Abstract
Objective
To compare estimated survival times of patients who had received maintenance monotherapy with gemcitabine (GEM), or an immuno-oncology (IO) drug (i.e., pembrolizumab or avelumab) or both therapies (one after the other) following platinum-based combination chemotherapy for metastatic urothelial carcinoma (UC) in a real-world setting.
Methods
For this retrospective study, we included consecutive patients with metastatic UC who had received first-line platinum-based chemotherapy followed by second-line treatment at our centre from March 2008 to June 2020.
Results
Of the 74 patients identified, 58 had received monotherapy as second line treatment, and 16 had received combination chemotherapy (i.e., non-monotherapy). The estimated median duration of survival was significantly longer in the monotherapy group compared with the non-monotherapy group (29 vs 7 months). Multivariate analysis showed that the outcome of the first-line chemotherapy treatment was the most important prognostic factor for survival. There was no significant difference in survival times between monotherapy with GEM or IO drugs. In addition, survival was significantly prolonged when GEM therapy was administered following IO drugs compared with GEM therapy alone.
Conclusion
Monotherapy following primary chemotherapy for advanced UC significantly prolonged survival times, and IO drug therapy remained effective when followed by GEM single agent maintenance therapy.
Introduction
Urothelial carcinoma (UC) is a malignant tumour of the upper (i.e., renal pelvis, and ureters) and lower (i.e., bladder, and urethra) urinary tract and is the sixth most common tumour in developed countries. 1 The most common type of UC is bladder cancer. 2 Metastatic UC is known to progress rapidly and has a poor prognosis with estimated median overall survival of approximately a year.3,4 The mainstay of treatment for metastatic UC is platinum-based combination chemotherapy, and gemcitabine (GEM) plus cisplatin is the most widely used first-line therapy. 5 Generally, UC is highly sensitive to chemotherapy and the initial response rate has been reported to be around 50–70%. 5 In patients with impaired renal function, carboplatin has been used instead of cisplatin and response rate of approximately 40% have been observed with survival rates equivalent to that of cisplatin.6,7 Because cisplatin causes cumulative nephrotoxicity and neurotoxicity, the use of GEM plus cisplatin as primary chemotherapy has been limited to six cycles.5,8 Interestingly, one study showed that five or more cycles of the initial platinum-based chemotherapy did not prolong overall survival compared with four cycles, and suggested that four cycles of chemotherapy may be sufficient. 9
Following platinum-based chemotherapy, there is no internationally accepted standard of care for second-line therapy. 10 The development of immuno-oncology (IO) agents, such as pembrolizumab and avelumab have added to the options available in the second-line setting.10,11 However, before the introduction of IO drugs, monotherapy with cytotoxic agents was used as follow-up treatment until disease progression.12,13 In our department, we have often used GEM as maintenance monotherapy after primary chemotherapy in advanced UC.
The aim of our retrospective study from a real-world setting was to compare estimated survival times of patients who had received maintenance monotherapy with GEM, or an IO drug (i.e., pembrolizumab or avelumab) or both therapies (one after the other) following platinum-based combination chemotherapy for metastatic UC. In addition, we investigated potential prognostic factors for survival.
Methods
We retrospectively reviewed data from consecutive patients with metastatic UC who had received platinum-based chemotherapy at our centre from March 2008 to June 2020. We separated patients into those who had received further combination therapy, or monotherapy with GEM and/or IO drug (pembrolizumab or avelumab) following their first line treatment.
GEM dosing was based on the standard infusion dose (1000 mg/m2) and dosing intervals (Day 1 and Day 8/every 3 weeks), pembrolizumab was given at 200 mg IV every three weeks, and avelumab at 10 mg/kg IV every two weeks. At the discretion of the attending physician, the dosing interval and doses were adjusted according to the patient's condition. None of the patients had a history of systemic anticancer therapy for advanced or metastatic disease. Treatment results for metastatic sites following first-line chemotherapy were evaluated based on the Response Evaluation Criteria in Solid Tumours (RECIST) guidelines (i.e., complete response [CR], partial response [PR], stable disease [SD], or progressive disease [PD]). 14 Termination of each therapy was at the discretion of the attending physician following observation of any side effects or clinical progression.
The reporting of this study conforms to STROBE guidelines, 15 and was approved by the Institutional Review Board and the Research Ethics Committee of Nihon University School of Medicine (RK-190611-3). Informed consent was obtained based on an opt-out method and patient details were de-identified.
