Abstract
Premature ejaculation (PE) is one of the major causes of sexual dysfunction. Levosulpiride is an off-label medicine used to treat PE, but no review on its efficacy exists. A systematic review and meta-analysis was performed to determine the efficacy of levosulpiride in treating PE. Databases PubMed, Science Direct, and Google Scholar were searched. Randomized control trials (RCTs) comparing levosulpiride with placebo or other medicine were selected. Odds ratio (OR) of improved intravaginal ejaculation latency time (IELT) was calculated. A total of 97 articles were retrieved from database search, of which only four RCTs containing 203 men met the selection criteria. All four RCTs were included in systematic review while only two were included in meta-analysis. A high selection and detection bias was found in both of these studies. Meta-analysis also showed the odds of improving IELT in PE patients using levosulpiride to be significantly higher (
Introduction
Premature ejaculation (PE) is among the most common sexual dysfunctions of men (Porst et al., 2007; Waldinger et al., 2004). It affects more than 50% of men in some populations (Irfan et al., 2020). PE has negative impacts on these men’s quality of life, as well as their sexual partners (Rosen & Althof, 2008; Sridharan et al., 2018). The International Society for Sexual Medicine (ISSM) defines PE based on three criteria: the men who have from the first intercourse persistently occurring ejaculation in ≤ 1 min of intercourse (lifelong PE) or significantly reduced ejaculation time (≤ 3 min) later in life (acquired PE), failed to delay ejaculation almost all the time of sexual intercourse, and have developed negative personal and mental conditions (e.g., bother, frustration, distress) and eventually sexual avoidance (Serefoglu et al., 2014).
PE may be caused by neurobiological or psychological defects (Hellstrom, 2011), but recently, organic causes have also been discovered, identified locally in the penis and central or peripheral nervous system (Greco et al., 2002). Upon stimulation, the glans penis relays sensory information to three ejaculatory centers (medial preoptic area, paraventricular nucleus, and periaqueductal gray) in the brain, which integrate the peripheral events of the seminal emission, ejaculation, and orgasm. The nucleus paragigantocellular is then modulates the efferent output of dopamine (DA), which exerts inhibitory effects on the serotonergic neurons responsible for inhibiting ejaculation (Bathla et al., 2020). In this way, the release of DA may inhibit the release of serotonin and ultimately cause ejaculation.
The central control of the ejaculatory pathway is primarily governed by serotonergic and dopaminergic neurons, whereas other chemical factors like acetylcholine, nitric oxide, neuropeptides, norepinephrine, gamma-aminobutyric acid, and oxytocin are secondarily involved (Bathla et al., 2020; Peeters & Giuliano, 2008). The release of DA is followed by the stimulation of DA receptors (Peeters & Giuliano, 2008). DA when binds to D2 receptors induces ejaculation (Kalejaiye et al., 2017). D2 receptor agonists are reported to decrease latency time and ejaculatory threshold and increase the frequency of ejaculation in rats (Kalejaiye et al., 2017; Peeters & Giuliano, 2008; Rosen et al., 1999). The damage to dopaminergic neurons increased the latency time of ejaculation (Rosen et al., 1999).
Selective serotonin reuptake inhibitors (SSRIs) could be an effective treatment of PE, but they impose adverse side effects, such as fatigue, dry mouth, dizziness, nausea, headache, and sleep disturbances (Liu et al., 2020). They can even cause other types of sexual dysfunction, such as anorgasmia and decreased sexual desire, leading to their discontinuation (Bathla et al., 2020).
Another approach to PE treatment is the inhibition of the synthesis or release of DA and/or D2 receptors. In this regard, levosulpiride (N-[2-methoxy-5-sulfamoyl benzamide]), a levorotatory isomer of sulpiride, derived from benzamide serves as an antagonist of D2 receptors on the dopaminergic neurons of the central and peripheral nervous system (Gong et al., 2014; O’Connor & Brown, 1982; Rossi & Forgione, 1995). Sulpiride is mainly used to treat psychosis associated with schizophrenia and major depressive disorder, and rarely anxiety and mild depression. Sulpiride is approved in most Asian, European, South African, and Central and South American countries. The U.S. Food and Drug Administration (FDA) has also approved amisulpride that is used as an alternative to sulpiride. Levosulpiride is approved in various Asian (India, Pakistan, South Korea, and China) and European (Italy, Spain, Cyprus, Malta, and Romania) countries. It is not approved in the United States, Canada, and Australia.
