Abstract
Hepatobronchial fistula is an uncommon but potentially life-threatening complication that can occurs following microwave ablation for hepatocellular carcinoma. The complication is associated with a significant mortality rate. We report the case of a patient with alcoholic cirrhosis and recurrent hepatic carcinoma who developed a delayed pyogenic liver abscess and hepatobronchial fistula and was treated using an uncommon treatment approach. The abscess in segment VIII, adjacent to the diaphragm, progressed to hepatobronchial fistula due to infection caused by Citrobacter freundii. Despite septic shock, acute liver injury, and delayed abscesses, the patient achieved complete oncologic and infectious resolution through ultrasound-guided percutaneous transhepatic drainage combined with lavage, thereby avoiding the need for high-risk surgery. Overall, the present case report highlights the viability of minimally invasive strategies for treating hepatobronchial fistula. In addition, this report emphasizes the significance of diaphragmatic protection during microwave ablation and the need for individualized treatment for postoperative complications.
Introduction
Pyogenic liver abscess (PLA) is a significant infectious condition that affects the hepatobiliary system. PLA is commonly associated with a high prevalence of diabetes among patients with biliary tract disorders.1,2 The incidence of PLA is low, ranging from 1.1/100,000 individuals in Europe to 17.6/100,000 in Asia. However, the mortality rate can be as high as 40%.3,4 The mortality rate of PLA is associated with several factors, including the general health condition of the patient, abscess size, and treatment approach. Additionally, it has been reported that iatrogenic factors can be significantly involved in PLA development. The implementation of locoregional therapies for hepatocellular carcinoma (HCC), such as microwave ablation (MWA) and transarterial chemoembolization (TACE), has identified PLA as a significant complication.5,6 PLA, which is commonly located deep near the diaphragm and is associated with multidrug-resistant pathogens and the immunocompromised status of patients, frequently progresses to hepatobronchial fistula (HBF), triggering invasive complications and substantially increasing the mortality risk. HBF is one of the most severe complications of PLA, which is characterized by the presence of bilioptysis and refractory pleural infections.7,8 In the majority of HBF cases with hepatic necrosis, emergency hepatectomy via a combined thoracoabdominal approach is the only viable treatment approach, despite its technical complexity and the lack of long-term outcome data. However, in 2021, Ammirabile et al. conducted a systematic literature review encompassing 64 studies and 75 patients and demonstrated that percutaneous interventional therapy plays a significant role in HBF management; this study underscores the efficacy and safety of percutaneous treatments for HBF, offering a progressive treatment pathway from minimally invasive techniques to surgery. 9 This approach is particularly suitable for patients with cirrhosis or malignancies who are poor candidates for major surgery. The present study is the first to describe a case of delayed HBF following MWA in a patient with HCC. Furthermore, this case validates and expands the application of percutaneous transhepatic drainage—a minimally invasive technique—in the scenario of complex liver abscess complicated by HBF.
Case presentation
A man in his early 60s presented to the emergency department of Chengde Medical University Affiliated Hospital in February 2024 with intermittent dull pain in the right upper quadrant, jaundice, high-grade fever (peak temperature: 40°C), chills, and respiratory distress. His medical history included alcoholic cirrhosis with esophagogastric varices (9-year history), type 2 diabetes mellitus (9-year history) managed with 250 mg/day metformin, and HCC. He had undergone right posterior segmentectomy for HCC 9 years ago. Recurrent HCC was treated using ultrasound-guided MWA at PLA 302 Hospital in January 2024 (Figure 1).

Pre- and post-MWA magnetic resonance imaging. The tumors in segment (a) V and (b) VIII of the liver (arrows). The areas of post-MWA in segments (c) V and (d) VIII of the liver and the middle hepatic vein (arrows). The arrows indicate the (e) hepatic artery branches and (f) portal vein branches supplying segment VIII of the liver post-MWA. MWA: microwave ablation.