Statistical analyses
Statistical analyses were performed using SPSS Statistics (IBM Japan, Tokyo, Japan) and JMP® version 14 (SAS Institute Japan, Inc., Tokyo, Japan). A
Results
Of the 74 patients with UC who had received first-line platinum-based chemotherapy at our centre during the study period, 21 had received neoadjuvant/adjuvant therapy, 19 were classed as PD and 34 were classed as CR/PR/SD (Figure 1). Of the 74 patients, 58 subsequently received monotherapy as second line treatment, and 16 received further combination chemotherapy (i.e., non-monotherapy; Figure 1). While there was no difference between groups in mean age (69

Patient flowchart.
Patient characteristics and outcomes for those who received first-line chemotherapy (

Data are expressed as, mean ± standard deviation, or
ns, not statistically significant.
The estimated median duration of survival was significantly longer in the monotherapy group compared with the non-monotherapy group (29 vs 7 months,
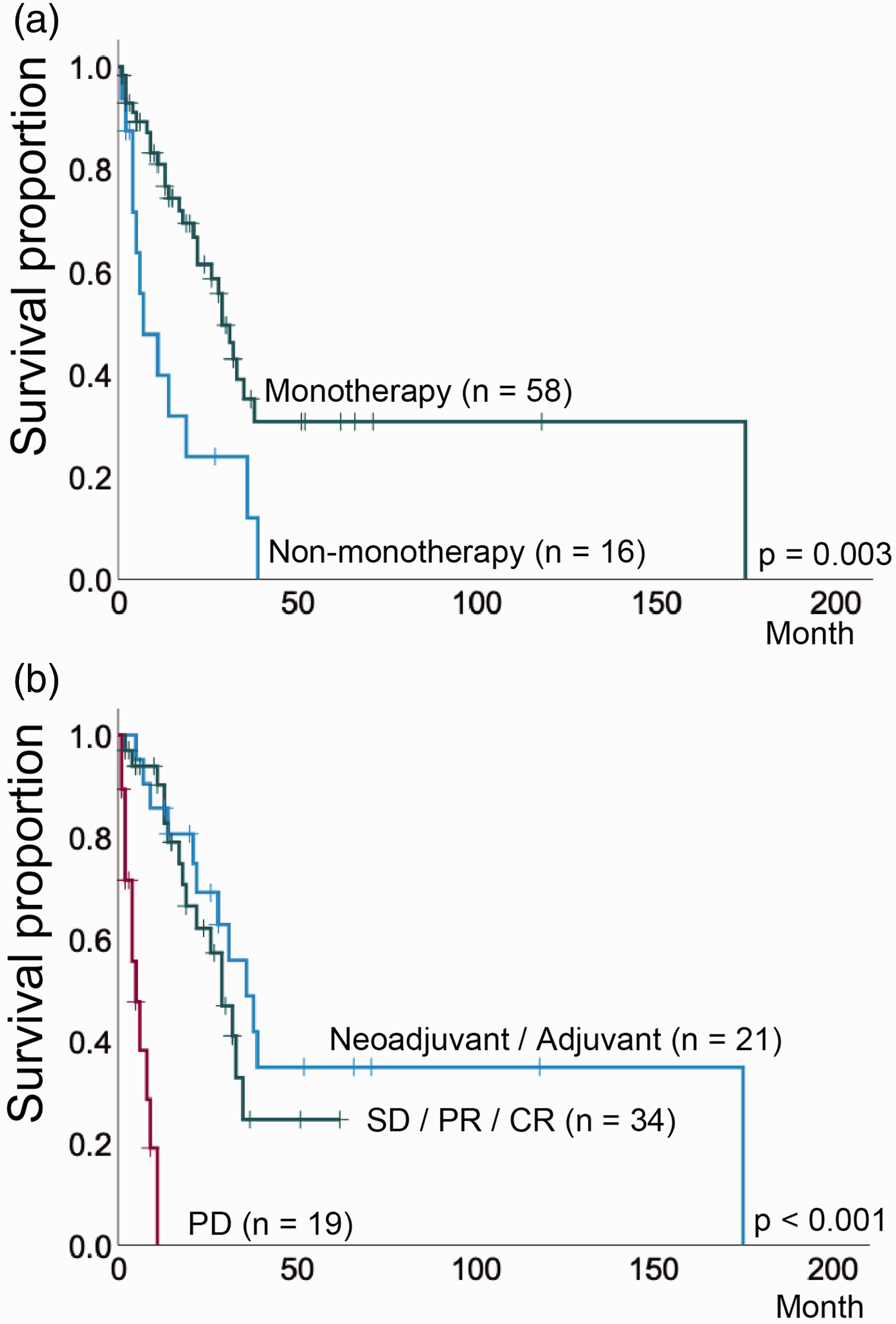
(a). Survival in patients who received either monotherapy or non-monotherapy (i.e., combination therapy) following first-line chemotherapy (