However, levosulpiride has shown therapeutic effects primarily in psychiatric and motor disorders of the upper gastrointestinal tract, such as depression and schizophrenia (Mucci et al., 1995), gastroesophageal reflux disease, irritable bowel syndrome, dyspepsia and emesis (Corazza & Tonini, 2000), and vertigo, and also PE (Gong et al., 2014). Some previous studies have reported improvements in intravaginal ejaculation latency time (IELT) in men with PE when they were treated with levosulpiride (Greco et al., 2002; Hussain et al., 2010). However, a few studies have reported no significant improvement after levosulpiride treatment (Bathla et al., 2020; Mohan, 2019). This review aims to report the efficacy of levosulpiride in the treatment of PE and improvement of IELT.
Method
This review was conducted by following the Cochrane Handbook for Systematic Reviews of Interventions (J. P. T. Higgins et al., 2020). The Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) was used to evaluate the quality of evidence (Guyatt et al., 2008).
Search Strategies
The databases PubMed, PubMed Central, Science Direct, and Google Scholar were searched using the keywords “levosulpiride,” “premature ejaculation,” and “treatment.”
Eligibility Criteria
Original articles on randomized control trials (RCTs) published in English and that compared the efficacy of oral levosulpiride doses with a placebo or other medicines were included in the review. For the meta-analysis, only RCTs comparing the efficacy of oral levosulpiride doses with a placebo were selected. There was no restriction on age or type of PE.
Study Selection
The articles’ titles and abstracts were screened by two authors (AA and MI). Subsequently, full texts were also screened if an article met the eligibility criteria. Study eligibility was assessed independently by both authors, who documented reasons for exclusion. Any discrepancy related to study selection was resolved via discussion.
Data Extraction
Author information, the year of the study, study region, subject age, method of diagnosis, number of participants, therapy duration, and occurrence of adverse events were independently collected from the eligible studies. The primary outcome was IELT, whereas the secondary outcome was adverse effects. IELT of < 1 min was a criterion for PE (Serefoglu et al., 2014).
Risk of Bias Assessment
Risk of bias was assessed using Review Manager 5.4 (RevMan 2020) based on the presence of a blinded randomization method, blinding of the outcome assessors, allocation concealment, completeness of the outcome data, the selectivity of the outcome reporting, and other biases. Each article’s risk of bias was then categorized as “low,” “high,” or “unclear.” Any discrepancy was resolved via discussion.
Statistical Analyses
Treatment effect was calculated as an odds ratios (OR) with 95% confidence intervals (CIs). Using a random-effect model, heterogeneity was assessed and reported first at face value by comparing the populations, interventions, settings, and outcomes, and then with the
To investigate the impact of the risk of bias for sequence generation and the included studies’ allocation concealment, a sensitivity analysis was performed. For the studies deemed sufficient, the construction of funnel plots assessed possible reporting bias, small study biases, or both.
Grading the Quality of Evidence
The GRADE approach was used to assess the quality of evidence for the primary as well as secondary outcome. Quality was classified as “high,” “moderate,” “low,” or “very low” based on the presence of the risk of bias, indirectness, inconsistency, imprecision, and publication bias. GRADEpro software was used to visualize the quality of evidence for each of the two outcomes, and the assessment was compiled with a Summary of Findings table.
Results
Study Selection
A total of 97 studies were retrieved by searching three electronic databases. After removing duplicates, the titles and abstracts of 89 records were screened. The full texts of nine studies were then assessed. Finally, four studies were included in the qualitative analysis (Bathla et al., 2020; Greco et al., 2002; Hussain et al., 2010; Mohan, 2019), and two were included in the meta-analysis (Figure 1; Greco et al., 2002; Hussain et al., 2010).