Physical examination revealed hepatic percussion tenderness. His laboratory results were notable; his hemoglobin level was 122.0 g/L, and he had an elevated neutrophil ratio of 95.80%. His total bilirubin level was 218.43 μmol/L, and serum creatinine level was 318.66 μmol/L. In addition, arterial blood gas analysis showed the following results: pH, 7.45; partial pressure of oxygen (pO2), 107 mmHg; partial pressure of carbon dioxide (pCO2), 20 mmHg; base excess, −9.90 mmol/L; and lactate level, 4.80 mmol/L. Furthermore, liver function tests revealed an alanine aminotransferase (ALT) level of 1937 U/L, aspartate aminotransferase (AST) level of 1628 U/L, γ-glutamyl transferase (GGT) level of 159 U/L, and cholinesterase level of 2656 U/L. Coagulation studies indicated that his prothrombin activity was 40.6%, and the N-terminal pro-brain natriuretic peptide (NT-proBNP) level was 5230 pg/mL.
In addition, computed tomography (CT) revealed a gas-containing abscess in segment VIII, with obliteration in the hepatic pedicle of the segment and disappearance of the middle hepatic vein (MHV) (Figure 2(a)). The MWA-treated lesion in segment V appeared intact with no signs of infection (Figure 2(b)). Additionally, the elevation of the right diaphragm resulted in compressive atelectasis of the middle and inferior lobes of the right lung (Figure 2(c) and (d)). Therefore, the patient was diagnosed with PLA, accompanied with septic shock, acute-on-chronic liver failure, acute kidney injury (AKI), and decompensated cirrhosis (Child-Pugh class B; APACHE II score, 11; and predicted mortality, 17.42%).
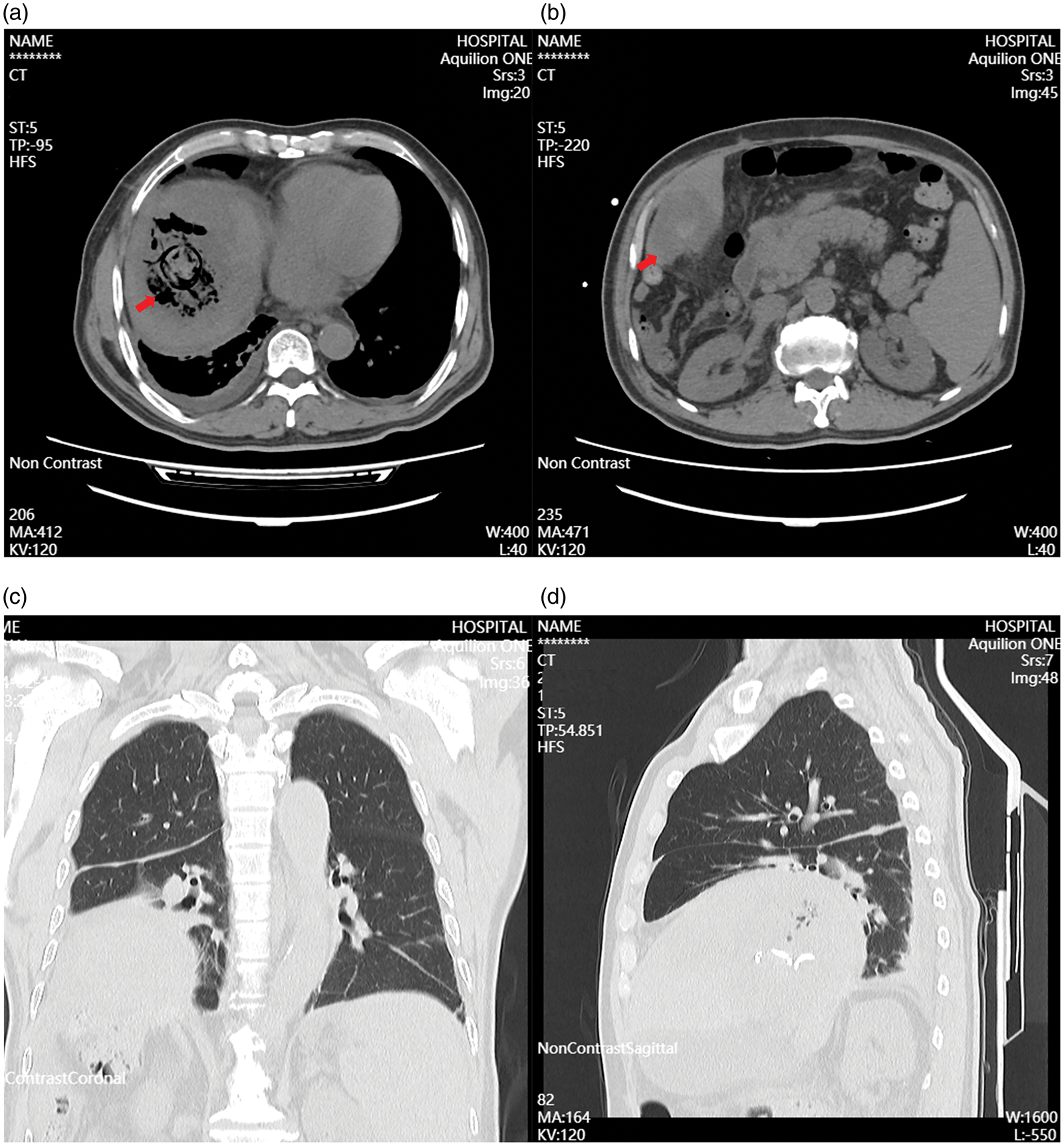
CT during the first admission. (a) A gas-forming pyogenic liver abscess was identified in the VIII segment of the liver (arrow). (b) The necrotic area of the V segment of the liver (arrow). (c) Coronal and (d) axial CT in the lung window. CT: computed tomography.