We analysed prognostic factors for survival based on patient characteristics for advanced UC. These were: age (<70 vs ≥70 years); resected primary tumour; TNM classification (≤2 vs >2); regional lymph nodes (≥N1); distant metastases (0 vs 1); outcome following first-line therapy (Others vs PD); monotherapy vs non-monotherapy. Multivariate analysis showed that the outcome of first-line chemotherapy was the most significant prognostic factor for survival (hazard ratio 15.5, 95% CI, 4.9–49,
Univariate and multivariate proportional hazard analyses of prognostic factors for survival (
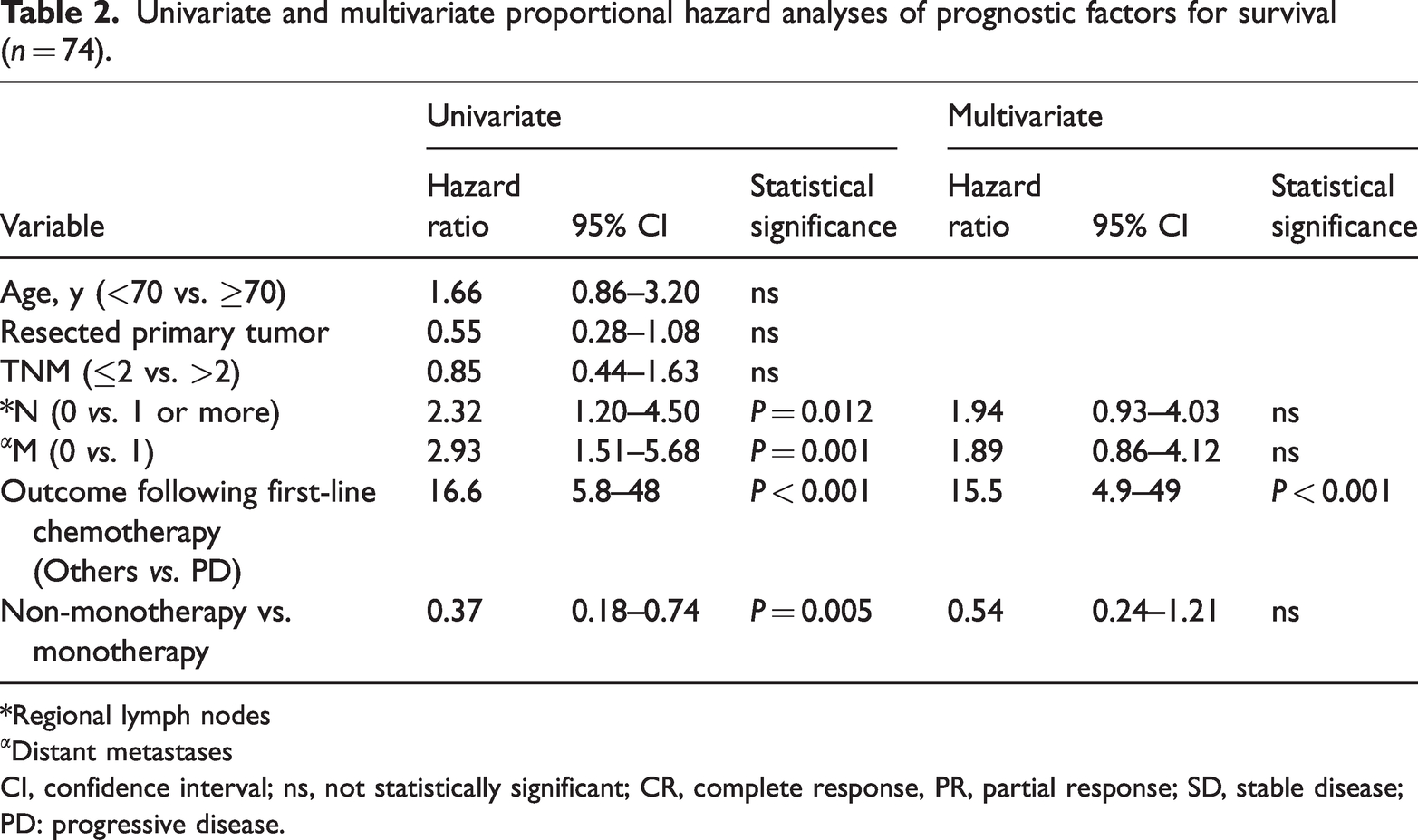
*Regional lymph nodes
Distant metastases
CI, confidence interval; ns, not statistically significant; CR, complete response, PR, partial response; SD, stable disease; PD: progressive disease.
We compared differences in patient characteristics and outcomes among patients that had received GEM (
Comparison between monotherapy treatment groups (

Data are expressed as, mean ± standard deviation, or
*Pembrolizumab or avelumab.
GEM, gemcitabine; IO, immune-oncology; ns, not statistically significant.

