Abstract
Prostate abscess, a rare condition often associated with prostate bacterial infections, often occurs in immunosuppressive individuals and manifests as fever and lower urinary tract symptoms. Clinical practice lacks standardized diagnostic and treatment protocols for prostate abscesses, resulting in predominantly empirical approaches with uncertain outcomes. This study presents a case of a giant prostate abscess, diagnosed in a patient exhibiting fever, lower urinary tract symptoms (including dysuria, urinary frequency, urgency, and weakness), and anal pain. The diagnosis was confirmed through prostate magnetic resonance imaging and transrectal color ultrasound examinations. Treatment included targeted anti-infective therapy (based on the urine culture results), urine flow diversion (suprapubic bladder puncture stomy), ultrasound-guided perineal puncture drainage of the prostatic abscess, intermittent abscess cavity irrigation, and urethral electroprostatectomy. The patient experienced a complete recovery and significantly improved quality of life. This successful case underscores several key points: (1) the importance of targeted anti-infective therapy based on etiological findings in prostate abscess treatment; (2) early urine flow diversion, precise puncture drainage, and intermittent abscess cavity irrigation may be one of crucial elements in abscess management; (3) the potential significance of transurethral prostate resection following abscess resolution in preventing recurrence. It is hoped that this case report offers new valuable insights for diagnosing and treating prostate abscesses. Slightly different from previous treatment experience, we extra used early urine diversion, intermittent abscess cavity irrigation, and etiological electroprostatectomy, which might also hold promise as potential therapies.
Introduction
A prostate abscess is a rare condition constituting approximately 0.5% of all urinary diseases, with mortality ranging between 1% and 16% (Ha et al., 2008). Typically, prostate abscess arises as a complication of severe prostate infection, often triggered by the retrograde flow of infected urine into the prostate duct during urination. Left untreated, prostate abscess can progress to urogenic sepsis and septic shock, potentially resulting in a fatal outcome (Ackerman et al., 2018). Individuals with poor glycemic control, compromised immunity, bladder outlet obstruction, neurogenic bladder dysfunction, postbiopsy prostate surgery, or underlying voiding dysfunction face an increased risk of developing prostate abscess (Brede & Shoskes, 2011; Nomura et al., 2021; Tiwari et al., 2011). Contemporary studies have highlighted gram-negative bacteria as the predominant pathogens responsible for prostate abscesses, with Escherichia coli accounting for over 70% of cases, followed by Klebsiella, Pseudomonas, Proteus, Enterobacter, Serrella, and Enterococcus. In addition, Staphylococcus aureus might cause prostate abscesses via hematogenous dissemination (Ackerman et al., 2018; Lee et al., 2016).
Given the absence of established guidelines for diagnosing and treating prostate abscesses, a range of therapeutic options are available. Based on prior clinical experience, typical interventions include conservative antibiotic therapy, ultrasound-guided puncture drainage of prostate abscesses, and transurethral deroofing (Lee et al., 2016). Existing literature suggests that smaller abscesses (diameter <2 cm) respond favorably to medical management, whereas larger abscesses (diameter >2 cm) often benefit from surgical unroofing, transurethral prostate resection, or similar drainage procedures. Such interventions have demonstrated the potential to reduce antibiotic dosage, shorten hospital stays, and improve urinary dysfunction (Alnadhari et al., 2020; Brede & Shoskes, 2011; Chou et al., 2004). Drawing on previous treatment experience and considering the patient's unique circumstances and preferences, a standardized treatment was administered to a patient with a giant prostate abscess. Finally, the patient achieved a full recovery and expressed high satisfaction with the treatment outcome. This article recounts the diagnostic and treatment process in the hope of contributing some valuable references for the future development of standardized diagnostic and treatment protocols for prostate abscess.