The patient was initially admitted to the intensive care unit (ICU), where he was subjected to aggressive fluid resuscitation and hemodynamic monitoring. He was also treated with intravenous meropenem (1 g every 8 h) combined with teicoplanin (400 mg daily). Coagulation support was also provided by transfusion of fresh frozen plasma (200 mL/day for 3 days). Furthermore, ultrasound-guided drainage was performed by placing two 8F pigtail catheters into the dependent portion of the abscess cavity. The initial drainage yielded 520 mL of reddish-brown purulent fluid. The microbiological cultures were positive for Citrobacter freundii, which is sensitive to commonly used broad-spectrum antibiotics. Following clinical stabilization, antibiotics were de-escalated to piperacillin–sulbactam (4.5 g every 8 h). The patient was discharged on hospital day 16 with an indwelling drain due to persistent purulent output. He was provided standardized education on drain maintenance protocols emphasizing tube patency, secure fixation, and scheduled follow-up upon drainage cessation.
However, the patient was re-admitted to the emergency department of Chengde Medical University Affiliated Hospital in March 2024, with recurrent fever (peak temperature: 39.8°C), chills, and epigastric pain. Laboratory tests revealed an increased procalcitonin level (2.820 ng/mL), reduced hemoglobin level (70.0 g/L), neutrophil ratio of 88.80%, and albumin level of 26.57 g/L. Subsequent abdominal CT demonstrated a persistent, gas-containing heterogeneous abscess in segment VIII of the liver (Figure 3(a)). In addition, acute emphysematous cholecystitis with gallbladder distension, wall thickening, and sediment-like gallstone was observed (Figure 3(b)).
