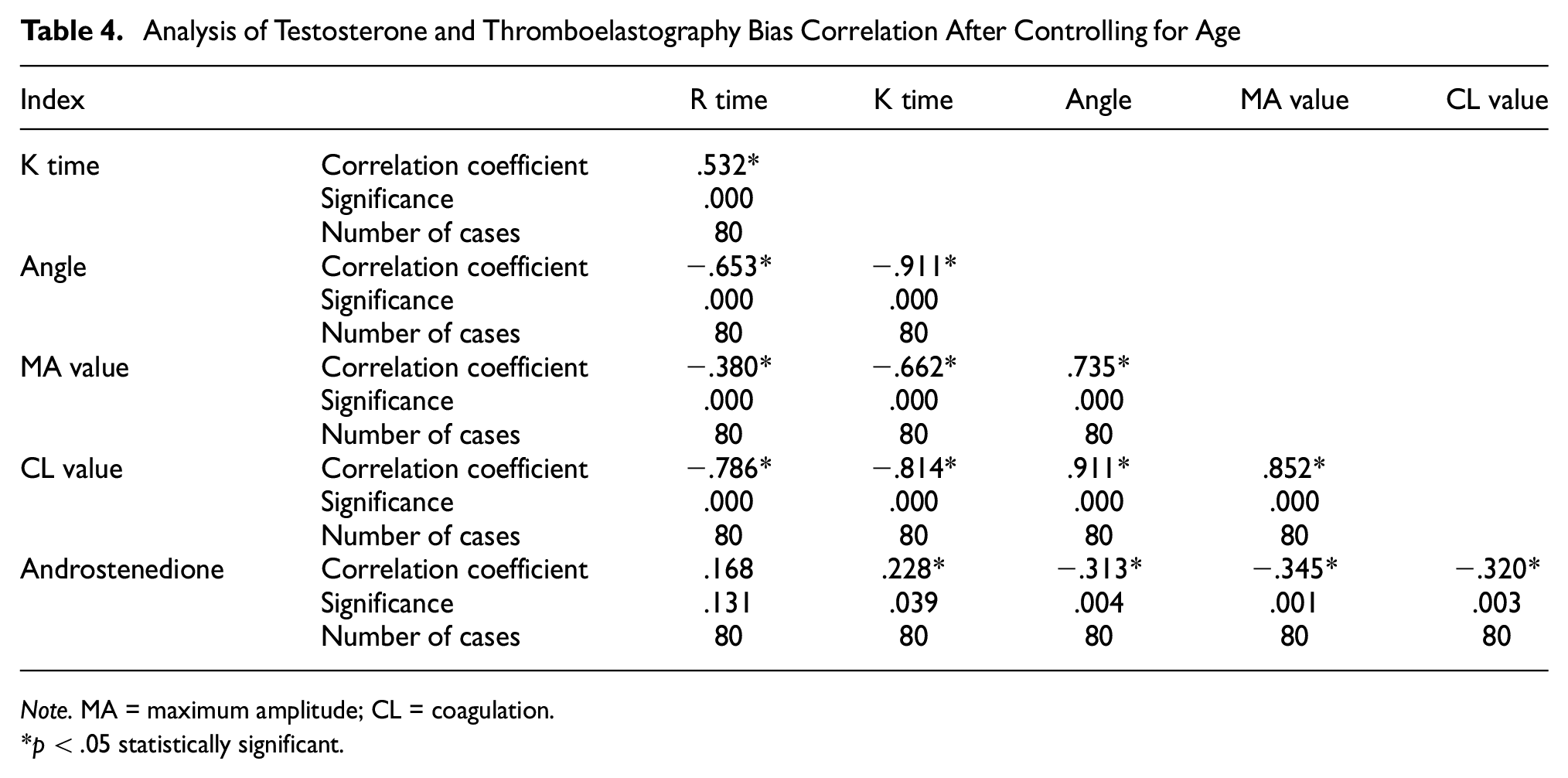
Note. MA = maximum amplitude; CL = coagulation.
p < .05 statistically significant.