Case Presentation
A 75-year-old male patient with a history of prostatic hyperplasia for over a decade presented with lower urinary tract symptoms. These symptoms included dysuria, a diminished urinary stream, weak urine flow, frequent urination, and increased nocturnal urination. The patient did not undergo pharmaceutical or surgical interventions during this period and had no history of prostatitis, immune deficiency, diabetes, hypertension, coronary heart disease, cerebral infarction, trauma, or surgery. Five months ago (1 month before admission), the patient’s lower urinary tract symptoms worsened, accompanied by the emergence of urgency and urinalgia. As these symptoms did not severely disrupt his quality of life (QOL) at the time, the patient did not seek immediate medical attention. Four months ago, the patient’s condition deteriorated, marked by pronounced urinalgia, chills, and high fever (maximum body temperature 39.0°C). Experiencing considerable fatigue, he sought care at a local community hospital, where he received cefazolin-based anti-infection treatment for 5 days, the effect was poor. This treatment yielded limited efficacy, with the patient’s body temperature ranging between 37.5°C and 39.0°C and the onset of anal pain. Subsequently, the patient was admitted to our hospital.
Upon the patient’s admission, clinical observations revealed a fever, with the highest recorded body temperature reaching 38.5°C, a heart rate of 96 beats/min, blood pressure measuring 135/90 mm Hg, and a blood oxygen saturation level of 96%. During the specialist physical examination, significant lower abdominal distension and suprapubic tenderness were observed, without any apparent rebound pain or muscle tension. The enlarged prostate was accessible through digital rectal examination, characterized by a soft texture without touching the tubercle. Palpation revealed the presence of the left lobe, which exhibited an undulating surface and elicited tenderness. The patient experienced urinary difficulty and noticeable subpubic distension. As an urgent measure, aseptic catheterization was performed, and a 16-Fr catheter was retained. Approximately 500 mL of turbid yellow urine, containing numerous white flocs, was successfully drained.
Upon the patient’s admission, the laboratory findings were as follows: white blood cell (WBC) count: 21.4 × 109/L, neutrophil percentage at 83.4%, lymphocyte percentage at 11.5%, platelet count at 169 × 109/L, hemoglobin at 115 g/L, procalcitonin at 1.49 ng/mL, aspartate aminotransferase at 17.4 IU/L, alanine aminotransferase at 25.4 IU/L, creatinine at 53.8 µmol/L, total prostate-specific antigen at 7.62 ng/mL, urinary WBC at 368.1/µL, urinary bacteria at 2915.8/µL, and urine pH at 5.5. The patient’ blood culture yielded negative results. However, the urine culture indicated infections with multidrug-resistant bacteria, specifically Enterobacter cloacae and Klebsiella pneumoniae, the results of pus culture also showed pathogenic bacteria was K. pneumoniae, with piperacillin/tazobactam being one of the effective antibiotics. A chest computed tomography ruled out pneumonia or bronchitis but revealed a low-density mass on the left side of the prostate. A subsequent enhanced prostate magnetic resonance imaging (MRI) displayed swelling of the left lobe of the prostate, with cystic formations and T1 and long T2 signals spanning 5.0 cm × 4.0 cm. The diffusion weighted imaging (DWI) exhibited high signal intensity in the liquid area and low signal intensity in the cyst wall. Honeycomb and circular enhancement patterns were observed on the enhanced scan, suggesting the possibility of a prostate abscess.