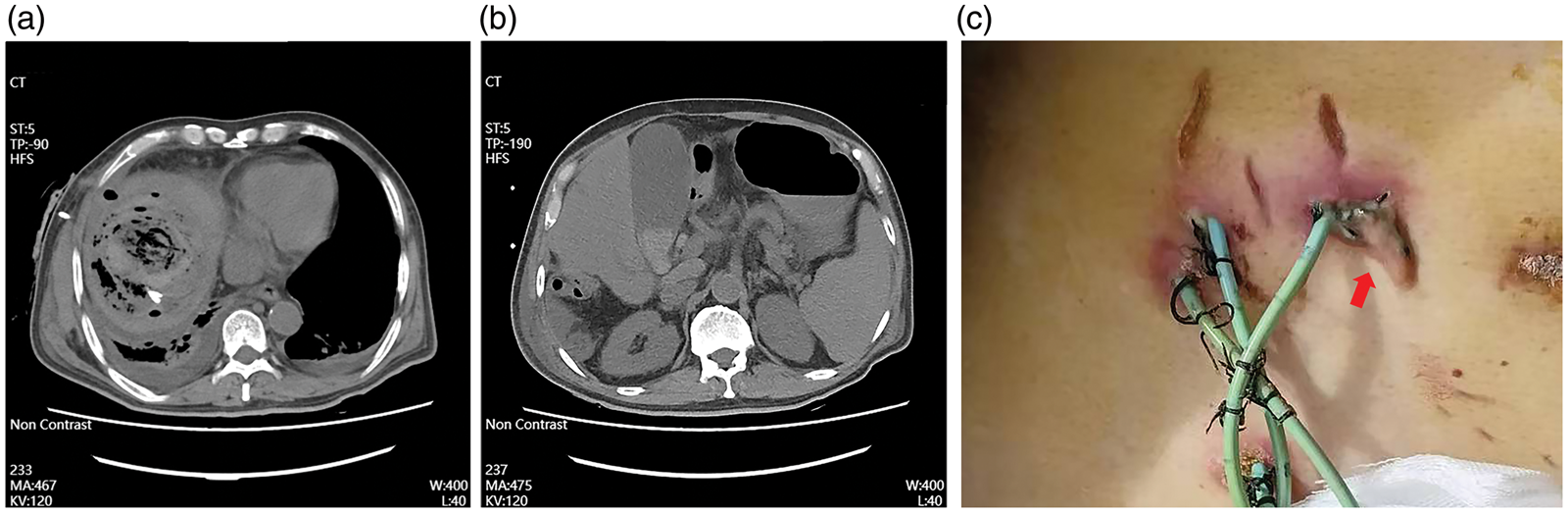
Computed tomography during the second admission and images of the drainage tube. (a) A large gas-forming pyogenic liver abscess within the drainage tube. (b) Cholecystitis and (c) an occluded percutaneous transhepatic liver abscess drainage catheter (arrow).
During the second admission, an occluded drainage tube was removed (Figure 3(c)), while two 8F catheters were inserted under ultrasound guidance. The bile cultures were again positive for C. freundii. However, the antibiotic susceptibility test indicated that the bacterium was resistant to piperacillin, ceftriaxone, ceftazidime, cefazolin, and aztreonam. An 8F percutaneous cholecystostomy tube was also placed, and the bile cultures showed co-infection with Enterococcus faecium. Therefore, ceftoperazone–sulbactam (3 g every 12 h) was initiated, and clinical resolution was achieved within 10 days.
In April 2024, the patient again presented to the emergency department of Chengde Medical University Affiliated Hospital with symptoms of hemoptysis, hematemesis, and melena, with noticeable bleeding from the abscess drainage catheter and the surrounding area (Figure 4(a)). His hemoglobin level further declined to 42.0 g/L, while CT revealed that the PLA had invaded the middle and low lobes of the right lung, verifying the development of HBF. Additionally, given the anatomical proximity and persistent symptoms, the presence of an HBF between the liver abscess and the right middle lobe bronchus was suspected. This finding is highly suggestive of a communication that may explain the patient’s clinical course (Figure 4(b) to (d)). A multidisciplinary management approach was employed. Accordingly, the patient was treated with continuous intravenous infusions of esomeprazole (8 mg/h) and octreotide (25 μg/h); for bleeding control, six units of red blood cells were transfused. Repeat ultrasound-guided abscess drainage was performed, and 10% hypertonic saline and metronidazole (500 mg in 100 mL saline) were instilled twice daily into the abscess cavity to treat both PLA and HBF. Postural drainage was performed and nebulized budesonide was administered to prevent aspiration pneumonia. Moreover, during irrigation of the hepatic abscess cavity via the drainage tube with metronidazole combined with hypertonic saline, the patient’s cough and sputum production markedly increased. Specifically, the patient reported expectoration of a large volume of salty tasting sputum during hypertonic saline irrigation and a large volume of bitter-tasting sputum during metronidazole irrigation. These clinical observations unequivocally confirm the communication between the liver abscess and bronchial tree, definitively establishing the diagnosis of HBF. The integrated therapeutic approach alleviated HBF-related symptoms, as evidenced by the reduced bloody drainage and resolution of hemoptysis. Intravenous ceftoperazone–sulbactam (2 g every 12 h) was initiated. Following hemoglobin stabilization (82.0 g/L) and recovery of hematemesis and bleeding drainage, the patient was discharged.