Flowchart of Studies Selection
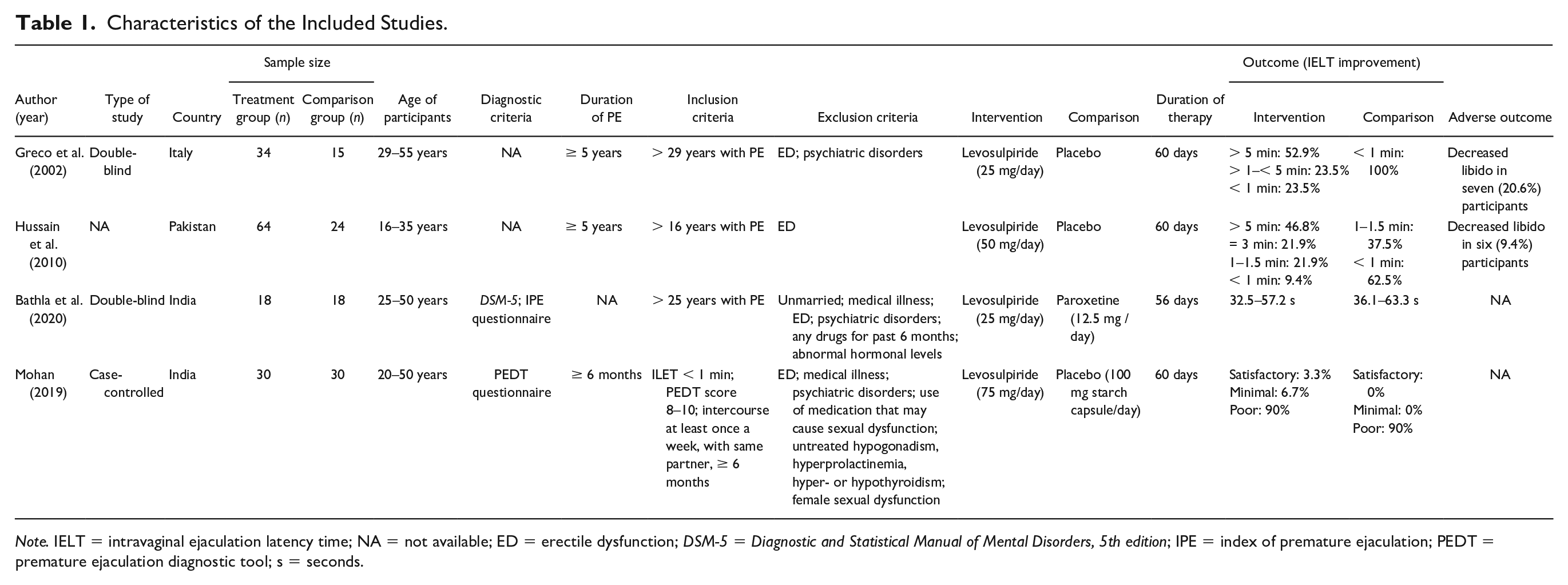
Characteristics of the Included Studies
The studies (
Characteristics of the Included Studies.
Systematic Review
The first double-blinded RCT reported that men with PE (
The second study reported on a 5-year study that included men with PE (
The third double-blinded study included 18 men with PE (25–50 years old) treated with 25 mg of levosulpiride and another 18 patients treated with 12.5 mg of paroxetine once daily for 8 weeks. IELT improved from 32.5 to 57.2 s in the levosulpiride group, whereas it improved from 36.1 to 63.3 s in the paroxetine group (Bathla et al., 2020).
The last study featured men suffering from PE for the past 6 months (
Meta-Analysis
Only two RTCs were included in the meta-analysis that fulfilled the eligibility criteria (Greco et al., 2002; Hussain et al., 2010).
Participants
The first study was conducted on men (29–55 years old) suffering from PE for at least the past 5 years (Greco et al., 2002), as did the second study, albeit with a younger age range (16–35 years old; Hussain et al., 2010). Neither of these studies stated the setting from which the participants were recruited.
Intervention
The participants in both studies were randomized into the intervention and control groups. One study used levosulpiride as an intervention drug in a daily 25 mg of dose (Greco et al., 2002), and the other prescribed levosulpiride in 50 mg of daily doses (Hussain et al., 2010). The control group was treated with a placebo in both studies (Greco et al., 2002; Hussain et al., 2010). Both studies assessed their outcomes after 60 days.
