Abstract
Male accessory breast cancer is an extremely rare tumor. There is no report about its monotherapy and subsequent outcome prior to 2022. The current study presents the case of a 76-year-old male patient with a hard mass in the left axilla. Histopathologic examination of an excisional specimen indicated an adenocarcinoma compatible with breast carcinoma. Immunohistochemical analysis demonstrated that the mass was estrogen receptor (ER) (−), progesterone receptor (PR)(−), and human epidermal growth factor receptor type 2 (HER2) (−). A diagnosis of breast cancer originating from the accessory mammary gland in the axilla was made. Two years following surgery, the patient presented with a pulmonary lesion. Core needle biopsy was performed, and the lesion was found to be ER (−), PR(−), and HER2 (3+). The patient was successfully treated with single-agent trastuzumab. Single-agent trastuzumab could be a reasonable regimen for metastatic accessory breast cancer patients with HER2 overexpression for whom chemotherapy and endocrine therapy are not suitable.
Introduction
Accessory breasts are observed in 2% to 6% of the general population, and these tissues may present in various positions along the milk line but most frequently in the axillary region. An accessory breast carcinoma is a form of breast cancer occurring in the axilla or inguinal region, where there are abundant lymph nodes and capillaries (Bi et al., 2015; Eguchi et al., 2021; Gutermuth et al., 2006). Male accessory breast cancer is rare tumor. Its treatment and effects have been poorly reported. The present study reports the case of a 76-year-old male accessory breast cancer patient who underwent surgical excisions of primary ectopic breast cancer. Two years later, the patient developed a pulmonary metastasis. He was successfully treated with single-agent trastuzumab.
Case Presentation
A 76-year-old man first noticed a small subcutaneous nodule in the left axillary area in 2013. The nodule gradually increased in size, and he was referred to our hospital in May 2014. Clinical examination revealed an irregular hard mass, measuring roughly 92 mm × 68 mm in the left axilla (Figure 1). Computed tomography (CT) showed an exposed and lobulated 82 mm × 62 mm soft tissue density mass and enlarged lymph nodes in the left axilla. Magnetic resonance imaging (MRI) and ultrasonography (US) revealed no primary lesion in the ipsilateral breast tail and bilateral mammary gland. In addition, positron emission tomography (PET/CT) did not detect any lesions other than the axillary one. The mass was located apart from the normal mammary gland.

An Irregular and Hard Mass, Measuring Roughly 92 mm×68 mm, Exposed in the left Axilla
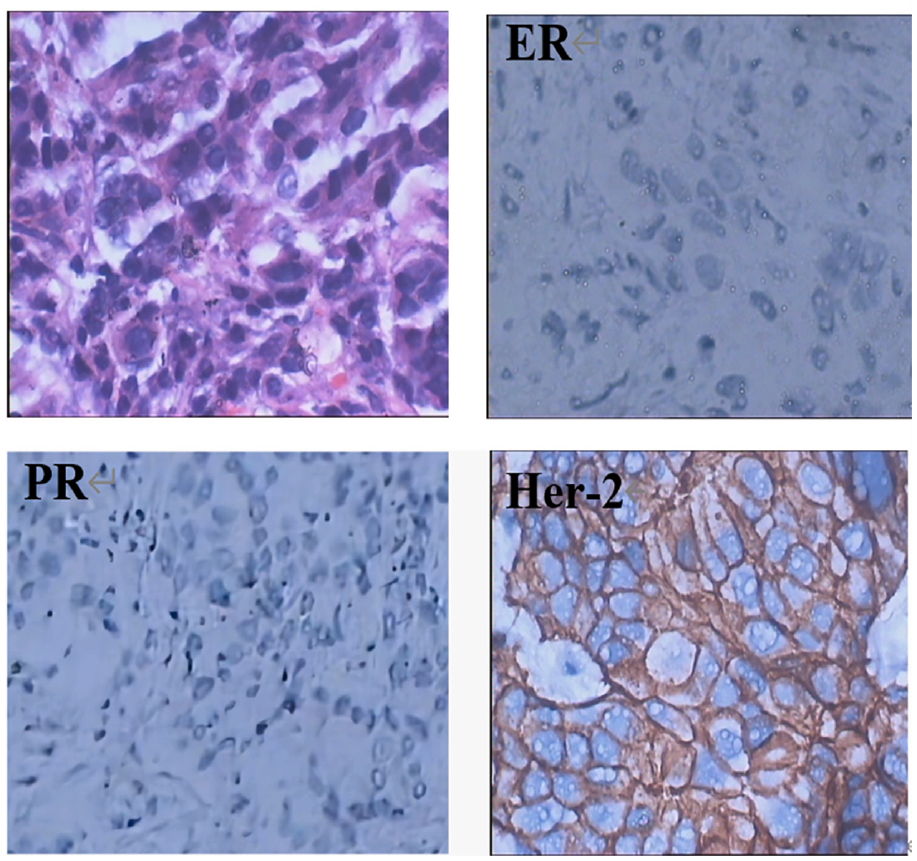
The patient had neither an apparent tumor in the breast nor a history of breast carcinoma. The tumor in the axilla was excised with a 2 cm horizontal margin, and left axillary lymph node dissection was performed. The size of the excised mass was 119 mm × 86 mm.(Figure 2). The macroscopic appearance of the lesion indicated the proliferation of atypical cells of variable size in the subcutis. The cells had enlarged and irregular nuclei and formed a luminal structure unconnected with the epidermis, indicating an adenocarcinoma compatible with breast carcinoma. Immunohistochemical analysis demonstrated that the mass was estrogen receptor (ER) (−), progesterone receptor (PR)(−), and human epidermal growth factor receptor type 2 (HER2) (−) (Figure 3). According to the clinical performance and pathological findings, the mass was considered to be an adenocarcinoma from the accessory breast tissue. A total of five out of the 16 dissected axillary lymph nodes were positive for metastatic carcinoma. The patient was diagnosed with left accessory breast carcinoma (pT3N2M0). Subsequently, he was subjected to 3 courses of FEC chemotherapy (5-Fu at 500mg/m2, epirubicin at 75mg/m2, and cyclophosphamide at 500mg/m2) once every 3 weeks, and sequential docetaxel chemotherapy at 75/m2 was continued by three cycles along with radiation therapy.