CT findings of the HBF and drainage tube. (a) Peritubular hemorrhage surrounding the drainage tube. (b) Axial CT showing the HBF (arrow). (c) Coronal CT depicting the HBF (arrow) and (d) HBF in the sagittal CT (arrow). CT: computed tomography; HBF: hepatobronchial fistula.
The patient presented to the emergency department of Chengde Medical University Affiliated Hospital again in June 2024 with a palpable, tender mass in the right upper abdomen, accompanied with intermittent fever (body temperature up to 39.2°C). Contrast-enhanced CT revealed regression of the abscess in segment VIII of the liver, with revascularization of the portal vein (PV), hepatic artery branches, and MHV (Figure 5(a) to (c)). In addition, a new abscess was identified in segment V, which had ruptured into the abdominal cavity and invaded the subcutaneous tissue (Figure 5(d) to (f)). Right-sided pleural thickening and minimal effusion were also detected. Therefore, an 8F catheter was inserted into the segment V abscess under ultrasound guidance for drainage. Intravenous ceftoperazone–sulbactam (2 g every 12 h) was administered, combined with metronidazole lavage (500 mg twice daily) through the abscess drainage to achieve resolution.

CT during the third admission. (a) Portal vein branches (arrow). (b) Hepatic artery branches (arrows) and (c) middle hepatic vein (arrows). (d) The liver abscess in segment V of the liver (arrow). (e) The abscess invading the subcutaneous tissue and (f) CT showing the subcutaneous abscess. CT: computed tomography.
Based on the National Comprehensive Cancer Network (NCCN) guidelines and China’s liver cancer diagnosis and treatment protocols, the patient underwent regular surveillance every 3 months postoperatively without receiving any specific therapy from December 2024 to May 2025. A follow-up CT in December 2024 revealed that both PLAs in segments V and VIII were completely healed. Therefore, all drainage tubes were removed, the right lower lung lobe was expanded, and the diaphragmatic volume was normalized (Figure 6(a) and (b)). Furthermore, magnetic resonance imaging (MRI) performed in March 2025 confirmed the eradication of abscess and complete remission of HCC (Figure 6(c)). Serum C-reactive protein and procalcitonin levels normalized (Figure 7(a) and (b)), while the serum alpha-fetoprotein level remained within the normal range (Figure 7(c)). Currently, the patient is in good mental, physical, and nutritional states, with free mobility, and has recovered to the health status he had before disease onset. Figure 8 presents a timeline illustrating the patient’s medical history, annotated with symptoms, key findings, and management measures. All patient details have been removed to ensure his privacy. Written informed consent was obtained from the patient prior to treatment. Additionally, the patient signed a data publication consent form. The reporting of this study conforms to the Case Report (CARE) guidelines. 10

The manifestations of patient recovery. (a) Appearance of the puncture incision. (b) Computed tomography showing the healed PLA and HBF and (c) representative enhanced magnetic resonance imaging results without tumor recurrence. PLA: pyogenic liver abscess; HBF: hepatobronchial fistula.

Laboratory results during the treatment process. (a) C-reactive protein levels. (b) Procalcitonin levels and (c) Alpha-fetoprotein levels.

Comprehensive timeline of the patient during each hospitalization. RUQ: right upper quadrant.