Outcomes
Both studies reported the primary outcome as changes in IELT (Greco et al., 2002; Hussain et al., 2010).
Quality of the Studies
The proportion of studies with a “high,” “low,” or “unclear” risk of bias for each indicator is presented in Figure 2, while the risk of bias indicators themselves are presented in Figure 3.

Risk of Bias Graph: Review Authors’ Judgments About Each Risk of Bias Item Presented as Percentages Across All Included Studies

Risk of Bias Summary: Review Authors’ Judgments About Each Risk of Bias Item for Each Included Study
Allocation
Neither of the two studies reported its method of participant allocation or randomization (Greco et al., 2002; Hussain et al., 2010).
Blinding
Only one of the two studies was double-blinded (Greco et al., 2002), and the other study did not report blinding (Hussain et al., 2010).
Incomplete Outcome Data
There were no dropouts in either of the two studies (Greco et al., 2002; Hussain et al., 2010).
Selective Reporting
Both studies reported their outcomes as specified in their methods sections (Greco et al., 2002; Hussain et al., 2010).
Efficacy in Improving IELT
Studies (RCTs)
The total number of improved PE patients treated with levosulpiride or a placebo, the accompanying ORs, and the CIs are presented in Figures 4 and 5. The odds of improving IELT for the PE patients using levosulpiride were significantly higher (

Forest Plot and Individual and Pooled Odds Ratios (95% CI) of Patients Who Improved IELT to That Poorly Improved/Not Improved (< 1 min) IELT When Treated With Placebo or Treatment

Forest Plot and Individual and Pooled Odds Ratios (95% CI) of Patients Who Improved IELT > 5 min to That Improved < 5 min IELT When Treated With Placebo or Treatment
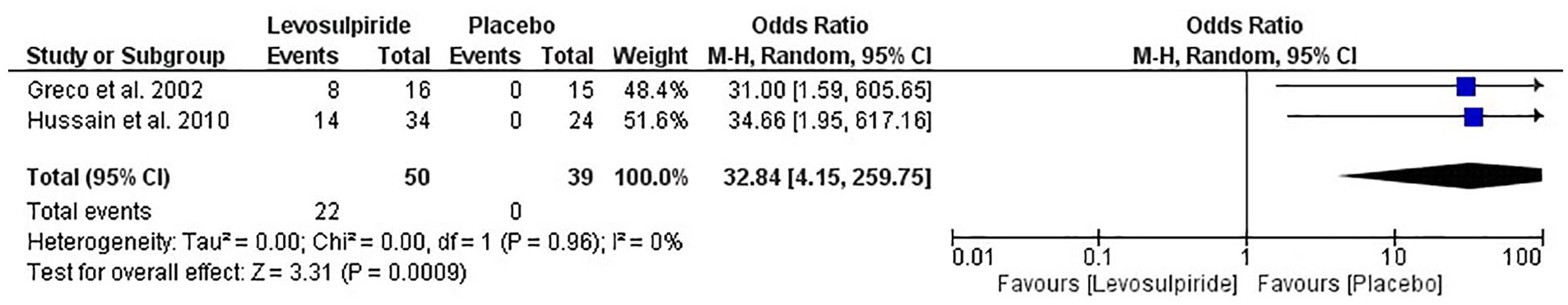
Forest Plot and Individual and Pooled Odds Ratios (95% CI) of Patients Who Improved IELT > 1 min But < 5 min to That Poorly Improved/Not Improved (< 1 min) IELT When Treated with Placebo or Treatment
Adverse Events
The first study reported a fall in libido in seven out of 34 patients (Greco et al., 2002), and the second reported a fall in libido in six out of 64 patients (Hussain et al., 2010).
Quality of the Evidence
The overall quality of the evidence was high (Table 2). The risk of bias was considered serious for method of participant randomization and allocation concealment bias. One of the studies did not report blinding either. The selection and detection biases were also high in both studies. Inconsistency, indirectness, and imprecision were not serious, effects were very large and no publication bias was detected that made overall risk of bias low.
Summary of Findings.