The Excised Mass, Size of the Mass Is 119 mm × 86 mm

The Microscopic Appearance of the Incisional Lesion Revealed That the Cell Had Enlarged and Irregular Nuclei and Formed a Luminal Structure Unconnected With the Epidermis, Which Is Compatible With Breast Carcinoma (×200) Immunohistochemistry of an Accessory Breast Tumor (×400)
In Jun 2016, CT demonstrated a 32 mm × 30 mm metastatic foci incidentally in the left lung. A core needle biopsy was performed for it. The hematoxylun–eosin (HE) staining of the biopsy specimen revealed adenocarcinoma metastatic from the breast tissue. Immunohistochemical staining yielded the following results: CA199-9 (−), carcinoembryonic antigen (CEA)(−), PeriodicAcid-Schiff stain (PAS)(−), ER(−), PR(−), and HER-2(3+) (Figure 4). Both US and MRI revealed no evidence of a tumor in the bilateral mammary glands. These findings strongly suggested metastatic accessory breast cancer. This patient was not a candidate for chemotherapy due to performance status or comorbidities. Due to his HER-2(3+), single-agent trastuzumab (8mg/kg at a loading dose and a maintenance dose of 6 mg/kg every 3 weeks) has been ongoing since Jun 2016, as a first-line treatment. Following four cycles of treatment, a partial response was confirmed by CT (Figure 5) in November 2016. At the latest follow-up in February 2017, the foci remained stable with no evidence of progression. During treatment with trastuzumab, no adverse effects, including cardiotoxicity, were observed, and an ejection fraction of more than 55% was confirmed by ultrasound cardiography. In June 2019, lung CT confirmed that the metastasis was stable, (Figure 5) and no other metastases were found. Due to the COVID-19 pandemic, patients are not able to come to the hospital for systematic examination. We cannot obtain the latest condition of this patient.
Histology and Immunohistochemistry of an Metastasis (×400)

CT Revealed Lung Metastases of the Accessory Breast Cancer: (A) CT Showed a Metastatic Foci in the Left Lung (June 2016); (B) After Trastuzumab Monotherapy, the Tumor Was Reduced, By 50% in Diameter (November 2016); (C) CT Showed the Patient’s Metastases Is Stable (June 2019)
Discussion
Accessory breast carcinoma in males is extremely rare, with the most common clinical manifestation being accessory breast carcinoma of the axilla (Fentiman, 2006; Grossl, 2000). Generally, accessory breast cancer must be pathologically demonstrated to be located adjacent to normal breast ducts or lobules that are not connected with the proper mammary gland, and it is also necessary to exclude the possibility of a metastatic lesion from another primary cancer (Yerra et al., 1997).
In our case, systemic examinations including US, CT, MRI, and PET/CT showed no occult lesions or other primary malignant tumors, only the axillary tumor. Histopathologic examination of the HE-stained specimen revealed a tumor growth pattern compatible with breast cancer, and Immunohistochemical analysis demonstrated it to be ER(−), PR(−), and HER2(−). We diagnosed this case as primary breast cancer, arising in an accessory mammary gland.