The patient was admitted with acute urinary retention, prompting a thorough evaluation that included clinical examination and physical examination. Initially, prostatitis, apart from the possibility of a prostate abscess, was considered. Aseptic catheterization was performed using a 16-Fr urinary catheter, coupled with empirical anti-infective treatment with fluoroquinolone (levofloxacin). During treatment, the patient repeatedly reported urethral and anal pain and discomfort, leading to an inability to tolerate the indwelling catheter. Following extensive consultations and discussions with the doctors and the patient, a decision was made to divert the patient’s urinary flow through suprapubic bladder puncture stomy, while retaining a 16-Fr urinary tube. A subsequent MRI examination of the patient’s prostate revealed a huge prostate abscess in the left lobe, confirming the diagnosis of a prostate abscess. Urine culture results indicated the presence of two types of multidrug-resistant bacteria, E. cloacae and K. pneumoniae. Consequently, antibiotic therapy was adjusted, replacing the previous regimen with piperacillin/tazobactam. As a result, the patient’s body temperature gradually normalized, and urinaldynia symptoms significantly improved. However, anal pain persisted, significantly affecting the patient’s QOL. Once the infection was initially controlled, an ultrasound-guided perineal prostatic abscess puncture and drainage procedure were performed. A single 8-Fr J-tube was inserted, and during the surgery, the pus cavity was irrigated with piperacillin/tazobactam in conjunction with 0.9% normal saline. Based on pus culture results, this regimen was continued, employing intermittent flushing, until the drainage tube was devoid of pus. Subsequent reassessment of the prostate via MRI indicated clear signs of abscess absorption. Finally, transurethral electroresection of the prostate was performed, with no pus drainage observed during the procedure (preoperative MRI image is presented in Figure 1, an intraoperative view of the prostate abscess and the placement of the postoperative drainage tube is presented in Figure 2, and a follow-up prostate MRI is presented in Figure 3).

Pretreatment Magnetic Resonance Imaging Examination of the Prostate Revealed a Large Abscess of the Prostate (The Size of Prostate Abscess is 5.0 cm × 4.0 cm × 4.3 cm): (a) Axial View Showing a Huge Abscess in the Left Lobe of the Prostate, (b) Coronal View, and (c) Sagittal View.

(a, b) Ultrasound-Guided Perineal Puncture for Draining the Prostatic Abscess, it Is a Milky White Purulent Discharge; (c) Percutaneous Transperineal Drainage Achieved by Placing an 8-Fr Tube.
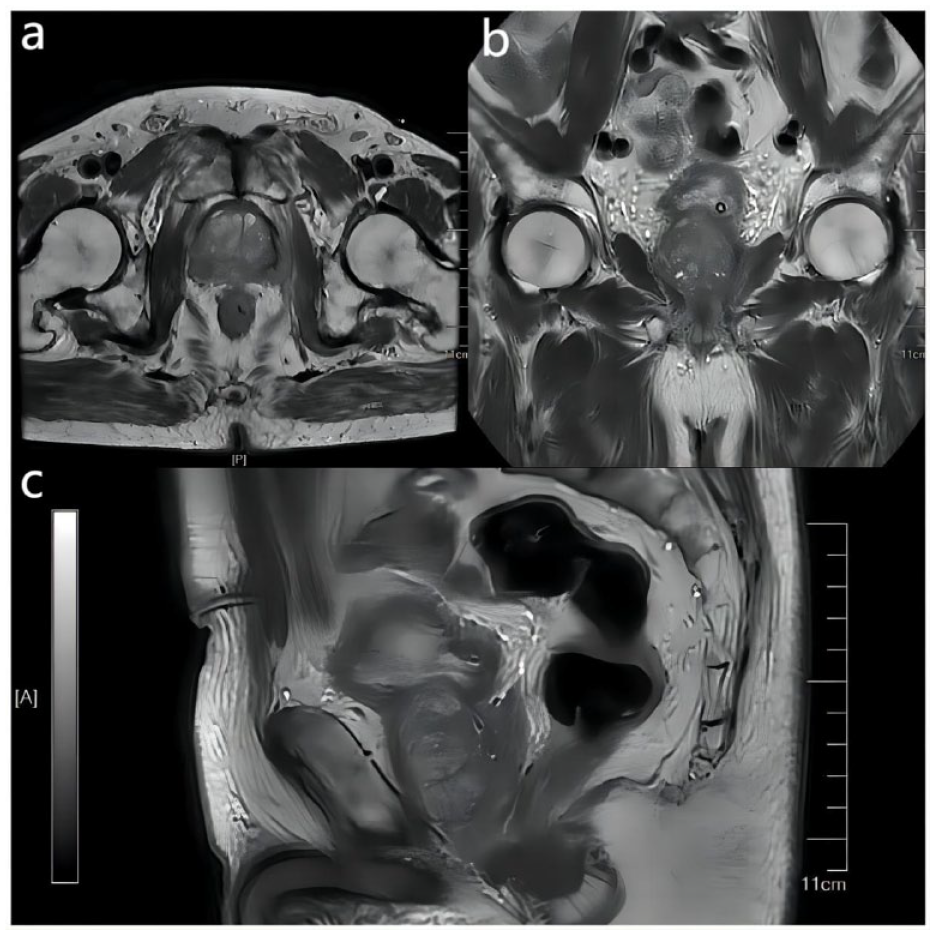
Posttreatment Magnetic Resonance Imaging Examination of the Prostate Revealed Complete Absorption of the Prostate Abscess: (a) Axial View, (b) Coronal View, and (c) Sagittal View.