Discussion
Correlation Between Testosterone and Glycated Hemoglobin
T2DM is an endocrine disease with a predominantly metabolic disorder (A. Wang et al., 2018). Some animal experiments have identified that male mice with low serum testosterone levels developed obesity, hyperglycemia, and insulin resistance (Dubois et al., 2016). Related literature suggests that older men with low testosterone levels not only have varying degrees of glucose and lipid metabolism disorders but also have a higher risk of developing diabetes mellitus (Yao et al., 2018). The present study reported that the observation group, which consisted of male patients with T2DM, had significantly lower testosterone levels than the control group, which consisted of non-diabetic male patients. A cross-sectional study of 104 type 2 diabetic men and 101 age-matched healthy non-diabetic men in Nigeria illustrated a significantly lower mean total testosterone in type 2 diabetic men compared to non-diabetic controls (Sepu et al., 2021), which is consistent with the present study. Reduced androgens can lead to disorders of glucose and lipid metabolism in men and induce the development and progression of obesity and its related diseases, and low androgen levels are closely related to the development of type 2 diabetes and are mutually causal (Yin & Wang, 2018). The main reasons considered are (1) hyperglycemia affects the hypothalamic–pituitary–gonadal axis, causing morphological changes and a decrease in the number of testicular mesenchymal cells, leading to a decrease in the secretion of testosterone (T; Kelly & Jones, 2013); (2) hyperglycemia can also lead to the release of large amounts of reactive oxygen species (ROS) from testicular mesenchymal cells, leading to a significant decrease in testosterone levels (Hackett et al., 2016). However, this paper did not report a correlation between testosterone and glycated hemoglobin, while a study of 110 male type 2 diabetic patients admitted from Jiading District Anting Hospital in Shanghai reported a negative correlation between their HbA1c levels and serum testosterone (T) levels (Liao & Shen, 2019). The present study is not fully consistent with its results, which may be due to experimental bias caused by low sample size or failure to perform multicenter sample extraction and the different clinical characteristics of study subjects.
Correlation Between Low Testosterone and Increased Stress Response and Cortisol Production in Male Patients With Diabetes Mellitus
This study reported that urinary free cortisol was higher in the T1 group than in the T2 group, and there was a linear negative correlation between testosterone and 24-h urinary free cortisol. A study of male college students with exercise habits from Shenyang Sports College reported a significant negative correlation between serum testosterone concentrations and serum cortisol concentrations in the quiet state (Lu, 2013). The results of the present study were approximately the same. This may imply that low testosterone is associated with increased stress response and cortisol production in male patients with diabetes mellitus.
Correlation Between Testosterone and Body Mass Index
The present study did not report a correlation between testosterone and BMI. A 14-year follow-up study of 423 men with type 2 diabetes reported a significant negative correlation between testosterone levels and BMI (Heald et al., 2020). Wu Fanglai et al. investigated the changes of different BMI on serum testosterone levels in men with T2DM and divided the study population into a normal weight group, overweight group, and obese group according to the cut points of BMI values of 18.5, 24, and 28 kg/m2. Multiple regression analysis reported that BMI was an independent influencing factor for the decrease of testosterone levels, that is, testosterone levels in men with T2DM decreased gradually with increasing BMI (Wu et al., 2021). The results of the present study did not report the same results, which may be due to selection and experimental bias caused by low sample size or not performing multicenter sample extraction.