In the previous literature, at the time of diagnosis, the majority of patients who had accessory breast cancer were diagnosed at an advanced clinical stage, with unresectable tumor size or nodal metastasis. The prognosis of accessory breast cancer may be worse than breast cancer, although no long-term follow-up data regarding the prognosis of accessory breast cancer is available(Nihon-Yanagi et al., 2011; Yamamura et al., 2012). In several studies, treatment regimens for accessory breast carcinoma followed the guidelines for breast cancer (Kahraman-Cetintas et al., 2008). Similar to breast cancer, accessory breast cancer is also surgically treated and supplemented with chemotherapy, radiotherapy, and hormonal therapy (Nguyen et al, 2022). Recently, some scholars have also conducted a systematic analysis of the treatment of male breast cancer. They concluded that at present, male parabreast cancer is mainly treated by surgical resection, supplemented with chemotherapy, radiotherapy, endocrine therapy, and other comprehensive treatments (Pang et al., 2021). There is no report on the use of trastuzumab monotherapy in metastatic male accessory breast cancer and its effect until 2022.
Trastuzumab is a humanized monoclonal antibody that is directed against the extracellular domain of the HER-2 receptor protein. HER 2 status was assessed by immunohistochemistry or dual-color fluorescence in situ hybridization (FISH). The patient’s immunohistochemistry indicated HER2 (3+), which can be directly judged to be positive for HER-2. If the immunohistochemistry test result is HER-2(2+), a positive FISH test should be performed for confirmation. FISH testing: FISH testing is performed in a qualified laboratory. According to the American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) criteria, if the HER-2/CEP17 ratio is ≥2.0 or the HER-2 gene copy number is ≥6, the test result is considered HER-2 positive. (Jensen et al., 2020). As an effective method, combined chemotherapy with trastuzumab has become a standard treatment for HER-2 overexpressing breast cancer. Despite there being little high-level medical evidence supporting trastuzumab monotherapy as a treatment for metastatic breast cancer, significant clinical benefits have been highlighted in a phase ãÀ clinical study for treatment of advanced breast cancer with a HER2 immunohistochemical score of 3+. In most cases, HER2 expression is compatible between the primary and metastatic breast cancer, and a meta-analysis reported that discordance of HER2 status between the primary breast tumor and its metastasis was 5.54% (Houssami et al., 2011). To date, most of the published studies of trastuzumab have assessed its role in the treatment of breast cancer with HER2 overexpression or amplification.
The present case was a patient with metastasis in the left lung 2 years following surgery for accessory breast cancer. A core needle biopsy was performed, and the morphologic and histopathologic features of the lesion supported a diagnosis of metastatic accessory breast cancer which was confirmed to be ER (−), PR (−), and HER2 (3+), indicating that although HER2 expression was negative in the primary lesion, it had become positive in the metastatic tumor. The individual, in this case, was a patient with HER2 overexpressing accessory breast cancer in the secondary pulmonary metastatic lesion. Because the tumor was ER−/PR−, it would not be sensitive to endocrine therapy, and due to the age of the patient, chemotherapy would pose a significant health risk. We considered an evaluation of the rationality of trastuzumab monotherapy to be essential before administration. Due to the HER2 overexpression, trastuzumab monotherapy may be appropriate for him, following the guidelines for breast cancer treatment. Although there has been no report concerning the response rate of trastuzumab monotherapy for metastatic male accessory breast cancer, Tsutani et al. (2006) reported one case of complete remission in a patient with a breast cancer lung metastasis treated with trastuzumab alone. Inspired by this report, we investigated the usefulness of single-agent trastuzumab for this HER2-overexpressing lung metastasis of accessory breast cancer. Following the tentative administration of trastuzumab monotherapy in the present case, the pulmonary foci were reduced gradually, and then it was stable for 15 months, which was likely to significantly prolong survival and enhance the patient’s quality of life.
Infusion reactions such as chills, fever, pain, fatigue, and cardiac dysfunction were not observed in our patient. Therefore, trastuzumab monotherapy is an effective and tolerated therapy for elderly patients who are unsuitable for chemotherapy.
In conclusion, the present study reported a case of male accessory breast cancer in a patient with pulmonary metastasis; however, he was successfully treated with trastuzumab monotherapy. Currently, the patient is under follow-up observation without any progression of the metastatic lesion prior to the latest follow-up in 2019. Single-agent trastuzumab could be a reasonable regimen for metastatic accessory breast cancer patients with HER2 overexpression in who chemotherapy and endocrine therapy are not suitable. This article also has some shortcomings, such as the lack of updated information on the current status of the patient due to the COVID-19 pandemic.
Footnotes
Acknowledgements
The authors would like to acknowledge the patient for allowing them to use his medical records in their case report and allowing the case to be published.
Authors’ Note
All authors have read and approved the information.
Authors’ Contributions
W.G. designed the study, researched the literature, and drafted the manuscript. W.J. participated in study design and coordination, and helped to collect data. All authors have read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent for Publication
The ethics committee of Dalian Central Hospital has agreed to perform the study and publish the results.
Informed Consent
Written informed consent was obtained from the patient for the publication of this case report.
Availability of Data and Materials
The data and material are freely available upon requests. Please contact Dr. Wenbin Guo (