Following the removal of the perineal drainage tube, the patient continued to receive oral furantoin treatment, as determined by the urine culture results, for 3 weeks. Subsequently, the patient returned to our hospital for a follow-up visit, presenting without fever, and a prostate MRI revealed clear signs of abscess absorption. Further assessment of the patient’s condition was conducted. Considering the patient’s previous history of prostatic hyperplasia, the International Prostate Symptom Score (IPSS) was recorded at 30, with a QOL score of 6. The urodynamic evaluation indicated concurrent bladder outlet obstruction and a slightly weakened detrusor muscle. Finally, the patient underwent transurethral electroresection of the prostate. Postoperative histopathological examination revealed prostatic hyperplasia combined with acute and chronic inflammatory cell presence. The patient was successfully discharged, and a week later, the urinary tube and suprapubic vesicostomy tube were removed. After surgery, the patient reported minor occurrences of urinary frequency and urgency, without experiencing dysuria or pain. Three months later, a telephonic follow-up was conducted, revealing no reported adverse events. The patient’ condition was reassessed, with the IPSS recorded at 12, the QOL score at 2, and the patient expressing high satisfaction with the treatment outcome.
Discussion
An abscess, typically resulting from an intense inflammatory response during infection, constitutes a collection of suppurative material comprising cellular debris, liquefied tissue from the infection source, bacteria, WBCs, and enzymes. Prostate abscesses commonly arise as complications of acute bacterial prostatitis following a severe infection, with the first reported case dating back to 1842, described by Allison, which unfortunately led to the patient’s demise (Allison, 1842). Untreated prostate abscesses, if not promptly and appropriately managed, can progress to severe urogenic sepsis and septic shock, ultimately resulting in fatality (Lee et al., 2016). Due to the widespread use of antibiotics today, the incidence of prostate abscesses has significantly decreased. However, the condition still prevails in developing countries and among high-risk individuals. Globally, the management of prostate abscesses remains challenging due to the lack of clear diagnostic and treatment guidelines (Abdelmoteleb et al., 2017; Shigemura & Fujisawa, 2018). This case report is based on previous diagnostic and treatment experiences, along with the patient’s specific clinical presentation. Through urinary diversion, endovascular irrigation, and electroprostatectomy, a large prostate abscess was successfully cured, and subsequent follow-up assessments revealed no recurrence of the abscess. This report is shared in the hope of providing some valuable references for the future development of guidelines for diagnosing and treating prostate abscesses.