Discussion
This is the first case study to document an instance of PLA complicated by delayed HBF following MWA for recurrent HCC. Emerging evidence has suggested that PLA is observed in approximately 0.8%–1.7% of patients undergoing MWA.5,6 However, the occurrence of HBF, a serious complication historically associated with a high mortality rate, following PLA is rare.11,12 Its high mortality rate is associated with its propensity to trigger mediastinitis, empyema, and refractory sepsis, which can progress to multiorgan failure despite aggressive intervention. 13 Herein, the direct transdiaphragmatic invasion of the subphrenic PLA was the primary anatomical factor involved in HBF formation. In addition, the abscesses located in segment VIII exerted sustained pressure, mechanically disrupting the diaphragmatic layers. This disruption allowed continuous bacterial invasion into the pleural space.
Herein, we reported the case of a patient with sepsis due to a gas-forming PLA caused by C. freundii, a gas-producing bacterium. The increasing pressure from the abscess led to the development of a productive cough and chest pain. These symptoms were associated with the formation of HBF, which was ultimately involved in the onset of hemoptysis and pneumonia. Isolation of C. freundii from a PLA promoting the development of HBF has rarely been reported. 14 Although C. freundii is considered an opportunistic pathogen, it is a gram-negative bacterium associated with several severe infections, 15 possibly due to its multidrug resistance potential. C. freundii exhibits several different resistance mechanisms, such as AmpC lactamase, broad-spectrum beta-lactamase, extended-spectrum beta-lactamase (ESBL), or plasmid-mediated reduction in influx or enhancement of efflux of quinolone or carbapenem.16,17 Citrobacter infections commonly occur in neonates, debilitated individuals, or immunocompromised patients and can affect multiple organs, such as the urinary tract, respiratory tract, wounds, bones, peritoneum, endocardium, meninges, and bloodstream. 15 In the current case, a mutation in C. freundii was identified, which occurred during the continuous administration of antibiotics. To the best of our knowledge, this phenomenon has not been previously reported in the context of PLA therapy. Moreover, the role of E. faecium co-infection in PLA has garnered increasing attention. Studies indicate that E. faecium is among the most prevalent pathogens in PLAs, with vancomycin resistance observed in one-third of these cases. 18 This antimicrobial resistance profile complicates therapeutic management, particularly in the context of multidrug-resistant strains. For PLA management, it is crucial to monitor the infection source to prevent antibiotic overuse, reduce the risk of secondary infections, and maintain the balance of the intestinal flora. In addition to the contribution of anatomical factors, it is necessary to acknowledge the role of iatrogenic factors stemming from surgical interventions in HBF development.
Currently, the increasing popularity and extensive application of ablation techniques for treating HCC have resulted in the rise of iatrogenic lesions, which in turn can promote the development and progression of PLA and HBF.19,20 Thermal injury is the most common type of ablation-related damage, particularly affecting the diaphragmatic and intrahepatic vasculature. A previous study demonstrated that compared with radiofrequency ablation (RFA), MWA with high power could generate more extensive and consistent ablation zones with lower rates of local tumor progression, even in highly vascularized tissues. 21 However, MWA could also cause thrombosis of larger vessels, resulting in serious complications in patients with cirrhosis or compromised liver function. 22 In the current case, postoperative MRI suggested that the PV and hepatic artery branches supplying the VIII segment of the liver, along with MHV, were occluded due to MWA-mediated thermal injury. Damage to the intrahepatic vessels could play a vital role in the persistence and prolonged course of the PLA over several months. In addition, mechanical injury to the diaphragm, caused by the percutaneous transhepatic drainage of the PLA and percutaneous MWA procedure, could be considered as another potential iatrogenic factor associated with disease progression. Furthermore, CT revealed that the ablation margins extended to the diaphragm and were accompanied with pleural effusion, suggesting thermal injury to the diaphragm, which eventually resulted in HBF development. Overall, the above findings suggest that the complex pathogenesis of PLA and HBF is attributed to a synergistic interaction between bacterial virulence, anatomical vulnerabilities, and iatrogenic factors.