(a) Kaplan-Meier estimates of survival times between gemcitabine (GEM) monotherapy and immuno-oncology (IO) drug treatment groups. There was no significant difference between these two groups (GEM, 29 months; IO, 28 months;
Discussion
Advanced and metastatic UC account for the majority of deaths associated with this disease. 16 In a large retrospective study on metastatic upper UC, the most common site of metastasis was identified as the distant lymph nodes, and liver metastasis was found to be a poor prognostic factor, whereas primary tumour resection and chemotherapy were predictors of better survival outcome. 17 The development of cisplatin-based combination chemotherapy has extended median survival from several to 11–15 months. 16 However, the prognosis for metastatic patients with no response to primary chemotherapy is poor. 18 In addition, long term treatment with platinum may result in severe adverse effects. The combination of GEM and cisplatin combination has been reported to be less toxic than the combination of methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) but with similar efficacy. 19 Moreover, we have often used GEM as maintenance monotherapy after primary chemotherapy in advanced UC. The emergence of immune checkpoint inhibitors, such as pembrolizumab and avelumab, has also provided alternative treatments for second-line therapy in metastatic UC.10,11
We conducted a retrospective, real world review of patients who had received monotherapy following platinum-based combination chemotherapy for metastatic UC at our centre over a 12-year period. We investigated estimated survival times and possible prognostic factors for survival. Maintenance monotherapy was GEM, or an IO drug (i.e., pembrolizumab or avelumab) or both (i.e., one treatment after the other). We found that patients who received monotherapy had a significant increase in survival times compared with those that had received further combination therapy. While at the time of diagnosis, the combination (i.e., non-monotherapy) group had a higher rate of advanced disease than the monotherapy group, there was no difference between groups in overall response to primary chemotherapy. However, RECIST-defined progressive disease (PD) following first-line treatment was found to be an important prognostic factor for survival.
Despite more PD cases in the IO drug group, there was no difference in survival times between the IO drug group and the GEM group. These data suggest that advanced UC is highly sensitive to IO agents. However, one study found that the average duration of prolonged prognosis following pembrolizumab used as second-line therapy for UC was as short as three months. 10 Monotherapy with cytotoxic agents, such as GEM, has shown favourable results in previous studies.12,13 While median survival for GEM maintenance chemotherapy was reported to be approximately 12–15 months,12,13 we found the median survival time was significantly longer at 29 months in GEM group and 28 months in IO drug group. The difference between our study and previous results may be due to the longer observation period of our study compared with others.
We found that patients treated with IO drugs followed by GEM had a longer survival time compared with GEM monotherapy, suggesting a favourable relationship between GEM and the IO drugs. In previous studies, it has been reported that a decrease in cytidine, which is the main effect of GEM, activates interferon (IFN)-responsive genes. 20 Furthermore, IFN-α has been shown to be involved in conflicting immune responses (e.g., activating T lymphocytes while promoting the PDL1 expression) suggesting a possible explanation for the efficacy of IFN-α and PD-1/PD-L1 antibody combination therapy against the PD-1/PD-L1 antibody-resistant cancer. 21 Our findings suggest that GEM monotherapy as a second-line therapy may affect subsequent PD-1/PD-L1 antibody sensitivity.
Our study had several limitations including its retrospective design and its low number of patients. In addition, we included patients who received adjuvant/neoadjuvant chemotherapy as part of first-line treatment. As shown by our findings, these patients tend to have a longer survival rate than patients who do not receive adjuvant/neoadjuvant therapy. Nevertheless, this is the first study to demonstrate the benefit of GEM monotherapy for advanced UC in the advent of IO drugs. Moreover, our findings suggest that second-line IO drug monotherapy remains effective if followed by GEM monotherapy.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231173319 - Supplemental material for Real-world retrospective review of monotherapy following platinum-based chemotherapy for metastatic urothelial cancer
Supplemental material, sj-pdf-1-imr-10.1177_03000605231173319 for Real-world retrospective review of monotherapy following platinum-based chemotherapy for metastatic urothelial cancer by Hideaki Uchida, Daisuke Obinata, Shogo Takada, Tsuyoshi Yoshizawa, Junichi Mochida, Kenya Yamaguchi and Satoru Takahashi in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231173319 - Supplemental material for Real-world retrospective review of monotherapy following platinum-based chemotherapy for metastatic urothelial cancer
Supplemental material, sj-pdf-2-imr-10.1177_03000605231173319 for Real-world retrospective review of monotherapy following platinum-based chemotherapy for metastatic urothelial cancer by Hideaki Uchida, Daisuke Obinata, Shogo Takada, Tsuyoshi Yoshizawa, Junichi Mochida, Kenya Yamaguchi and Satoru Takahashi in Journal of International Medical Research
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by JSPS KAKENHI (grant numbers JP19H03793 and JP19K09740).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