Prostate abscesses are more frequently observed in individuals with poor blood glucose control and compromised immunity. Additional risk factors encompass bladder outlet obstruction, neurogenic bladder dysfunction, postbiopsy prostate surgery, and potential voiding dysfunction (Brede & Shoskes, 2011; Nomura et al., 2021; Tiwari et al., 2011). Although this patient does not belong to the diabetic and immunocompromised group, he has a history of prostatic hyperplasia, a condition often associated with dysuria, weak urinary flow, frequent urination, and urgency. Urodynamic assessments revealed signs of bladder outlet obstruction and an increased residual urine volume. The patient also reported a low daily water intake, which adds to the risk factors for developing a prostate abscess. The patient’s lower urinary tract symptoms worsened progressively 5 months ago, though they did not receive adequate attention until chills and fever, coupled with severe voiding dysfunctions, prompted medical evaluation. It was only after evaluation that the diagnosis of a prostate abscess was considered. Our hypothesis suggests that the patient might have experienced prolonged prostatic hyperplasia, resulting in bladder outlet obstruction, impaired urination, and residual urine. Initially, the patient presented with a urinary tract infection, followed by acute prostatitis, which progressed to a prostate abscess due to untreated prostatic hyperplasia. Therefore, it is believed that the root cause of this patient’s condition involves resolving the bladder outlet obstruction caused by prostatic hyperplasia.
Previous studies have underscored the pivotal role of anti-infective therapy and drainage in the treatment of prostate abscesses, necessitating the choice of appropriate antibiotics based on etiological findings. Common drainage strategies encompass ultrasound-guided puncture drainage, transurethral drainage, transurethral holmium laser removal of prostate abscess, and various open surgical drainage methods (Abdelmoteleb et al., 2017; Lee et al., 2016). In the case of this patient, who presented with urinary retention combined with urinary tract infection, treatment was initiated by inserting an indwelling urinary catheter, followed by a suprapubic bladder puncture stomy to fully drain the infected urine and redirect urine flow, thereby preventing further irritation to the prostate and exacerbation of the inflammatory response. Subsequently, ultrasound-guided perineal drainage of the prostatic abscess was performed, followed by intermittent irrigation of the pus cavity to achieve complete pus evacuation. Finally, after the patient’s prostate abscess underwent absorption, transurethral electroresection of the prostate was performed. In this case, transurethral surgery was delayed performed after infection was controlled by percutaneous perineal drainage and anti-infective therapy. We thought this patient had a severe infection, and if the infection was not well controlled, transurethral surgery may could lead a massive release of abscesses, causing sepsis or even septic shock because of the a high-pressure environment and a long operation time. Moreover, after communicating with patient and his families, they want to take relatively less risky treatment measures. We thought these strategies might be an appropriate personalized treatment for this patient. This choice was motivated by our objective to address the patient’s outlet obstruction, improve urination function, and minimize the risk of complications such as urinary system infections. The patient’s prognosis validated this approach, as follow-up assessments revealed significant improvements in urination function without any recurrence of prostate abscess. The patient expressed high satisfaction with this favorable outcome.
The pathogens investigated in this study were identified as E. cloacae and K. pneumoniae. Urine culture results indicated the susceptibility of these pathogens to piperacillin/tazobactam and furantoin, one of the sensitive antibiotics for these strains. It is possible that inadequate antibiotic intensity and duration of treatment administered at local hospitals contributed to the development of multidrug-resistant bacteria. Without the prompt initiation of sensitive antibiotics, the prostate abscess might progress further, potentially leading to the infection of distant organs (Chrystle et al., 2021; Kam et al., 2019; Mücke et al., 2017). Hence, the judicious selection of sensitive antibiotics, coupled with adequate dosage and duration of treatment, is of paramount importance in the management of prostate abscesses.
This case underscores that the key to the treatment of prostate abscess lies in targeted anti-infective therapy and comprehensive pus drainage. Preventing the recurrence of prostate hinges on addressing the underlying cause of the abscess. In this patient, the root cause was attributed to prostatic hyperplasia. Our therapeutic approach in this case, encompassing urinary diversion and intermittent pus cavity irrigation, yielded favorable results. However, due to the inherent limitations of case reports, further research is warranted to prove its effectiveness in the future. It is hoped that the insights shared through our case report can offer some valuable guidance for the diagnosis and treatment of prostate abscess.
Footnotes
Acknowledgements
The authors thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All interventions were part of standard health care practices; thus, ethical approval was neither required nor sought.
Informed Consent
Written informed consent for the publication of this case report was obtained from the patient.