HBF symptoms include bilioptysis, fever, pleuritic chest pain, and abdominal tenderness. 19 In severe cases, there may be complications, including hemoptysis, sepsis, and pneumothorax, requiring urgent medical interventions. 23 Imaging examinations, such as CT and MRI, can reveal indirect signs of HBF, such as subphrenic liver abscesses, pleural effusion, and atelectasis. Furthermore, bronchoscopy is a commonly used and effective method for visualizing HBF in particular cases. 24 Percutaneous transhepatic drainage under digital subtraction angiography (DSA) guidance remains the diagnostic gold standard as it allows the dynamic visualization of contrast agent leakage from intrahepatic lesions into the bronchial tree. 25 Herein, HBF was diagnosed based on clinical symptoms, such as hemoptysis and cough, particularly during antibiotic irrigation, as well as indirect findings on CT indicators. The patient did not present bilioptysis or bile-tinged sputum, as neither PLA nor HBF is associated with the bile duct.
Surgical interventions and supportive therapies are commonly utilized to treat HBF. Supportive therapy can only be used for patients with mild symptoms, while nonsurgical treatment options can be considered for those with poor overall health. Endobronchial techniques, such as the Watanabe plug or Amplatzer device, have also been proven to be effective minimally invasive treatment strategies for HBF. 26 Percutaneous transhepatic drainage is used to manage PLA and HBF, commonly combined with antibiotics to alleviate symptoms. 19 A comprehensive review of the existing literature indicates that for patients with massive liver abscesses, indwelling catheter drainage demonstrates superior efficacy compared with simple needle aspiration. Furthermore, repeated intermittent aspiration necessitates multiple procedures, subjecting patients to the discomfort of successive interventions and resulting in poor tolerance and compliance.27,28 Although nonsurgical methods can be effective in particular circumstances, surgical intervention remains the preferred approach for the majority of patients, particularly for complex cases. The surgical management of HBF commonly includes the resection of the necrotic tissues, such as the infectious parts of the liver, diaphragm, or lungs, and repair of the fistula. 29 Although early diagnosis and surgical intervention can improve prognosis, individualized therapy remains essential due to the rare incidence of postoperative PLA and HBF as well as the lack of standardized guidelines. 30 In the present case, the successful resolution of PLA complicated by HBF and concurrent cholecystitis was achieved via ultrasound-guided percutaneous transhepatic drainage accompanied with external antibiotic irrigation. This approach challenged traditional surgical intervention, thereby emphasizing the potential of minimally invasive techniques, even in complex cases, and was ultimately considered a significant advancement in the treatment of PLA and HBF.
Taken together, this is the first case report of delayed PLA and HBF caused by C. freundii infection following MWA for recurrent HCC. Despite life-threatening complications, the patient achieved complete oncologic remission and successful fistula resolution through minimally invasive percutaneous transhepatic drainage in combination with antibiotic irrigation, without the need for major invasive surgery. HBF is a serious complication of MWA for HCC. Comprehensive preoperative evaluation, improvement in surgical techniques, and personalized management of postoperative complications are crucial for improving patient prognosis. However, further studies are needed to establish standardized protocols.
Footnotes
Acknowledgments
The authors would like to thank Dr Lilong Yan from the Hepatology department of PLA 302 Hospital for verifying and reviewing the treatment process, which improved the manuscript.
Authors’ contributions
Haizhao Yi and Hua Fu wrote the main manuscript text and provided important intellectual content. Mengze Song contributed to the treatment process and prepared the figures. Xiangning Lu, Jinlong Liu, and Jian Li contributed to study supervision. All authors contributed to writing, critical review, and final approval of the submitted manuscript.
Data availability statement
The datasets utilized and/or analyzed during this study can be obtained from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare no competing interests.
Ethics statement
The study was approved by the Clinical Research Ethics Committees of the Affiliated Hospital of Chengde Medical University (Approval No. CYFYLL2024075). The patient provided written informed consent for publication of their data.
Funding
No funding support was received for this research.