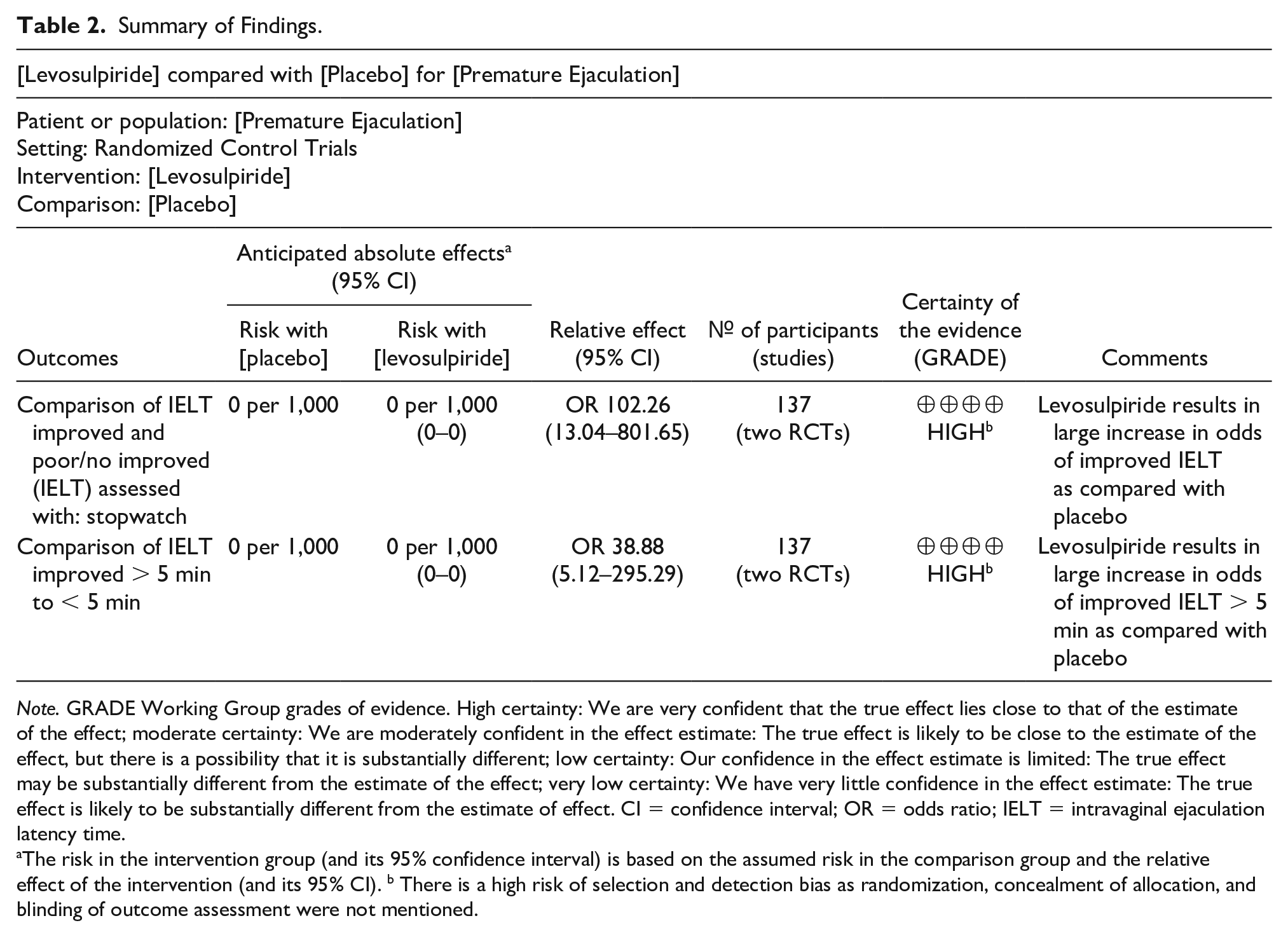
The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). b There is a high risk of selection and detection bias as randomization, concealment of allocation, and blinding of outcome assessment were not mentioned.