Thromboelastography
TEG is a method to assess the coagulation status of blood by measuring the viscoelastic properties of a clot as it forms and lyses under low shear stress (Wang et al., 2020). R time is significantly more sensitive to endogenous coagulation pathways than exogenous coagulation pathways (Wang et al., 2020). K time indicates the time required for the clot strength to reach a certain level during blood coagulation and reflects the activity of fibrinogen, which is proportional to the rate of clot intertwining and forming (Ye et al., 2020). MA value is the best indicator of fibrin and platelets cross-linking each other to form clots using GP IIb/IIIa as a bridge (Li, 2020). The MA value of TEG is mainly influenced by two factors, platelets and fibrinogen, of which the role of platelets accounts for 80%, and the change in MA value reflects any abnormality in platelet quantity and quality, which can be calculated to visually express platelet function and is mostly used to guide the use of anti-platelet drugs (Weitz et al., 2017). CL value represents the percentage of clot lysis after reaching maximum clot strength, which reflects the balance between fibrin formation and fibrinolysis (Ye et al., 2020). TEG is more valuable in the assessment of coagulation dysfunction (Hum et al., 2020), and TEG can accurately report the coagulation status of patients (Sakai, 2019). Blood in type 2 diabetic patients can be hypercoagulable, while platelet function can be enhanced in patients (A. Wang et al., 2018). This study reported a correlation between age and both testosterone and TEG. In combination with previous studies claiming that testosterone levels in men reach their highest level at approximately 30 years of age and then gradually decrease at a rate of 1% to 2% per year (Herring et al., 2013). Age is an acquired thrombogenic factor that is closely associated with pathological and physiological changes (Akrivou et al., 2022). Biased correlation analysis after controlling for age suggested that as testosterone decreased in men with diabetes, K values decreased and Angle, MA values, and CL values increased. Increased MA values suggested an increased risk of thrombosis, consistent with the results of previous studies (Chen et al., 2020). Considering that reduced testosterone levels can lead to increased blood coagulation activity and inhibition of fibrinolytic activity, hypercoagulation occurs (Si & Li, 2005). When the blood in the body is in a hypercoagulable state, it is prone to the formation of a thrombus, which in turn causes ischemia and hypoxia in the corresponding tissues and organs (Jiang & Luo, 2021), and early detection of the factors affecting the patient can help to give active therapeutic measures at an early stage (Zhou et al., 2020).
Correlation Between Sex Hormones and Obesity in Men With Diabetes
Some studies have reported altered body fat distribution in the presence of low testosterone levels and elevated E2, leading to abdominal obesity (Sattar et al., 2021). Relevant literature reported that exogenous supplementation with androgens significantly reduces visceral fat mass, waist-to-hip ratio, insulin resistance levels, and the risk of developing obesity and aortic stenosis (AS) (Cheung et al., 2015). The present study reported an increase in BMI with estradiol in men with diabetes, further suggesting that sex hormones are associated with obesity in men with diabetes, in line with the results of previous studies (Smith et al., 2022). Another study reported that marked reductions in circulating testosterone are uncommon in middle-aged men with diabetes. Increasing BMI and age are dominant drivers of lowered total and cFT, respectively, independent of the presence or absence of diabetes (Ng Tang Fui et al., 2013).
Our study still had some limitations. First, this study was a single-center study with a small sample size, so the conclusions obtained from this study still need to be further confirmed in a multicenter, large sample size study. Moreover, due to the cross-section design, this study could not detect the cause–effect relationship between sex hormones and hypercoagulation, and longitudinal studies are necessary to verify our results in the future.
Conclusion
In summary, there is a tendency for men with diabetes to develop lower testosterone levels. Also, the decrease in testosterone levels is one of the risk factors for a hypercoagulable state. Elevated estradiol and increased insulin resistance index are both influential factors for increased BMI, indicating that more attention should be paid to the levels of sex hormones in male patients with T2DM. Further studies with large sample sizes and longitudinal designs should be conducted to explore the causality and provide evidence for clinical practice.
Footnotes
Acknowledgements
Not applicable.
Authors’ Contributions
C.X.J. conceived of the study, participated in its design and data analysis and statistics, helped to draft the manuscript, read, and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: 2020 Program for Introducing Innovative and Entrepreneurial Talents.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of our hospital (No. 2020-YF-02). We obtained signed informed consent from the participants / legal guardians in this study.
Consent for Publication
Not applicable.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