Discussion
PE is generally diagnosed when a man climaxes and ejaculates before his desire to do so. The causes of PE may be physiologic, psychogenic, or behavioral (Aggarwal et al., 2017; Hellstrom, 2011). The sexual response cycle in men has four phases, including a sequential phase, a plateau phase, ejaculation, and orgasm phase and a resolution phase (Georgiadis & Kringelbach, 2012). The sequential phase starts with the swelling of the genitalia, the onset of erection on stimulation and sexual thoughts. The plateau phase is characterized by delayed ejaculation and sexual intercourse. After the plateau phase, the sexual pleasure rises and reaches its peak resulting in the onset of ejaculation and orgasm phase (Gillman & Gillman, 2019).
The process of ejaculation is a spinal reflex action modulated by the cerebral, parasympathetic, and sympathetic pathways and characterized by two consecutive processes that are emission and expulsion (Clement & Giuliano, 2016; Giuliano & Clement, 2012). These processes are responsible to propel semen into the urethra, through, and out of the urethra. The process of emission is responsible for forwarding semen into the posterior urethra while the process of expulsion characterized by rhythmic muscular contraction propels semen through the urethra and out of penis (Clement & Giuliano, 2016). The nerve impulses that during orgasmic plateau reach to the brain through ascending sensory pathways stimulate interneurons to send signals back to the concerned glands muscles to release and expulse semen (Aggarwal et al., 2017; Peeters & Giuliano, 2008). The DA, serotonin, and nitric oxide and their receptors are found abundant in the ejaculatory neuronal pathways (Giuliano & Clement, 2012). These neurotransmitter act in antagonistic manner, serotonin and nitric oxide being inhibitory delay ejaculation while DA being excitatory stimulate the process of ejaculation (Kalejaiye et al., 2017; Rosen et al., 1999). The ejaculatory phase is then followed by the resolution phase with genital detumescence and flaccid penis. In patients with PE, the excitement phase may become steeper, plateau phase becomes shorter with early ejaculation (Buvat, 2011).
Although PE is considered among one of the most prevalent sexual dysfunctions the status still is controversial due to its subjective nature. The self-description of PE varies from patient to patient that may have resulted in varied published epidemiological data (Irfan et al., 2020; Porst et al., 2007; Waldinger et al., 2004). Therefore, now, it is required to confirm the diagnosis of PE on the basis of all three criteria of decreased delay in ejaculation (IELT ≤ 1 min) in the plateau phase, less or no control to delay ejaculation and presence of distress and bothering to the early ejaculation (Serefoglu et al., 2014). Premature ejaculation diagnostic tool (PEDT) is the most used and reliable tool to diagnose PE (Symonds et al., 2007).
Similarly, the treatment options for PE are also varied and controversial. A number of psychological, behavioral, and pharmacotherapies are available for PE but all are considered off-label as none of these is approved by FDA (Shindel et al., 2022). The common and first-line treatments of PE are behavioral therapies, counseling, strengthening exercises of pelvic floor muscles, decreasing sensitivity using condoms, or topical anesthetics, medications, such as antidepressants, analgesics, and phosphodiesterase-5 inhibitors (N. Gillman & Gillman, 2019). The traditional practitioners may also suggest diversion of mind during vaginal intercourse, which is not only useless in this case, but also brings other sexual dysfunctions like detumescence of the penis leading to erectile dysfunction and anorgasmia. According to the guidelines of the American Urological Association, the first-line pharmacotherapy should be on-demand clomipramine or dapoxetine (SSRIs) and penile anesthesia. If a patient failed first-line, may be treated with tramadol (opioid) or α1-adrenoreceptor antagonists (Shindel et al., 2022).
The most common and effective pharmacological treatment of PE are antidepressants, such as SSRIs, but they have adverse side effects (Bathla et al., 2020; Liu et al., 2020). SSRIs are designed for chronic use that block pre-synaptic membrane serotonin transporters and thus inhibit the serotonin reuptake and breakdown resulting in increased availability of the serotonin for the postsynaptic receptors of serotonin that may delay the process of ejaculation (A. Higgins et al., 2010). However, even most modern SSRIs could not be used for a longer time due to associated threats of degraded mental health and other sexual dysfunctions, such as erectile dysfunction, anorgasmia, and hypoactive sexual desire disorder. Sudden discontinuation may also bring withdrawal effects (Aggarwal et al., 2017; Giuliano & Hellstrom, 2008). The modern SSRIs are also suspected to have a much lower ejaculation delaying effect compared with the traditional SSRIs (Aggarwal et al., 2017).
Another approach to PE treatment, the inhibition of the excitatory pathway through inhibition of DA and its receptors could be useful. Very few studies are done on this aspect of delaying ejaculation using levosulpiride an antagonist of D2 receptors with contradictory results. This review aims to report the efficacy of levosulpiride in the treatment of PE and the improvement of IELT.
The present review aimed to include all RCTs on the efficacy of levosulpiride as a PE treatment. Only four RCTs were available for the review that compared an intervention group of PE patients who took levosulpiride and a control group that took a placebo or paroxetine. The reason for this scarcity could be that levosulpiride, an antipsychotic drug, is prescribed mainly for the management of psychiatric disorders, such as schizophrenia and depression, and motor disorders of the upper gastrointestinal tract, such as irritable bowel syndrome, gastroesophageal reflux disease, nausea, vomiting, and dyspepsia. It is only used as an off-label medication for PE. Although the outcome measure was the determination of improvement to IELT, in all of the trials, the definition of IELT improvement differed. None of the studies defined PE or separated lifelong or acquired PE according to the definitions provided by ISSM. The sample size was also low. In addition, patient age, drug administration duration, and dosage were different, thereby limiting this review’s generalizability.
Of the four, only two RCTs fulfilled the criteria for a meta-analysis. Despite this limited inclusion and the issue of generalizability, the meta-analysis’s results are important in showing a significant increase in IELT in an increased proportion of PE patients treated with levosulpiride compared with a placebo. The quality of evidence in these two studies was also high. Some assessments had an unclear and high risk of bias considered serious risk of bias. There was also a lack of information on randomization, blinding of the outcome assessment, and allocation concealment in both studies, leading to treatment effect bias, detection bias, and selection bias, respectively (Greco et al., 2002; Hussain et al., 2010). In one study, lack of double-blinding also posed a high risk of performance bias (Hussain et al., 2010). Reporting bias due to selective reporting, attrition bias due to incomplete outcome data, and other biases were low in both of the included studies (Greco et al., 2002; Hussain et al., 2010). Multiple databases were searched and reference lists of all related articles were screened to find out related studies to reduce the publication bias.
There was very low heterogeneity among both RCTs in the meta-analysis (Greco et al., 2002; Hussain et al., 2010). Using the GRADE approach, the assessment of the overall confidence level that contributed to the present review was of high quality. The individual effects of significant improvements in patient IELT reported by both studies are strengthened by the pooled effect that treating PE with standard doses of levosulpiride is very effective than placebo. However, two other studies have suggested that levosulpiride does not improve IELT (Bathla et al., 2020; Mohan, 2019).
The trials included in meta-analysis reported mild side effects, including a fall in libido, perhaps due to ejaculation delays or hyperprolactinemia, as in one study that reported increased prolactin concentrations in nine out of 34 patients (Greco et al., 2002). The frequencies of side effects were almost similar in the two treatment groups. These side effects did not influence patient participation or the studies’ completion (Greco et al., 2002; Hussain et al., 2010). These side effects are less in number and less severe compared with those of SSRIs, such as paroxetine or dapoxetine, which are highly effective but have more adverse side effects, including fatigue, dry mouth, dizziness, nausea, headache, and disturbed sleep, and can even cause other types of sexual dysfunction, such as anorgasmia (Bathla et al., 2020; Liu et al., 2020).
Limitations
Although the present meta-analysis was the first to demonstrate that levosulpiride may significantly improve IELT, a subgroup analysis based on dosage and treatment duration could not be performed due to having only two available studies. In addition, none of the studies defined PE or separated lifelong or acquired PE according to the definitions provided by ISSM (Serefoglu et al., 2014). The sample size was also low. It is suggested to further evaluate the effectiveness of levosulpiride for improving IELT in PE patients compared with placebo groups in double-blinded RCTs.
Conclusion
The current review included all RCTs (
Footnotes
Acknowledgements
The authors would like to thank Ms. Ayesha Bibi, PhD scholar, PMAS-Arid Agriculture University, Rawalpindi, Pakistan, for her assistance with database searches.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
